Abstract
Background:
Prostate cancer (PC) has a serious public health impact, and its incidence is rising due to the aging population. There is limited evidence and consensus to guide the management of PC in Southeast Asia (SEA). We present real-world data on clinical practice patterns in SEA for advanced PC care.
Method:
A paper-based survey was used to identify clinical practice patterns and obtain consensus among the panelists. The survey included the demographics of the panelists, the use of clinical guidelines, and clinical practice patterns in the management of advanced PC in SEA.
Results:
Most panelists (81%) voted prostate-specific antigen (PSA) as the most effective test for early PC diagnosis and risk stratification. Nearly 44% of panelists agreed that prostate-specific membrane antigen positron emission tomography-computed tomography imaging for PC diagnostic and staging information aids local and systemic therapy decisions. The majority of the panel preferred abiraterone acetate (67%) or docetaxel (44%) as first-line therapy for symptomatic mCRPC patients. Abiraterone acetate (50%) is preferred over docetaxel as a first-line treatment in metastatic castration-sensitive prostate cancer patients with high-volume disease. However, the panel did not support the use of abiraterone acetate in non-metastatic castration-resistant prostate cancer (nmCRPC) patients. Apalutamide (75%) is the preferred treatment option for patients with nmCRPC. The cost and availability of modern treatments and technologies are important factors influencing therapeutic decisions. All panelists supported the use of generic versions of approved therapies.
Conclusion:
The survey results reflect real-world management of advanced PC in a SEA country. These findings could be used to guide local clinical practices and highlight the financial challenges of modern healthcare.
Introduction
Prostate cancer (PC) is the second most commonly diagnosed malignancy after lung cancer and the fifth leading cause of cancer death in men worldwide, accounting for 1,414,000 new cases and causing 375,304 deaths (3.8% of all cancer deaths in men) in 2020.1,2 PC is the fifth most commonly diagnosed cancer and the seventh leading cause of cancer mortality in Asia. 2 The International Agency for Research on Cancer predicts that the overall number of new cancer cases in Asia will reach 11.5 million by 2030. 3 According to World Health Organization data on PC incidence across 53 nations, only 30% of cases occurred in low-resource areas like Southeast Asia (SEA). 4 There are substantial differences in cancer mortality-to-incidence ratios between Asian countries in PC epidemiology. Developing SEA countries such as the Philippines, Malaysia, and Thailand had a mortality-to-incidence ratio that was more than twice as high as Japan and South Korea.5,6 Obesity, older age, race, family history, diabetes mellitus, dietary patterns, and vitamin E supplementation are all identified as risk factors for PC. 7 The number of new cases is expected to reach 22.9 million by 2040, with little variance in mortality. Mortality is expected to grow by 1.05% due to population aging. 2
PC is commonly detected by elevated plasma levels of prostate-specific antigen (PSA), a glycoprotein normally expressed by prostate tissue. According to the US Preventive Service Task Force (USPSTF), PSA screening has the potential to reduce PC and mPCa mortality in males aged 55–69 years (USPSTF C recommendation). However, PSA screening is not recommended in men aged above 70 years old owing to the potential harms of screening, and unless their life expectancy is >10 years, because of the slow-growing nature of the disease. Non-judicious screening may result in additional testing including prostate biopsy leading to overdiagnosis of clinically insignificant cancers and overtreatment, with increased treatment complications, such as incontinence and erectile dysfunction.8–10 With the 2012 USPSTF Grade D recommendation against PSA screening, PC-specific mortality for localized and metastatic PC increased among men aged 60 and older; GRADE D recommendations are still in effect.9–14 Prior to the recommendation, PSA-based screening had been adopted in several developed countries, and a decline in PC mortality rates had been observed in several countries, including the United States of America (USA), Canada, the United Kingdom (UK), and Japan.13,15,16 However, it is also noted that there has been some contention on the utility, level, and type of PSA thresholds to determine further investigations (compared to other methods such as risk-adapted or age-adapted PSA), and also poor interchangeability of PSA results obtained from different assays. 8 In view of known false-positive PSA results, 14 a tissue biopsy has been adopted as the gold standard for confirming the presence of cancer. 15
The PSA testing rate in Asian countries is lower as compared to Western countries. The increased usage of PSA testing has been linked to an increase in the incidence of PC in both Japan and Taiwan.16–18 Furthermore, the variation in PC incidence may be attributed to the fact that the genomic features of PC differ between Asian and Western populations, as well as across different regions and countries in Asia.1,18–20 SEA countries are economically diverse, resulting in differences in healthcare funding and health insurance systems, diagnostic and treatment facilities, and access to innovative treatments. Treatment practices and recommendations also vary by country. Patients in countries or regions where PSA screening is not available tend to have more advanced disease than patients in countries or regions where PSA screening is available.21,22
In recent years, treatment options for patients with advanced PC have expanded. In addition to chemotherapy, novel hormonal treatment (NHT) which includes apalutamide, enzalutamide, darolutamide, and abiraterone acetate, increases overall survival (OS) in men with advanced PC and may be administered in individuals who cannot tolerate chemotherapy.21,23 It has been observed that the use of newer medicines is inconsistent across SEA, and long-term real-world data concerning their use and impact outside controlled studies are not yet available. 24 Given the variety of therapies available for advanced PC, determining the outcomes of the disease and its treatment has become critical for treatment decision-making. Such knowledge gaps cannot be filled solely by data obtained from clinical trials conducted in homogeneous settings that do not reflect real-world practice, such as the availability or unavailability of certain treatment options or diagnostic procedures [prostate-specific membrane antigen positron emission tomography (PSMA PET) scan, for example] in certain countries. Therefore, we developed a survey designed to collect real-world data to better inform on country-specific patterns of advanced PC in SEA. Establishing an informed set of high-value clinical practice patterns is necessary to lay the foundation for the effective management of advanced PC in SEA. The aim of this study was to identify clinical practice patterns for advanced PC management in SEA countries by collaborating with clinical experts from healthcare organizations in Brunei, Indonesia, Malaysia, the Philippines, Singapore, Thailand, and Vietnam.
