Abstract
Primary hepatobiliary cancers (PHCs), which mainly include hepatocellular carcinoma (HCC) and biliary tract cancers (BTCs), are mostly diagnosed in the advanced stage and are not candidates for curative surgery or ablation, resulting in a dismal prognosis. Targeted therapies with or without programmed death receptor 1 (PD-1)/PD-L1 inhibitors have been incorporated into first-line treatments for advanced HCC. Systemic chemotherapy is still the mainstay treatment for advanced BTCs, and combining it with PD-1/PD-L1 inhibitors has resulted in prolonged patient survival. Intra-arterial therapies, including trans-arterial chemoembolization, selective internal radiation therapy, and hepatic arterial infusion chemotherapy (HAIC), have been explored and used for advanced hepatobiliary cancers for many years with positive results, particularly when combined with systemic treatments. Recently, an increasing number of phase II/III trials have demonstrated the efficacy and safety of HAIC for the treatment of advanced HCC with portal vein tumor thrombosis and/or a large tumor burden, for the neoadjuvant and adjuvant treatment of HCC with high-risk factors, and for treating advanced intrahepatic and perihilar cholangiocarcinoma. However, the techniques and regimens used for HAIC are diverse and differ greatly between various regions and centers worldwide. This review focuses on these diverse techniques and regimens, as well as the updated evidence on HAIC regarding the treatment of PHCs.
Introduction
Primary hepatobiliary cancers (PHCs) mainly include hepatocellular carcinoma (HCC) and biliary tract cancers (BTCs). HCC is the third leading cause of cancer-related death worldwide, with an incidence of 10.1 cases per 100,000 person-years. 1 Most cases of HCC occur in Africa and eastern Asia due to chronic hepatitis B and aflatoxin B1 exposure, while non-alcoholic fatty liver disease is becoming the main risk factor of HCC in developed countries.2–4 BTCs, which account for <1% of all cancers worldwide, comprise cholangiocarcinoma and gallbladder cancer (GBC). 5 The incidence of BTCs in southeast Asia and China, especially in regions where liver fluke infection is common (85 cases per 100,000 for cholangiocarcinoma), is higher than that in Europe and the United States (0.3–3.5 cases per 100,000 for cholangiocarcinoma and 1.6–2.0 cases per 100,000 for GBC). 6
Ultrasonography, contrast-enhanced computed tomography (CT)/magnetic resonance imaging (MRI), and tumor marker tests [alpha fetoprotein (AFP) and carcinoembryonic antigen (CEA)/carbohydrate antigen 19-9 (CA19-9)] are the most commonly used techniques for the surveillance and diagnosis of PHCs. Surgical resection and ablation are the main curative methods for early-stage PHCs. However, most patients are diagnosed with PHCs at an advanced stage, at which point they are not candidates for curative resection or ablation, resulting in dismal prognosis.7,8
Targeted therapies alone or combined with programmed death receptor 1 (PD-1)/PD-L1 immune checkpoint inhibitors are currently recommended as first-line treatments for advanced HCC, with the median overall survival (OS) ranging from 6.5 to 22.1 months, while grade 3 or higher adverse events (AEs), such as hand-foot syndrome, hypertension, and diarrhea, were observed in 52–80.9% of participants.8–12 Although combination therapy using atezolizumab plus bevacizumab is considered a preferred first-line therapy for advanced HCC, the survival benefit of this combination is severely compromised in patients with HCC and high-risk factors, such as those with high liver tumor burdens or main portal vein tumor thrombosis (PVTT), with a median OS of only 7.6 months. 11 Gemcitabine plus cisplatin is still the cornerstone of systemic treatment for advanced BTCs, with median progression-free survival (PFS) and OS of 8.0 and 11.7 months, respectively. 13 The addition of PD-1/PD-L1 inhibitors to this combination was recently reported to prolong the median OS for advanced BTCs slightly, ranging from 12.7 to 12.9 months.14,15 However, 60.9–70.7% of participants presented with grade 3 or higher AEs, including decreased white blood cell count, decreased neutrophil count, and abnormal liver function.13–15 Regarding to second-line treatment, although the median OS ranged from 8.5 to 10.6 months following treatment with regorafenib, cabozantinib, or ramucirumab for advanced HCC, the incidence of grade 3 or higher AEs was more than 60%.16–18 Likewise, FOLFOX chemotherapy was the only effective second-line treatment as confirmed by a phase III trial for advanced BTCs, with a median OS of 6.2 months, while more than 70% of participants presented with grade 3 or higher AEs. 19 Thus, it is crucial to explore safer and more effective treatment methods for advanced PHCs.
Hepatic arterial infusion chemotherapy (HAIC) was first reported in the late 1980s. Based on the pathophysiological theory that most hepatobiliary malignant tumors, including HCC, intrahepatic cholangiocarcinoma (iCCA), and perihilar cholangiocarcinoma (pCCA), receive their blood supplies mostly from hepatic arteries, while normal liver parenchyma is mostly supplied by the portal vein, HAIC delivers high concentrations of chemotherapeutic agents directly to hepatobiliary tumors, achieving high tumor response while avoiding excessive exposure of normal liver parenchyma to chemotherapeutic agents. 20 Therefore, HAIC is a theoretically alternative treatment method for PHCs.
Recently, multiple phase II/III trials have demonstrated the efficacy and safety of HAIC alone, or combined with systemic therapies, as first-, second-line, neoadjuvant, and/or adjuvant treatment, for HCC, with the median OS ranging from 10.6 to 23.1 months.21–26 The objective response rate (ORR) and disease control rate (DCR) ranged from 35.3% to 61.5% and 76% to 97.4%, respectively. In addition, the safety profile is acceptable, with the incidence of grade 3 or higher AEs ranging from 19% to 59%. Likewise, the efficacy and safety of HAIC with or without systemic therapies for advanced BTCs have also been verified, with the median OS ranging from 13.5 to 30.8 months.27–35 The ORR and DCR ranged from 16% to 67.6% and 65% to 98%, respectively, while only 14.7–35% of participants presented with grade 3 or higher AEs.
