Abstract
Background:
Afatinib is indicated for patients with advanced-stage non-small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) mutations, including uncommon mutations. However, the differences in survival benefits between patients with different types of EGFR mutations remain unclear.
Objectives:
This study aimed to compare the effectiveness of afatinib treatment in patients harboring the EGFR-G719X mutation with that in patients carrying other uncommon EGFR mutations.
Design:
This was a retrospective study.
Methods:
Ninety-two patients with locally advanced and metastatic NSCLC, of whom 49 patients with EGFR-G719X mutations that were both single and compound, and 43 patients harbored other uncommon EGFR mutations, who were treated with afatinib as first-line treatment. The patients were followed up and evaluated every 3 months or when there were symptoms of progressive disease. The endpoints were the objective response rate (ORR), time-to-treatment failure (TTF), and overall survival (OS).
Results:
The average ages of patients with the EGFR-G719X and uncommon EGFR mutations were 62.7 years and 63.1 years, respectively. There were no significant differences in sex or smoking history between the two groups. In total, 28.6% of patients with the G719X mutation and 23.3% of patients with other mutations had brain metastases. The ORR of patients with the G719X mutation was 79.6%, which was 10% higher than that of patients with other EGFR mutations. Patients harboring the EGFR-G719X mutation had median TTF and median OS periods of 19.3 months and 31.4 months, respectively, which were significantly higher than those of patients carrying other mutations at 11.2 months. Subgroup analysis showed that TTF and OS benefits were observed in female patients, patients without brain metastasis, and patients with good performance status who harbored the G719X mutation.
Conclusion:
Patients with the EGFR-G719X mutation achieve significantly better TTF and OS benefits than those with other uncommon EGFR mutations.
Background
In patients with non-small cell lung cancer (NSCLC), epidermal growth factor receptor (EGFR) mutations related to the response to tyrosine kinase inhibitors (TKIs) belong to 4 exons 18–21 of the EGF gene and were divided into two groups. The group of mutations that increase the sensitivity of cancer cells to targeted therapy includes 85%–90% of exon 19 deletion mutations and L858R point mutations in exon 21. The second group of uncommon EGFR mutations includes G719X, S768I, L861Q, and 20 insertion mutations. 1 In a review by Zhang et al. in patients with NSCLC harboring uncommon EGFR mutations from five studies, the authors noted that the rate of G719X mutation on exon 18 was the highest at 26%, followed by the L861Q mutation on exon 21 with 17%, exon 20-S768I mutation with only 9%, and exon 20 insertion with 19%. 2 Some studies evaluating the first-line treatment effectiveness of EGFR-TKI in patients with NSCLC and uncommon EGFR mutations have been conducted, with all three generations of TKIs. First-generation TKIs seem to be less effective for this group of patients, with an objective response rate (ORR) of only 40%–50% and a median progression-free survival (PFS) period of only 6–7.7 months. 3 The analysis from three studies, LUX-lung 2, 3, and 6, showed that patients carrying G719X, L861Q, and S768I mutations had a median PFS period of 8.2–14.7 months and a median OS period of 16.9–26.9 months. 4 Real-world studies in Asia have demonstrated the effectiveness of afatinib for patients with NSCLC and uncommon EGFR mutations, with a median PFS period of 11–19.7 months.5,6 However, results from these studies have not shown any difference in response or survival time between patients carrying different types of uncommon EGFR mutations.4 –6 Recently, studies on the effectiveness of osimertinib in patients with NSCLC and uncommon EGFR mutations have also shown that third-generation TKIs are effective in this group of patients.7,8 It seems that patients with the L861Q mutation have a better survival benefit than those with the G719X and S768I mutations, particularly for G719X, ORR was 47% and mPFS 8.8 months, for L861Q, ORR was highest at 80% and mPFS 16 months. 7 In Vietnam, the number of patients with NSCLC participating in international, multicenter clinical trials on the efficacy of EGFR-TKIs was limited, thus, the effectiveness of these drugs in the Vietnamese patient population is unclear. In addition, first- and second-generation TKIs are still the main drugs indicated for patients with NSCLC and EGFR mutations, and afatinib is a TKI often prescribed for patients with uncommon EGFR mutations. In a recent study, we found that patients harboring the EGFR mutation of G719X (single and compound) had a median time-to-treatment failure (TTF) period of 19.3 months, which was significantly longer than that of patients carrying other mutations, at 11.2 months (p < 0.001). 9 However, whether patients with the G719X mutation will benefit from both TTF and OS remains unclear. Therefore, this study aimed to compare the effectiveness of afatinib treatment in patients harboring the EGFR-G719X mutation with that in patients carrying other uncommon EGFR mutations.
