Abstract
The identification of Epidermal Growth Factor Receptor (EGFR) mutations in lung adenocarcinoma has facilitated the development of personalized medicine based on oncogenic drivers. EGFR-Tyrosine Kinase Inhibitors (TKIs) are part of the targeted therapy; they impede the phosphorylation of the intracellular tyrosine kinase component of EGFR and consequently block signal transduction pathways. These drugs inhibit the proliferation and survival of tumor cells, leading to long-term progression-free survival and overall survival. Diarrhea is one of the most frequent adverse events associated with EGFR-TKIs, affecting at least 18% of patients and reaching up to 95% in some cases. Diarrhea should be managed carefully given its association with important complications, treatment interruptions, and dose reductions. Moreover, nutritional status and quality of life (QoL) can deteriorate due to severe diarrhea. Changes in diet, such as increment of fiber, supplementation with glutamine, and use of probiotics, may contribute to a decrease in the incidence of diarrhea. Improving the control of diarrhea can provide a significant benefit to the QoL of patients.
Introduction
Lung cancer (LC) is one of the most lethal cancers in the world. Non-small cell lung cancer (NSCLC) is the most frequent, accounting for 80% of cases. 1 Lung adenocarcinoma (LUAD) is the main subtype of NSCLC, which harbors a unique molecular profile and has allowed treatment to move from traditional chemotherapy toward personalized medicine. 2 Targeted therapy has been proven to be effective in patients with advanced NSCLC and oncogenic drivers2,3 The identification of Endothelial Growth Factor Receptor (EGFR) mutations, such as in-frame deletions of the amino acids LREA (leucine, arginine, glutamic acid, and alanine) within exon 19 and the L858R substitution mutation, has changed the landscape of NSCLC. 4 They are detected overall in 10–20% of Caucasian patients, although the frequency is higher in East Asia (around 45%) as well as in Latin American countries. The latter has reported somatic EGFR mutations ranging from 14.4% in Argentina to 24.7–34.3% in Mexico and Colombia, and up to 51.1% in Peru2,3,5–7
EGFR mutations are among the most widely studied signal transduction networks and are known to promote cancer cell proliferation and tumor invasion. 8 Among the current targeted therapies, small-molecule tyrosine kinase inhibitors (TKIs) have been developed to treat different solid tumors, including breast cancer, head and neck cancer, renal cell carcinoma, gastrointestinal stromal tumor, and NSCLC. Most of them work by binding to the intracellular adenosine triphosphate domain of tyrosine kinase, preventing downstream signaling and subsequent cell division and growth. 9 EGFR-TKIs have been developed as part of targeted therapy and, overall, they impede the phosphorylation of the intracellular tyrosine kinase component of EGFR and consequently block the signal transduction pathways. These effects affect the proliferation and survival of tumor cells.2,10 Furthermore, patients with LUAD harboring EGFR mutations are sensitive to EGFR-TKIs, whose benefits have been proven as monotherapy. They can lead to long-term progression-free survival (PFS) and overall survival. 4 Of note, specific EGFR polymorphisms including −216G > T and variable cytosine-adenine (CA repeat have been associated with the outcome of TKI-based NSCLC treatment.11,12 Interethnic differences in the distribution of EGFR polymorphisms and mutations can also affect the response to treatment. 7 To date, no predictive biomarkers for the efficacy and toxicity of EGFR-TKIs in the treatment of NSCLC have been identified. 13 Potential biomarkers are needed to identify patients who will be long responders and will have the greatest benefit with treatment.