Thus, HAIC plays an increasingly significant role in the treatment of PHCs, such as decreasing tumor burden, reducing the risk of recurrence, and prolonging survival, as first-, second-line, neoadjuvant, and/or adjuvant treatments. However, the techniques used for HAIC, including percutaneously implanted port-catheter system, surgically implanted pump, and temporary indwelling hepatic artery catheter, vary greatly between different regions and centers worldwide, as do the chemotherapy regimens, which include oxaliplatin and 5-fluorouracil, cisplatin-based, floxuridine (FUDR), oxaliplatin, raltitrexed, etc. The most common regimen used for HAIC for PHCs is oxaliplatin and 5-fluorouracil, which was mostly used in China and Europe, while a cisplatin-based regimen is the main HAIC regimen for HCC in Japan, and FUDR is mostly used in HAIC for BTCs in the United States and other western countries. This review describes the diverse techniques and regimens across the world of HAIC and discusses up-to-date evidence regarding the efficacy of HAIC for PHCs.
HAIC techniques
Percutaneously implanted port-catheter system
The percutaneously implanted port-catheter system, mostly used in Japan, China, and Europe, is cost-effective for patients who need multiple cycles of HAIC or continuous administration of time-dependent chemotherapeutic agents, such as 5-fluorouracil.36–40 This technique can achieve whole-liver perfusion during HAIC while also being simpler, safer, and faster than a surgically implanted pump. Nevertheless, the implantation of a percutaneously implanted port-catheter system should be performed at a specialized interventional radiology center, and the port-catheter system should be regularly maintained with heparin to prevent thrombosis in the system.
The most common access for the port-catheter system is the femoral artery. Although the subclavian, axillary, and brachial arteries are alternative accesses, the increased risk of cerebral complications should be considered when using them. 41 Celiac and superior mesenteric angiographies should be performed to visualize hepatic arterial variations. Extrahepatic and intrahepatic blood flow redistribution are vital procedures that should be performed before port implantation.37,39 Extrahepatic blood flow redistribution refers to the procedures used to embolize all of the extra-hepatic arteries originating from the hepatic artery, which supply the gastroduodenum [such as the right gastric artery, accessory left gastric artery, pancreaticoduodenal artery, and/or gastroduodenal artery (GDA)] with micro-coils, to avoid drug infusion to the gastrointestinal tract during HAIC treatment and thus reduce acute gastroduodenal mucosal toxicity (Figure 1). Intrahepatic blood flow redistribution refers to the redistribution of multiple hepatic arteries into one hepatic artery to supply the whole liver, ensuring drug infusion to the whole liver via HAIC (Figure 2).
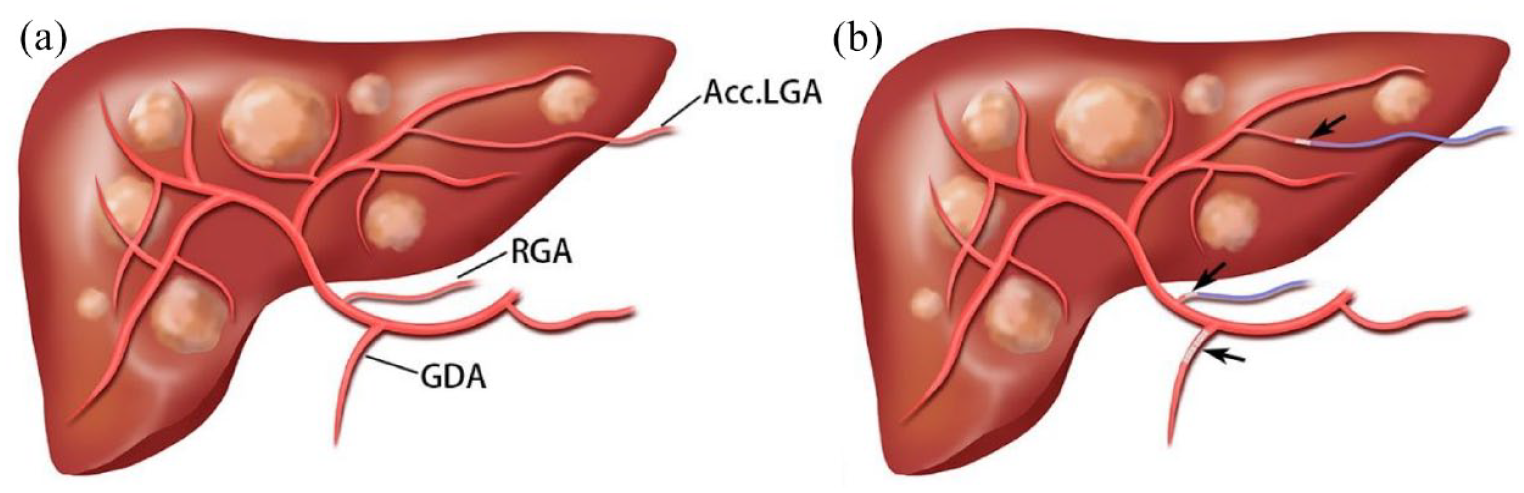
Procedure of extrahepatic blood flow redistribution. (a) The Acc.LGA and RGA were indicated by angiography. (b) The Acc.LGA, RGA, and GDA were embolized using micro-coils (arrows) to avoid drug infusion to the gastrointestinal tract.