Methods
Patients
Patients with advanced-stage NSCLC harboring the G719X mutation and other uncommon EGFR mutations received first-line afatinib treatment and were monitored at nine hospitals in Vietnam from April 2018 to June 2022.
The following patients were included in this study: (1) patients with NSCLC stage IIIB, IIIC, and IV according to the American Joint Committee on Cancer version 8, 2017, or recurrence after surgery or local treatment, (2) patients with uncommon EGFR, single or compound with other EGFR mutations include two uncommon mutations or one common and one uncommon mutation, (3) patients aged > 18 years, (4) patients not receiving any previous systemic treatment, (5) patients with a predicted survival period of at least 12 weeks, and (6) patients with complete record.
The following patients were excluded from this study: (1) patients with NSCLC stage I–IIIA; (2) patients with a severe allergy to afatinib; (3) patients with an exon 19 deletion EGFR mutation and an L858R point mutation in exon 21 but no double mutations or other uncommon mutations; (4) patients with hepatitis, cirrhosis, kidney failure, and combined interstitial lung disease; (5) patients with other combined cancers; and (6) patients with insufficient patient information during treatment and follow-up.
Methods
This study had a retrospective design and used convenience sampling. Information on the patients’ diagnosis, treatment, and follow-up was obtained from the medical records in a uniform manner from all centers and uploaded to KoboToolbox.org. EGFR mutations were tested in tissue or serum samples using polymerase chain reaction or next-generation sequencing. Uncommon EGFR mutations were divided into (i) G719X and (ii) other uncommon mutations, including 20 insertion mutations.
The EGFR-TKI afatinib (Giotrif) was started at a dose of 40, 30, or 20 mg, taking one tablet per day. The appointment of the therapeutic dose was based on the age of the patient and performance status (PS) according to the Eastern Cooperative Oncology Group (ECOG). Patients with ECOG PS of 0–1 were considered to have good PS and patients with ECOG 2–4 were considered to have poor PS. For patients using a dose of 40 or 30 mg, if there is a recurrence of grade 2, 3, or higher side effects, the dose can be reduced every 10 mg. Conversely, in patients who were initiated at 20 or 30 mg, if well tolerated, PS improvement may increase to a higher dose. Dose adjustment was determined by the investigator based on the specific patient. The patients were monitored and evaluated every 3 months of treatment or when symptoms of disease progression were assessed by clinical examination, computed tomography of the chest and abdomen, brain magnetic resonance imaging, and bone scan.
The patients were evaluated for treatment response using Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 in 2009. 10 Afatinib-resistant patients were identified with resistance mutations. Subsequently, the patients were treated with third-generation TKIs if T790M was positive and treated for other mutations, if any. Patients without resistance mutations received chemotherapy with or without an immune checkpoint inhibitor, depending on their PS. Adverse effects were assessed according to the National Cancer Institute Common Terminology Criteria for Adverse Events standards version 5.01-2017. 11 The closing times for analysis of TTF and OS data were December 31, 2022, and April 2024, respectively.
The endpoint criteria included the ORR, TTF, and OS. TTF was defined as the time from the date of initiation of afatinib treatment to the date of cessation of afatinib because of disease progression according to RECIST 1.1 or the presence of a severe adverse event, adjusted, and the combination not relieving symptoms or the patient not wanting to continue treatment. ORR was calculated according to RECIST 1.1 criteria and included patients with complete and partial responses. OS was defined as the time from the initiation of afatinib treatment to the date of death. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (STROBE checklist; Supplemental Material). 12
Data processing was performed using the SPSS software (version 22.0; IBM Corp., Armonk, NY, USA). TTF and OS were calculated based on the Kaplan–Meier method to determine the medians and 95% confidence intervals (CIs). The relationship between TTF and OS and clinical and paraclinical characteristics was analyzed using Cox regression. Differences between categorical variables were determined using the chi-squared or Fisher’s exact test. For continuous variables, differences were calculated using t-tests. Differences were considered statistically significant at p < 0.05. A forest plot of hazard ratio (HR) for TTF and OS was constructed using the R software.
