Abstract
The NeoRAS phenomenon is defined as the conversion of tumor RAS status from mutant-type (MT) to wild-type (WT) after systemic chemotherapy in metastatic colorectal cancer (mCRC). Cetuximab, an anti-epidermal growth factor receptor (EGFR) antibody, is effective in patients with RAS WT mCRC but ineffective in those with RAS MT mCRC; however, its outcome in patients with NeoRAS WT mCRC is unclear. Herein, we report two cases of NeoRAS WT mCRC that responded clinically to anti-EGFR treatment. The first was a 40-year-old man with synchronous peritoneal metastatic rectosigmoid cancer. The first RAS testing on tumor tissue revealed a KRAS G12C mutation, which was converted to RAS WT after two lines of chemotherapy, as assessed by liquid biopsy. After initiating irinotecan plus cetuximab treatment, a computed tomography (CT) scan revealed that malignant ascites had resolved. The treatment was discontinued after 4 months because of disease progression. The second was a 68-year-old male patient with synchronous liver metastasis from sigmoid colon cancer. The KRAS G12D mutation, initially detected in tumor tissue, was not detected by liquid biopsy after six lines of chemotherapy. Cetuximab monotherapy was initiated, and the liver metastases shrank significantly. The patient continued cetuximab monotherapy for 8 months without disease progression. Our cases demonstrate the efficacy of anti-EGFR therapy for NeoRAS WT mCRC and highlight the importance of capturing the gene mutation profile throughout the clinical course for optimal treatment selection.
Introduction
Metastatic colorectal cancer (mCRC) is the second leading cause of cancer death worldwide. 1 Its prognosis remains poor, but the development of molecular targeting agents has extended the survival time of patients with mCRC. Currently, it is essential to evaluate the multiple gene alterations of an individual’s tumor and select appropriate molecular targeting agents for the treatment of mCRC.
RAS mutations (KRAS or NRAS exons 2, 3, and 4) are found in approximately 50% of patients with mCRC. 2 Previous studies have shown that anti-epidermal growth factor receptor (EGFR) antibodies, cetuximab and panitumumab, have lower survival benefits for patients with RAS mutant-type (MT) mCRC than for those with RAS wild-type (WT) tumors.3,4 Because RAS mutations are negative predictors of anti-EGFR antibody efficacy, international practice guidelines recommend RAS testing prior to initiating anti-EGFR therapy in patients with mCRC.5–7 Furthermore, the KRAS G12C mutation, which occurs in 3% of patients with mCRC, 8 has been identified as not only a negative predictor of anti-EGFR antibodies but also a novel treatment target for solid tumors, including mCRC.9,10
Previously, RAS testing was typically performed on tumor tissue obtained through biopsy or surgery. However, such tumor sampling is usually invasive and difficult to repeat. Thus, monitoring RAS mutational status has been difficult during chemotherapy. In addition, the results of RAS testing using tumor biopsy may be limited by spatial tumor heterogeneity. Tumor heterogeneity refers to the notion that a single tumor consists of numerous subclone cells and that analysis using a portion of the tumor tissue may not reflect the genetic abnormalities of the entire tumor within the patient. 11
Recent studies have demonstrated that analyzing circulating tumor DNA (ctDNA) in blood samples is a remarkable surrogate for tumor biopsy for detecting mutations and overcoming spatial tumor heterogeneity. Because such liquid biopsy using ctDNA is also a noninvasive method compared to conventional tissue sampling, it allows for monitoring of gene mutational status changes through the clinical course.12,13 In previous clinical trials, a comparison between the use of beads, emulsion, amplification, and magnetic digital polymerase chain reaction (PCR; BEAMing) technology to determine RAS mutational status in plasma ctDNA and the reference method of tumor tissue DNA revealed concordance rates ranging from 86% to 93%.13–17
The development of liquid biopsy using ctDNA has gradually revealed dynamic changes in RAS mutational status during chemotherapy in patients with mCRC. Several studies have revealed that the RAS mutational status of some patients with mCRC changed from RAS WT to MT during anti-EGFR therapy. 18 The emergence of RAS MT subclones is well described as a result of clonal evolution under treatment-induced selection pressure, resulting in acquired resistance to anti-EGFR treatment.19–21
On the other hand, reversion of RAS MT to WT was thought to be rare because of the expected evolutionary advantage of RAS MT clones during tumor evolution. 22 However, the reversion of RAS MT to RAS WT during chemotherapy, known as the NeoRAS phenomenon, has gained popularity in recent years. The mechanisms of the NeoRAS phenomenon remain unclear, and its frequency varies greatly among reports, ranging from 10.7% to 83.3%.23–29 It is worth considering whether patients with NeoRAS WT mCRC can benefit from anti-EGFR treatments as much as those with RAS WT mCRC, because treatment options for patients with RAS MT mCRC are limited in comparison to those for patients with RAS WT mCRC. However, few reports have described the clinical outcomes of anti-EGFR treatments for patients with NeoRAS WT mCRC. Herein, we report two cases of NeoRAS WT mCRC that achieved clinical response to anti-EGFR therapy, as well as a literature review.