Methods
We followed a three-step process: (1) collection and analysis of recently published evidence on the PC treatments and guidelines, (2) development of the survey questionnaire, and (3) survey administration to panelists and response collection. A four-member core scientific committee (CSC) initiated the first step. A systematic literature review (SLR) was performed to critically analyze the data on the efficacy and safety of the therapies used in the treatment of advanced and metastatic PC in Asian patients. The findings from this SLR in the form of a manuscript have been published elsewhere. 25 PC diagnosis and treatment guidelines and consensus papers which were thoroughly reviewed included the US National Comprehensive Cancer Network (NCCN), 26 American Urological Association (AUA), 27 American Cancer Society (ACS), 28 American Society of Clinical Oncology (ASCO), 29 Canadian Urological Association-Canadian Urologic Oncology Group,30,31 National Institute of Health and Care Excellence (NICE), 32 European Association of Urology (EAU), 33 European Society of Medical Oncology (ESMO), 34 Asian Oncology Summit, 35 Singapore Cancer Network (SCAN), 36 Japanese Urological Association (JUA), 37 Chinese guidelines, 38 Hong Kong Urological Association and Hong Kong Society of Uro-Oncology (HKSUO), 39 and Advanced PC Consensus Conference (APCCC). 40
In the second step, the survey questionnaire was developed after a thorough analysis of the literature and guidelines. A total of 81 distinct questions were framed, which covered the different aspects of the clinical practice patterns relevant to SEA countries. The different themes covered in the questionnaire included general practice, PC guidelines, diagnosis and risk stratification, genetic testing, localized PC, advanced PC, metastatic castration-sensitive prostate cancer (mCSPC), non-metastatic castration-resistant prostate cancer (nmCRPC), mCRPC, bone targeting agents, and access and availability.
In the last step, the survey was administered to 16 panelists, including 4 CSC members and a panel of 12 experts from the SEA countries. The panel members were selected based on their expertise in the field of advanced PC management and comprised urologists, medical oncologists, radiation oncologists, and nuclear medicine oncologists. Panelists were invited through an email to take part in this survey. Ethics Committee or Institutional Review Board approval was not required for such type of study. However, all participants were informed of the survey’s objectives and the nature of the activity. Participants were also informed that the survey results would be published in the form of an article in a peer-reviewed publication. Before sharing the questionnaire, all panelists provided formal confirmation of their participation. All panelists completed a paper-based survey form. Panelists were asked to choose choices depending on their clinical practice patterns.
Data collected included panelists’ information encompassing age, gender, specialty, type and region of practice, years in the practice, how many patients they attend in a month, and how they keep themselves updated about the latest developments in the PC field. Information on the diagnosis and treatment of localized and advanced diseases such as mCSPC, nmCRPC, and mCRPC, innovator and generic drugs approved in their countries for the treatment of advanced disease was also collected. Responses obtained from panelists for each question were analyzed in Microsoft Excel and results were presented in the form of descriptive statistics (numbers and/or percentages).
Results
Panelist’s characteristics
Table 1 provides the characteristics of the panelists who participated in this survey. The study included 16 specialists from 7 SEA countries. The majority of participants (81%) were men. The median age of the panelists was 49 years (range: 36–71). Participants had an average of 20 years of clinical practice experience. Over half (53%) were from government hospitals/institutions, with the remaining (33%) from both government and private hospitals/institutions. Panelists were mostly from urban settings (80%) and attended 50–100 PC patients in a month. They received patients mostly through internal referrals from hospital clinicians (39%) and external referrals from other hospitals (32%). Among all the participants, 94% mentioned that they keep themselves updated with peer-reviewed articles and local/international guidelines on advancement in PC diagnosis and management.
Characteristics of the 16 panelists who participated in the survey.

PC, prostate cancer.
Responses of the 16 panelists to the survey of 81 questions on aPC management in SEA provided in Supplemental Table S1 summarizes the overview of the results obtained from the survey.
