Abstract
Ewing’s sarcoma (ES) is a bone and soft tissue tumor that mainly occurs at a young age. The underlying cause of Ewing’s sarcoma is the formation of fusion proteins between FET family genes and ETS family genes. Tumors with FET/ETS fusion genes can have defects in the DNA damage response and are sensitive to PARP inhibitors (PARPi). However, several studies have shown that PARPi alone is not sufficient to induce a meaningful antitumor response and that combinations of DNA-damaging agents with PARPi are required to achieve efficacy. Accordingly, preclinical studies have reported dramatic responses to PARPi treatment in combination with DNA-damaging agents such as temozolomide or irinotecan. Similarly, it has been previously reported that by generating reactive oxygen species, high-dose intravenous vitamin C (IVC) can induce DNA damage. This suggests that the combination of IVC with PARPi may increase genotoxic stress and enhance the antitumor response. In addition, unlike chemotherapeutic agents, IVC induces DNA damage selectively in cancer cells, and the side effects are significantly milder than those of chemotherapy. As ETS fusion-positive ES is deficient in faithful DNA repair, partly due to the interaction between ETS fusion products and PARP1, a PARPi plus IVC seems to be a logical and effective combination for the treatment of ETS fusion-positive ES. This paper reports significant responses to IVC (1–1.5 g/kg) in combination with PARPi (olaparib 300 mg BID or talazoparib 1 mg/day) in two patients with metastatic Ewing’s sarcoma. The observations highlight an unmet therapeutic need for patients with advanced metastatic ES. The combination of PARPi with a selective DNA-damaging agent was effective in these cases. This case experience suggests that IVC may be incorporated into PARPi-based therapeutic strategies. Further studies are needed to confirm the efficacy of this combination in the treatment of Ewing sarcoma with ETS fusions.
Plain language summary
Ewing’s sarcoma is a type of bone and soft tissue tumor that commonly affects young people and it is often resistant to conventional therapy. In this study, clinical cancer scientists and oncologists investigated a new approach to treating this cancer by combining high-dose vitamin C with PARP inhibitors. High-dose vitamin C can damage the DNA of cancer cells and PARP inhibitors block the damaged DNA sites so they can’t be repaired and eventually this leads to cancer cells dying. The researchers found that when these two treatments were used together, there were significant improvements in two patients with advanced Ewing’s sarcoma. Importantly, the combination led to fewer side effects compared to standard chemotherapy, suggesting it might be a more tolerable treatment option. These findings suggest that combining high-dose intravenous vitamin C with PARP inhibitors could be a promising treatment for Ewing’s sarcoma. More research is needed to confirm these results, but this approach shows potential for helping patients with advanced forms of this type of cancer. This is the first clinical report demonstrating the benefits of using high-dose vitamin C with PARP inhibitors and the study emphasizes the importance of exploring more treatment options for this aggressive type of cancer and suggests that further investigations into this combined approach could lead to more effective and tolerable treatments for Ewing’s sarcoma.
Introduction
Ewing’s sarcoma (ES) is the second most common bone tumor at a young age. 1 ES is typically caused by pathognomonic fusion events between FET and ETS family genes, which are the hallmarks of the disease. 2 The most common chromosomal rearrangement is t(11;22) (q24;q12), which results in the production of the EWSR1-FLI1 fusion (80–90%); the second most common rearrangement is t(21;22)(q22;q12), which results in EWSR1-ERG (5–10%), which is considered to play a crucial role in the pathogenesis of ES through transcriptional dysregulation. 1 However, these fusion genes are currently not directly druggable, necessitating targeting of indirect vulnerabilities in these tumors. 1 Although intensive systemic chemotherapy and local control measures can increase the 5-year survival rate for patients with localized tumors to 70%, those with recurrent or metastatic ES have poor prognosis.3,4 There is no consensus on the standard of care for effective treatment of these patients, and complete responses are rare. Therefore, 5-year event-free and overall survival rates for patients with metastatic ES remain dismal at approximately 20%.5,6 Thus, targeting FET-ETS fusion-driven tumorigenesis is a crucial part of treatment strategies.