TKIs have been developed since 2001 and their use has continued to expand rapidly. 14 Three generations of EGFR-TKIs have been developed as first- or second-line therapies to treat advanced LUAD with EGFR mutations. The first (gefitinib, icotinib, and erlotinib), second (afatinib and dacomitinib), and third-generation (osimertinib, aumolertinib, lazertinib, and furmonertinib) agents are generally better tolerated, can be used for longer periods of time, and the method of administration is more convenient when compared with chemotherapy, and there is strong evidence to support improvement in quality of life (QoL).15,16 However, regardless of the chosen EGFR-TKI, the safety profile remains a major concern, and severe adverse effects have limited their clinical application. 14 Besides dermatological adverse effects, diarrhea is the most common side effect associated with TKI’s and it could be severe enough to require dose reduction or discontinuation of treatment. 17
Diarrhea induced by EGFR-TKIs can be an important dose-limiting toxicity and cause an increase in therapeutic costs. 18 Overall, cancer therapy-induced diarrhea is associated with significant economic loss. Recent reports suggest that it may incur additional costs of up to $25,000 (USD) per cycle. These costs are attributed to a high risk of infection, longer hospital stay, and the need for supportive care measures.18,19 It is crucial to consider that EGFR-TKI therapy is often administered for longer periods than chemotherapy, so persistent low-grade diarrhea can remarkably affect the nutritional status and QoL of patients. Effective and proactive strategies focusing on management based on the understanding and assessment of diarrhea induced by EGFR-TKIs should result in improved adherence, response rate, survival, and quality of life (QoL). In addition, it could reduce costs related to complications secondary to diarrhea. 20 The objectives of this review are to provide an overview of the appropriate management of diarrhea based on the mechanisms involved in the pathogenesis induced by EGFR-TKIs.
Diarrhea induced by EGFR-TKIs
EGFR-TKI-associated diarrhea can lead to severe diarrhea, and if not managed in a timely manner, it can lead to severe complications, dose reduction, and treatment interruption. Medical oncologists may underestimate mild diarrhea while tolerable; however, treatment lasts several months or even years, and mild toxicity causes a quantifiable deterioration in the QoL. 17 The main mechanism related to TKI diarrhea described in the literature is the excessive secretion of chloride, which promotes nutritional deficiencies, dehydration, fatigue, kidney failure, and a higher risk of infection. 17 Diarrhea can affect social function and performance status, induce fatigue and sleep disturbances, and cause the person to become housebound owing to embarrassment and discomfort. Additional information should be considered (personal history, biochemical evaluation, comorbidities, nutritional status, diet, and symptoms). The Common Terminology Criteria of Adverse Events guidelines are commonly used to determine the severity of diarrhea. Diarrhea can be classified as grade 1 (mild) if the patient has less than four stools, grade 2 (moderate) if they have four–six stools, grade 3 (severe) if more than seven stools over baseline, immediate attention is required, and grade 4 is considered life-threatening and urgent intervention is needed. Grade 5 refers to death 20 (Supplemental Table 1).
Pathophysiology of diarrhea induced by EGFR-TKIs
Some possible mechanisms for EGFR-TKI-induced diarrhea have been elucidated, but the definite pathogenesis is yet to be elucidated 21 (Figure 1)

Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitors (EGFR-TKIs)-induced diarrhea may be caused by different mechanisms, though, none has been proven clinically. (1). EGFR-TKIs may induce diarrhea by secretory mechanisms resulting in dysfunction in water homeostasis in the colon. When EGFR receptors are no longer able to negatively control chloride secretion, this could result in constant deposit of chloride in the lumen and subsequently, lead to water accumulation (16–19). (2) The inhibition of EGFR could also favor changes in the intestinal architecture leading to a reduction in the absorption of nutrients and electrolytes in the gut (6, 17, 18, 20, 21). (3) Several factors such as bowel dysfunction, pelvic radiotherapy (RT), gastrointestinal (GI) surgery, diet, and antibiotics may induce a shift in the composition of the gut microbiota. Commensal bacteria decrease and pathogenic species increase, favoring a pro-inflammatory state. Mutations in cystic fibrosis transmembrane conductance regulator can also lead to changes in the microbiota (8, 13, 14, 17, 21–24).