PC guidelines
For the diagnosis and management of PC, 56% of panelists reported having local or country-specific guidelines, while 44% reported the lack of such guidelines. NCCN, EAU, and ESMO were the very frequently or frequently referenced guidelines used for the diagnosis and/or management of PC. Notably, the use of Asian guidelines was very limited.
General
According to 94% of panelists, the prevalence of PC has increased in the last 5–10 years, and opportunistic screening, followed by general health screening, was the most popular screening approach for PC. Population-based screening was sometimes preferred by 19% of panelists, while no population-based screening was recommended and was strongly discouraged by 77% of panelists.
PC diagnosis and risk stratification
Panelists strongly agreed that PSA (81%), biopsy (69%), and magnetic resonance imaging (MRI) (50%) were the most preferred techniques for early PC diagnosis, followed by digital rectum examination (DRE), which was somewhat supported by 31% panelists. A large percentage of panelists preferred targeted biopsy with MRI fusion (69%) for PC biopsy, followed by non-targeted transrectal biopsy (19%) and non-targeted transperineal biopsy (19%). Nearly 56% of panelists chose 12-core (non-targeted) biopsy as the minimal standard biopsy in the initial scenario of raised PSA and normal/abnormal DRE, followed by MRI-targeted biopsy alone (25%) and MRI-targeted with systematic biopsy (25%). All panelists stated that they routinely risk stratifying PC patients. Among all the panelists, approximately 63% of participants indicated that the risk groups defined by the NCCN guideline was their first preference for localized PC risk classification. Harvard and EAU criteria were the top choices for 31% and 25% of panelists, respectively. Nearly 50% of panelists said EAU was their second preference, whereas 38% said AUA was their third preference. According to specialty subgroups (Figure 1), the NCCN guideline was the first preferred option among medical oncologists, while Harvard criteria was preferred among urologists.

The first preferred guideline is to stratify localized PC patients into low-, intermediate-, and high-risk categories, by specialty.
Genetic testing
Out of 16 panelists, 93% stated that understanding gene mutations is vital for family genetic counseling and better evaluation of the risks of PC patients. However, because of high cost and lack of awareness among patients only 20% of all panelists consistently use genetic testing in their practice. About 43% of those surveyed said they would recommend genetic counseling ‘after the tests’ or ‘both before and after the testing’.
Over 47% of panelists normally performed somatic gene mutation testing first, followed by germline testing (if somatic is positive); while 26% and 20%, respectively, performed only germline and somatic testing. Over 79% of panelists suggest somatic genetic testing at the mCRPC stage, while 69% and 54% recommend such testing at the mCSPC and localized high-risk or locally progressed PC stages, respectively. According to 47% of panelists, archival tissue and new fresh biopsy tissue were sent for somatic mutation testing. According to most panelists (69%), testing for BRCA1/2 and ATM gene mutation was always recommended for their patients, followed by the 15 homologous recombination repair (HRR) mutation gene panels according to the PROfound study (43%). A full next-generation sequencing (NGS) panel is rarely advised as stated by 50% of the panelists.
Localized PC
According to 53% of panelists, the most preferred treatment option for patients with low-risk localized PC was active surveillance, followed by radical prostatectomy (27%), low-dose-rate (LDR) brachytherapy (20%), and external beam radiotherapy (EBRT) (13%). According to specialty subgroups (Figure 2), medical oncologists preferred radical prostatectomy and EBRT, followed by active surveillance, whereas urologists preferred active surveillance followed by radical prostatectomy.