Procedure of intrahepatic blood flow redistribution. (a) The Acc.LHA originates from the LGA and Acc.RHA arises from the SMA, indicated by angiography. (b) The Acc.LHA and Acc.RHA were embolized using micro-coils (arrows) so that the whole-liver arterial blood flow from a single artery (PHA) was reconstructed.
Then, the indwelling catheter with a side hole is inserted into the GDA. The tip of the catheter is fixed in GDA using micro-coils to reduce the potential for dislocation of the catheter and thrombosis of the hepatic artery, with the side hole located at the origin of the proper hepatic artery (PHA) to achieve whole-liver perfusion during HAIC (Figure 3).36,37,39,42,43 Alternatively, the catheter tip can be inserted into the hepatic artery as distally as possible, with the side hole located at the PHA. The stability and patency of the catheter in the latter technique were demonstrated in a published study. 44

The fixed-catheter-tip technique of port-catheter system. (a) The tip of the indwelling catheter is fixed in the GDA (arrows) using micro-coils, with the side hole located at the origin of the PHA. (b) Chemotherapeutic agents could be infused via the side hole of the catheter, into the whole liver.
The inserted catheter is then connected to the port, which can be implanted subcutaneously at the medial anterior superior iliac crest or the chest wall. Moreover, the HAIC is performed through a port punctured with a Huber needle. Subsequently, digital substraction angiography (DSA), CT hepatic arteriography, or 99mTc-MAA SPECT is performed through the port to confirm the whole-liver perfusion without extrahepatic perfusion, and the port-catheter system should be checked in the same way before each cycle of HAIC.
Surgically implanted pump
The surgically implanted pump is used more frequently in the United States for HAIC, and it can also achieve whole-liver perfusion. However, the implantation procedure, which requires either a laparotomy or minimally invasive techniques, should be performed by an experienced surgeon at a specialized center. As a key step before implantation, exploration is performed to detect the presence of any extrahepatic disease, and cholecystectomy is performed routinely. 45 A subcutaneous pocket, which is commonly placed in the left abdomen, is created to accommodate the pump. The pump is secured on the fascia, and the catheter is inserted through the peritoneum behind the pump.
Arteries, such as common hepatic artery (CHA), GDA, and superior mesenteric artery, are isolated circumferentially to detect the extrahepatic supplying arteries and hepatic arterial variations. Arteries that supply extrahepatic organs, such as the right gastric artery, should be ligated to prevent the exposure of extrahepatic organs to chemotherapeutic agents. Accessory or replaced hepatic arteries should be ligated as well to reduce competitive intrahepatic blood flow that could lead to the preferential flow of chemotherapeutic agents to only half of the liver. 45 The catheter is then inserted and secured to the GDA/PHA using non-absorbable ties.
It is important to flush the catheter after each tie to keep the catheter unobstructed and to promptly identify any ties that may be occluding flow through its lumen. Undiluted blue dye or angiography can be used to confirm whole-liver perfusion and rule out the possibility of extrahepatic perfusion after implantation.
Temporary indwelling hepatic artery catheter
Temporary hepatic artery catheter indwelling is less invasive than other techniques, and it could serve as an alternative technique for HAIC when patients refuse to receive port implantation or when the implanted port-catheter system is obstructed. However, this requires repeated artery punctures and catheterization. Following intrahepatic and extrahepatic blood flow redistribution as mentioned above, a micro-catheter is temporarily indwelled for HAIC for several hours with its tip located in PHA/CHA to achieve whole-liver perfusion. HAIC is performed through the catheter, and the catheter must be removed when each HAIC treatment is finished. However, the repetitive puncture of the artery and the requirement of a long time in bed can compromise patient compliance with this technique.
Indications for HAIC
(1) As a trans-arterial whole liver local treatment, HAIC is generally recommended for patients with locally advanced HCC with large tumors, a high tumor burden, or diffuse tumors, especially with portal vein/hepatic vein/biliary tract tumor thrombosis, in case that the tumor is confined within the liver or exhibits only limited extrahepatic metastasis.40,46
(2) HAIC recently has been recommended as a neoadjuvant/adjuvant therapy for patients with HCC and high-risk factors of recurrence, such as those high tumor burden and/or microvascular invasion.
(3) HAIC may also serve as a better choice for patients with compromised liver function (Child-Pugh B) and/or an elevated total bilirubin level so long as it is less than five times the upper limit of normal who are unsuitable for systemic therapy.31,47
(4) HAIC is recommended as an alternative and salvage treatment of systemic chemotherapy for locally advanced iCCA, pCCA, or GBC with or without liver metastasis.28,29,31,48
Rationale of HAIC combined with systemic therapy
As mentioned above, HAIC delivers high concentrations of chemotherapeutic agents directly to hepatobiliary tumors for local tumor control due to the first-pass effect but it has very limited efficacy for extrahepatic tumors. In 2006, Kemeny et al. 49 demonstrated that HAIC could increase the time to hepatic progression compared to systemic therapy, while systemic therapy was associated with longer time to extrahepatic progression. Furthermore, multiple phase II/III trials demonstrated that HAIC combined with systemic therapy was associated with longer survival for patients with PHCs when compared to systemic therapy alone.21,24,32,50 Thus, for patients with advanced PHCs and extrahepatic metastasis, combining HAIC with systemic therapy is recommended.
Meanwhile, an improvement in vascular normalization and vascular permeability can be induced by anti-vascular endothelial growth factor (VEGF) target treatment, which benefits the delivery of chemotherapeutic agents.51,52 The immune system can also be modulated by chemotherapeutic agents via direct immune-stimulatory mechanisms, downregulation of the immunosuppressive microenvironment, and increased immunogenicity.53,54 Recently, the combination of chemotherapy and immunotherapy has been demonstrated to be more effective than chemotherapy or immunotherapy alone in lung cancer, as well as in esophageal and gastric cancers.55,56 Moreover, Mei et al. 57 demonstrated that HAIC combined with PD-1 inhibitors is effective for advanced HCC, with median PFS and OS of 10.0 and 18.0 months, respectively.