Secretory mechanisms
The pharmacokinetics of TKIs may not be precisely associated with diarrhea, according to previous studies, suggesting that the origin could be predominantly luminal. 8 Epithelial cells in the gastrointestinal tract express EGFR, specifically at the basolateral membranes, which maintain the negative regulation of chloride secretion. 21 The mechanisms of chloride secretion involves several membrane transport pathways. 22 The basolateral membrane potassium channels favor chloride ion secretion by activating the Na+/K+ pump, and they enter the cell through the sodium, potassium, and chloride cotransporters. Moreover, calcium-activated chloride channels (CaCCs) contribute to chloride secretion, which also participate in the activation of cystic fibrosis transmembrane conductance regulator (CFTR) and Sk4 calcium-activated potassium channels. After chloride ions accumulate in the lumen through these mechanisms, they provide the electrochemical driving force for the paracellular movement of sodium. The presence of sodium chloride in the lumen then provides an osmotic gradient for water to pass to the latter. Any breakdown in this pathway, such as an episode of diarrhea, disrupts the homeostasis of the system and can lead to several complications (Figure 1); Therefore, EGFR-TKIs may induce diarrhea by secretory mechanisms, resulting in dysfunction of water homeostasis in the colon. When ErbB receptors are no longer able to negatively control chloride secretion, a constant deposit of chloride occurs in the lumen, which subsequently leads to water accumulation (secretory diarrhea). In ex vivo models, it has been suggested that the primary mechanism of this alteration is the activation of basolateral membrane potassium and apical membrane chloride channels in intestinal epithelial cells. Murine models have reported that a daily dose of EGFR-TKIs induces severe diarrhea, whereas blocking CaCC activation effectively decreases intestinal fluid secretion levels and alleviates diarrhea. 23
Histopathological changes: Impaired epithelium generation and barrier leakage
Another prevailing hypothesis is that the inhibition of EGFR signaling diminishes the capacity of epithelial cells to heal, as well as their growth and proliferation in the gastrointestinal tract, leading to direct mucosal and villi atrophy. 8 EGFR pathways have stimulatory effects on enterocyte proliferation and electrolyte transport. When altered, structural and functional changes may occur. Changes in intestinal architecture can contribute to atrophy and reduction in gut absorption (nutrient and electrolyte transport). 22 In a preclinical study, dacomitinib led to severe histopathological injury localized in the ileum, which coincided with increased levels of monocyte chemoattractant protein-1. The damage was characterized by severe villus atrophy, and in the congested lamina propria, there was more inflammatory infiltrate. 24 Dacomitinib did not cause direct cytotoxicity, which supports previous studies that suggest that EGFR-TKI-induced diarrhea does not induce direct cell death. 8 Similarly, in two mouse models that evaluated gefitinib and erlotinib, there was significant atrophy of the small-intestinal wall and a decreased absorptive surface area. 25 The latter has not been proven completely with osimertinib; animal models have not exhibited significant intestinal histopathologic damage, suggesting a possible mechanism of diarrhea independent of intestinal tissue damage, such as lapatinib-induced diarrhea.8,23
Barrier dysfunction: Endoplasmic reticulum stress-mediated injury
As observed in preclinical models, EGFR-TKIs promote endoplasmic reticulum stress. Erlotinib can cause barrier dysfunction in small-intestinal epithelial cells by increasing their permeability and diminishing E-cadherin expression. It also induced endoplasmic reticulum stress in a concentration-dependent manner. In parallel, gefitinib and icotinib arrested the cell cycle at the G0/G1 phase by increasing cyclin D1 and p27 in the same intestinal cells. They trigger the endoplasmic reticulum stress response by activating the protein kinase R-like ER kinase pathway. These findings suggest that endoplasmic reticulum stress-mediated injury may contribute to TKI-induced diarrhea.18,26
Dysfunction of efflux transporters
Most EGFR-TKIs show an affinity for ATP-binding cassette, subfamily B (ABCB) efflux transporters, which, if abnormal, could lead to dysregulation of the cellular availability of these drugs. In turn, there can be greater accumulation in the intestine, leading to gastrointestinal toxicity.
27
Polymorphisms in efflux transporters might affect their clearance, which might cause excessively high plasma concentrations and a potential increase in adverse events.
28
Inflammation and gut microbiota
In this context, inflammation and gut microbiota may also play a role in the development of diarrhea. Given its role as a mediator of intestinal inflammation, the gut microbiome may play a key role. The microbiome has been proposed to mediate inflammatory responses through the innate immune system. 13
Recently, it has been recognized that pathway alterations can be associated with the remodeling effects of TKIs, including compositional shifts of the gut microbiota. Microbial metabolites promote alterations in host physiology. Erlotinib, sorafenib, sunitinib, and anlotinib influence microbiota-related pathways, including altered metabolites, such as bile acids, indoles (e.g. hydroxyindoleacetic acid, indole, indoleacrylic acid, serotonin, and indolelactic acid), and polyamines (e.g. spermidine). These metabolite alterations may increase susceptibility to adverse effects of TKIs, such as diarrhea. 14 Dysbiosis of the gut microbiome can alter the levels of metabolites such as butyrate, which can induce regulatory T cells that maintain immune tolerance. 17 However, changes promoted by the gut microbiota associated with TKI’s continue to be exploratory.
