Abstract
A 71-year-old non-smoker woman was admitted to our hospital because of left front chest pain. A computed tomography scan showed a large mass of >7.0 cm in the lower left part of the lung and multiple organ metastases in the liver, brain, bone, and left adrenal gland. Pathological analysis of a resected specimen obtained by bronchoscopy revealed keratinization. In addition, p40 was positive and thyroid transcription factor-1, synaptophysin, CD56, and chromogranin A were negative by immunohistochemistry. Programmed cell death ligand 1 expression was 1%–10%, and exon 19 deletion was detected. We diagnosed the patient with stage IVB lung squamous cell carcinoma and administered osimertinib. Osimertinib was later replaced with afatinib because of grade 3 skin rash. Overall, the size of the cancer was decreased. Furthermore, her symptoms, laboratory data, and computer tomographic findings markedly improved. In summary, we experienced a case of epidermal growth factor receptor–positive lung squamous cell carcinoma that was responsive to epidermal growth factor receptor tyrosine kinase inhibitors.
Keywords
Introduction
Epidermal growth factor receptor (EGFR) mutations are uncommon in lung squamous cell carcinoma (LSCC), and several researchers reported that the effectiveness of epidermal growth factor receptor–tyrosine kinase inhibitors (EGFR-TKIs) is poor in EGFR mutation–positive LSCC.1,2 Nevertheless, the Japanese guidelines recommend kinase inhibitors for LSCC patients with driver oncogenes. 3 A comparative study has not been conducted using EGFR-TKIs in EGFR-mutated LSCC patients because the frequency is reported to be less than 5%. 4 The frequency of EGFR mutations in LSCC is 3.3% in Caucasians and 4.6% in Asians, unlike lung adenocarcinoma, which occurs in 19.2% of Caucasians and 47.9% of Asians. 5 However, subgroup data from prospective trials and pooled analyses reported an overall response rate (ORR) of 9.1%, 2 while several retrospective analyses reported ORRs of 25%–32% and median progression-free survival of 1.4–3.9 months. On the basis of this evidence, it appears that kinase inhibitors may be successful, albeit with limited efficacy, in LSCC patients with driver oncogenes.1,2,6 In addition, never-smoking patients have more frequent EGFR mutations than ever-smoking patients. 7 We experienced a case of EGFR-positive LSCC that was responsive to EGFR-TKIs.
Case
A 71-year-old woman was admitted to our hospital because of left front chest pain. She was a non-smoker and had no medical history. Physical findings were low-grade fever, a non-painful bulge on her right back, and tenderness in the left lower part of her chest. Her Eastern Cooperative Oncology Group performance status was 1, and she was a thin woman; her height was 159.0 cm, her body weight was 41.0 kg, and her body mass index was 16.2. There were no weight-loss periods. The concentration of serum cytokeratin-19 fragments (CYFRA) was 32.3 ng/mL, higher than the normal upper limit, whereas serum carcinoembryonic antigen (CEA) and sialyl Lewis X were within normal levels. A computed tomography scan showed a large mass of >7.0 cm in the lower left part of the lung (cT4), and multiple enlarged lymph nodes were observed in the left hilar region and