Regimens and updated evidence for HCC
Oxaliplatin and 5-fluorouracil
In first-line treatment settings
HAIC alone
As a trans-arterial local treatment, HAIC has been confirmed to be effective and safe for advanced HCC. In 2018, the FOXAI study showed that HAIC with oxaliplatin and 5-fluorouracil presented a median time to progression (TTP) and ORR of 6.1 months and 28.6% [Response Evaluation Criteria in Solid Tumor (RECIST)]/40.8% [modified RECIST (mRECIST)], respectively, for advanced HCC, with most toxicities being minor (grade 1/2 versus grade 3/4: 91% versus 11%). 58 In particular for advanced HCC with major PVTT, HAIC showed much better efficacy than trans-arterial chemoembolization (TACE)/trans-arterial embolization, with the ORR, DCR, median OS, and PFS of 59.1% versus 22.7% (p = 0.014), 90.9% versus 50.0% (p = 0.002), 20.8 versus 4.0 months (p < 0.001), and 9.6 versus 1.5 months (p < 0.001), respectively. 59
Although TACE is considered the standard treatment for HCC in the Barcelona Clinic Liver Cancer (BCLC) B-stage, HAIC also showed superior efficacy in cases of large tumor sizes (⩾7 cm) in a randomized, multi-center phase III trial, with the median OS of 23.1 versus 16.1 months (p < 0.001) in the intent-to-treat (ITT) population and the median PFS of 9.6 versus 5.4 months (p < 0.001). 23 The symptomatic PFS was also significantly longer in the HAIC group (17.9 versus 10.4 months, p < 0.001), which also had a better ORR [46% versus 18% (RECIST 1.1, p < 0.001), 48% versus 33% (mRECIST, p = 0.004)], DCR [82% versus 61% (RECIST 1.1, p < 0.001), 82% versus 65% (mRECIST, p = 0.001)], and conversion rate (24% versus 12%, p = 0.004). There were no severe vascular complications in the HAIC group, while more participants in the TACE group experienced serious treatment-related AEs (30% versus 19%, p = 0.03).
In 2021, Lyu et al. 22 randomly assigned 262 participants, among which 65.5% presented with macrovascular invasion and 49.2% presented with high-risk factors (Vp4 PVTT and/or >50% liver occupation), into HAIC and sorafenib groups in a multicenter phase III trial. In the ITT population, the median OS was 13.9 versus 8.2 months (p < 0.001), with median PFS and intrahepatic tumor PFS of 7.8 versus 4.3 months (p < 0.001) and 9.1 versus 4.5 months (p < 0.001), respectively. For the participants with high-risk factors, the median OS and PFS were 10.8 versus 5.7 months (p < 0.001) and 7.7 versus 2.9 months (p < 0.001), respectively. The ORR and DCR based on the RECIST 1.1/mRECIST criteria were 31.5% versus 1.5% (p < 0.001)/35.4% versus 5.3% (p < 0.001) and 77.7% versus 58.3% (p = 0.001)/77.7% versus 61.4% (p = 0.004), respectively. Furthermore, more participants in the sorafenib group experienced grade 3/4 treatment-related AEs (48.1% versus 20.3%). Thus, HAIC was superior to sorafenib for patients with advanced HCC, even for those with PVTT and high-risk factors.
HAIC plus systemic therapy
The targeted therapy agent sorafenib has been a standard treatment for BCLC C stage HCC for many years. Unfortunately, the survival benefit of sorafenib alone is very limited for patients with advanced HCC and PVTT. HAIC plus sorafenib has therefore been investigated for patients with advanced HCC and PVTT, showing better tumor control and survival benefits.
In 2019, a total of 247 participants with HCC and PVTT were randomly assigned to a sorafenib plus HAIC group (125 participants) and a sorafenib group (122 participants). 21 The median OS was 13.37 versus 7.13 months (p < 0.001), and the median PFS was 7.03 versus 2.6 months (p < 0.001). The ORRs were 40.8% versus 2.5% (RECIST) (p < 0.001) and 54.4% versus 5.7% (mRECIST) (p < 0.001). More grade 3/4 neutropenia [9.68% versus 2.48% (p = 0.03)], thrombocytopenia [12.9% versus 4.96% (p = 0.04)], and vomiting [6.45% versus 0.83% (p = 0.04)] were observed in the combination group, and 10 participants experienced thrombosis or dislocation of the catheter tip.
This combination therapy was also shown to be more beneficial for advanced HCC with major PVTT. A total of 64 participants with advanced HCC and major PVTT (Vp3/Vp4) were randomly assigned to be treated either by sorafenib plus HAIC or sorabenib alone (400 mg twice daily). 24 In the ITT population, the median OS and PFS were 16.3 versus 6.5 months (p < 0.001) and 9.0 versus 2.5 months (p < 0.001), respectively, and the ORRs were 41% versus 3% (RECIST 1.1) (p < 0.001) and 50% versus 3% (mRECIST) (p < 0.001). However, grade 3/4 treatment-related AEs were more frequent in the sorafenib plus HAIC group (59% versus 25%).
Along with the combination of targeted therapy and immunotherapy gradually becoming a standard first-line treatment for HCC, a triple combination of HAIC, targeted therapy, and immunotherapy has been increasingly investigated recently and has shown better tumor responses and potential survival benefits.