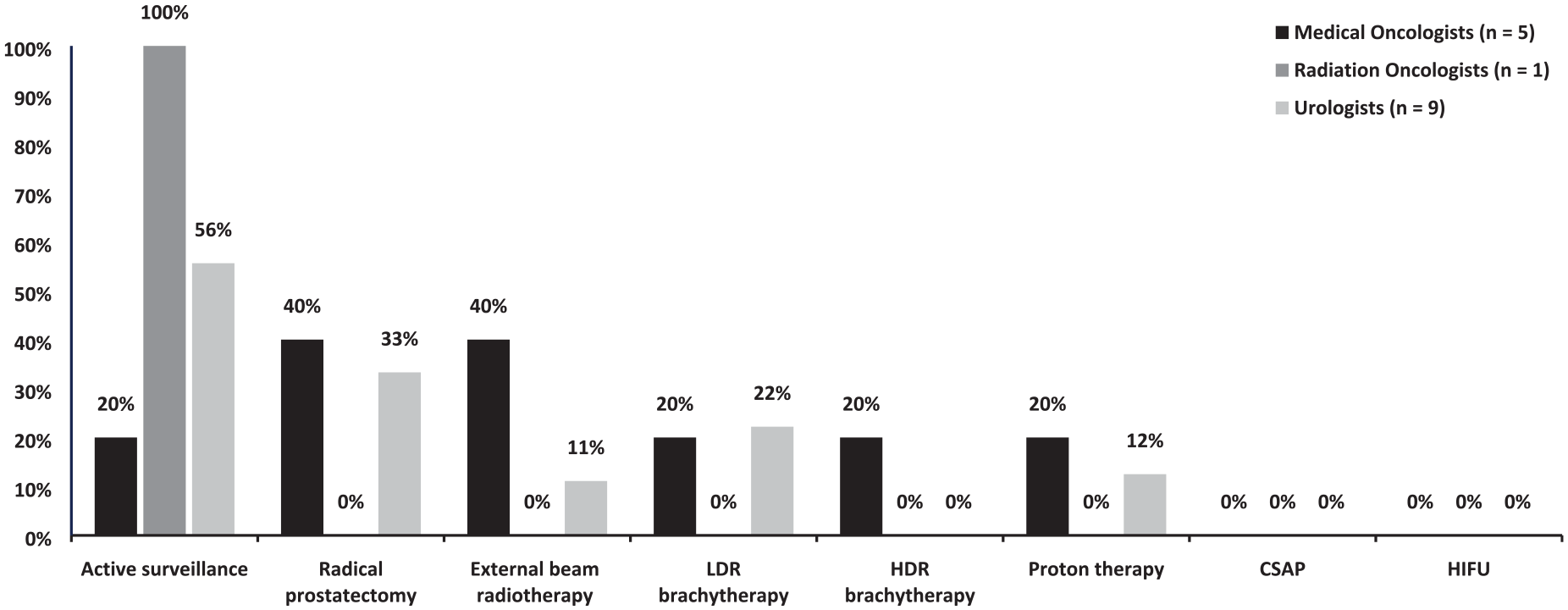
Strongly agreed preferred treatment option for patients with low-risk localized prostate cancer, by specialty.
In patients with intermediate risk of localized PC, all panelists (100%) stated radical prostatectomy as the most preferred treatment option, followed by EBRT and high-dose rate (HDR) brachytherapy. All panelists (100%) selected EBRT as the most preferred treatment option for high-risk localized PC patients, followed by radical prostatectomy (88%).
Following radical prostatectomy, 57% of panelists preferred a PSA level of 0.2 ng/mL to begin early salvage radiation therapy. When asked to define biochemical recurrence after radical radiotherapy, nearly two-thirds (63%) said a PSA rise ⩾2 ng/mL higher than the nadir PSA (Phoenix criteria).
Oligometastatic PC
There was a clear, unified definition of oligometastatic PC, according to 44% of panelists. When asked about a number of metastases a patient must have to be categorized as having oligometastatic PC, most panelists (80%) answered ‘no visceral metastasis and three or less skeletal bone lesions’.
Advanced PC
In Figure 3, according to 94% of panelists, androgen deprivation therapy (ADT) is the primary systemic treatment for advanced PC. To achieve ADT, LHRH agonist is the preferred strategy (86%), followed by LHRH antagonist (7%) and maximum/combined androgen blockade (7%). Three-fourths of panelists mentioned that serum testosterone level <50 ng/dL was the accepted castration level.

Most preferred method of treatment for patients with advanced prostate cancer.
mCSPC
In mCSPC patients with low-volume disease, abiraterone acetate was chosen as the first preferred treatment by 60% of panelists, followed by apalutamide (33%) and ADT alone (19%) (Figure 4). None of the panelists recommended the use of docetaxel and nearly 75% of the panelists proposed using metastasis-directed therapy (MDT) in addition to standard therapy, in patients with mCSPC with low-volume disease (Figure 4). Approximately 87% of the panelists opined that the addition of NHT to ADT would be most effective in causing PSA reduction compared to docetaxel-ADT or ADT alone, in patients with mCSPC with low-volume disease [Figure 4(4.4)]. According to panelists, among high-volume mCSPC patients, abiraterone acetate was recommended as the first preferred option by 53% of panelists, followed by ADT plus docetaxel with or without abiraterone acetate (33%), while ADT plus apalutamide, ADT plus enzalutamide, and ADT plus docetaxel were preferred by a minority of the panelists (Figure 4).

Most preferred method of treatment for patients with mCSPC – results from the survey.

Most preferred method of treatment for patients with mCSPC – results from survey.
With regards to the use of docetaxel in mCSPC patients, 40% of the panelists said they utilized the standard docetaxel regimen (75 mg/m2), while another 40% said they used docetaxel between 60 and 75 mg/m2 (Figure 4). According to 88% of panelists, patients showing a good response to docetaxel should receive up to six cycles of docetaxel chemotherapy.
As shown in Figure 5, to reduce the risk of docetaxel-related febrile neutropenia, 62% of panelists reported using GCSF as a primary prophylactic. More than 80% of panelists considered factors such as the performance status of the patient, high-volume versus low-volume disease, efficacy, and safety while administering docetaxel in patients with mCSPC, while 63%, 56%, and 53% considered comorbidities, cost, and patient preference, respectively.
Approximately 70% of panelists preferred CT scans combined with bone scans to guide treatment for mCSPC patients, while 44% chose PSMA-PET scans (Figure 5). One panelist claimed that if PSMA-PET scans became available in his country, he would use them for patients to guide treatment for mCSPC.