Case 1: A 40-year-old male patient with synchronous peritoneal metastasized rectosigmoid cancer
A 40-year-old male patient presented with constipation and was diagnosed with synchronous peritoneal metastasized rectosigmoid cancer (T4aN2bM1c Stage IVC according to the Union for International Cancer Control TNM, 8th edition). Due to the rectal obstruction, an emergency laparoscope-assisted high anterior resection was performed. The primary tumor was pathologically evaluated and showed a well-differentiated adenocarcinoma. Mutational analysis of the primary tumor tissue was performed using the MEBGEN RASKET-B kit™ and the PCR-rSSO method (Medical & Biological Laboratories Co., Tokyo, Japan) and revealed the KRAS G12C mutation, NRAS, and BRAF WT. Microsatellite instability (MSI) testing using a PCR-based method was negative. In addition to the aforementioned gene alterations, genetic analysis of primary tumor tissue using the OncoGuide™ NCC Oncopanel System (Sysmex Corporation, Hyogo, Japan) revealed several oncogenic gene alterations (Figure 1). Following surgery, the patient was started on capecitabine, oxaliplatin, and bevacizumab (CapOX+BEV) as first-line systemic chemotherapy. Partial response (PR) was the best tumor response as assessed by Response Evaluation Criteria in Solid Tumors 1.1. Fifteen months after the initiation of CapOX+BEV, a computed tomography (CT) scan revealed an increase in ascites and suggested disease progression. Subsequently, the fluorouracil, leucovorin, irinotecan, and ramucirumab (FOLFIRI+RAM) regimen was initiated as second-line chemotherapy, but it was discontinued 1.5 months later due to disease progression. At the end of each first- and second-line chemotherapy, a liquid biopsy was performed using the OncoBEAM™ RAS CRC Kit (Sysmex Corporation, Hyogo, Japan), which is based on BEAMing technology. Although the KRAS G12C mutation was detected via mutational testing on primary tumor tissue, neither of the liquid biopsies detected the RAS mutation. Irinotecan plus cetuximab was started as third-line chemotherapy with the patient’s consent. After 2 months, a CT scan revealed that the ascites had vanished [Figure 2(a) and (b)]. There were no severe adverse events during the treatment. Four months after initiating cetuximab, peritoneal metastasis progressed. Subsequently, trifluridine/tipiracil (FTD/TPI) +BEV and regorafenib were started, both of which caused disease progression. This patient is currently receiving the best supportive care.