In 2022, Lai et al. 60 enrolled 36 treatment-naïve participants with advanced HCC to receive a combination therapy of HAIC, lenvatinib, and toripalimab (a PD-1 inhibitor) in a single-center, single-arm, phase II trial. The 6-month PFS rate was 80.6%, meeting the primary endpoint. The PFS and OS were 10.4 and 17.9 months, respectively, with an ORR of 63.9% (RECIST)/66.7% (mRECIST). Notably, the OS and PFS were 17.4 and 10.4 months, respectively, in participants with high-risk factors. Grade 3/4 treatment-related AEs and immune-related AEs were experienced by 72.2% and 11.1% of the participants, respectively.
In 2023, Zhang et al. 61 reported a phase II trial evaluating the efficacy and safety of HAIC combined with Apatinib (a targeted therapy agent) and Camrelizumab (a PD-1 inhibitor) for BCLC C stage HCC. The ORR was 77.1% (RECIST 1.1)/88.6% (mRECIST), with the DCR of 97.1%. Impressively, the median PFS was 10.38 months, and the 12-month OS rate was 87.4%. However, 74.3% of the participants experienced grade 3 or higher treatment-related AEs, with the predominant AEs being decreased lymphocyte count (37.1%).
Adjuvant/neoadjuvant settings
HAIC with oxaliplatin and 5-fluorouracil has also been shown to have promising advantages in both adjuvant and neoadjuvant treatment settings for patients with HCC and high risks of recurrence after resection.
In 2022, Li et al. 25 reported their randomized phase III trial, comparing the efficacy and safety of adjuvant HAIC and routine follow-up for patients with HCC and microvascular invasion. A total of 315 participants were enrolled, with the primary endpoint of disease-free survival (DFS) in the ITT population. In the ITT population, the median DFS was 20.3 versus 10.0 months (p = 0.001), while the median OS was similar (p = 0.130). In the per-protocol (PP) population, a significantly longer median DFS was also achieved in the HAIC group (19.3 versus 8.9 months, p < 0.001). In the safety profile, all of the participants in the HAIC group experienced HAIC-related AEs, most of which were grade 0–2 (146/148, 98.6%).
In 2023, Wei et al. 26 demonstrated that HAIC represents an excellent neoadjuvant treatment for patients with BCLC A/B stage beyond the Milan criteria. A total of 487 participants were enrolled and assigned to either the neoadjuvant group (treatment group) or the operation without neoadjuvant treatment group (control group). In the ITT population, the median PFS was 17.4 versus 9.8 months (p < 0.001), and the median OS in the treatment group was significantly longer than that of the control group (p = 0.032). Similar results were also observed in the PP population [median PFS: 22.7 versus 10.2 months (p < 0.001), median OS (p = 0.001)]. Grade 0–2 HAIC-related AEs were observed in 97.9% of participants in the treatment group, and the operation-related complications were similar in both groups (p = 0.265). The updated evidence of HAIC with oxaliplatin and 5-fluorouracil for HCC was summaried in Table 1.
HAIC with oxaliplatin and 5-fluorouracil for HCC.

HAIC, hepatic arterial infusion chemotherapy; HCC, hepatocellular carcinoma; mOS, median overall survival; mPFS, median progression-free survival; ORR, objective response rate; DCR, disease control rate; AEs, adverse events; PVTT, portal vein tumor thrombosis; RECIST, Response Evaluation Criteria in Solid Tumor; mRECIST, modified RECIST; TACE, trans-arterial chemoembolization; TAE, trans-arterial embolization.
Cisplatin-based regimens
In first-line treatment settings
HAIC alone
In 2018, Choi et al. 62 compared the efficacy and safety of HAIC to sorafenib for HCC with PVTT. A total of 58 participants with HCC and PVTT were enrolled and assigned (1:1) to either the HAIC group or the sorafenib group. A port-catheter system was implanted for HAIC, with the regimen of cisplatin (60 mg/m2 for 2 h on day 2) and 5-fluorouracil (500 mg/m2 for 5 h on days 1–3). The median OS and TTP were 14.9 versus 7.2 months (p = 0.012) and 4.4 versus 2.7 months (p = 0.010), respectively, with the ORR of 27.6% versus 3.4% (p = 0.001). However, more participants in the HAIC group experienced grade 3/4 AEs (62.0% versus 51.7%).
In 2021, Ahn et al. 63 found that HAIC with cisplatin and 5-fluorouracil was superior to sorafenib in patients with HCC and major PVTT, with a median TTP of 6.2 versus 2.1 months (p = 0.006) and a DCR of 76% versus 37% (p = 0.001); however, the median OS was similar (10.0 versus 6.4 months, p = 0.139), and more patients in the HAIC group experienced hematologic AEs. Taken together, these results suggest that HAIC with cisplatin and 5-fluorouracil represents an alternative treatment method for patients with advanced HCC and PVTT, even for those with major PVTT.
HAIC plus systemic therapy
HAIC with a cisplatin-based regimen plus sorafenib has also been investigated for advanced HCC for many years. However, several prospective trials reported that the addition of HAIC did not present significantly better survival benefits over sorafenib alone.
In 2016, Ikeda et al. 50 compared the efficacy and safety of sorafenib plus HAIC to sorafenib alone for advanced HCC. The median OS was 10.6 versus 8.7 months (p = 0.073), and the response rate was 21.7% versus 7.3% (p = 0.09). However, the combination group presented better OS benefits in participants with serum AFP levels of <400 ng/ml (median OS: 14.8 versus 8.7 months, p = 0.042). Treatment-related AEs were more frequent in the combination group but all were well tolerated.
In the SCOOP-2 trial, Kondo et al. 64 reported that the efficacy of HAIC with cisplatin plus sorafenib was similar to that of sorafenib alone for HCC, with the median OS of 10.0 versus 15.2 months (p = 0.78), as well as a similar median TTP (2.8 versus 3.9 months, p = 0.60). The most frequent HAIC-related AEs was elevated aspartate aminotransferase (AST)/alanine aminotransferase (ALT) (9%), and one patient discontinued HAIC due to an unacceptably elevated total bilirubin level.