Risk factors associated with diarrhea induced by EGFR-TKIs
Retrospective studies have identified particular characteristics that make patients more susceptible to severe diarrhea, especially with afatinib. These include low weight (<50 kg), low body surface area, low body mass index (BMI), low hemoglobin, female sex, malnourishment, decreased performance status, and baseline kidney impairment. 29 The product information for afatinib also suggests that female sex, low body weight, and kidney failure are associated with a higher exposure to afatinib, which may lead to higher toxicity. 30 Other factors, such as genetic landscape, antibiotic history, nutritional status, comorbidities, and concurrent therapies, should be considered as risk factors for the development of diarrhea as well. (Figure 2).
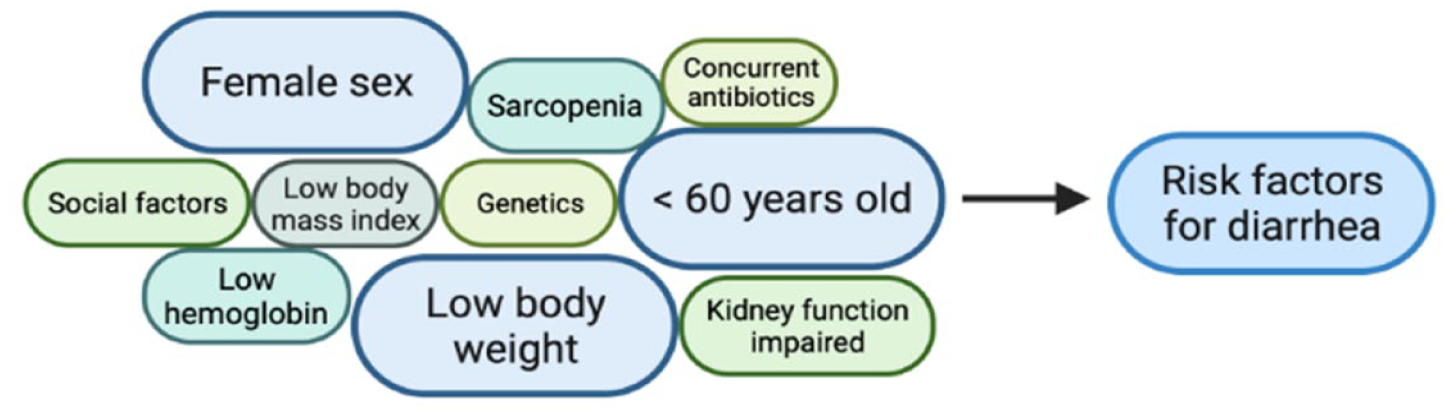
Risk factors associated with a high risk of developing diarrhea induced by Tyrosine Kinase Inhibitors (TKIs) that could help to select the patients that may benefit from adjustments in the dose of the TKI before starting treatment (29–36).
In this regard, patients with cancer have important nutritional alterations in 25–70% of cases, which directly affect many spheres of patient care, including QoL, treatment toxicity, and survival outcomes. 31 Moreover, sarcopenia is highly prevalent among patients with LC, with a prevalence of approximately 45%, and has been associated with dismal outcomes regardless of the stage of disease or type of treatment. 32 Sarcopenia is an important risk factor for severe gastrointestinal toxicity. Early recognition of a deficient nutritional status can help prevent cancer cachexia and identify patients at potential risk of serious AE.29,33
A large study evaluated predictors of severe diarrhea pretreatment by pooling data from afatinib clinical trials. It included 1151 patients, of whom 16% had severe diarrhea. The incidence of severe diarrhea has been reported to be between 6% and 37% among the studies. The factors included were age, sex, functional status, weight, BMI, estimated glomerular filtration rate, and hemoglobin level. Multivariate logistic regression analysis identified that female sex, low body weight (less than 45 kg), and older age (>60 years) were significant independent predictors of severe diarrhea (p < 0.01). Interestingly, a simplified risk score was constructed that included these three major factors. 34 Prospective evidence is warranted to identify patients at high risk of developing severe diarrhea, and an individualized approach is currently recommended.
