Abstract
Background:
Afatinib is indicated for advanced-stage non-small-cell lung cancer (NSCLC) with Epidermal Growth Factor Receptor (EGFR) and uncommon mutations. However, real-world studies on this topic are limited. This study aimed to evaluate afatinib as first-line therapy for locally advanced and metastatic NSCLC with uncommon EGFR mutations.
Patients and methods:
A retrospective study included 92 patients with advanced NSCLC with uncommon and compound EGFR mutations, treated with afatinib as first-line therapy. Patients were followed up and evaluated every 3 months or when symptoms of progressive disease arose. The endpoints were objective response rate (ORR), time-to-treatment failure (TTF), and adverse events.
Results:
The G719X EGFR mutation had the highest occurrence rate (53.3% for both monotherapy and the compound). By contrast, the compound mutation G719X–S768I was observed at a rate of 22.8%. The ORR was 75%, with 15.2% of patients achieving complete response. The overall median TTF was 13.8 months. Patients with the G719X EGFR mutation (single and compound) had a median TTF of 19.3 months, longer than that of patients with other mutations, who had a median TTF of 11.2 months. Patients with compound EGFR mutations (G719X and S768I) demonstrated a median TTF of 23.2 months compared to that of 12.3 months for other mutations. Tolerated doses of 20 or 30 mg achieved a longer median TTF of 17.1 months compared to 11.2 months with 40 mg. Median TTF differed between patients with and without brain metastasis, at 11.2 and 16.9 months, respectively. Rash (55.4%) and diarrhea (53.3%) were the most common adverse events, primarily grades 1 and 2. Other side effects occurred at a low rate.
Conclusion:
Afatinib is effective for locally advanced metastatic NSCLC with uncommon EGFR mutations. Patients with G719X, compound G719X–S768I mutations, and tolerated doses of 20 or 30 mg had a longer median TTF than those with other mutations.
Background
Lung cancer is the most common malignancy and ranks as the leading cause of cancer-related deaths worldwide, including in Vietnam. In 2020, Globocan reported 26,262 new cases of lung cancer in Vietnam in both sexes, constituting 14.4% of all cancers, with 23,797 associated deaths, accounting for 19.4% of all deaths in both sexes. 1 In recent years, there have been considerable changes in non-small-cell lung cancer (NSCLC) treatment, and the 5-year survival rate of patients has changed substantially. For patients receiving targeted therapy or immunotherapy, the 5-year survival rate is approximately 15–50% depending on the type of biomarker. 2 Globally, phase III multi-center clinical trials show favorable responses to tyrosine kinase inhibitors (TKIs) in patients with advanced-stage NSCLC with EGFR mutations. Among these, afatinib demonstrates a better treatment effect than chemotherapy. In the Lux-Lung 3 and Lux-Lung 6 studies, patients treated with afatinib experienced significantly longer median progression-free survival (PFS) afatinib of 11.1 and 11 months, respectively, than that of the chemotherapy-treated patients with PFS of 6.9 and 5.6 months.3,4 Real-world studies in Asia confirm the effectiveness of afatinib for patients with NSCLC with sensitive EGFR mutations (exon 19 deletion mutation and L858R point mutation in exon 21) and in patients with uncommon EGFR mutations, with a median PFS of 11–19.7 months.5,6 First-generation TKIs appear less effective for this group of patients, with an objective response rate (ORR) of only 40–50% and a median PFS of 6–7.7 months. 7 For patients with uncommon EGFR mutations (mutations G719X, L861Q, and S768I), analysis of three studies (LUX-lung 2, 3, and 6) revealed a median PFS of 8.2–14.7 months and median overall survival (OS) of 16.9–26.9 months. 8 Currently, osimertinib is indicated as a first-line treatment for patients with advanced NSCLC with uncommon EGFR mutations. 2 However, in Vietnam, the first- and second-generation TKIs remain the predominant drugs recommended for patients with NSCLC patient and EGFR mutations. Numerous real-world studies have been conducted to evaluate the therapeutic efficacy of afatinib.9,10 The objective of this study was to evaluate the outcomes of first-line afatinib treatment in patients with advanced NSCLC with uncommon EGFR mutations.
