Abstract
Background:
The exosome-focused translational research for afatinib (EXTRA) study is the first trial to identify novel predictive biomarkers for longer treatment efficacy of afatinib in patients with epidermal growth factor receptor (EGFR) mutation-positive nonsmall cell lung cancer (NSCLC) via a comprehensive association study using genomic, proteomic, epigenomic, and metabolomic analyses.
Objectives:
We report details of the clinical portion prior to omics analyses.
Design:
A prospective, single-arm, observational study was conducted using afatinib 40 mg/day as an initial dose in untreated patients with EGFR mutation-positive NSCLC. Dose reduction to 20 mg every other day was allowed.
Methods:
Progression-free survival (PFS), overall survival (OS), and adverse events (AEs) were evaluated.
Results:
A total of 103 patients (median age 70 years, range 42–88 years) were enrolled from 21 institutions in Japan between February 2017 and March 2018. After a median follow-up of 35.0 months, 21% remained on afatinib treatment, whereas 9% had discontinued treatment because of AEs. The median PFS was 18.4 months, with a 3-year PFS rate of 23.3%. The median afatinib treatment duration in patients with final doses of 40 (n = 27), 30 (n = 23), and 20 mg/day (n = 35), and 20 mg every other day (n = 18) were 13.4, 15.4, 18.8, and 18.3 months, respectively. The median OS was not reached, with a 3-year OS rate of 58.5%. The median OS in patients who did (n = 25) and did not (n = 78) receive osimertinib during the entire course of treatment were 42.4 months and not reached, respectively (p = 0.654).
Conclusions:
As the largest prospective study in Japan, this study confirmed favorable OS following first-line afatinib in patients with EGFR mutation-positive NSCLC in a real-world setting. Further analysis of the EXTRA study is expected to identify novel predictive biomarkers for afatinib.
Trial registration:
UMIN-CTR identifier (UMIN000024935, https://center6.umin.ac.jp/cgi-open-bin/ctr/ctr_his_list.cgi?recptno=R000028688
Introduction
Recent advances in epidermal growth factor receptor (EGFR)-targeted therapy for nonsmall cell lung cancer (NSCLC) have improved survival in precision medicine. As a result, three generations of EGFR tyrosine kinase inhibitors (TKIs) have been approved in Japan as first-line treatments for patients with EGFR mutation-positive NSCLC: first-generation reversible TKIs (erlotinib and gefitinib), second-generation irreversible TKIs (afatinib and dacomitinib), and third-generation mutant-selective TKIs (osimertinib). 1 The FLAURA phase III study recently demonstrated significantly prolonged overall survival (OS) in patients with EGFR mutation-positive NSCLC treated with first-line osimertinib (n = 279) compared with first-generation EGFR–TKIs [gefitinib (n = 183) or erlotinib (n = 94)]. 2 Osimertinib has thus been established as the standard treatment for previously untreated common EGFR mutation-positive NSCLC. However, its efficacy in the FLAURA study was not definitive, with hazard ratios (HRs) for OS of 1.00 [95% confidence interval (CI): 0.75–1.32] and 1.00 (95% CI: 0.71–1.40) in Asian and EGFR L858R-mutated patients, respectively, suggesting limited benefit of osimertinib over first-generation EGFR–TKIs in these subgroups. 2 Considering that race and EGFR mutation subtypes were just two stratified factors in the randomization of the FLAURA study, the results could indicate that these two factors had independent negative impacts on the clinical benefit of osimertinib.
Regarding first-line afatinib, the LUX-Lung 7 phase IIb study showed a trend toward better OS in patients treated with afatinib (n = 160) compared with gefitinib (n = 159). 3 Furthermore, the retrospective Gio-Tag study, which included real-world clinical patients treated with first-line afatinib followed by second-line osimertinib, found that the median duration of sequential afatinib and osimertinib treatment was 37.1 months, and the median OS was 44.8 months in Asian patients (n = 50), compared with 27.6 and 36.7 months, respectively, in non-Asians (n = 137). 4 Similarly, the Up-SwinG study, which had a similar study design, found a median duration for sequential afatinib and osimertinib treatment of 28.8 months and median OS of 42.3 months in Asian patients (n = 118), compared with 25.5 and 31.3 months, respectively, in non-Asians (n = 73). 5 These data thus indicated that first-line afatinib followed by second-line osimertinib might prolong the total duration of EGFR–TKI therapy, especially in Asian patients, thus improving OS. However, the efficacy of first-line afatinib, like other EGFR–TKIs, varies, with some patients benefiting from long-term efficacy while others do not. There is thus a need to identify biomarkers for afatinib efficacy in actual clinical settings.6,7
As previously reported, the EXTRA (EXosome-focused Translational Research for Afatinib) study protocol aims to explore novel biomarkers for afatinib efficacy by matching data from multi-omics analyses of peripheral blood samples in patients treated with first-line afatinib to clinical efficacy data (Supplemental Figure S1). 8 We conducted a prognostic survey and locked the clinical data 3 years after the final enrollment in this trial, and then started to carry out proteomic, genomic, metabolomic, and epigenomic analyses. Prior to the publication of the results of these four omics analyses, the current study aimed to analyze the clinical efficacy data based on first-line afatinib treatment. This report may be considered to reflect the latest real-world data on first-line afatinib, which has been used in Japan since 2014.