In 2018, Kudo et al. 65 compared the efficacy and safety of sorafenib plus HAIC with low-dose cisplatin and 5-fluorouracil to sorafenib alone for the treatment of advanced HCC in the SILIUS trial. The median TTP of the combination group was significantly longer than that of the sorafenib group (5.3 versus 3.5 months, p = 0.004), and the combination group also presented better ORR based on RECIST criteria (36% versus 18%, p = 0.003). The trial, however, did not meet its primary endpoint (OS), with the median OS of 11.8 versus 11.5 months (p = 0.955). The median PFS was also similar between the groups (4.8 versus 3.5 months, p = 0.051). In the safety profile, more grade 3/4 treatment-related AEs were observed in the sorafenib plus HAIC group than in the sorafenib group. Grade 3 and grade 4 port-related complications were observed in 11% and 1% of the participants, respectively, in the combination group.
More recently, the combination of HAIC with cisplatin and Lenvatinib has also been explored as an alternative treatment for advanced HCC. In 2021, Ikeda et al. 66 reported the LEOPARD trial, which evaluated the efficacy and safety of HAIC with cisplatin plus lenvatinib for advanced HCC categorized as Child-Pugh class A. The results revealed an ORR of 64.7% (mRECIST)/45.7% (RECIST 1.1) and the median PFS and OS of 6.3 and 17.2 months, respectively. In the safety profile, elevated AST (34%), hyponatremia (25%), leukopenia (22%), elevated ALT (19%), and hypertension (11%) were the most frequently observed grade 3/4 treatment-related AEs.
Neoadjuvant settings
More rarely, HAIC with cisplatin has been investigated as a neoadjuvant treatment for HCC, with unsatisfactory results. In 2021, Oyama et al. 67 explored the efficacy and safety of HAIC with cisplatin before radiofrequency ablation for HCC. A total of 70 participants were enrolled and assigned (1:1) to HAIC and non-HAIC groups. HAIC was performed via an intra-arterial catheter, with the regimen of cisplatin (65 mg/m2). The 1- and 3-year recurrence-free survival rates were 82.9% and 54.3%, respectively, in the HAIC group and 74.3% and 34.4%, respectively, in the non-HAIC group, without statistically significant differences (p = 0.094). Only two participants in the HAIC group experienced treatment-related AEs, which were both grade 2. The updated evidence of HAIC with cisplatin-based regimens for HCC was summaried in Table 2.
HAIC with cisplatin-based regimen for HCC.

HAIC, hepatic arterial infusion chemotherapy; HCC, hepatocellular carcinoma; mOS, median overall survival; mPFS, median progression-free survival; ORR, objective response rate; DCR, disease control rate; AEs, adverse events; PVTT, portal vein tumor thrombosis; RECIST, Response Evaluation Criteria in Solid Tumor; mRECIST, modified RECIST; RFA, radiofrequency ablation.
Regimens and updated evidence for BTCs
FUDR
FUDR has shown a high liver extraction rate of 95% during HAIC, resulting in liver tumors being exposed to 400 times the concentration of chemotherapeutic agents compared to the systemic infusion. 68 Thus, in the United States, FUDR via a surgically implanted pump has been the most frequently used HAIC regimen for patients with colorectal carcinoma liver metastases and iCCA for many years. Dexamethasone has been found to decrease the biliary toxicities of FUDR when added to the infusion. 69
HAIC alone
Recently, HAIC with FUDR was demonstrated to be not inferior to surgical resection for patients with multifocal iCCA. Wright et al. 33 found that surgical resection did not prolong survival compared to intra-arterial therapies, which included HAIC via pump, TACE, and selective internal radiation therapy in 2018, with a median OS of 20 versus 16 months (p = 0.627). However, the HAIC group had the longest OS when compared to the surgical resection and TACE groups (39 versus 20 versus 15 months, p = 0.002). In 2022, Franssen et al. 35 also found that patients with multifocal iCCA showed similar OS after being treated by HAIC with FUDR or surgical resection (20.3 versus 18.9 months, p = 0.32). However, more patients in the HAIC group had bi-lobar diseases (88.0% versus 34.3%), large tumors (median: 8.4 versus 7.0 cm), and four or more lesions (66.7% versus 24.2%) in this study, and the 30-day mortality associated with the treatment was higher in the surgical resection group (6.2% versus 0.8%, p = 0.01).
HAIC plus systemic chemotherapy
HAIC combined with systemic chemotherapy has also been explored for iCCA, and the efficacy of this combination may be superior to systemic chemotherapy alone.
In 2016, Konstantinidis et al. 32 found that HAIC with FUDR plus systemic chemotherapy presented better survival benefits when compared to systemic chemotherapy alone, with the median OS of 30.8 versus 18.4 months (p < 0.001). The response rate was also higher in the combination group, although this difference did not achieve statistical significance (59% versus 39%, p = 0.11).
In 2020, Cercek et al. 34 evaluated the efficacy and safety of HAIC with FUDR (0.12 mg/kg × kg × 30/pump flow rate) combined with systemic gemcitabine (800 mg/m2) and oxaliplatin (85 mg/m2) for unresectable iCCA in a phase II trial. In all, 42 participants without distant metastatic disease were enrolled, most of whom were chemo-naïve. The 6-month PFS was 84.1%, and the median PFS and OS were 11.8 and 25.0 months, respectively. Notably, 58% of the participants achieved a partial radiological response, and the DCR at 6 months reached 84%. The safety profile was acceptable, and the most frequent grade 3/4 treatment-related AEs were liver dysfunction. The updated evidence of HAIC with FUDR for BTCs was summaried in Table 3.