The most common chromosomal rearrangements, EWSR1-FLI and EWSR1-ERG fusions have similar clinical features and common biological properties (Figure 1). 1 The fusion products disrupt the cell’s regular transcription program, leading to upregulation of oncogenes and downregulation of tumor suppressor genes. 7 Dysregulation of transcription and chromatin remodeling by EWSR1-FLI1 and EWSR1-ERG fusions are pivotal components of ES tumorigenesis. 8
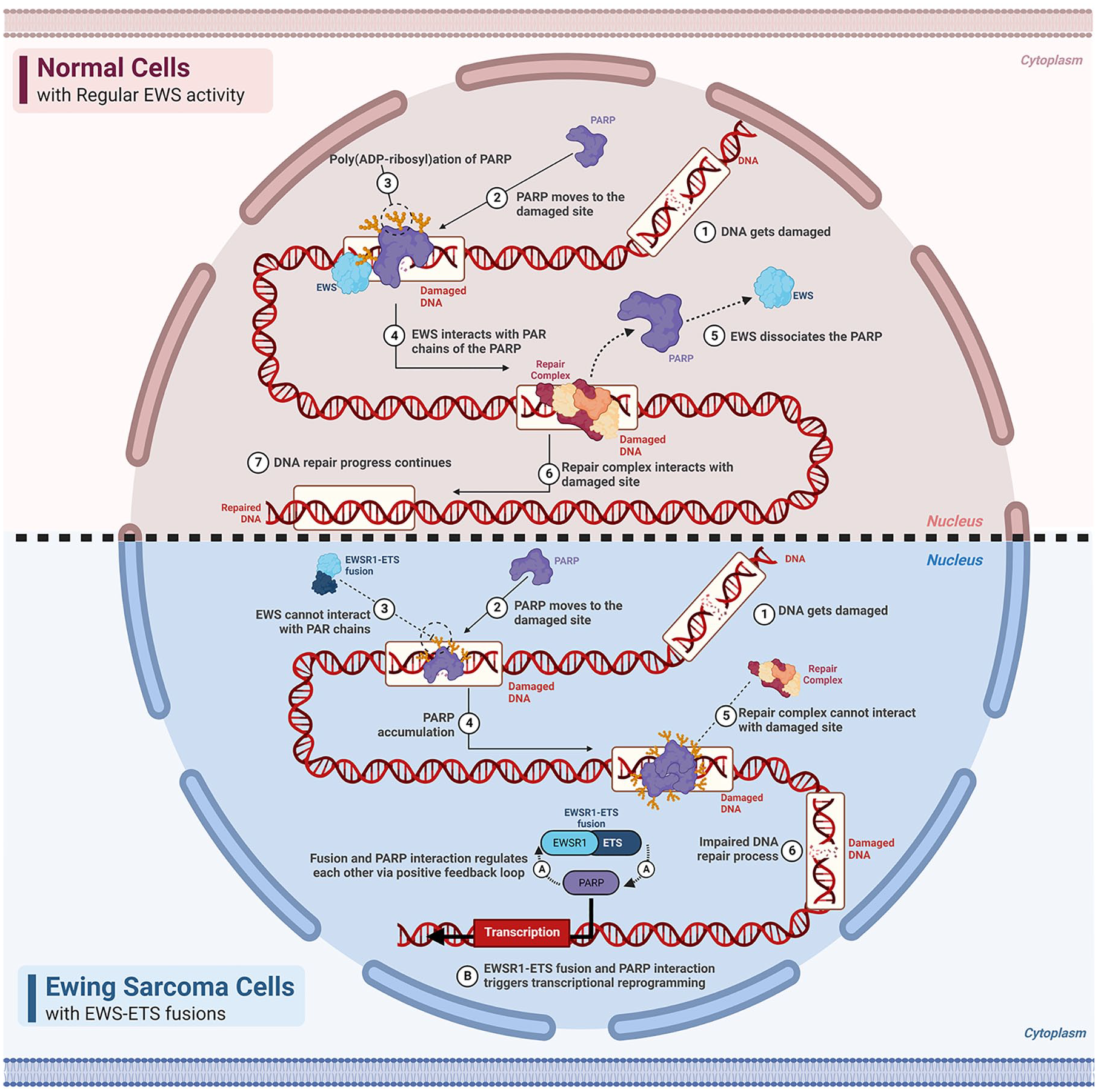
Comparison of cells containing normal EWSR1 and EWSR1-ETS fusion gene.