Results
There were 49 patients with EGFR-G719X mutations, both single and compound, and 43 patients harboring other uncommon EGFR mutations who were treated with afatinib as first-line treatment. There were no statistically significant differences in age, sex, smoking history, PS, or disease stage between the two groups. Brain and adrenal metastases were more common in patients with the G719X mutation, whereas patients with other mutations had higher rates of bone, liver, and pleural metastases, although the differences were not statistically significant. Patients in both groups were primarily treated with tolerated doses <40 mg. The results are summarized in Table 1.
Characteristics of patients.

Only one patient received a starting dose at 20 mg for each group.
One patient in the G719X group and five patients in the other uncommon mutations group received a tolerable dose at 20 mg.
Patients with the EGFR-G719X mutation had an ORR of 79.6%, which was higher than that of the remaining patient group (69.8%); however, the difference was not statistically significant (p = 0.28). The complete response rates in the two groups were 20.4% and 9.3%, respectively. In the G719X group, 93.9% of patients achieved at least stable disease when treated with afatinib, whereas this percentage in the non-G719X patient group was 83.8% (p = 0.12). The results are shown in Figure 1.
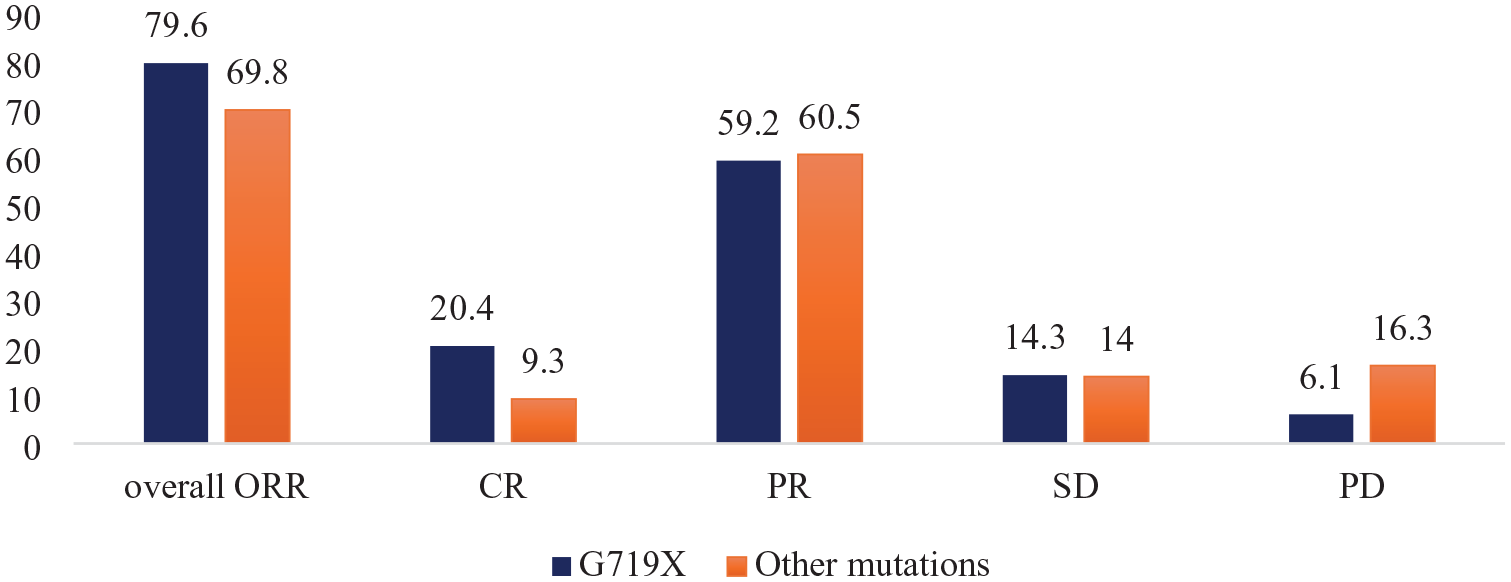
Objective response rate of G719X and other EGFR mutations.
The median TTF period of patients with G719X was 19.3 months (95% CI, 14.6–23.9 months), which was significantly longer compared with that in patients carrying other EGFR mutations, with a median TTF period of 11.2 months (95% CI, 8.9–13.4 months) (HR, 0.39; 95% CI, 0.23–0.66; p < 0.001). In addition, the median OS period of patients with G719X mutation was better than that of patients with other uncommon EGFR mutations, with 31.4 months (95% CI, 23.7–39.1 months) and 21.9 months (95% CI, 8.6–35.2 months), respectively (HR, 0.57; 95% CI, 0.33–0.98; p = 0.044). The 3-year OS rate of patients with the G719X mutation was 40.3%, and that of patients with other uncommon EGFR mutations was 18.7%. The results are shown in Figure 2(a) and (b).