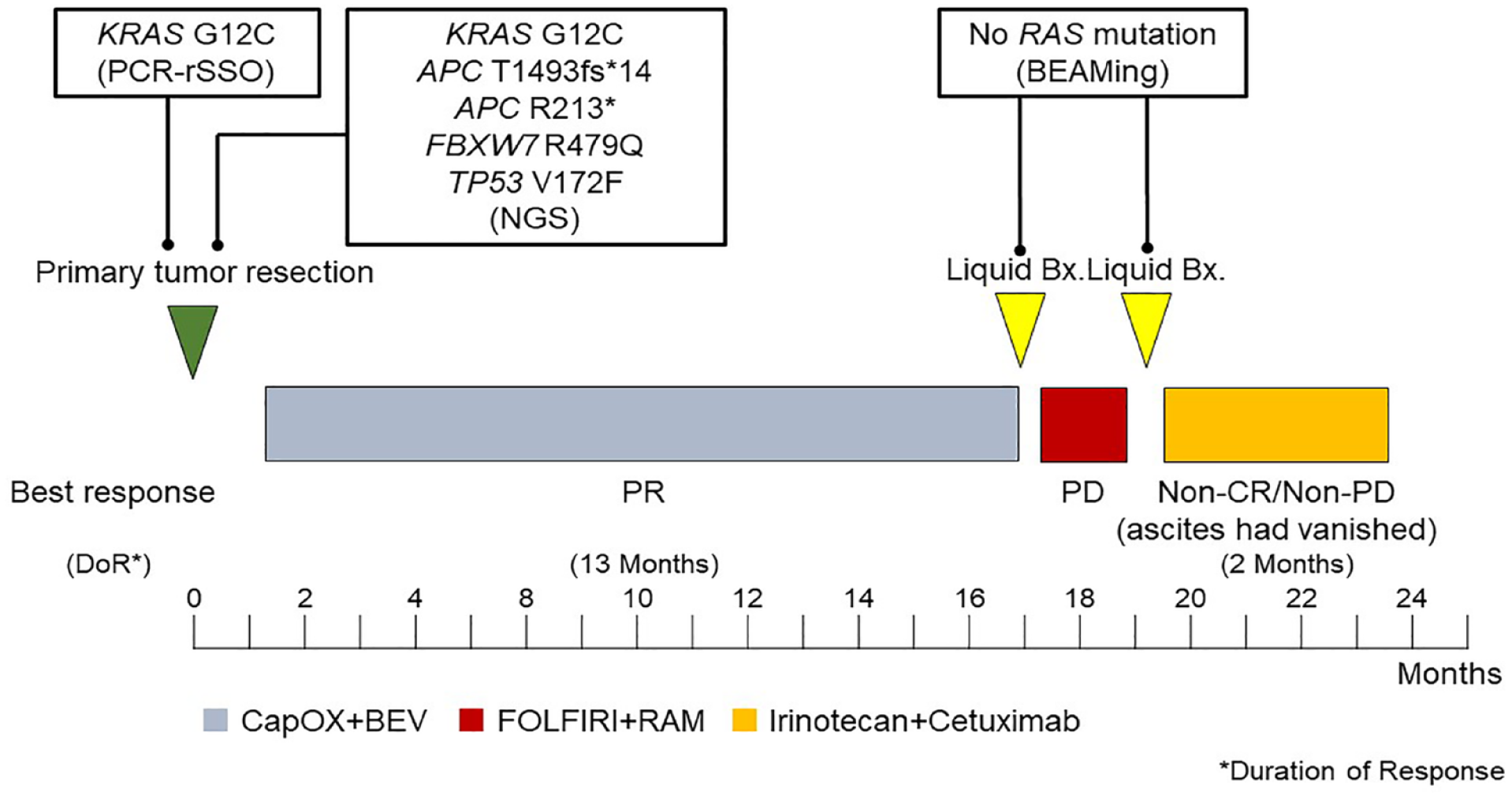
The clinical course of the case 1.

(a) The CT scan before administration of irinotecan plus cetuximab and (b) After 2 months, a CT scan revealed the ascites had vanished (arrowhead).
Case 2: A 68-year-old male patient with synchronous hepatic metastasized sigmoid colon cancer
A 68-year-old male patient was diagnosed with sigmoid colon cancer with multiple liver and lymph node metastases (T3N2bM1b Stage IVB according to the Union for International Cancer Control TNM, 8th edition). The primary tumor was pathologically diagnosed as a well-differentiated adenocarcinoma. Mutational analysis of the primary tumor tissue using the MEBGEN RASKET-B kit revealed the KRAS G12D mutation, NRAS, and BRAF WT. MSI testing using a PCR-based method yielded a negative result (nonMSI-High). After the primary tumor was resected, the patient was treated with six lines of systemic chemotherapy: FOLFOX (fluorouracil, leucovorin, and oxaliplatin) plus BEV, FOLFIRI+BEV, FTD/TPI+BEV, regorafenib, FOLFOX+BEV re-introduction, and investigational immunotherapy. The tumor response was PR during FOLFOX+BEV application as first-line chemotherapy. However, other treatment regimens did not result in tumor shrinkage, and all ended in disease progression. During the FOLFOX+BEV re-introduction treatment, comprehensive genomic analysis was performed using the Foundation One Liquid CDx™ Assay (Foundation Medicine, Cambridge, MA, USA). It revealed several actionable genomic alterations and a high tumor mutation burden, but not the KRAS G12D mutation (Figure 3). After the completion of investigational immunotherapy, the KRAS mutational status was reassessed using the OncoBEAM™ RAS CRC Kit, which revealed RAS WT, and cetuximab monotherapy was initiated as seventh-line therapy with the patient’s consent. After 3 months, a CT scan revealed remarkable shrinkage of the liver metastasis [Figure 4(a) and (b)]. The patient continued cetuximab monotherapy for 8 months without disease progression. No severe adverse events were observed during the treatment. However, he died of cancer progression 10 months after cetuximab initiation.

The clinical course of the case 2.