Methods
Patients
Patients with advanced-stage NSCLC with uncommon EGFR mutations received first-line afatinib treatment and were monitored at nine hospitals in three regions (North, Central, and South Vietnam) from April 2018 to June 2022.
Inclusion criteria
(1) NSCLC stages IIIB, IIIC, and IV as per the American Joint Committee on Cancer version 8, 2017, or recurrence after surgery or local treatment. (2) Patients with uncommon EGFR mutations and compound mutations. (3) Age >18 years. (4) No prior systemic treatment received. (5) Predicted survival of at least 12 weeks. (6) Complete medical records.
Exclusion criteria
(1) NSCLC stages I–IIIA. (2) Severe allergy to afatinib. (3) Exon 19 deletion EGFR mutation and an L858R point mutation in exon 21, excluding double mutations or other uncommon mutations. (4) Presence of hepatitis, cirrhosis, kidney failure, and combined interstitial lung disease. (5) Coexistence with other cancers. (6) Insufficient patient information during treatment and follow-up.
Methods
This study adopted a retrospective design with convenience sampling. Information on the diagnosis, treatment, and follow-up of patients was uniformly collected from medical records across all centers and uploaded to KoboToolbox.org. EGFR mutations were tested in tissue or serum samples using polymerase chain reaction or next-generation sequencing. Uncommon EGFR mutations were divided into (i) the main group (G719X, S768I, and L861Q), (ii) compound mutations with exon 19 deletion and L858R, and (iii) other uncommon mutation variables, including exon 20 insertion.
Patients received afatinib (Giotrif) at initial doses of 40, 30, or 20 mg, taken once (one tablet) daily. Dose selection depended on the age of the patient and performance status (PS) according to the Eastern Cooperative Oncology Group (ECOG); ECOG 0–1 denoted good PS, and ECOG 2–4 denoted poor PS. Patients on 40 or 30 mg could undergo dose reduction by 10 mg in case of grade 2, 3, or higher side effects recurrence. Conversely, patients who were initiated on 20 or 30 mg, if well tolerated, might see an increase in dose with improved PS. Dose adjustment was determined by the investigator based on the individual circumstances of the patient. Patients were monitored and evaluated every 3 months of treatment or when symptoms of disease progression were assessed by clinical examination, computed tomography of the chest and abdomen, brain magnetic resonance imaging, and bone scan.
Evaluation of treatment response was according to the Response Evaluation Criteria in Solid Tumors (RECIST 1.1). 11 Afatinib-resistant patients were identified by the presence of resistance mutations. Subsequently, the patient was treated with third-generation TKI if T790M was positive and other mutations, if any. Patients without resistance mutations underwent chemotherapy if PS was allowed. Adverse effects were assessed according to the National Cancer Institute Common Terminology Criteria for Adverse Events Standards, version 5.01, 2017. 12 The closing date for data analysis was 31 December 2022.
The primary criteria were time to treatment failure (TTF) and ORR. The secondary endpoints were disease control rate (DCR) and adverse events. TTF was defined as the duration from the initiation of afatinib treatment to the discontinuation date because of disease progression according to RECIST 1.1, a severe adverse event, adjusted, ineffective symptom relief with combination therapy, or patient’s preference to cease treatment. ORR and DCR were calculated according to the RECIST 1.1 criteria, in which ORR included patients with complete response and partial response, and DCR included patients with complete response, partial response, and stable disease.
Data processing was performed using SPSS software (version 22.0; IBM Corp., Armonk, NY, USA). The TTF was calculated using the Kaplan–Meier method to determine the median and 95% confidence intervals. The relationship between the TTF and clinical and paraclinical characteristics was analyzed using Cox regression. Differences between categorical variables were determined using the chi-square or Fisher’s exact tests. For continuous variables, differences were calculated using t-tests. Differences were considered statistically significant at p < 0.05.