High plasma concentrations of afatinib have been associated with an increased incidence of severe diarrhea. Afatinib concentrations of AUC0–24> 823.5 ng·h/mL and C0 > 28.5 ng/mL could be used as cutoff values for the incidence of grade 2 diarrhea 34 A second study reported that a cutoff value of 21.4 ng/mL for the trough plasma concentration of afatinib was useful for earlier prediction of severe adverse effects such as diarrhea, oral mucositis, and rash. 35 Monitoring trough plasma concentrations could be effective in reducing the risk of diarrhea.
Patients treated with antibiotics while using EGFR-TKIs had a higher incidence of grade 3–4 diarrhea (15.9% versus 1.7%, p = 0.008), probably related to their alteration on long-term microbial shifts, and had a poor PFS (10.1 months versus 6.6 months, p = 0.02).36–38
Genetic polymorphisms and pathogenic variants have been associated with a significantly higher concentration of EGFR-TKIs in the plasma, which would explain the higher incidence of AE, including severe diarrhea, in carrier patients. Genetic polymorphisms in the Janus Kinase-Signal Transducer and Activator of Transcription (JAK-STAT) pathway have been studied. STAT3 rs1053023, rs1053005, and STAT6 rs324011 may be potential predictors of AE severity in NSCLC patients treated with EGFR-TKI. 39 An association between polymorphisms in −216G > T and −191C > A and grade >1 diarrhea has also been reported (p < 0.01). 39 However, ethnic variability in EGFR-TKI pharmacokinetic determinants has the potential to result in interethnic differences in toxicity, as previously described in the literature. For example, East Asians are more susceptible to erlotinib- and gefitinib-related AE than Europeans. The ABCG2 421C>A allelic variant correlated with gefitinib-induced grade >2 diarrhea (p = 0.0046) and erlotinib-induced diarrhea (p = 0.035). It was also associated with higher gefitinib plasma concentration at steady state, which represents an independent risk factor for diarrhea (p = 0.006). This variant is more prevalent in East Asians and contributes to toxicity. In addition, African Americans have higher erlotinib clearance and lower erlotinib systemic exposure than other races, which correlates with a lower incidence of AE. 40 Recently, a systematic review and meta-analysis examined the association between EGFR polymorphisms and TKI-associated toxicity. Data associated with diarrhea from rs11568315 were analyzed, but the difference was not significant (p = 0.661). 41
Bacterial overgrowth can also be a risk factor for diarrhea. It can be promoted by previous radiotherapy, colectomy, or altered gut motility derived from concomitant therapies 42 and medications (prokinetic agents, laxatives, proton pump inhibitors, trimebutine, and acid reducers or comorbidities such as irritable bowel syndrome, inflammatory bowel disease, and gastrointestinal surgery).43,44
Incidence of diarrhea according to type of EGFR-TKIs and concomitant therapy
The incidence of diarrhea is highly variable among the three generations of EGFR-TKIs currently available. The prevalence of diarrhea varies from 1 in every 3 patients to 1 in every 1045–63 (Table 1), and so does the onset and duration. 21 Diarrhea can occur as early as 2 days after starting EGFR-TKIs and may persist for more than 7 days, often resulting in therapy interruption or discontinuation. This depends on the patient’s characteristics, type of EGFR-TKI, and whether it is used as a monotherapy or in combination with other therapies. 64
CT, chemotherapy; LL, lux-lung; ND, not determined; NNH, number needed to harm (all-grade diarrhea); NR, not reported; OD, once daily.