around the left bronchus, and the left mediastinal lymph node was prominent (cN2). Multiple organ metastases to the liver, brain, bone (both ribs, thoracic spine, right ilium, and left pubis), and left adrenal gland were also evident. Furthermore, brain magnetic resonance imaging showed multiple brain metastases with punctate, nodular, and ring-shaped contrast effects, and some lesions were accompanied by changes that appeared to be edema in the surrounding area (cM1c). Pathological analysis of a resected specimen obtained by bronchoscopy revealed a keratinizing tendency; in addition, p40 was positive, and thyroid transcription factor-1 (TTF-1), synaptophysin, CD56, and chromogranin A were all negative by immunohistochemistry (Figure 1). The tumor proportion score for the expression of programmed cell death ligand 1 (PD-L1), which was determined using the anti-PD-L1 antibody clone 22C3 (BML, Tokyo, Japan), was 1%–10%; however, exon 19 deletion was detected in an outsourced genetic analysis by next-generation sequencing (Oncomine multi-CDx FFPE; BML, Tokyo, Japan). This was pre-determined panel and also revealed negativity for BRAF mutations, ALK fusion genes, and ROS1 mutations. The duration of this analysis was estimated to be 11–14 days, in contrast to a lung cancer multi-gene polymerase chain reaction (PCR) panel test, which took 4–7 days at a municipal hospital. We diagnosed the LSCC stage IVB tumor as cT4cN2cM1c (liver, brain, bone, and adrenal gland). She was initially fit, but her activities of daily living gradually declined because of pain associated with bone metastasis after hospitalization. We requested that an orthopedic team reduce the pressure caused by Th8 metastasis to the spinal canal, and a posterior thoracic fusion was performed. Furthermore, the patient underwent whole-brain radiotherapy for the multiple brain metastases and stereotactic radiotherapy for the multiple bone metastases. At the same time, she was administered osimertinib 80 mg once daily. After 27 days, osimertinib was withdrawn because of common terminology criteria for adverse events (CTCAE) version (v) 5.0 8 grade 3 skin rash, and erythema multiforme with pruritus was observed mainly on the trunk. We prescribed oral minocycline and fexofenadine and applied clobetasol propionate ointment. We changed osimertinib to afatinib 40 mg once daily, as directed by a dermatologist. As a result, these skin rashes disappeared. Nevertheless, CTCAE v 5.0 grade 3 diarrhea occurred after 8 days and afatinib treatment was halted; consequently, we decreased the dose to 20 mg once daily. The size of the primary lesion was reduced from 7 cm to 0 cm, while one of the liver metastases was reduced from 2.8 cm to 0.8 cm in 8 months. Overall, the EGFR-TKIs decreased the size of the cancer alongside stereotactic radiotherapy for the multiple bone metastases (Figure 2). Furthermore, the patient’s symptoms, laboratory data, and computer tomographic findings markedly improved. She attained partial remission for approximately 1 year.
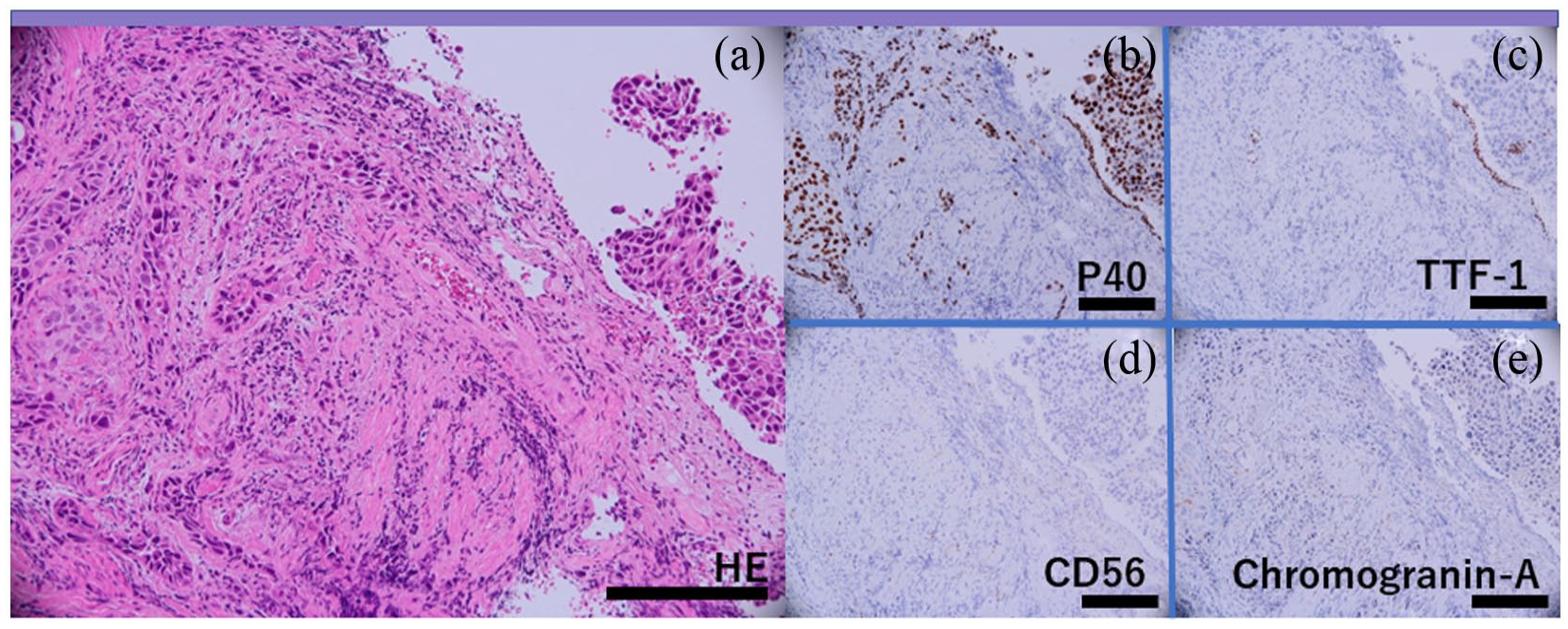
Histology of transbronchial biopsy specimen. (a) Hematoxylin–eosin staining, (b) P40, (c) TTF-1, (d) CD56, and (e) chromogranin A staining by immunohistochemistry. Scale bars, 200 µm.