Nearly 70% of panelists agreed that additional imaging is required in patients with mCSPC beyond baseline and disease progression. According to 88% of panelists, additional imaging in mCSPC patients should be done at PSA rise or symptoms (Figure 5). Triple therapy was the new standard of care for de novo high-volume mCSPC patients, according to 50% of the panelists, while 25% of panelists agreed on triple therapy as the new standard of care for all high-volume mCSPC. Ninety-four percent of panelists reported fitness for chemotherapy, whereas 80% indicated cumulative cost, patient desire, and toxicity as the barriers to triple therapy.
nmCRPC
As shown in Figure 6, apalutamide was selected as the first preferred treatment by 75% of participants in patients with nmCRPC, followed by enzalutamide and darolutamide. According to 69% of panelists, docetaxel has no role in patients with nmCRPC. To guide treatment decisions, 50% of panelists said they use a CT scan plus a bone scan or a whole-body MRI scan to differentiate between nmCRPC and mCRPC, and none recommended the use of a PSMA PET CT scan.

Most preferred method of treatment for patients with nmCRPC – results from the survey.
mCRPC
As shown in Figure 7, most panelists (88%) defined castration-resistant PC (CRPC) as ‘two elevations above PSA nadir by 50%, and PSA >2 ng/mL’. Seventy-five percent of panelists identified imaging progression as CRPC, while 56% identified biochemical progression.

Most preferred method of treatment for patients with mCRPC – results from the survey.
When it comes to treatment for mCRPC, 80% of panelists said that prior treatment and its response was the most important consideration, followed by patients’ symptoms (56%). The second preferred factors were patient comorbidities and performance status (44%), and time to CRPC (36%).
The majority of the panelists preferred abiraterone acetate over apalutamide and enzalutamide as first-line therapy in asymptomatic or mildly symptomatic mCRPC patients who received ADT alone in the mCSPC scenario. Sixty-seven percent of panelists selected enzalutamide as their second choice.
Abiraterone acetate was preferred by 80% of panelists as first-line therapy in asymptomatic or minimally symptomatic mCRPC patients who have previously received docetaxel in the mCSPC condition, followed by enzalutamide (33%) and apalutamide (13%).
In symptomatic mCRPC patients who received ADT alone in mCSPC, 67% of panelists preferred abiraterone acetate as first-line therapy, followed by docetaxel (44%). Only one panelist stated another option that includes radiotherapy as a recommended route of treatment in symptomatic mCRPC patients receiving ADT alone in mCSPC.
As shown in Figure 8, in mCRPC patients, 50% of panelists stated that they routinely utilize ‘5 mg BID’ prednisone/prednisolone with abiraterone acetate. Thirty-eight percent of the panelists chose 5 mg QD. Docetaxel 75 mg/m2 was the routine docetaxel regimen used by 69% of panelists for patients with mCRPC. Forty-four percent of panelists said they would prescribe ‘up to 6 cycles’ or ‘6–10 cycles’ of docetaxel to patients with mCRPC who respond show satisfactory responses. Around 63% of panelists reported frequent utilization of prophylactic use of GCSF in mCRPC patients to reduce the risk of docetaxel-induced febrile neutropenia.

Most preferred method of treatment for patients with mCRPC – results from the survey.
In asymptomatic or mildly symptomatic mCRPC patients who had progressed on or after docetaxel, 74% of panelists favored abiraterone acetate as the second-line therapy, followed by enzalutamide (38%) if the patients did not have prior exposure to apalutamide, enzalutamide, or abiraterone acetate. Over 80% of panelists stated ‘docetaxel’ was their first choice for second-line therapy in symptomatic mCRPC patients who had progressed on or after first-line enzalutamide or abiraterone treatment. Notably, 13% of panelists chose poly-ADP ribose polymerase (PARP) inhibitors followed by radium-223 as their first preference for their second-line therapy in symptomatic mCRPC patients who had progressed on or after first-line treatment with enzalutamide, or abiraterone acetate.
As per Figure 9, cabazitaxel and PARP inhibitors were mentioned as the first recommended medication in third-line therapy by about 60% of panelists. According to 63% of the panelists, very few mCRPC patients receive second- or third-line cabazitaxel treatment.

Most preferred method of treatment for patients with mCRPC – results from the survey.
When discontinuing abiraterone acetate or chemotherapy in mCRPC patients, 38% of panelists would taper corticosteroid medication gradually in 1–2 weeks and 31% in 3–4 weeks. However, 25% reported an immediate cessation of corticosteroids. Approximately 81% of the panelists said they utilize 177Lu-PSMA in the later line of treatment for patients with mCRPC. Forty-four percent of panelists stated that they do not use immunotherapy (e.g. pembrolizumab) in mCRPC, whereas 37% stated that they do.
As shown in Figure 10, almost 56% of panelists said they use pembrolizumab in the second or third line of therapy, and 73% said they always advocate biomarker testing before using immunotherapy in mCRPC patients. Pembrolizumab was used in mCRPC MSI-high patients, according to 56% of panelists, followed by high PD-L1 expression, according to 31% of panelists. NGS is now the top technology in clinical practice for enhancing patient care, according to 50% of panelists.