First-generation EGFR-TKIs are more selective than afatinib or dacomitinib. Diarrhea presented later and was less severe. Diarrhea with first-generation TKIs develops in less than 10% of patients and appears around day 13 (7–18 days) with erlotinib and day 18 (10–19 days) with gefitinib.45,47,50,51,56,58,60,64,65 Dacomitinib and afatinib are associated with a higher incidence of diarrhea because they have a broad spectrum of activity, blocking several members of the ErbB family. 64 Afatinib may confer the highest rate of all-grade diarrhea (up to 95%) among EGFR-TKIs and typically occurs during the first week of treatment in 50–62% of patients and in 71% by day 14.17,54,55,65–67 Dacomitinib is also associated with a higher incidence of all-grade diarrhea (87%) and its appearance is usually within the first week of treatment. 68 Third-generation EGFR-TKIs exhibit greater affinity and resistance to mutations than wild-type EGFR. This higher selectivity for mutated EGFR may be associated with a decreased incidence of toxicities compared with the first and second generations.16,46,52,53,57,61,62,69
Combination strategies with first-generation EGFR-TKI have consistently shown no difference in the rate of diarrhea. Two randomized trials explored first-generation EGFR-TKIs administered in combination with platinum-based chemotherapy in the first-line setting and did not find an increase in the incidence of all-grade diarrhea, ranging from 35.3 to 42%.59,63 Moreover, the inhibition of vascular endothelial growth factor receptor plus EGFR inhibitor showed similar rates of diarrhea ranging from 47% to 63%, and less than 7% had grade 3 diarrhea.70,71
Impact of diarrhea on EGFR-TKIs treatment
Diarrhea is a dose-limiting toxicity for second-generation EGFR-TKIs, and its severity is dose dependent. According to clinical trials, the proportion of patients requiring dose reduction of 40 mg is 28–53%. The standard recommended dose of afatinib is 40 mg/day and can be reduced by 10 mg decrements to a minimum of 20 mg if not tolerated.54,67,72 This information is in line with real-world studies, which report that more than half of the patients need dose modification due to AE. 73 In a recent meta-analysis that included 24 RCTs (16 phase III and 8 phase II), it was found that the use of EGFR-TKIs increased the risk of all-grade diarrhea (RR 3.45; 95% CI, 2.94–4.06; p < 0.00001) and high-grade diarrhea (RR 8.22;95% CI, 6.02–11.23; p < 0.00001). Afatinib was associated with the highest risk of all-grade (RR = 9.42) and high-grade (RR = 66.67) diarrhea, whereas gefitinib had the lowest risk of both (RR = 2.99 and 3.15, respectively). 74 A second meta-analysis showed that 40% of patients who received first- and second-generation EGFR-TKIs experienced grade 3–4 AE. The risk of these events was lower for gefitinib (29.1%) than for erlotinib (54.1%) or afatinib (42.1%). 75
Due to the high rate of AE associated with second-generation EGFR-TKIs, different strategies have been explored to improve tolerance without affecting outcomes. Dose reduction according to tolerability, even before starting treatment, has been studied with excellent results. Post hoc analysis of two RCTs confirmed that the efficacy of second-generation EGFR-TKIs after dose reduction due to toxicity was equivalent to that of standard doses. In 2016, a post hoc analysis of the LUX-LUNG (LL) three and six trials reported that dose reductions occurred in 53.3% and 28% of patients in each of the studies. Most cases (>80%) occurred within the first 6 months of treatment. Dose reductions occurred more frequently in females and in those with a lower body weight (<50 kg). PFS was similar in patients whose dose was reduced during the first 6 months versus those who did not [LL3:11.3 versus 11 months (hazard ratio (HR) 1.25); LL6:12.3 versus 11 months (HR 1.00)]. They concluded that tolerability-guided dose adjustment of afatinib reduced the incidence and severity of AE without affecting the treatment efficacy. 76 A subsequent analysis of ARCHER 1050 showed that dose reductions due to toxicity occurred in 150 patients (66.1%) taking dacomitinib at 45 mg/day. In total, 87 patients (38.3%) were reduced to 30 mg and 63 patients (27.8%) were further reduced to 15 mg. Female patients required greater dose reductions, and the median weight was lower in the dose reduction group. Median PFS was similar between groups (14.7 versus 16.6 months). Notably, the incidence of grade 3 diarrhea decreased from 11.3% to 4% after the dose reduction. This analysis also supports that dose modifications helped manage toxicity without the need for permanent discontinuation in most patients while maintaining efficacy. 77 Tolerability-guided dose modification is a feasible strategy that may enable patients to continue treatment without affecting their effectiveness.