Results
Characteristics of patients
In all, 92 patients participated in this study, with a median age of 63 years. The oldest was 86 and the youngest was 31. Of the patients, 43.5% were 65 years or older and 67.4% were men. Sixty-three percent of patients had no smoking history. Most patients had a good PS with an ECOG score of 0–1 (90.2%); only 9 patients (9.8%) had a poor PS with an ECOG score of 2–4. Of the patients with brain metastases, 26.1% were treated with afatinib, and most were diagnosed with stage IV disease (89.1%). G719X alone had the highest percentage of uncommon EGFR mutations (28.3%), followed by L861Q (14.1%), and the combination of G719X and S768I had the highest percentage (22.8%). Only 3.3% of patients had a single S768I mutation. Other EGFR mutations occurred at lower rates. The total number of patients harboring the G719X mutation (alone or in combination) was 53.3%. The results are summarized in Table 1.
Characteristics of patients.
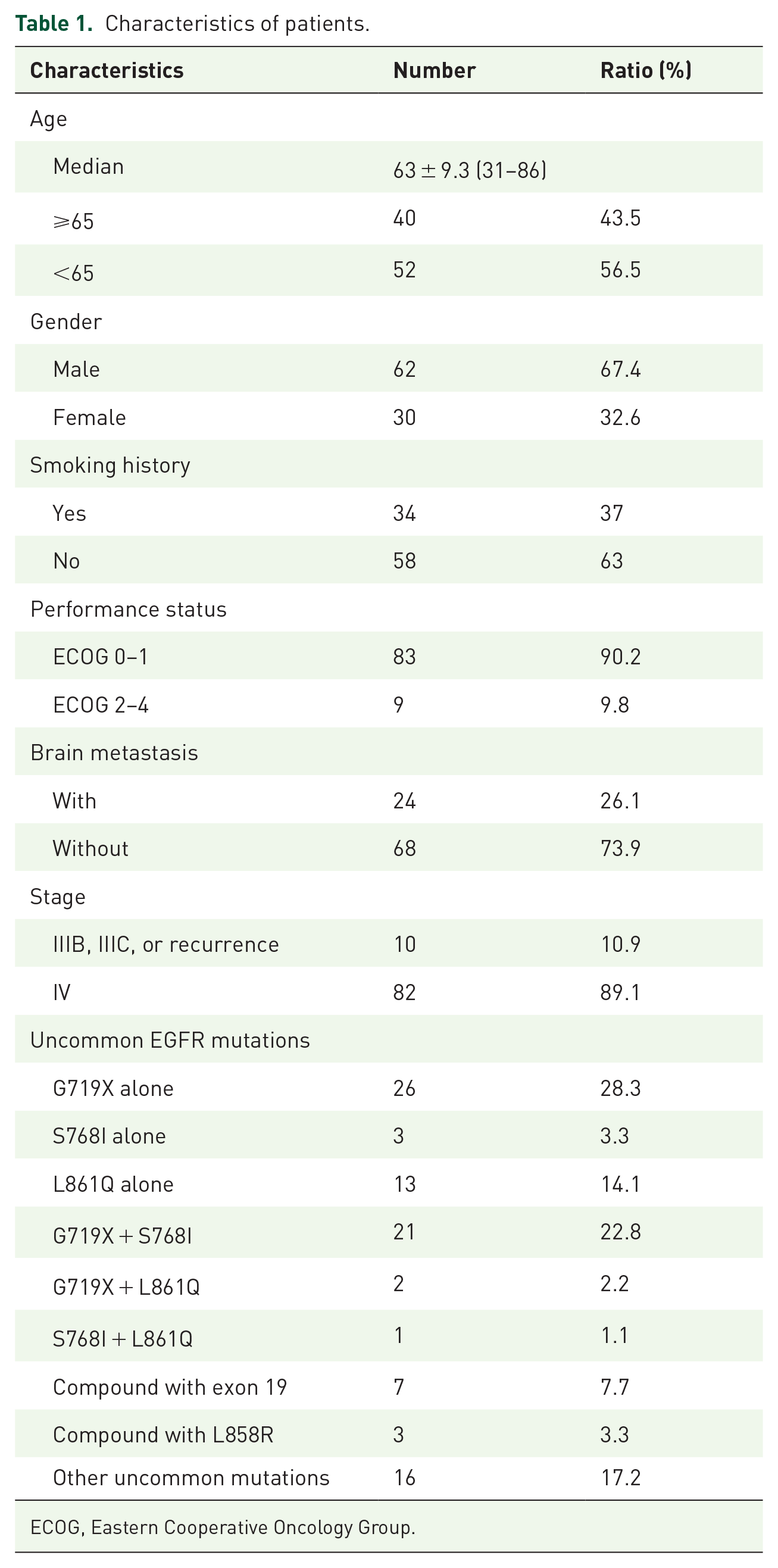
ECOG, Eastern Cooperative Oncology Group.