Clinical course. Black solid box, terms of radiotherapy; striped box, terms of EGFR-TKIs; Mo, months; CYFRA 21-1, serum cytokeratin-19 fragments.
Discussion
The positive rate of EGFR mutations in LSCC is lower than that in lung adenocarcinoma.9,10 EGFR-mutant LSCC and lung adenocarcinoma patients have a similar objective response rate, but LSCC patients have a lower disease control rate and significantly shorter median progression-free survival and median overall survival (OS). 11 EGFR-TKIs are less effective in EGFR-mutant LSCC than in lung adenocarcinoma, but there is still a clinical benefit for LSCC patients. Fukuoka et al. 12 reported that non-smoking women with histological adenocarcinoma better responded to gefitinib. Moreover, Huang et al. 7 reported that both non-smokers and women with LSCC might have more EGFR mutations than smokers with LSCC. In fact, EGFR mutation-positive LSCC is most common in women and non-smokers.13,14 However, Shoji et al. 15 reported that even a man with a 45-pack-year smoking history had partial responses to osimertinib. Thus, it is important to identify which LSCC patients are EGFR-positive and whether they can benefit from EGFR-TKIs. Although different from this case, we should also consider the mixed pattern of EGFR-mutated adenocarcinoma cells in LSCC cases, and we might choose EGFR-TKIs as first-line treatment if patients have the following factors: female sex, never-smoker, high CEA serum levels, and pathologically TTF-1-positive. Conversely, male sex, current smoker, high CYFRA serum levels, and pathological p63 positivity indicate that platinum doublets and/or immune checkpoint inhibitors (ICIs) are necessary. 16 In addition, OS was significantly longer in non-smokers or in those with exon 19 deletion among 639 LSCC patients. 9 Progression-free survival was significantly longer in patients aged ⩾65 years than in younger patients aged <65 years. 17 This was reflected in the characteristics of the current patient who achieved a good result with a partial remission. Although other immunotherapeutic options might also have suppressed her cancer because her PD-L1 expression was 1%–10%, 18 we may expand the treatment options and improve mortality if EGFR mutations are confirmed in non-smoking female LSCC patients. ICIs are effective as second-line therapy for EGFR mutation-positive lung cancer if PD-L1 expression is high. 19 The relevance of ICI efficacy and the rate of expression of PD-L1 in EGFR-positive patients are unclear; however, EGFR- and anaplastic-lymphoma-kinase-positive patients with ⩾25% of tumor cells exhibiting PD-L1 expression seemed to be recovered with durvalumab treatment. 20 Common EGFR mutations have been discussed, but we should also consider patients with minor EGFR mutations. EGFR-TKIs are reported to be less effective in patients with minor mutations, such as G719X. 21 Accumulating evidence for EGFR major/minor mutation-positive LSCC might shape therapeutic strategies in the future.
Conclusion
The first-line strategy worldwide for LSCC is currently a combination of chemotherapy and immunotherapy. However, a search for EGFR mutations in LSCC patients might broaden treatment options and improve prognosis, although a positive rate of EGFR mutations is low in these patients. Treatment with an EGFR-TKI could be expected following the assessment of clinical and pathological findings in some patients.
Footnotes
Acknowledgements
Author contributions
S.M. and S.A. designed the study, N.K., M.M., and H.O. collected the data, S.M. and S.A. analyzed the data, and S.M. and S.A. prepared the manuscript. Y.K. diagnosed and prepared the pathological images. Y.I. and N.H. supervised and validated this study. All authors approved the final version for submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