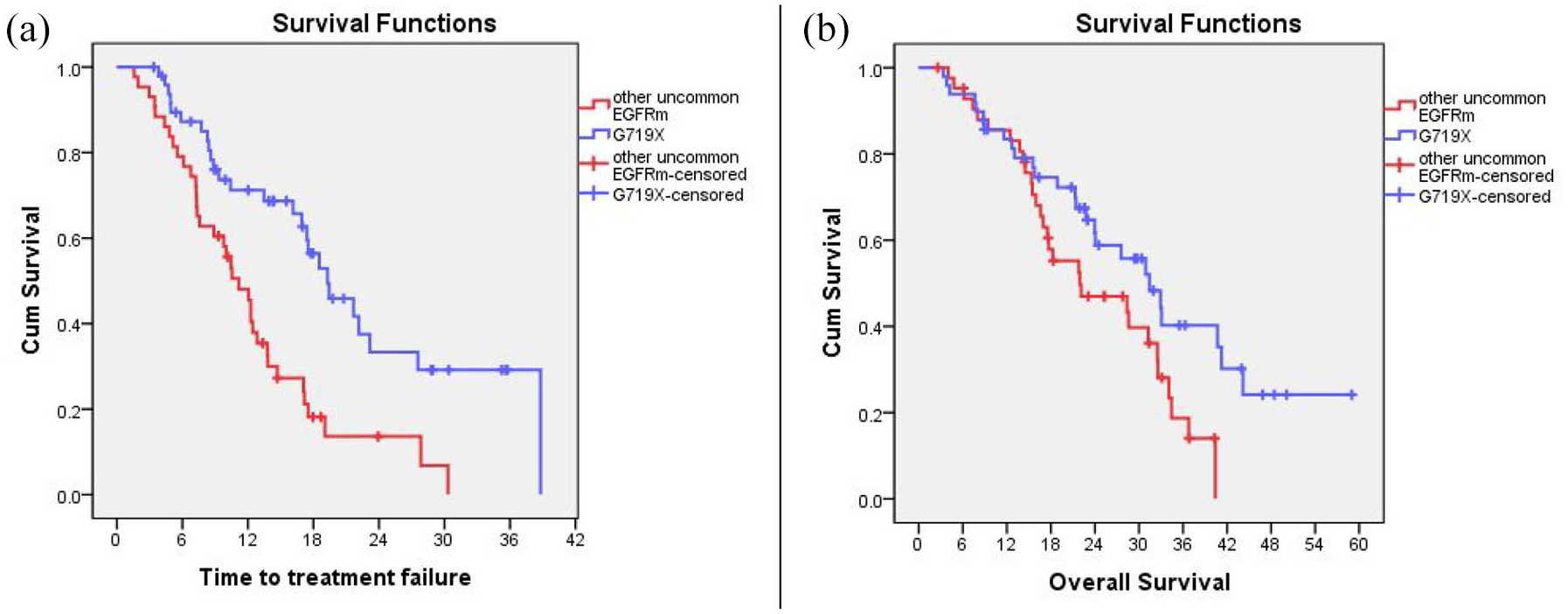
TTF (a) and OS (b) according to G719X and other uncommon EGFRm.
In the without brain metastasis group, patients harboring the G719X mutation achieved a median TTF and OS of 21.7 months and 40.7 months, respectively, which were statistically significantly longer than patients carry other uncommon EGFR mutations with a median TTF of 12.1 months (p = 0.000) and a median OS of 28.4 months (p = 0.025; Figure 3(a) and (b)). In the brain metastasis group, there was no statistically significant difference between the G719X mutation-carrying group and the remaining patients in both TTF and OS (Figure 3(c) and (d)).