Results of targeted therapy with afatinib
Patients predominantly initiated treatment with a 30 mg dose, which constituted the highest proportion (59.8%), followed by 40 mg (38%). In all, 18 patients (19.6%) had an adjusted dose after 1 month of treatment, of which nine patients required a dose reduction from 40 to 30 or 30 to 20 mg, and nine escalated from 20 to 30 or 30 to 40 mg daily. A total of 20.7% of patients required dose reduction owing to toxicity during treatment. The tolerated dose of 40 mg accounted for only 24.1%, and the tolerated doses of 20 and 30 mg amounted to 73.9%. Local treatment of the brain was performed in eight patients, of whom three underwent radiosurgery and five received whole-brain radiation therapy. The median follow-up time was 22.2 months, ranging from 3.8 to 51.6 months. The complete and partial response rates were 15.2% and 59.8%, respectively. The proportion of patients with stable disease was 14.1%. The ORR was 75% and the DCR was 89.1%. The results are presented in Tables 2 and 3.
Treatment characteristics.

WBRT, whole-brain radiation therapy.
Patient responsibilities.

CR, complete response; DCR, disease control rate; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease.
The total number of events was 61. Median TTF was 13.8 ± 2.13 months (95% CI: 9.7–18 months). The estimated 2-year TTF rate was 23.6% (Figure 1). In all, 49 patients had the G719X EGFR mutation, including single and combined, with a median TTF of 19.3 ± 2.37 months (95% CI: 14.64–23.96 months), significantly longer than that of patients carrying other EGFR mutations with a median TTF of 11.2 ± 1.15 months (95% CI: 8.9–13.43 months), p = 0.000. Patients with the EGFR mutation compound G719X and S768I exhibited a median TTF of 23.2 ± 4.5 months (95% CI: 14.33–32 months), whereas the remaining group had a median TTF of 12.3 ± 1.55 months (95% CI: 9.23–15.3 months), p = 0.01. The median TTF of patients starting with a dose of 40 mg was 16.1 ± 2.86 months (95% CI: 10.52–21.74 months) and the median TTF of patients starting with a dose <40 mg was 13.8 ± 2.36 months (95% CI: 9.18–8.42 months), p = 0.18. After dose adjustment, only 24 patients tolerated the 40 mg dose with a median TTF of 11.2 ± 1.3 months (95% CI: 8.63– 3.7 months). This was noticeably shorter than the median TTF of the patient group that tolerated 20 or 30 mg dose, which was 17.1 ± 2.07 months (95% CI: 13.03–21.17 months), p = 0.029. The results are shown in Figure 2.

Time-to-treatment failure.

TTF based on G719X (a), G719X compound S768I (b) with other mutations, according to the starting dose (c) and tolerable dose (d).
The median TTF of patients with brain metastases was 11.2 ± 2.5 months (95% CI: 6.22–6.1 months) and the median TTF of patients without brain metastasis was 16.9 ± 1.73 months (95% CI: 13.54–20.32 months), p = 0.16. Multivariate analysis showed that the G719X mutation, starting dose, and tolerated dose were predictors of TTF, viz., HR: 0.51, p = 0.045 for G719X; HR: 0.24, p = 0.002 for the starting dose; and HR: 5.3, p = 0.001 for the tolerated dose (Table 4).