Dose reduction in real-world studies
Real-world data available on afatinib were also consistent with those of clinical trials. Observational studies have reported that different doses of afatinib do not affect its effectiveness.29,78–90 (Table 2) A meta-analysis that included 12 real-world studies (n = 1290 patients) evaluated the effectiveness and safety of afatinib dose reduction in patients with NSCLC. Overall, there were no differences in the PFS between the two dose schedules of afatinib, except in the study by Tan et al. 87 that included a cohort of patients with stage IV disease with brain metastases (BM), who had a better PFS with 40 mg of afatinib. In addition, the objective response rates reported in four of these studies were similar between the two regimens. These results consistently suggest that the 30- and 40-mg afatinib regimens could be similar without affecting the PFS in patients without BM, and lower doses could help reduce the rate of AE, including diarrhea. Nonetheless, no data or label indication currently supports the commencement of afatinib at a dose lower than 40 mg/day; thus, identifying patients at a high risk of AE and assessing the possibility of starting treatment at a lower dose, followed by a close evaluation of tolerability, could improve a patient’s QoL and reduce the need for treatment suspension.
Reduced-dose afatinib: 20–30 mg per day. Full-dose afatinib: 40 mg per day; NS: no significant; NR: not reported. Reduced-dose erlotinib: <100 mg per day, Full-dose erlotinib: 150 mg per day.
BM, brain metastases; CI, confidence interval; HR, hazard ratio; PFSm, median progression-free survival; TTF, time to treatment failure.
Recommendations and general management of diarrhea induced by EGFR-TKIs
EGFR-TKI-induced diarrhea should include pharmacological and nonpharmacological strategies to control bowel movements, assess the response to therapy, and rule out organic causes of diarrhea that can require urgent intervention and increase costs.21,91,92 In 2004, consensus-based guidelines recommend classifying diarrhea into two groups: uncomplicated (without risk factors) or complicated (does not respond to initial treatment after 48 h or has additional risk). 93 Patients without risk factors should receive nonpharmacological measures and a standard dose of oral loperamide (initial dose 4 mg followed by 2 mg after each loose stool or every 4 h, maximum dose a day of 16 mg) can be used as needed. Patients who progress to severe diarrhea might require hospital admission and additional treatment. Intravenous hydration and appropriate multidisciplinary therapy should be initiated as soon as possible.21,91,92,94 (Figure 3, Supplemental Table 2, and Figure 1).

Algorithm of management according to the severity of diarrhea (21, 91, 92).
Antidiarrheal medications
The severity of symptoms dictates specific recommendations regarding EGFR-TKI modifications, the dose of loperamide, and how to prescribe it. 21 It is very important to instruct patients to record the number of stools, characteristics, and symptoms, if present. Loperamide is the agent of choice because it has local activity in the gut and is not related to systemic effects (minimum absorption, only 0.3% systemic bioavailability). It acts as an agonist of opioid receptors in the intestinal wall, reducing gut motility by inhibiting the release of acetylcholine and limiting ion transport. The standard dose of loperamide for mild to moderate (grade 1 or 2) uncomplicated diarrhea is an initial 4 mg dose, followed by 2 mg every 4 h or 2 mg after each loose stool (maximum daily dose 16 mg per day). After 24–48 h if diarrhea persists, a high dose of loperamide and/or adding a second agent should be considered.21,91,92 A high-dose loperamide regimen (initial dose of 4 mg, followed by 2 mg every 2 h; maximum daily dose 16 mg) is preferred for patients with grade 3–4 diarrhea and persistent grade 1–2 diarrhea or those complicated by symptoms. Additional agents can be considered including racecadotril, which is an enkephalinase inhibitor proven effective in secretory diarrhea by blocking epithelial cyclic adenosine monophosphate (AMP)-mediated secretion, and has no central nervous system effects21,95, somatostatin analogs such as octreotide. The latter is favored in this context because, in animal and clinical studies, octreotide prolongs intestinal transit time and decreases the loss of fluids and electrolytes. The starting dose is 100–150 mcg subcutaneously or intravenously three times a day, and the dose can be titrated up to 500 mg three times a day or selection of continuous infusion may be an option (25–50 mcg/h). 92