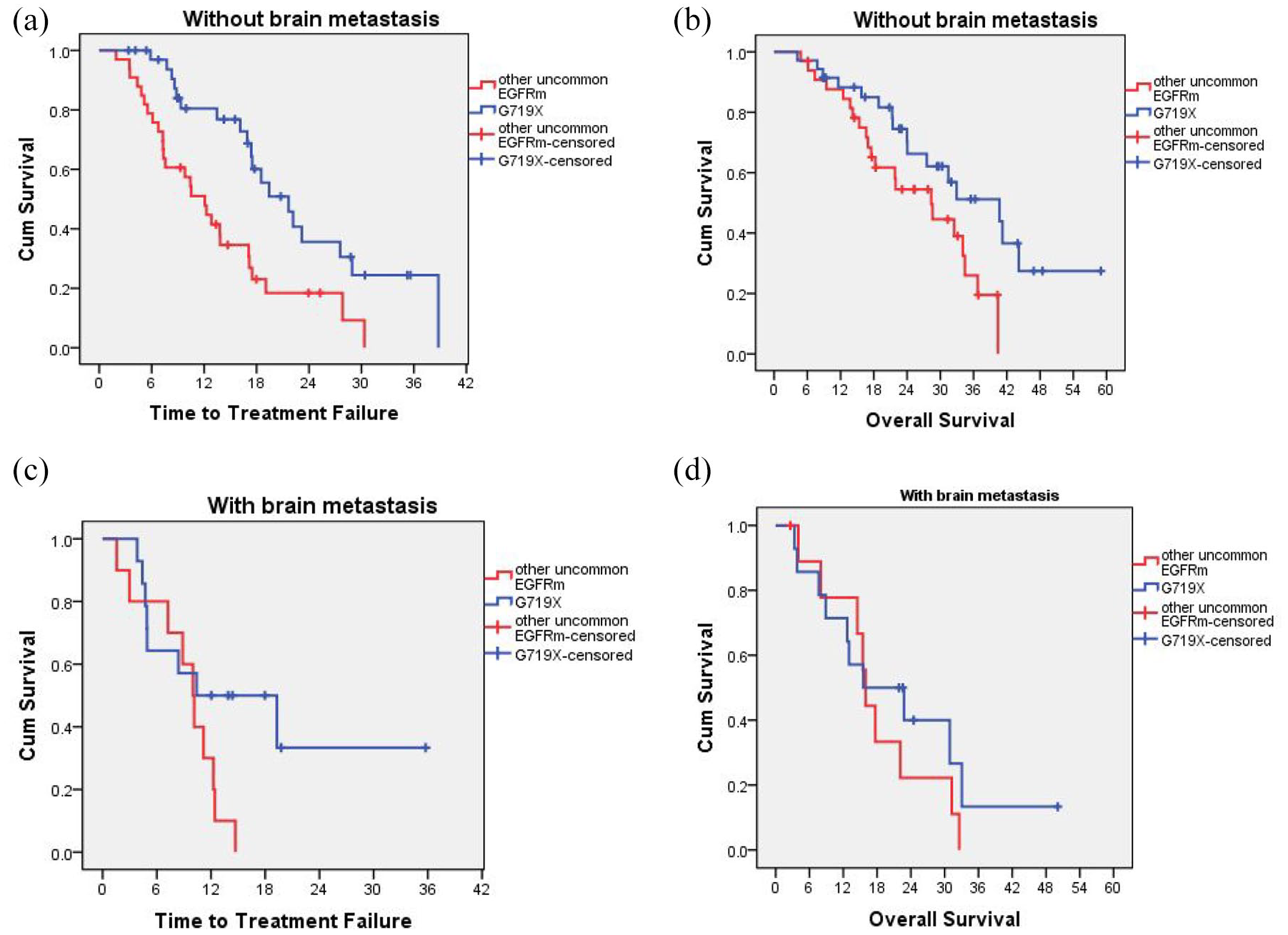
Time-to-treatment failure and overall survival according to without brain metastasis (a, b) and with brain metastasis (c, d) of G719X and other uncommon epidermal growth factor receptor mutations.