Poly (ADP-ribose) polymerase (PARP) is a crucial component of the DNA damage repair system, and PARP1 is the most active enzyme of the PARP family. 9 PARP is involved in DNA damage repair by activating homologous recombination (HR) and suppressing nonhomologous end joining (NHEJ). 9 Therefore, PARPi can disrupt the DNA repair mechanism in homologous recombination deficiency by trapping PARP and thus prevent the progression of cancer. 10 In 2005, two research groups discovered a synthetic lethal interaction between PARP inhibition and mutations in BRCA1 and BRCA2.11,12 In addition to disruption of chromatin remodeling and transcriptional activity by EWSR1-ETS fusion genes, ES cells exhibit deficiency in DNA repair mechanisms. 10 Mechanistically, recruitment of EWS to PARP1 regulates the dissociation of PARylated PARP1 from DNA-damaged sites, as achieved through interaction between EWS and PARP1, which promotes DNA repair in normal cells. Therefore, a lack of EWS-PARP1 interaction in Ewing’s sarcoma leads to the accumulation of PARP1 at DNA damage sites and impairs the DNA repair process. 13 On the other hand, EWSR1-ETS fusion proteins lead to an increase in PARP expression, and PARP binds to the N-terminal part of the EWSR1-ETS fusion gene and increases the transcriptional activity of the fusion protein, ultimately promoting tumorigenesis. It can be said that PARP and EWSR1-ETS fusion genes exert positive feedback on each other. 14
ES cells are deficient in DNA damage repair; yet, the antitumor efficacy of PARPi has limited efficacy in both preclinical and clinical studies. 15 Although the underlying mechanisms of low PARPi efficacy are not well understood, preclinical studies have shown that combining PARPi with genotoxic agents such as chemotherapeutics may be a promising new strategy. For instance, in a study conducted by Stewart et al., EWS cell lines were up to 1000 times more sensitive to PARP inhibitors after DNA-damaging chemotherapeutic agents were introduced. 10 However, a PARPi plus genotoxic chemotherapy may act as a double-edged sword that can produce high levels of toxicity. Because of this high risk of toxicity, their combined use is limited. An alternative way to induce genotoxic stress in cancer cells is the use of high-dose intravenous vitamin C (IVC) (Figure 2). Mechanistically, IVC has been shown to have multiple deleterious effects on cancer cells. First, by producing hydrogen peroxide (H2O2), IVC leads to the formation of ROS, resulting in the selective killing of cancer cells because noncancerous cells express the catalase enzyme that converts hydrogen peroxide to water and prevents the formation of ROS. 16 Second, IVC can increase the activity of ten eleven translocation enzyme (TET) enzymes, which leads to DNA demethylation and epigenetic reprogramming, upregulating the expression of tumor suppressors. 16 Third, IVC may inhibit hypoxia-inducible factor-1-alpha (HIF1α) activity and suppress tumor growth by increasing HIF hydroxylase, reversing the epithelial–mesenchymal transition and hindering invasion. 16

Mechanism of high-dose vitamin C and PARPi on EWS cell.
We hypothesized that the antitumor activity of PARPi can be enhanced by combining them with additional DNA-damaging agents in the treatment of ES. Due to the toxicity limitations of combined genotoxic chemotherapy with PARPi, IVC as a genotoxic stressor was used in combination with PARPi in the treatment of two ES patients in this case study. Both patients showed dramatic tumor regression with the combination treatment. Overall, this study suggests that the combination of IVC with PARPi may offer a new clinical solution for metastatic and/or refractory ES.
Methods
Two patients with stage IV recurrent progressive ES that was deemed incurable by conventional methods or who refused standard options were admitted to our clinic. Detailed evidence to support the rationale for the use of a PARPi plus IVC was presented to the patients. The patients were informed that no clinical data have proven the efficacy of single-agent PARPi or PARPi plus IVC combination therapy against ES but that preclinical data have illustrated that combinations of PARPi and DNA-damaging agents (e.g. temozolomide, irinotecan) are effective. No formal research protocol was submitted to an institutional review board, but a consent form was signed by the investigator and each patient.