Analysis of the relationship between TTF and some clinical and paraclinical characteristics.
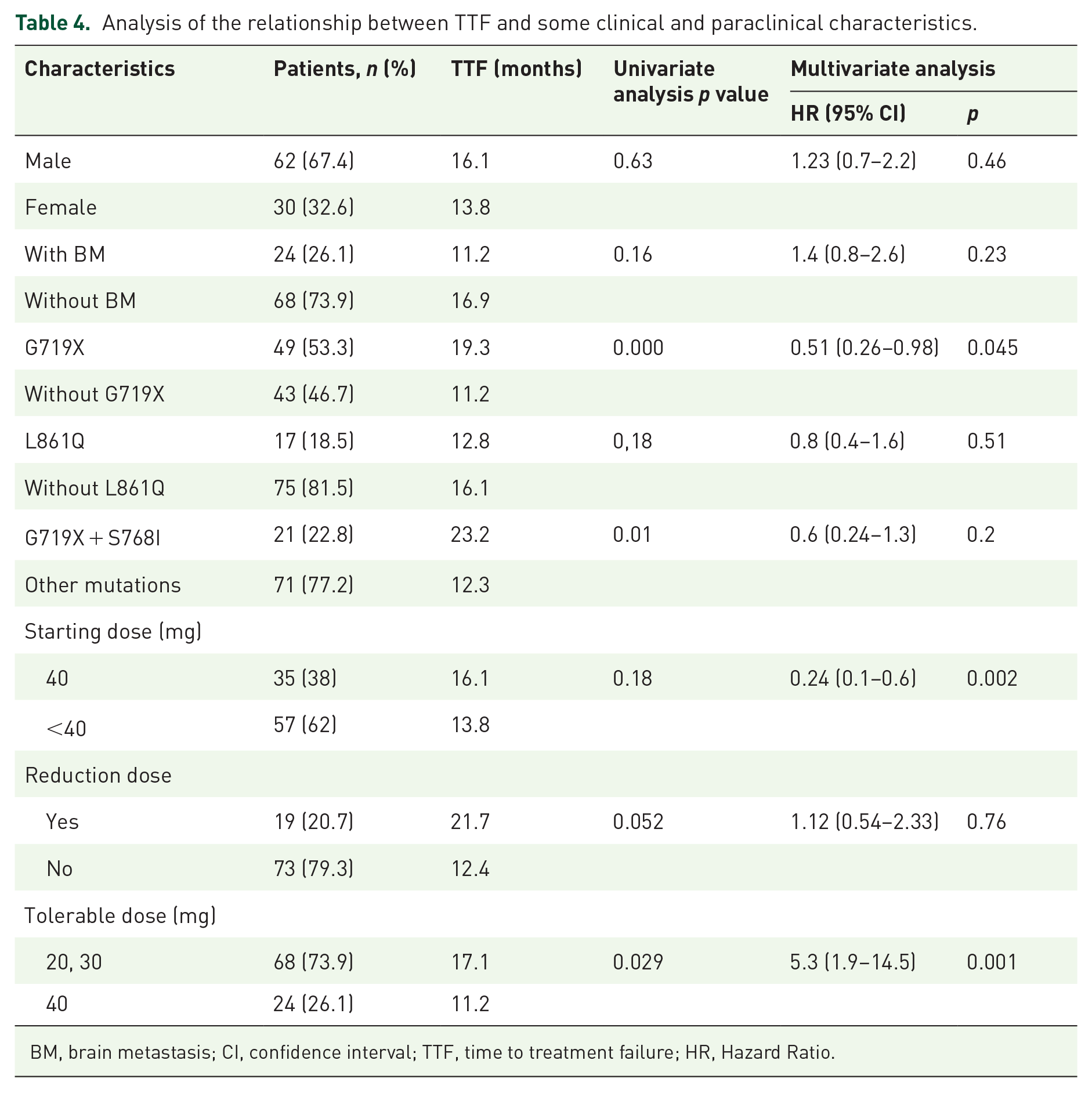
BM, brain metastasis; CI, confidence interval; TTF, time to treatment failure; HR, Hazard Ratio.