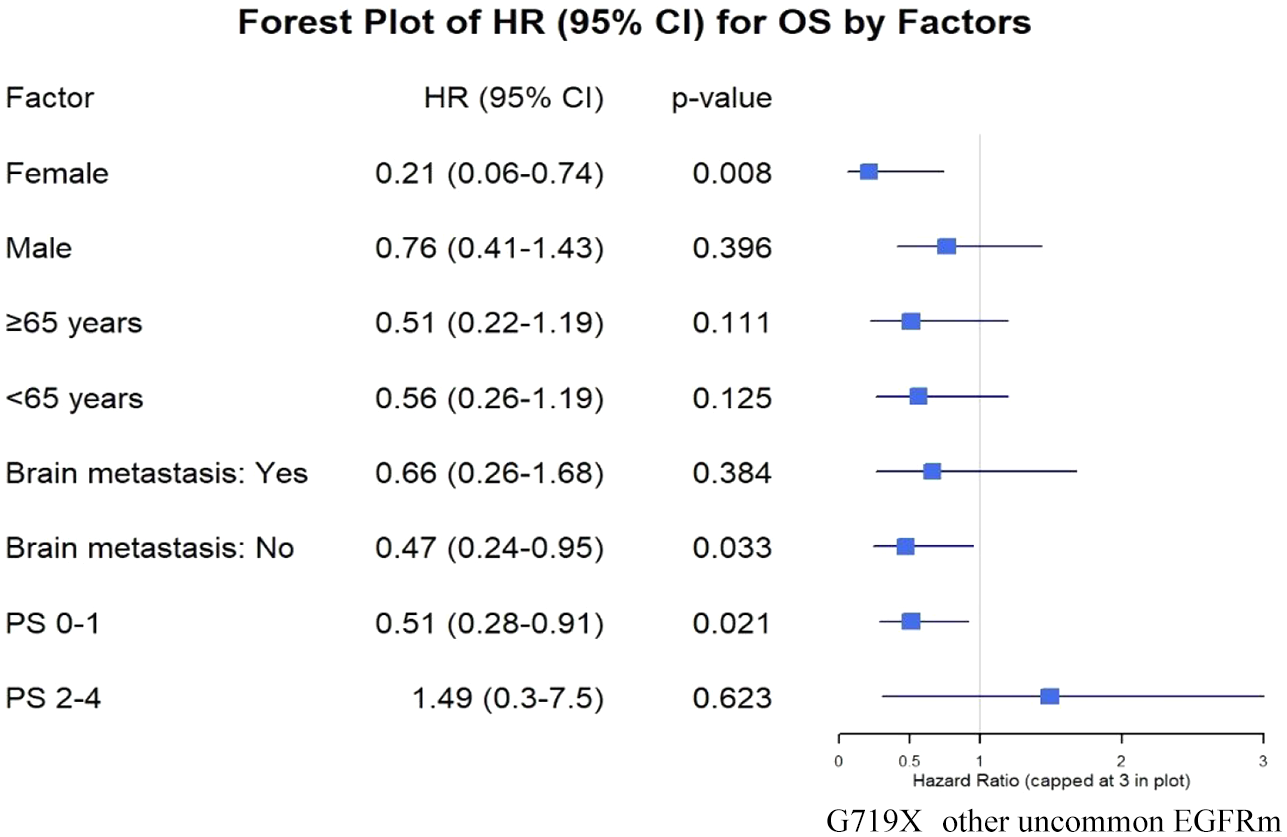
Subgroup analysis showed that the TTF benefit in patients harboring the G719X mutation was better than that in patients with other uncommon EGFR mutations in both women (p = 0.001) and men (p = 0.036), in patients aged ⩾ 65 years (p = 0.008) and <65 years (p = 0.01), patients without brain metastasis (p = 0.001), and patients with good PS (p < 0.001). However, in the OS analysis, we found that statistically significant differences were only observed in women (p = 0.008), patients without brain metastases (p = 0.033), and patients with good PS (p = 0.021) when they had the G719X mutation compared with other uncommon EGFR mutations (Figures 4 and 5).

Analysis of the relationship between TTF and some clinical and paraclinical characteristics of G719X and other uncommon EGFR mutations.
Analysis of the relationship between OS and some clinical and paraclinical characteristics of G719X and other uncommon EGFR mutations.
Discussion
Recently, we reported the efficacy of afatinib in patients with advanced NSCLC harboring an uncommon EGFR mutation. In this analysis, we found that patients with the G719X mutation, including both single and compound mutations, had a better TTF benefit than patients with other uncommon EGFR mutations. 9 In this analysis, we further assessed whether patients with the G719X mutation had a TTF, OS, and ORR benefit. In 49 patients with the G719X mutation and 43 patients without the G719X mutation, we did not find statistically significant differences in age, sex, smoking history, PS, disease stage, distant metastasis characteristics, or treatment dose of patients treated in the two groups. In a pooled analysis of the Lux-Lung 2, 3, and 6 studies, Yang et al. evaluated the efficacy of afatinib as a first-line treatment for patients with NSCLC carrying uncommon EGFR mutations, and the ORR of patients with the G719X mutation was 78%, which was lower than that of patients with the S768I mutation, with an ORR of 100%, however, the sample size in this study was small. 4 In this study, we found that the ORR of patients with the G719X mutation was 10% higher than that of patients with other uncommon EGFR mutations and that the rate of patients achieving a complete response was twofold higher in those with the G719X mutation compared to patients with other mutations. In a data analysis of 693 patients treated with afatinib, with 315 patients previously untreated with EGFR-TKIs, when analyzing 265/315 patients with ORR data, patients with the G719X mutation achieved an ORR of 63.4%, which was higher than that in patients with L861Q (59.6%) and S768I (62.5%). 13 Although the difference was not statistically significant, a benefit could be observed in patients with the G719X mutation, whether single or double mutation.