Before starting treatment, the diagnosis of ES and genomic alterations were confirmed by NGS (FoundationOne® Heme). Imaging studies [positron emission tomography (PET), computed tomography (CT), and/or magnetic resonance imaging] and hematological and biochemical analyses were performed at the beginning of treatment. Furthermore, treatment response was assessed using the Response Evaluation Criteria in Solid Tumors (Version 1.1). Treatment-related toxicities were evaluated using the National Cancer Institute’s Common Terminology Criteria for Adverse Events (Version 5.0). The Eastern Cooperative Oncology Group performance status was 0 in the first patient (Case 1) and 3 in the second patient (Case 2).
This article was written using the CARE case reporting guidelines. 17
Intravenous vitamin C
According to the Riordan IVC protocol, which is the standard IVC method, the initial IVC dose was set at 15 g to evaluate tolerability. 18 High-dose intravenous vitamin C can cause serious side effects in people with renal insufficiency and G6PD deficiency. Therefore, we checked the kidney function and G6PD enzyme of both patients before starting treatment, and both had normal kidney function and normal G6PD enzyme, and the target dose was calculated to be in the range of 1–1.5 g/kg. Dose escalation was titrated up to a therapeutic range of 65–100 g per infusion. Treatment with a PARPi and IVC was started the same week, and IVC was administered 2–4 times a week. Notably, High-dose intravenous vitamin C did not increase the rate of National Cancer Institute Common Terminology Criteria for Adverse Events version 3 (CTCAEv3) grade 3 or 4 toxicities, and no treatment-emergent grade 5 toxicities were observed. 19
PARP inhibitor
The starting dose of olaparib was 300 mg bid, and that of talazoparib was 1 mg/day. Dose adjustment was performed according to the appearance of side effects. Both patients started olaparib (initial assessment responses were obtained with olaparib), and the patient in Case 1 continued treatment with talazoparib (a second response was obtained in this patient, this change was due to its easy access).
Results
Case presentations
Demographic data, genomic variations, and the therapy schedule for the two patients are presented in Table 1.
Patient characteristics.

FISH, fluorescence in situ hybridization; IVC, intravenous vitamin C; NGS, next-generation sequencing; PARP, poly (ADP-ribose) polymerase; VAC/IE, vincristine sulfate, Adriamycin and cyclophosphamide followed by ifosfamide and etoposide alternating regimen.
means truncating mutation.
Case 1
A 19-year-old female patient was admitted to our clinic with left-sided chest pain and effort dyspnea. Her medical history included ES which appeared 7 years prior in the left distal femur. She received four cycles of the vincristine, adriamycin, cyclophosphamide, and ifosfamide and etoposide alternating regimen (VAC/IE protocol) as a neoadjuvant treatment followed by complete tumor resection. After the operation, the same regimen was administered to the patient for 13 more cycles. Eventually, lung metastasis was detected in 2015, and metastasectomy was performed. Subsequently, the patient refused further chemotherapy. In 2017, lung metastasis was detected at the same site, and the tumor was also operable. After complete resection, oral cyclophosphamide and complementary therapies were administered. In February 2019, a gross lung mass was detected again. Surgery was performed with near complete resection. Because of operative complications, chemotherapy was resumed in April 2019. The patient came to our clinic for a second opinion and was re-evaluated with CT and PET/CT, which revealed a lung mass larger than the previously resected lesion. Broad genomic profiling (Foundation One Heme) was performed, which revealed two alterations: EWSR1-ERG fusion and FANCD2 truncation at exon 17. Olaparib 300 mg bid and IVC (1.5 g/kg, body weight 51 kg, 75 g/day) were administered 2–3 times a week on consecutive days. After 2 weeks, symptomatic improvement in effort dyspnea and pain was observed. After 2 months, a response evaluation using CT and PET revealed a dramatic response [Figure 3(a) and (b)]. Then, she returned to her previous oncology center, which suggested that she should continue olaparib therapy only and that an IVC was unnecessary. Consequently, the patient stopped IVC therapy. Almost 3 months later, this patient returned to our clinic with chest pain. PET/CT revealed tumor progression [Figure 3(c)]. After reassessment, IVC therapy was again recommended, with a detailed explanation of the molecular mechanisms. After the resumption of IVC therapy, therapeutic response was achieved again [Figure 3(d)]. This result shows that a PARPi alone is insufficient for treatment.