In total, 71 of 92 patients treated with afatinib had adverse events (77.2%), with rash and acne accounting for the highest rate (55.4%), followed by diarrhea (53.3%) and paronychia (41.3%). In 19.6% of the patients, stomatitis and other side effects were present in a lower proportion. Most adverse events were grades 1 and 2; only 5.4% of the patients had rash and acne, 2.2% had diarrhea, and 6.5% had grade 3 paronychia. Two patients discontinued treatment because of adverse effects, equally divided between acne rash and diarrhea (Table 5).
Adverse events related to afatinib.
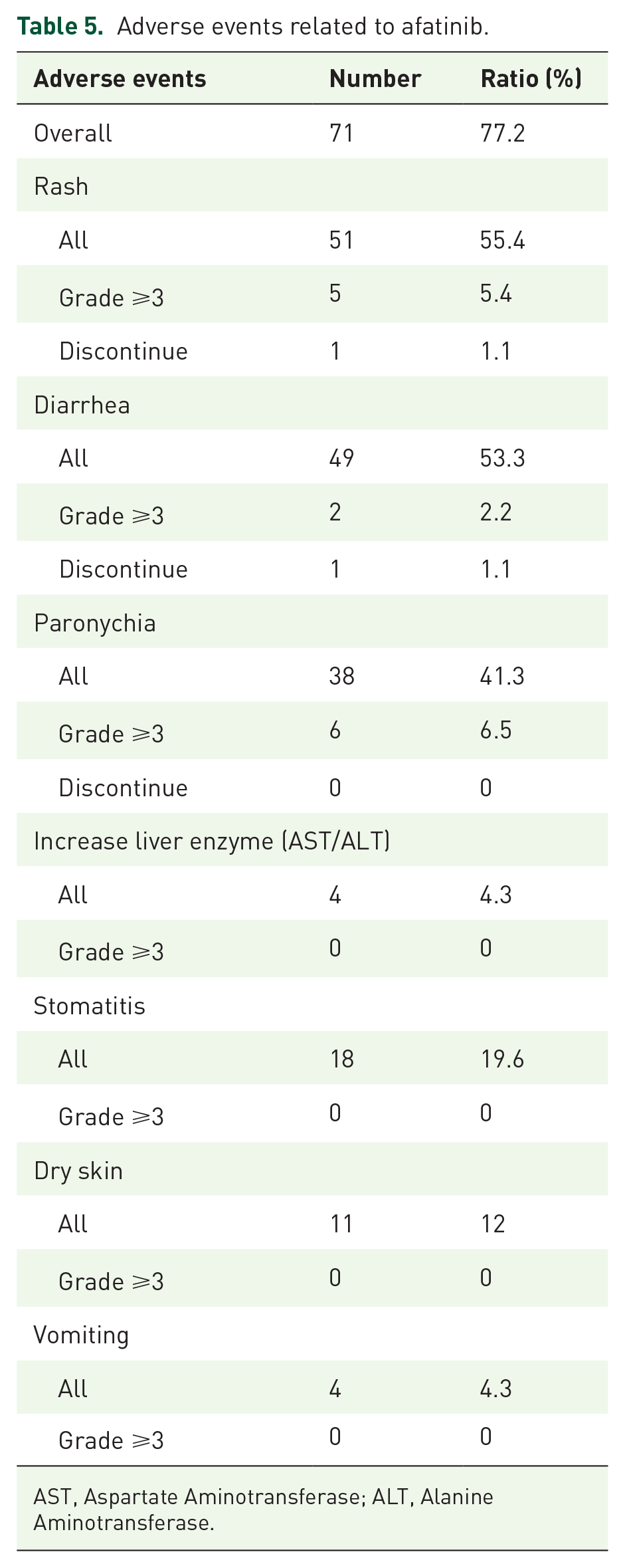
AST, Aspartate Aminotransferase; ALT, Alanine Aminotransferase.