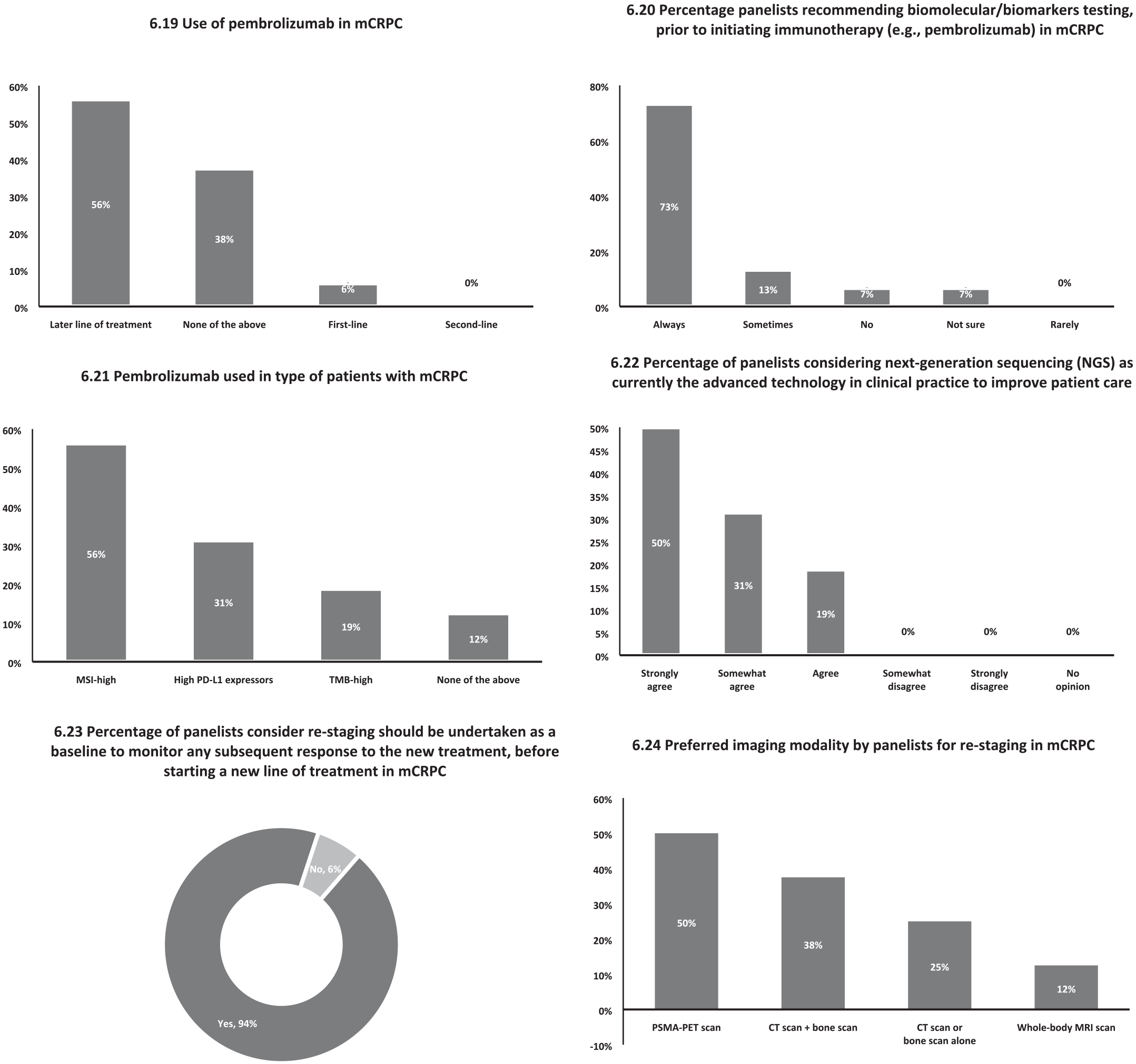
Most preferred method of treatment for patients with mCRPC – results from the survey.
Almost 94% of panelists agreed that re-staging should be performed at baseline before beginning a new line of treatment, and 50% reported that PSMA-PET scan is the most widely utilized imaging modality for re-staging, followed by CT scan plus bone scan (38% of panelists).
Bone targeting agents
According to 56% of panelists, most advanced PC patients show involvement of bone metastases. Denosumab is the first preferred bone-targeting drug, according to 87% of panelists; when denosumab is not available, zoledronic acid was the second preferred bone-targeting agent as per 69% of panelists. According to a panelist who chose the ‘other option’, alendronate and other bisphosphonates were commonly utilized bone targeting medicines. According to the majority of panelists (74%), bone-targeting agents have no role in mCSPC high-volume disease.
General practice
In terms of monitoring the treatment effect, blood tests every 3 months was the advisable practice, according to all panelists, followed by imaging, according to 81% of panelists. Slowing disease progression was the criterion as per all panelists to continue treatment, followed by quality-of-life benefits and minimal adverse effects as stated by at least 80% of panelists. Clinical deterioration was indicated by all panelists as the reason criteria for discontinuing current treatment, followed by radiographic progression by 88% of panelists.