Imaging studies in Case 1 during treatment. (a) Positron emission tomography/computed tomography at presentation (April – 2019). (b) Two months of treatment with olaparib plus IVC (75 g/day, 2–3 times a week; July – 2019). (c) Progression after continuing single-agent olaparib without IVC (November – 2019). (d) Retreatment with talazoparib plus IVC. Once again, a response was achieved, although it was not as strong as the first time (February – 2020).
Because of the COVID-19 pandemic, while the patient’s father died due to COVID-19 so the patient’s further treatment (PARP inhibitor plus high-dose intravenous vitamin C) was interrupted and progression (new bone metastases) was detected in May 2020. Radiation therapy (to eliminate bone metastases) plus PARPi combination therapy was planned. However, the patient remained progression-free for approximately 5 months (February–July 2020) after receiving high-dose vitamin C again (rechallenge) in addition to the PARP inhibitor. Finally, she died in July 2020.
Case 2
A 26-year-old female patient was admitted to our emergency department with severe dyspnea and abdominal pain that first appeared approximately 6 months previously. Physical examination and ultrasonography revealed massive ascites, a huge mass, and peritoneal carcinomatosis. Because she had a prior history of ES, this patient consulted a medical oncology clinic. After large-volume paracentesis and supportive care, imaging was performed. CT and PET revealed a gross abdominal mass approximately 30 cm in diameter and diffuse peritoneal involvement. Renal ES was diagnosed, and the VAC/IE protocol was implemented. After four cycles, the patient’s symptoms improved. Because of her improved condition, she had the belief that she was cured and refused chemotherapy because of its side effects. After 2 months, abdominal distention and pain reappeared. The VAC/IE protocol was restarted. After two cycles of chemotherapy, she experienced grade 3–4 hematologic and infectious complications, and therapy was discontinued. She was admitted to the emergency clinic of our hospital. The diagnosis and mutation status (FoundationOne®Heme) were re-evaluated, and ES with EWSR1-FLI1 and CPS1 alterations were confirmed. After providing informed consent, the patient received olaparib 300 mg bid. IVC was administered four consecutive days a week (1.5 g/kg, body weight 50 kg, 75 g/day). Four days after starting therapy, the patient’s uric acid, lactate dehydrogenase, and potassium levels increased, and allopurinol treatment and urinary alkalinization were initiated. The evaluation indicated that the tumor had responded to treatment. The patient’s uric acid and potassium levels returned to normal during the follow-up. Dramatic clinical and radiological responses were achieved 1 month later (Figure 4). After 2 months of therapy, the patient was admitted to the emergency department with sudden-onset abdominal pain and hypotension. CT revealed a suspicious intestinal perforation. The patient was admitted to the intensive care unit but subsequently developed septic shock and died.

Case 2 imaging studies during treatment. Initial 18 F-fluorodeoxyglucose positron emission tomography/computed tomography (CT) (a and b) for staging revealed solid-cystic bulky tumors (white arrows) with an extremely high maximum standardized uptake value extending to the retroperitoneum in the right upper quadrant of the abdomen. After treatment, follow-up CT (c) revealed marked shrinkage of the tumor (yellow arrows), which was considered a partial response.