According to an evaluation of the survival benefit of patients treated with afatinib, an analysis from three studies, Lux-Lung 2, 3, and 6, showed that in patients carrying G719X, L861Q, and S768I mutations, the median PFS periods were 8.2 months, 13.8 months, and 14.7 months, respectively, whereas the median OS periods were 26.9 months for patients carrying the G719X mutation and 17.1 months in patients carrying the L861Q mutation. However, a limitation of this analysis was that only 18 patients carried the G179X mutation, including single and double mutations, 16 patients carried the L861Q mutation, and only eight patients carried the S768I mutation. 4 In another study, with 272/315 patients having available TTF data, the authors also noted that the median TTF was longest in patients with the S768I mutation, with results of 14.7 months, 10.0 months, and 15.6 months in patients with G719X, L861Q, and S768I mutations, respectively. 13 However, in this study, we found that patients with G719X mutations, including single or compound, had a longer median TTF than patients without G719X, with 19.3 months compared with 11.2 months, respectively (HR, 0.39; 95% CI, 0.23–0.66; p < 0.001). In addition, the median OS period was 10 months better in patients with the G719X mutation than in patients with other uncommon EGFR mutations (HR, 0.57; 95% CI, 0.33–0.98; p = 0.044). The 3-year OS period of patients with the G719X mutation was 40.3%, whereas that of patients with other uncommon EGFR mutations was only 18.7%. Our results are in line with those of another study by Hsu et al., who analyzed 90 patients with uncommon EGFR mutations, with 47 patients having G719X mutations (37 single and 10 compounds), and found that the median PFS period of patients carrying the G719X mutation at 18.2 months tended to be higher than that of patients with other uncommon EGFR mutations at 13.1 months (p = 0.081), and the median OS periods of the two groups were 47.4 and 23.0 months, respectively (p = 0.051). However, multivariate analysis showed a statistically significant association between PFS and HR (0.578 and p = 0.027). In addition, the authors also found that if the patient did not have brain, bone, and liver metastasis, all had a longer median PFS period than patients with metastasis to these organs (p < 0.05). 14 Another trend was observed in the study by Li et al. on 42 patients with NSCLC carrying uncommon EGFR mutations. The median TTF was found to be longer in patients with the L861Q mutation, with results of 15.0, 11.7, and 16.6 months in patients with G719X, S768I, and L861Q mutations, respectively. 15 It can be seen that there are also different results on the efficacy of afatinib in patients with different uncommon EGFR mutations; however, most previous reports have small sample sizes, so studies showing different values of mutation types are very meaningful for clinical use. Currently, although osimertinib has also been approved for the treatment of patients with NSCLC carrying uncommon EGFR mutations, data from some studies seem to show that osimertinib’s effectiveness is better in the group of patients carrying the L861Q mutation.7,8 A retrospective study (UNICORN), including 60 patients, reported that the response rate and PFS were 78% and 15.7 months and 53% and 8.6 months in patients with L861Q and G719X, respectively. 7 According to a multicenter, open-label, phase II study in Korea on the efficacy of osimertinib in 37 patients with NSCLC and uncommon EGFR mutations (G719X, 19 patients; L861Q, 9 patients; S768I, 8 patients; and others, 4 patients), with 61% of patients receiving first-line therapy, ORR reached 50%, and the median PFS period was only 8.2 months. ORR was noted in 78% of patients with the L861Q mutation, followed by 53% with G719X, and 38% with S768I. Moreover, the PFS periods of patients with L861Q, G719X, and S768I mutations were 15.2 months (95% CI, 1.3–29.1 months), 8.2 months (95% CI, 6.2–10.2 months), and 12.3 months (95% CI, 0–28.8 months), respectively. 8 In a recent analysis, Fukuda et al. found that osimertinib tended to be superior to afatinib in patients with L861Q mutations alone. In contrast, afatinib was superior to osimertinib in patients with G719X or S768I mutations in both ORR and PFS. However, the authors noted that patients with the S768I mutation fared less well than patients with other mutations. In addition, the authors found that osimertinib was associated with longer PFS in patients with a combination of common and uncommon EGFR mutations compared to afatinib. 