In the overall management of PC at all phases, all panelists agreed or strongly agreed that a multidisciplinary team would provide better care for patients than individual practice. More than 80% of panelists mentioned they could discuss PC management with a multidisciplinary team. Approximately 46% of panelists indicated it was difficult or extremely difficult to build a multidisciplinary team in their practice or country.
Access and availability
Most panelists strongly agreed that the high cost of medications (75%) and limited coverage by public insurance schemes (69%) are the most significant barriers to the access and use of PC therapy, followed by drug non-inclusion in the essential medicines list (EML) (56%).
Based on the availability, generic versions of approved drugs were commonly prescribed for cancer patients, according to 69% of the panelists. Off-label drugs were rarely prescribed by 50% of panelists, whereas 38% of panelists stated they sometimes prescribe off-label medicines. All participants agree that improving the availability of valuable high-cost medications, increasing budget allocation for effective high-cost medicines, and reducing the time for approval and registration of cancer medicines will all assist in improving cancer medicine availability. All panelists strongly agreed that price negotiations, patient assistance schemes, and the availability of high-quality generic medicines/biosimilars may help improve cancer treatment costs. While 44% of panelists indicated compulsory licensing as one of the options that could assist in increasing cancer drug affordability.
Discussion
This article consolidates the real-world clinical practice patterns of clinicians involved in managing PC, based on survey responses by expert panelists in the SEA region, allowing for some consensus statements to be established for dissemination. With regards to PC diagnostics, it is interesting to note that only half the panelists listed MRI as a preferred diagnostic modality, the majority of the panelists preferred MRI fusion for PC biopsy. This could reflect that while the panelists understood that in men with positive MRI results and elevated PSA levels, an MRI-targeted biopsy combined with systematic biopsy may allow for higher diagnostic accuracy and a reduction of unnecessary biopsies by half, there may be financial and resource constraints toward the routine use of MRI scans in some parts of the region. 41 Alternative strategies to the routine use of MRI scans could include the use of nomograms, biomarkers, or PSA isoforms, for individual risk prediction and selective use of an expensive and resource-intensive MRI scan. 42
The NCCN risk-grouping system was the first preference to classify PC patients based on risk by nearly two-thirds of panelists in the survey, likely recognizing the NCCN criterion’s discrimination ability to categorize patients as low, intermediate, or high risk. 43
Poly (adenosine diphosphate-ribose) polymerase inhibitor (PARPi) treatment is currently recommended for patients with metastatic CRPC (mCRPC) who have HRR gene mutations. 44 It is interesting to note that while the majority of panelists favor genetic testing in the mCRPC stage, with some advocating its use in earlier stages of disease such as localized high risk or mCSPC, only 20% of all panelists consistently use genetic testing in their practice due to high cost and lack of patient awareness. This paradox well reflects the influence of nonclinical factors on clinical practice in the real world.
According to panelists, ADT plus NHT such as abiraterone acetate, apalutamide, and enzalutamide are the preferred standard treatments in low-volume mCSPC patients, with MDT as a treatment modality in addition to standard therapy. In high-volume mCSPC patients, it is interesting to note that more than 50% of panelists chose ADT plus abiraterone acetate as the preferred standard treatment, while only between 13% and 33% of panelists regarded ADT plus docetaxel in a doublet or triplet (with additional abiraterone acetate or darolutamide) regimen, as preferred options. This may be reflective of the concerns of potentially high rates of toxicities such as neutropenic sepsis, in Asian patients undergoing chemotherapy. 45 Poon et al. reported that Asian PC patients are particularly susceptible to docetaxel-related febrile neutropenia, and advocated primary GCSF prophylaxis in mCRPC and mHSPC patients undergoing chemotherapy, particularly in those with poor performance status. 46 This real-world concern could also explain why a higher percentage (62%) of panelists routinely used GCSF as primary prophylaxis to reduce the risk of docetaxel-induced febrile neutropenia. Panelists opined that patient’s performance status, high-volume versus low-volume disease, efficacy, and safety are all factors to consider when using docetaxel. NHTs such as apalutamide, enzalutamide, and darolutamide are now available for the treatment of nmCRPC. According to panelists’ responses, ADT plus apalutamide appears to be the preferred treatment option for patients with nmCRPC, among the three standard NHT options. It is also interesting to note that 44% of panelists opted to use abiraterone in nmCRPC patients although it is generally not approved for treatment in this indication, and a quarter of panelists agreed to the role of deocetaxel in nmCRPC patients, despite the lack of OS benefit data. This interesting observation may be reflective of cost, access, reimbursement considerations, and possibly region/country-specific registered indications or approval of treatment agents in different SEA countries. Abiraterone acetate is currently recommended as first and second line of treatment for patients with asymptomatic or mildly symptomatic and symptomatic mCRPC patients, including those who have not received docetaxel, with an approved dosing schedule for abiraterone was 1000 mg on an empty stomach.47,48 However, according to the survey, most physicians occasionally use a low dose of abiraterone in mCRPC patients, based on a small prospective phase II study which showed that low-dose abiraterone (250 mg) taken with a low-fat breakfast may be as effective (based on PSA response) as the full dose taken on an empty stomach. 49 This is likely a cost-mitigating measure for patients who would otherwise not be able to afford this treatment, given the need for out-of-pocket payments in some healthcare systems in the Southeast Asian region. The NCCN guidelines also listed this strategy as a potential option for such consideration. In mCRPC patients with bone metastases, bone health is a major concern, and 50 bone-targeted agents such as bisphosphonate zoledronic acid and the RANKL inhibitor denosumab are approved drugs in the treatment scenario of mCRPC with bone metastases for the prevention of skeletal-related events. 50 In the survey, the majority of the panelists had preferred denosumab over zoledronic acid for mCRPC patients with bone metastases, possibly due to reasons such as higher efficacy, ease of administration, and no need to adjust dosage with renal function for denosumab.