Other agents that can be used in this scenario include budesonide, which is a topically active steroid with a 90% first-pass effect in the liver, and therefore, with low systemic availability. The recommended dose is 9 mg once daily for 3–5 days. 96 Bile acid sequestrants (cholestyramine, colestipol, and colesevelam) may also help in the management of refractory diarrhea. Unabsorbed bile salts can cause diarrhea by stimulating colonic secretion and motility. If used, the diet should be adjusted to a low-fat diet to be a more effective and safe strategy. 91
It is not recommended to take antidiarrheal medication prophylactically because of constipation, which can also be complicated and exacerbated if patients are already under other medications such as opioids for pain control. 91 Glutamine, celecoxib, probiotics, activated charcoal, and absorbents have been suggested for the treatment of chemotherapy-induced diarrhea, but there is a lack of evidence regarding their efficacy in this context, especially related to diarrhea. 92
Nonpharmacologic support
Hydration and diet are crucial, and patients should drink 8–10 glasses of liquid daily (approximately more than 2 L per day) and reduce or avoid lactose products, high-fat foods, high-insoluble fiber, alcohol, and high-osmolar supplements. A stringent diet is intended to manage digestive symptoms by easily metabolizing and having the necessary energy intake, as well as food with a high requirement of minerals and vitamins that should help retain fluids. Patients were reassessed every 12–24 h to determine the follow-up. It is essential for patients to understand the high frequency of diarrhea, its possible implications, and the main treatment.92,94
QoL of the patients who are receiving EGFR-TKIs
QoL is an important treatment outcome because of the high incidence and mortality of LC. Several studies have demonstrated that patients complain about physical and psychological symptoms; therefore, it is necessary to evaluate not only the medical outcomes but also the impact on QoL. Patients treated with EGFR-TKIs have reported an improvement and a longer time to deterioration in their QoL and symptoms than patients treated with chemotherapy. 97 According to real-world studies, survivors of EGFR-TKI therapy present a clinically relevant high symptom burden for diarrhea and rash compared to survivors treated with non-TKI therapy. These results are in accordance with the findings of previous RTCs. 98 Therefore, optimal management is essential, and not less important than the treatment of chemotherapy-related AEs.30,98 Tolerability-guided dose modifications may enable patients to continue treatment without an impact on effectiveness.
Conclusion
Diarrhea, an AE of cancer treatment, can have negative effects on performance status, social impact, and the ability to complete therapy. In NSCLC, the development of EGFR-TKIs has improved clinical outcomes. Nonetheless, diarrhea induced by EGFR-TKIs is a well-known adverse event and dose-limiting, especially for second-generation agents. Diarrhea management is a crucial intervention, even before the start of treatment. Nonpharmacologic and pharmacologic strategies should be implemented on time to prevent complications, drug discontinuation, and deterioration of the QoL. A special focus on high-risk patients is relevant for minimizing and optimally controlling AE.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231192396 – Supplemental material for Management of diarrhea induced by EGFR-TKIs in advanced lung adenocarcinoma
Supplemental material, sj-docx-1-tam-10.1177_17588359231192396 for Management of diarrhea induced by EGFR-TKIs in advanced lung adenocarcinoma by Daniela Cárdenas-Fernández, Pamela Soberanis Pina, Jenny G. Turcott, Norberto Chávez-Tapia, Emilio Conde-Flores, Andrés F. Cardona and Oscar Arrieta in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-png-2-tam-10.1177_17588359231192396 – Supplemental material for Management of diarrhea induced by EGFR-TKIs in advanced lung adenocarcinoma
Supplemental material, sj-png-2-tam-10.1177_17588359231192396 for Management of diarrhea induced by EGFR-TKIs in advanced lung adenocarcinoma by Daniela Cárdenas-Fernández, Pamela Soberanis Pina, Jenny G. Turcott, Norberto Chávez-Tapia, Emilio Conde-Flores, Andrés F. Cardona and Oscar Arrieta in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