Discussion
This was the first study in Vietnam to evaluate the effectiveness of first-line afatinib treatment in 92 patients with advanced NSCLC and uncommon EGFR mutations, including those with brain metastases (26.1%). G719X was the most common among the uncommon EGFR mutations, followed by L861Q, with G719X and S768I being the most common. This aligns with recent studies highlighting G719X as the uncommon EGFR mutation with the highest rate.8,13,14
In clinical practice, afatinib dosing is often tailored according to the age and PS of the patient owing to the relatively common occurrence of side effects, especially severe diarrhea, which has been mentioned in current reports.3,4,15 However, in most real-world studies, 40 mg is the recommended dose for patients. Hsu et al. 14 showed that 66.7% of patients were assigned a starting dose of 40 mg and none were assigned a dose of 20 mg; however, 26.7% of patients had to reduce their dose from 40 to 30 mg. Kim et al. 16 initially treated patients with 40 or 30 mg afatinib; however, 67.8% of patients required one or two dose reductions, 80 patients tolerated a dose of 30 mg, and 32 tolerated a dose of 20 mg. In our study, the majority of patients started at 30 mg, with nine patients increasing their dose after 1 month of treatment owing to an improvement in PS, similar to the findings by Ho et al., where 10 patients (11.8%) experienced a dose increase. 17 Ultimately, 20% of patients required dose reduction because of toxicity. This rate was lower than those reported previously.14 –16 This difference may be attributed to 59.8% of patients starting at 30 mg instead of 40 mg. In contrast to the study performed by Kim et al., 16 where no patients tolerated the 40 mg dose, we observed that 24% of our patients tolerated it.
Yang et al. 8 conducted studies on the treatment response of Lux-Lung 2, Lux-Lung 3, and Lux-Lung 6 in patients with uncommon EGFR mutations and showed an ORR of 100% for the S768I mutation and 78% for the G719X mutation. In this study (median follow-up: 22.2 months), 15.2% of patients achieved complete response, with an ORR of 75%, and a DCR of 89.1%. A study conducted by Ho et al. 17 in Malaysia showed that the ORR in a group of patients with uncommon and compound mutations was 83.3%, which was higher than the ORR of patients with exon 19 deletion mutations (76.5%) or L858R (54.5%). Although our results were lower, the study conducted by Hsu et al. 14 on the three uncommon EGFR mutations, G719X, S768I, and L861Q, recorded an ORR of 63.3%, no complete responses, and a DCR of 86.7%. Evaluation of the survival time of patients and real-world effectiveness of afatinib for both common and uncommon EGFR mutations indicates positive outcomes for PFS, TTF, and OS.6,7,13,16 A study conducted by Tu et al. 6 in Taiwan showed that the overall median PFS was 12.2 months; however, a noteworthy aspect of the study is the marked median PFS of 19.7 months for patients with uncommon EGFR mutations. This result was higher than our results, with the median TTF of patients carrying the uncommon EGFR mutation at 13.8 months and less than a quarter of patients continued treatment at 2 years. Among patients with uncommon EGFR mutations (G719X, L861Q, and S768I), a pooled analysis of the studies of Lux-Lung 2, 3 revealed a median PFS of 8.2–14.7 months and a median OS of 16.9–26.9 months. 8 Shen et al. 5 showed that the median PFS of patients with uncommon EGFR mutations is 11 months. Assessing the relationship between survival time and some clinical and paraclinical characteristics, Hsu et al. found that patients carrying the G719X mutation tended to have a higher median PFS than non-G719X carriers with 18.2 and 13.1 months, respectively, p = 0.081, but multivariate analysis showed a statistically significant association with HR 0.578 and p = 0.027. In addition, the author found that patients without brain, bone, or liver metastases had a longer median PFS than patients with metastasis to these organs (p < 0.05). 