HAIC with floxuridine for BTCs.

HAIC, hepatic arterial infusion chemotherapy; FUDR, floxuridine; BTCs, biliary tract cancers; mOS, median overall survival; mPFS, median progression-free survival; ORR, objective response rate; DCR, disease control rate; AEs, adverse events; iCCA, intrahepatic cholangiocarcinoma.
Oxaliplatin and 5-fluorouracil
HAIC alone
HAIC with oxaliplatin and 5-fluorouracil was explored as a first-line treatment for pCCA in 2016, and it showed excellent efficacy and safety. 31 The HAIC was performed via a percutaneously implanted port-catheter system in 37 participants, with the regimen of oxaliplatin (40 mg/m2 for 2 h) and 5-fluorouracil (800 mg/m2 for 22 h), which circulated 3 days every 3–4 weeks. Notably, the ORR and DCR were 67.6% and 89.2%, respectively. The median OS was 20.5 months, and the median PFS was 12.2 months, with the median local PFS of 25.0 months. Meanwhile, participants with periductal infiltrating pattern experienced better survival benefits from HAIC than those with mass-forming pattern in this trial, with a median PFS of 26.2 versus 6.9 months (p < 0.001) and median local PFS of 26.2 versus 7.0 months (p < 0.001). The most frequent treatment-related AEs were grade 1/2 nausea and/or vomiting (83.8%) and oxaliplatin-related peripheral neuropathy (75.7%), while the most frequent grade 3/4 treatment-related AEs were anemia (16.2%), leukopenia (10.8%), and thrombocytopenia (13.5%).
In 2013, Sinn et al. 28 investigated the efficacy and safety of HAIC for unresectable BTCs in a phase II trial. A total of 37 participants were enrolled, most of whom were iCCA (32/37). The median OS and PFS were 13.5 and 6.5 months, respectively. However, the primary endpoint was not met, with the ORR of only 16%. The treatment-related AEs were mild, and the most frequent treatment-related AEs were grade 1/2 sensory neuropathy. In 2021, HAIC with oxaliplatin and 5-fluorouracil was demonstrated to be effective and safe for patients with advanced GBC who experienced disease progression after systemic chemotherapy or were contraindicated for systemic chemotherapy, with the ORR, DCR, median OS, and PFS of 69.2%, 92.3%, 13.5 months, and 10.0 months, respectively. 48 A total of eight patients experienced grade 3/4 treatment-related AEs, while no port-related complications were observed. Taken together, HAIC with oxaliplatin and 5-fluorouracil also presented satisfactory efficacy and safety for patients with advanced iCCA, pCCA, and GBC.
In 2021, Cai et al. 70 demonstrated that, compared to TACE, HAIC was more effective for unresectable iCCA. In this study, the median OS was 19.6 versus 10.8 months (p = 0.028), with 1- and 2-year OS rates of 60.2% versus 42.9% and 38.7% versus 29.4%, respectively. The median intrahepatic PFS in the HAIC group was significantly longer than that in the TACE group (9.2 versus 4.4 months, p = 0.026), although the median PFS was similar (3.9 versus 3.7 months, p = 0.641). The treatment-related AEs were mild and manageable with symptomatic treatment, while more patients in the HAIC group experienced treatment-related myelosuppression (p = 0.007) and vomiting (p = 0.006).
HAIC plus systemic therapy
Recently, the combination of HAIC and systemic therapy has been increasingly investigated and demonstrated to be both effective and safe for patients with advanced BTCs.
In 2022, Wang et al. 71 reported their phase II trial evaluating the efficacy of HAIC combined with bevacizumab and toripalimab (a PD-1 inhibitor) as a first-line treatment for advanced BTCs. A total of 32 participants were enrolled and treated by HAIC with bevacizumab (300 mg for 2 h, day 1), oxaliplatin (40 mg/m2 for 2 h, days 1–3), and 5-fluorouracil (800 mg/m2 for 22 h, days 1–3) plus intravenous toripalimab (240 mg, day 1) every 4 weeks. Impressively, the ORR was 84.3%, and the DCR was 96.9%. The median PFS and OS were not reached, while the 6-month PFS rate and OS rate were 78.5% and 89.9%, respectively. Grade 3/4 treatment-related liver dysfunction, the most frequent grade 3/4 treatment-related AEs, was observed in only 18.8% of participants.
In 2022, Zhang et al. 72 conducted a retrospective study evaluating the efficacy of HAIC combined with PD-1 inhibitor as a second- or more line treatment for advanced BTCs. The median PFS and OS were 3.7 and 8.8 months, respectively, in all 36 patients, with an ORR of 11.5% and a DCR of 76.9%. The investigators also found that early administration of the combination of HAIC and PD-1 inhibitor presented better survival benefits, with a median OS of 13.0 versus 7.6 months (p = 0.004). The treatment-related AEs were mild, with 44.4% of patients experiencing grade 3/4 treatment-related AEs. The updated evidence of HAIC with oxaliplatin and 5-fluorouracil for BTCs was summaried in Table 4.
HAIC with oxaliplatin and 5-fluorouracil for BTCs.

HAIC, hepatic arterial infusion chemotherapy; BTCs, biliary tract cancers; mOS, median overall survival; mPFS, median progression-free survival; ORR, objective response rate; DCR, disease control rate; AEs, adverse events; pCCA, perihilar cholangiocarcinoma; iCCA, intrahepatic cholangiocarcinoma; eCCA, extrahepatic cholangiocarcinoma; GBC, gallbladder cancer; TACE, trans-arterial chemoembolization.