Some concerns on maintaining standards of clinical practice raised by panelists include the challenge of forming multidisciplinary teams in many SEA countries. High drug costs, limited coverage by public insurance schemes, and non-inclusion on the EML, among other factors, are major barriers to access and use of PC medicine and cancer treatment in SEA countries. Physicians prescribe generic versions of approved PC medications as well as off-label medications. Improving the availability of valuable high-cost medicines, increasing budget allocation for effective high-cost medicines, and shortening the time for approval and registration of cancer medicines will all help to improve cancer medicine availability in the SEA region.
Despite clear evidence of clinical benefit, cost remains a major factor in whether clinicians consider and discuss these options with their patients. Patients in SEA countries, such as Malaysia, have limited access to high-cost drugs that are not covered by the publicly funded health system. Only 18% of Malaysian patients, particularly those from higher-income families, have personal insurance. Most men with mCRPC cannot afford abiraterone (US$ 2800/month) or enzalutamide (US$ 3400/month), which are more than twice as expensive as the country’s median monthly household income (US$ 1300). 51
The ACTION [Association of Southeast Asian Nations (ASEAN) Costs in Oncology] study recently revealed the degree of financial catastrophe (out-of-pocket health costs 30% of annual household income) and economic hardship (inability to make necessary household payments) experienced by cancer patients from SEA countries. 52 In Malaysia, for example, the 1-year risks of financial catastrophe and economic hardship following a cancer diagnosis were 48% and 45%, respectively.
These financial disasters were primarily attributed to medical costs for inpatient/outpatient care, as well as the purchase of drugs, medical supplies, and equipment. 52 To get through this economic downturn, 28% of affected families took out personal loans, and 60% used savings that had previously been set aside for other purposes.
The NCCN recently published PC management guidelines based on resource stratification 53 and in the SEA context. These resource-stratified guidelines could be a medium for providing management recommendations based on levels of healthcare resources, 35 and adherence to these guidelines could be further improved through a focused, clinician-centered education program at the regional level. Collaboration among various countries, particularly in the Asian and SEA region, may aid in the long run in overcoming some common challenges. 54
The approach employed in this study has some strengths and drawbacks. One of the study’s strengths is that response rates among the selected panel were relatively high, with 100% of participants completing the survey, signifying a high level of interest and engagement in the process. Another strength of this study is that experts from multiple countries and backgrounds/specialties were invited to participate, representing many points of view. This study also has a few limitations. The first is that the survey-based approach results can only be viewed as expert opinions including a small number of panelists; and there is no evidence of reliability that other expert panels would reach the same conclusions. Another limitation is that the outcome of this process has temporal validity, as it may change over time, and participant perspectives were not consistent, implying that the panel represented a range of perspectives. As a result, these findings should be evaluated rationally, and recommendations should be supported by clinical studies.
Conclusion
Based on the survey, the burden of PC has increased in the SEA countries in the last 5–10 years. Therefore, a consensus statement in the local setting is extremely valuable to increase clinician awareness of current evidence and challenges in managing patients with advanced PC. It also provides real-world guidance for clinicians and patients who are making healthcare access decisions under financial constraints. The outcomes of the consensus in the highlighted key areas need to be addressed to better serve patients, particularly in health economics and resource allocation.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231216582 – Supplemental material for Prostate cancer management in Southeast Asian countries: a survey of clinical practice patterns
Supplemental material, sj-docx-1-tam-10.1177_17588359231216582 for Prostate cancer management in Southeast Asian countries: a survey of clinical practice patterns by Edmund Chiong, Marniza Saad, Agus Rizal A.H. Hamid, Annielyn Beryl Ong-Cornel, Bannakij Lojanapiwat, Choosak Pripatnanont, Dennis Serrano, Jaime Songco, Loh Chit Sin, Lukman Hakim, Melvin Lee Kiang Chua, Nguyen Phuc Nguyen, Pham Cam Phuong, Ravi Sekhar Patnaik, Rainy Umbas and Ravindran Kanesvaran in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