16 The difference in efficacy of afatinib and osimertinib in patients with uncommon EGFR mutations may be explained by the fact that the G719X mutation is a member of the PACC mutation family along with L747X, S768I, L792X, and T854I and is predicted to alter the orientation of the P-loop or αC-helix. 17 When analyzing the effect of osimertinib on the PACC G719S and L718Q mutations, the authors suggested that changes in the orientation of the P-loop altered the position of the TKI stabilizing sites, thereby tilting the indole ring of osimertinib away from the P-loop, destabilizing drug binding. In contrast, second-generation TKIs such as afatinib do not interact with the EGFR P-loop and maintain the interaction sites in the hydrophobic cleft. At the same time, when comparing the selectivity of EGFR-TKIs for PACC mutations, the authors also found that second-generation TKIs were significantly more selective for PACC mutations than other TKIs. In addition, the authors found that PACC mutations combined with common EGFR mutations retained sensitivity to second-generation TKIs while they were resistant to third-generation TKIs. On the other hand, the authors noted that patients with common EGFR mutations who were resistant to osimertinib and had uncommon PACC mutations were still effective when treated with afatinib. Regarding the mechanism of resistance to third-generation TKIs, previous studies have reported that the C797S mutation confers resistance to third-generation TKIs even in the absence of T790M. 18 Similarly, the C797S mutation without T790M confers resistance to Ex20ins inhibitors, but not to first- or second-generation TKIs unless T790M is present. Thus, it can be seen that both primary and acquired PACC mutations are sensitive to second-generation TKIs and receive greater efficacy than third-generation TKIs. 17
Evaluating the relationship between TTF and OS and clinical and paraclinical characteristics, patients carrying G719X-EGFR mutations had an ECOG PS of 0–1 or had no brain metastasis; female sex was a factor that conferred statistically significant benefits in both TTF and OS compared with patients without the G719X mutation. Patients of any age and male sex also received better TTF benefits when they harbored the G719X mutation. To the best of our knowledge, these features have not been reported previously. Hsu et al. also analyzed the clinical characteristics of patients with and without the G719X mutation and found no statistically significant differences in age, sex, smoking history, PS, disease stage, or distant metastasis characteristics between the two groups. However, the authors did not analyze the relationship between these characteristics and the survival time of patients with G719X-EGFR mutations. 14
The limitation of this study is that it is a retrospective study, and the sample size is small, however, this may be a suggestion for further studies to be conducted with larger sample sizes and possibly prospective studies to more clearly see the benefits of patients with G719X mutation compared to patients with other uncommon EGFR mutations. In addition, in this study, we have not analyzed the role of some prognostic factors, such as combined diabetes. In future studies, we will analyze to clarify the prognostic value of comorbidities such as diabetes as well as some other factors, such as neutrophil-to-lymphocyte ratio or platelet-to-lymphocyte ratio, on the effectiveness of targeted therapy and immunotherapy.
Conclusion
Patients harboring the G719X mutation, both single and compound, received excellent benefits in terms of both TTF and OS compared with patients carrying other uncommon EGFR mutations. These results may be a contributing factor in guiding clinical practice as well as the basis for further studies to clarify this benefit.
Supplemental Material
sj-doc-1-tar-10.1177_17534666251341747 – Supplemental material for Excellent survival benefit achieved in patients with advanced-stage non-small cell lung cancer harboring the epidermal growth factor receptor-G719X mutation treated by afatinib: the real-world data from a multicenter study in Vietnam
Supplemental material, sj-doc-1-tar-10.1177_17534666251341747 for Excellent survival benefit achieved in patients with advanced-stage non-small cell lung cancer harboring the epidermal growth factor receptor-G719X mutation treated by afatinib: the real-world data from a multicenter study in Vietnam by Van Luan Pham, Cam Phuong Pham, Thi Thai Hoa Nguyen, Tuan Khoi Nguyen, Minh Hai Nguyen, Thi Anh Thu Hoang, Tuan Anh Le, Dinh Thy Hao Vuong, Dac Nhan Tam Nguyen, Van Khiem Dang, Thi Oanh Nguyen, Hung Kien Do, Ha Thanh Vu, Thi Thuy Hang Nguyen, Van Thai Pham, Le Huy Trinh, Hoang Gia Nguyen, Cong Minh Truong, Tran Minh Chau Pham and Thi Bich Phuong Nguyen in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