Complications and toxicities of HAIC
Device-related complications
Catheter dislocation has been one of the most frequent complications observed of HAIC, occurring in 2–44% of patients.45,73 Catheter dislocation can lead to the loss of chemotherapeutic agents in the tumor and may aggravate the gastrointestinal AEs associated with chemotherapy, such as nausea, vomiting, and gastrointestinal ulcer. Replacing or removing the catheter is necessary whenever the catheter dislocation is detected. 40
Catheter or hepatic artery occlusion due to thrombosis, fibrin sheath formation, or catheter kinking, which can prevent or interrupt the HAIC treatment, has been found in 4–17% of patients treated with HAIC.40,45,73 Thrombolytic therapy with tissue plasminogen activators is useful for acute catheter or hepatic artery occlusion due to thrombosis and fibrin sheath formation, while replacing and removing the catheter should be considered whenever catheter or hepatic artery occlusion due to catheter kinking or late thrombosis and fibrin sheath formation is found.73–75 Another viable alternative is to place a metallic stent into the occluded hepatic artery. 40
Erosion of the GDA or other abdominal viscera due to the inserted catheter is a rare complication in patients with surgically implanted pumps. 45 The erosion may cause infection, gastrointestinal bleeding, or abdominal pain. Thus, the catheter or pump should be removed whenever erosion is found.
Pump/port dislocation or rotation is rare and requires pump/port repositioning, either manually or via a new incision. 45 An appropriate pocket created before the implantation may be useful for preventing these complications.
Procedure-related complications
Infection is an undesirable complication that is mainly associated with the pump/port pocket and occurs in up to 25% of patients. 76 Sterile operation and early administration of antibiotics are necessary for preventing infection. Debridement and device removal should be considered whenever the infection is observed.45,77
Bleeding at the puncture site, pseudoaneurysm, seroma, hematoma, and nonunion of the wound at the pump/port pocket are rare procedure-related complications in 5–16% of patients.45,73,78 Aspiration is the most common method for small and non-expanding seromas and hematomas, while re-exploration should be considered whenever the pocket expands rapidly or there is apparent tension in the pocket. 45 Delayed suture removal may be useful for patients who experienced nonunion of the wound.
Lung or cerebral infarction due to thrombosis are rare complications that sometimes occur in patients receiving HAIC via an intra-arterial catheter, which often requires a relatively long time in bed. 40 The placement of a catheter via subclavian, axillary, or brachial arteries may increase the risk of cerebral infarction as well. 41 Thus, the use of anticoagulant agents should be considered during HAIC.
HAIC-related toxicities
HAIC-related toxicities, including both hematologic and non-hematologic toxicities, are mainly associated with the use of chemotherapeutic agents and may require medical intervention, dose modification, or treatment interruption. Leukopenia, thrombocytopenia, and neutropenia are the most frequently observed HAIC-related hematologic toxicities, while elevated AST/ALT, elevated bilirubin, and gastrointestinal reaction represent the most common non-hematologic toxicities. For patients who receive HAIC with FUDR, dexamethasone should be infused alongside FUDR to decrease the biliary toxicities. In addition, the less-frequently observed complication of peripheral neuropathy should also be considered in patients who received HAIC with oxaliplatin.
Chemotherapeutic agents may also cause vascular damage and gastrointestinal ulcers. 40 Extrahepatic blood flow redistribution should be performed to prevent the exposure of those organs to chemotherapeutic agents. Mixing hydrocortisone or other steroids with intra-arterial agents may represent an alternative management strategy for preventing vascular damage. 69
Conclusion
The techniques and regimens used for HAIC differ significantly between different regions and centers of the world. A percutaneously implanted indwelling port-catheter system with a fixed catheter tip following extrahepatic and intrahepatic blood flow redistribution has provided us with a simple, repeatable HAIC treatment is now recommended in many clinical applications. The efficacy and safety of HAIC for PHCs have been gradually confirmed by an increasing number of prospective studies. As a trans-arterial whole liver local treatment, HAIC is generally indicated for patients with locally advanced HCC and large tumors, a high tumor burden, and diffuse tumors, especially in those with portal vein/hepatic vein/biliary tract tumor thrombosis, as well as in cases where the tumor is confined within the liver or with limited extrahepatic metastasis. Moreover, HAIC has been recently confirmed to be a beneficial neoadjuvant/adjuvant therapy for patients with HCC and high-risk factors of recurrence, such as a high tumor burden and/or microvascular invasion. HAIC is also recognized as an alternative and salvage treatment of systemic chemotherapy for locally advanced iCCA, pCCA, or GBC with or without liver metastasis. When combined with systemic therapy, the addition of HAIC has presented greater survival benefits for patients with advanced PHCs compared to systemic therapy alone, with acceptable safety profiles. Conclusively, HAIC plays an increasingly significant role in the multidisciplinary treatment algorithms for patients with advanced PHCs.
Perspectives and future directions
Currently, many ongoing trials are evaluating and/or comparing the efficacy and safety of HAIC plus TACE, and the combination of HAIC, targeted therapy, and immunotherapy as first-line, neoadjuvant, or adjuvant treatments for BCLC B/C stage HCC, high-risk HCC, potentially resectable HCC, and advanced BTCs. Furthermore, a trial that seeks to explore HAIC as a neoadjuvant method for TACE for BCLC B stage HCC is currently recruiting. In the future, it will be worthwhile to further improve and/or modify the techniques of HAIC; explore the new regimens of HAIC, especially the use of novel agents, including nano-agents; and identify the optimal indications of HAIC. Meanwhile, exploring the optimal combination pattern of HAIC and systemic therapy or other local treatment methods, such as TACE, radiation, and ablation, as well as exploring the role of HAIC in the downstaging and/or conversion of PHCs, requires further phase III trials to provide more high-level evidence.