It is worth mentioning that since the patient could not be operated on, we could not prove intestinal perforation, but the clinical findings were consistent with intestinal perforation. The intestinal perforation is believed to be associated with diffuse peritoneal sarcomatosis and intestinal structural disorder, rather than the administration of high-dose intravenous vitamin C. This assumption is supported by the pharmacokinetic data. Moreover, it is noteworthy that the patient did not receive high-dose vitamin C for 1 week during the period when the intestinal perforation occurred. Notably, no existing publication has reported any association between high doses of vitamin C and intestinal perforation to date. In the phase III VITALITY trial in colorectal cancer, no instances of intestinal perforation related to high-dose vitamin C were reported. 20
Discussion
Metastatic and/or recurrent ES remains a challenge, with limited therapeutic options for patients, poor prognosis, and lack of effective standard of care. In this case report, we demonstrate dramatic responses to the combination of PARPi with IVC in two patients with metastatic and refractory ES. Olaparib (300 mg bid) or talazoparib (1 mg/day) plus high-dose vitamin C therapy was well tolerated in both patients. No dose reductions were required in either patient. Renal function and electrolyte levels (Na, Cl, K, Ca, Mg) were assessed twice weekly. Hypomagnesemia and hypokalemia were the most common adverse events, and additional replacement therapy was administered. The FANCD2 mutation was found in the comprehensive genomic profile of one of the patients (Case 1). As the exon 17 truncation in FANCD2 has not been characterized, its function is unknown. FANCD2 is a component of DNA damage repair by HR, and loss of FANCD2 function has been shown to sensitize tumor cells to PARP inhibition.21,22 Monoubiquitination of FANCD2 at S561, encoded by exon 27, is essential for its function in the DNA damage response and is unlikely to be directly affected by exon 17 truncation. Moreover, there are no known splice sites in FANCD2 exon 17; thus, it is possible that the transcription of FANCD2 is intact. Alterations that occur in exon 17 (aa516-552) of FANCD2 have not been associated with pathogenicity. In fact, the majority of them have been classified as benign in ClinVar. To the best of our knowledge, there is no clinical evidence demonstrating the sensitivity of FANCD2-altered cancer patients to PARP inhibitors. Therefore, although we cannot completely rule out the potential impact of FANCD2 alteration on patient response, it is unlikely to be the main determinant of sensitivity to PARP inhibitors.
ES cells are sensitive to DNA-damaging agents. This is partly due to the accumulation of PARP1 at lesions, as ES cells lack the canonical EWSR1–PARP1 interaction that mediates the dissociation of PARP1 from DNA-damaged sites. 13 Furthermore, Gorthi et al. showed that EWSR1 inhibits the phosphorylation of RNA polymerase 2 and prevents R-loop formation. 23 In ES cells, phosphorylation of RNA polymerase 2 is not inhibited due to the EWSR1-FLI fusion protein, which results in R-loop accumulation. 23 Taken together, these data suggest that the presence of EWSR1-ETS fusions sensitizes ES cells to genotoxic agents mainly by reducing the rate of PARP1 dissociation from DNA and by promoting R-loop formation.13,23 Moreover, the formation of R-loops is associated with BRCAness phenocopy. 23 Therefore, PARP inhibitors should have a synergistic effect as well. In addition, the EWS-FLI1 fusion protein inhibits HR by disrupting the interaction between BRCA1 and BARD1 by binding to BARD1, thereby enabling the tumor to acquire the ‘BRCAness’ phenotype. 24 In fact, DNA damage repair defects may be caused by the EWSR1-ETS fusion gene itself in ES. Briefly, PARP1 appears to be a convergence point of multiple DNA damage and repair pathways, making it an attractive therapeutic target in ES. 14 Iniguez et al. showed that the small molecules THZ1 and THZ531 when used in combination with PARPi in ES have a synergistic effect on each other without apparent toxicity because these small molecules inhibit CDK12 and thus can cause HR deficiency. 25 IVC causes cell death by reducing the expression levels of homologous recombination and non-homologous end joining-associated proteins. 26 A preclinical study showed that ovarian cancer cells with wild-type BRCA did not respond to olaparib treatment; however, with the addition of IVC, a significant tumor reduction was observed due to the downregulation of BRCA1/2 and RAD51 genes and generation of ROS. 26 Choy et al. designed a phase II study to evaluate the efficacy of single-agent olaparib in patients with pretreated metastatic ES. No objective response was observed, and the best outcome recorded in the study was stable disease. 15 This result was not surprising, as preclinical studies have shown that olaparib alone is not sufficient to achieve an objective response. Furthermore, we had a similar experience with one of our patients (Case 1), in whom a significant response was observed when the combination of a PARPi and IVC was initiated. However, this patient had progression when the IVC was discontinued and the PARPi alone was in place.