14 In this study, we found that patients with G719X mutations, including single or compound mutations, had a longer median TTF than those without this mutation in both univariate and multivariate analyses (HR 0.51, p = 0.045). The patients carrying the L861Q mutation achieved a median TTF of 12.8 months. A pooled analysis by Yang et al. 18 also found that the median TTF of patients carrying the G719X mutation was longer than that of the L861Q mutation carrier at 14.7 and 10 months. In this study, patients with G719X and S768I compound mutations also achieved better median TTF results than patients without (p = 0.01). However, we did not observe any differences between patients with and without brain metastasis. Although osimertinib has also been approved for the treatment of patients with NSCLC with uncommon EGFR mutations, data have shown that the efficacy of afatinib is more favorable than that of osimertinib in this group of patients. Wang et al. studied 71 patients with NSCLC and uncommon EGFR mutations treated with afatinib or osimertinib. Although the ORRs between these TKIs were not statistically significant (60.6% for afatinib and 50.3% for osimertinib), the median PFS for patients treated with afatinib was significantly higher at 11 months than that in the osimertinib-treated group at 7 months (p = 0.044). In addition, there were no significant differences in ORR and PFS between afatinib and osimertinib in patients with brain metastases. 19 A Korean multicenter, open-label phase II study on 37 patients with NSCLC and uncommon EGFR mutations (G719X 19, L861Q 9, S768I 8, and another four patients) revealed a 50% ORR, with a median PFS of only 8.2 months, where 61% received first-line therapy. 20
Ho et al. 17 showed that no statistically significant differences were found in the median PFS of patients between different doses and between dose-adjusted and unadjusted patients. In contrast to this study, we found that patients who started treatment with 40 mg had a median TTF of 16.1 months, which was longer than patients who started treatment with <40 mg for 13.8 months. Although univariate analysis showed no significant difference, multivariate analysis using Cox regression revealed a significant result with HR 0.24 and p = 0.002. By contrast, patients tolerating the <40 mg dose achieved a longer median TTF than those tolerating the 40 mg dose, and this difference was statistically significant in both univariate and multivariate analyses, with an HR of 5.3, and p = 0.001. However, we did not observe such results in this study. Tu et al. 6 and Yang et al. 21 found no difference in efficacy between the 40 and 30 mg doses.
Adverse events commonly observed in patients with NSCLC treated with TKIs include rash and acne, paronychia, anorexia, diarrhea, increased liver enzymes, and drug-induced interstitial pneumonia. The specific events may vary depending on the generation of TKIs. In Lux-Lung 3, diarrhea and rash were the two most common side effects in patients treated with afatinib, with ratios of 95.2% and 89.1%, respectively (14.4% and 16.2% for levels 3 and above). 3 In our study, among 92 patients treated with afatinib, side effects were observed in 71, with acne rash being the highest at 55.4%, followed by diarrhea (53.3%), paronychia (41.3%), and stomatitis (19.6%). The rate of patients with grade 3 toxicity was only about 6%. This trend was also seen in a study by Hsu et al. 14 ; however, the rate of adverse events in this study was higher, with rash, diarrhea, and paronychia accounting for 92.2%, 81.1%, and 72.2%, respectively. A study by Tu et al. also showed that the proportion of patients with diarrhea, rash, and paronychia was the highest; however, the majority were grade 1 and grade 2. The rate of grade 3 toxicity was approximately 6%, and no patients had grade 4 toxicity. 6 The incidence of adverse events in our study was lower than that in other studies, which may be explained by the fact that most patients were treated with lower doses than in the clinical trials, which were initiated with a dose of 40 mg. Early dose adjustments, including a 9.8% reduction after the first month and 20% of patients reducing their dose during treatment, helped minimize the occurrence of grade 3 side effects. No instances of decreased red blood cells, white blood cells, platelets, or interstitial lung disease were observed with afatinib. However, two patients discontinued treatment because of diarrhea, rash, and grade 3 acne.
Conclusion
Afatinib is an effective treatment option for patients with advanced NSCLC harboring uncommon EGFR mutations. In particular, the benefit of afatinib treatment was clearly observed in patients with the G719X EGFR mutation and those tolerating doses of 20 or 30 mg.