(a) The CT scan before administration of cetuximab monotherapy and (b) After 3 months, a CT scan revealed remarkable shrinkage of the liver metastasis (arrowhead).
Discussion
The use of anti-EGFR antibodies for NeoRAS WT mCRC remains controversial, but it is worth investigating because treatment options for RAS MT mCRC are limited. Several previous studies suggested the effectiveness of anti-EGFR antibodies in NeoRAS WT mCRC.23,30–34 However, all of these findings were obtained in combination with cytotoxic chemotherapy, and the extent to which anti-EGFR antibodies contributed to these findings is unclear. In Case 2, remarkable tumor shrinkage was observed following the initiation of cetuximab monotherapy. Our report is the first to show efficacy of anti-EGFR antibody monotherapy in NeoRAS WT mCRC. It more clearly suggests that anti-EGFR antibodies are effective, at least in some patients with NeoRAS WT mCRC. Several prospective clinical trials to evaluate the treatment outcomes of anti-EGFR antibodies for NeoRAS WT mCRC are currently underway, with results expected. 35
In our cases, there were discrepancies in the results of RAS gene mutation tests performed at different times. However, all the tests were properly performed according to the manufacturer’s instructions, and we do not believe that the discrepancies were caused by the testing technique. Though the mechanisms of such NeoRAS phenomenon are unclear, they may be explained by intratumor heterogeneity and chemotherapy-induced clonal evolution. In other words, systemic chemotherapy eliminates the initially predominant RAS MT subclones while increasing the proportion of the minor clone, the RAS WT subclone, resulting in the NeoRAS phenomenon. Indeed, Klein-Scory et al. demonstrated that RAS WT clones disappeared rapidly during chemotherapy and converted to RAS WT clones in more than 90% of patients with RAS MT mCRC who responded to chemotherapy. 36 Long RAS testing intervals, 37 as well as a good response to chemotherapy prior to second sampling,32,36 are significantly associated with the NeoRAS phenomenon, suggesting that clonal evolution by chemotherapy causes the NeoRAS phenomenon. From this perspective, the RAS MT subclone might be present in plasma samples from patients with NeoRAS WT tumors below the ctDNA assay limit of detection. 30 However, even if this were the case, it would not negate the efficacy of anti-EGFR treatment in NeoRAS WT mCRC because it has been demonstrated that patients with a low RAS mutant fraction (0.1–5%) might benefit from the addition of cetuximab to chemotherapy.4,38 Both of our two cases achieved PR according to the Response Evaluation Criteria in Solid Tumors during first-line chemotherapy, and the sampling interval for RAS testing was >15 months. The clinical courses of our cases support the existence of a link between chemotherapy-induced clonal evolution and the NeoRAS phenomenon. For future investigation, gene mutation analysis with deep sequencing using ctDNA at the baseline or tumor tissues from multiple lesions may be useful to clarify the involvement of intra-tumor heterogeneity and clonal evolution in the NeoRAS phenomenon.
Our two cases highlight the importance of repeating genetic analysis throughout the treatment course and understanding gene mutational profiles in real-time for patients with mCRC. In Case 1, the KRAS G12C mutation was detected in tumor tissue samples prior to chemotherapy induction, and the effectiveness of KRAS G12C inhibitors was anticipated.9,10 However, a liquid biopsy after first-line chemotherapies revealed that the tumor’s RAS status had converted to NeoRAS WT. Though the effectiveness of KRAS G12C inhibitors for NeoRAS WT mCRC is unclear, RAS testing just prior to treatment may be desirable for the indication of KRAS G12C inhibitors, taking the NeoRAS phenomenon into account. In Case 2, comprehensive genetic analysis prior to cetuximab administration revealed no negative predictors of anti-EGFR antibody efficacy, such as RAS MT, HER2 amplification, or BRAF V600E MT. 39 This case demonstrates that comprehensive genetic mutation analysis may be able to efficiently predict which patients will benefit from highly effective anti-EGFR antibodies.
In conclusion, we presented two cases of mCRC with RAS gene status divergence between tissue and blood sample testing. Both were initially diagnosed with RAS MT mCRC using primary tumor tissues, but liquid biopsy after systemic chemotherapy revealed RAS WT status. These patients with NeoRAS WT mCRC responded clinically to anti-EGFR therapy. Though the etiology and clinical significance of the NeoRAS phenomenon are not fully understood, it has been demonstrated that anti-EGFR antibodies are effective in some patients with NeoRAS WT mCRC. Furthermore, our cases highlight the importance of capturing the gene mutation profile throughout the clinical course, rather than just once, for optimal treatment selection.