Subsequently, the PARPi was used again in combination with IVC, and a response was achieved again. Brenner et al. also found in mouse xenografts that ES cells (RD-ES) treated with olaparib continued to grow, though at a significantly slower rate than untreated controls. The combination of olaparib and temozolomide was shown in the same study to induce dramatic tumor responses and a fairly pronounced durable complete response. 27 The most important finding was from the preclinical study by Stewart et al., which demonstrated that a PARPi alone is not sufficient to induce cytotoxicity. Statistically significant responses were observed in mice treated with irinotecan and/or temozolomide combinations, and complete and durable responses were achieved in more than 80% of mice with no tumor recurrence up to 12 weeks after discontinuation of therapy. 10 However, this strategy has a narrow therapeutic window in patients because the synergistic response achieved by PARPi is not selective for tumor cells. Instead, PARPi disrupts an important mechanism of DNA repair in normal cells and exacerbates the side effects of chemotherapy, such as myelosuppression. 28 Although lower-dose and intermittent PARPi administration strategies have been attempted to avoid hematologic toxicity, they are not usually tolerable. 29 In addition, the inhibitory effect of PARPi is short-lived and lost within a week, necessitating their continuous use to achieve long-term PARP inhibition and clinical efficacy. 30 As indicated by preclinical data, the combination of a PARPi with chemotherapy appears to be a novel and potentially therapeutically promising strategy for ES. Overall, these therapies appear to require continuous treatment with combinations of a full-dose PARPi and selective cytotoxic drugs such as IVC.
Yun et al. found that KRAS- and BRAF-mutant colon cancer cells can overexpress the glucose transporter GLUT1, through which DHA (oxidized form of vitamin C) can pass and be converted to ascorbate in the cells. As a result, ROS are generated, which accumulate and inactivate GAPDH inside the cells, leading to an energy crisis and cell death, which does not occur in KRAS and BRAF wild-type cells. These results suggest that IVC is cytotoxic rather than cytostatic. 31 In another study, Lv et al. demonstrated that ascorbate has antitumor effects on hepatocellular carcinoma and liver cancer stem cells. 32 Another important finding from preclinical studies is that IVC induces epigenetic reprogramming. Cimmino et al. found that IVC increases the cytotoxicity of PARP inhibition in TET2-deficient tumor cells. 33 However, no data have been reported on the efficacy of IVC as a single agent against ES. In another study, Schoenfeld et al. demonstrated that the addition of IVC to standard chemotherapy and radiotherapy is safe and that patients receiving IVC have improved response rates and survival. 34 In addition, Demiray has shown in a case series study that the combination of PARPi and IVC is well tolerated and improves outcomes. 35 The phase III VITALITY clinical trial also showed that KRAS-mutant colorectal cancer patients may benefit from high-dose vitamin C. 20 Our previous case series and observation of the two patients described here demonstrate that the use of integrative cancer therapies that follow the principles of personalized or molecular-based approaches can make a difference in the lives of patients.
The major limitation of this combination is the inability to determine the treatment regimen regarding how to dose and how often the IVC can be used. Weekly 2–4 IVC infusions in the outpatient setting may not be sustainable for patients in the long term, as this condition reduces their quality of life and therapy compliance. A high frequency (3–4 per week) of IVC infusions can be maintained until a good response is achieved. After achieving this response, the frequency of IVC infusions can be decreased, and other DNA-damaging agents (oral chemotherapeutics, etc.) can be added to improve therapy compliance. In our two patients, a good response was achieved during the first 4–8 weeks of treatment. To increase the effectiveness of IVC during treatment, non-toxic fasting mimic diets can be adopted by patients, as previously shown by Di Tano and colleagues. 36
Conclusion
Overall, we achieved remarkable responses in these two patients. Although PARPi may have a role in the treatment of ES, they do not induce sufficient anti-tumor response on their own. Accordingly, we need additional DNA-damaging agents to support PARPi activity. IVC can be used as a joker due to its cancer-specific DNA-damaging properties. Recent reports found that IVC was safe and well tolerated.31,32,33,35 The cancer specificity of IVC enables its combination with radiotherapy, chemotherapy, immunotherapy, and tyrosine kinase inhibitors. To our knowledge, this is the first clinical case report that demonstrates the benefit of the combination of IVC and PARPi in the treatment of ES. Further studies that evaluate IVC as a DNA damaging agent in combination with PARPi should be conducted.
Footnotes
Acknowledgements
We would like to thank Prof. Dr. Razelle Kurzrock (Medical College of Wisconsin) for her insights, Dr. Metin Çevener (Medicana International Istanbul Hospital) for the radiological reevaluation and image selection. We also thank Dr. Orhan Çömlek (General Manager of Teknopol Istanbul) and Prof. Dr. Cevdet Erdöl (President of Sağlık Bilimleri University) for providing us with vitamin C.