Patients and methods
Study design
The EXTRA study was designed as a prospective, single-arm, observational study to identify novel predictive biomarkers associated with longer OS in patients treated with first-line afatinib, via comprehensive genomic, proteomic, epigenomic, and metabolomic association analyses using serial peripheral blood samples (free molecules in serum/plasma and exosome-packaged molecules) (Supplemental Figure S1). 8 We planned to enroll 60 patients as the discovery cohort and 40 patients as the independent validation cohort.
Patient eligibility
The main inclusion criteria for registration were: age ⩾20 years; histologically or cytologically confirmed metastatic or locally advanced NSCLC; EGFR mutation (common or uncommon); Eastern Cooperative Oncology Group performance status (PS) of 0 or 1; adequate bone marrow, renal, and liver functions; and chemotherapy-naive. The main exclusion criteria were: interstitial pneumonia or pulmonary fibrosis; active infection or uncontrolled disease; and other active malignant disease.
Study treatment
Enrolled patients were initially treated with afatinib 40 mg/day, and the dose was adjusted according to toxicities observed by the investigators. Patients who developed drug-related grade ⩾2 adverse events (AEs) temporarily discontinued afatinib until recovery to grade 1, and then resumed afatinib treatment with a 10 mg dose reduction. The dose could be reduced by a further 10 mg in patients who developed drug-related grade ⩾2 AEs again despite the initial dose decrease. A total of three dose reductions were allowed, with a minimum dose of afatinib of 20 mg every other day.
Treatment was discontinued in patients who developed afatinib-induced grade ⩾1 interstitial lung disease and in patients who required a fourth dose reduction of afatinib. Treatment was continued until disease progression, unacceptable toxicity, or withdrawal of consent by the patient.
Assessment
Tumor response was assessed by thoracoabdominal and head computed tomography or head magnetic resonance imaging. Tumor assessment was performed every 8 weeks for the first 24 weeks of treatment and every 12 weeks thereafter until progressive disease (PD), treatment discontinuation, withdrawal of consent, or death, with the date of treatment initiation defined as the reference date. The tumor response was evaluated according to RECIST, version 1.1.
AEs were classified by the Medical Dictionary for Regulatory Activities, and their severities were assessed according to the Common Terminology Criteria for Adverse Events, version 4.0.
Statistical analysis
The primary endpoint was the identification of novel predictive biomarkers of afatinib efficacy associated with longer OS. The secondary endpoints were the following clinical indicators to be matched with the generated omics data: objective response rate (ORR), disease control rate (DCR), progression-free survival (PFS), OS, and AEs.
ORR was defined as the percentage of patients who had a complete or partial radiological response. DCR was defined as the percentage of patients who had a complete or partial radiological response, or stable disease. PFS was defined as the time from each registration to confirmation of PD or death from any cause. OS was defined as the time from the registration to death from any cause.
The 95% CIs for the proportions of ORR and DCR were calculated using the Clopper–Pearson method. Median PFS and OS and their 95% CIs were estimated using the Kaplan–Meier method. Between-group comparisons were performed using log-rank tests. The analyses were carried out using SAS ver. 9.4 (SAS Institute Inc., Cary, NC, USA).
Ethics
This study complied with all the principles in the Declaration of Helsinki (as revised in 2013), and was approved by the Ethical Review Board for Medical and Health Research Involving Human Subjects at Teikyo University (Approval No. 16-066). All enrolled patients provided written informed consent. This trial was registered with the University Hospital Medical Information Network clinical trial registry (No. UMIN000024935).
Results
Patient characteristics
A total of 103 patients (60 patients in the discovery cohort, 43 patients in the validation cohort) were enrolled from 21 institutions in Japan between February 25, 2017, and March 30, 2018. The patient characteristics are shown in Table 1. The median age was 70 years, with 32% aged ⩾75 years, 74% were female, and 50% were PS 1. About a quarter of patients (27%) had postsurgery recurrence, about a fifth (22%) had brain metastasis before afatinib treatment, and all patients (100%) had adenocarcinoma. Most patients (90%) had common EGFR mutations (exon 19 deletion and exon 21 L858R), and the other 10% had uncommon EGFR mutations.
Patient characteristics.
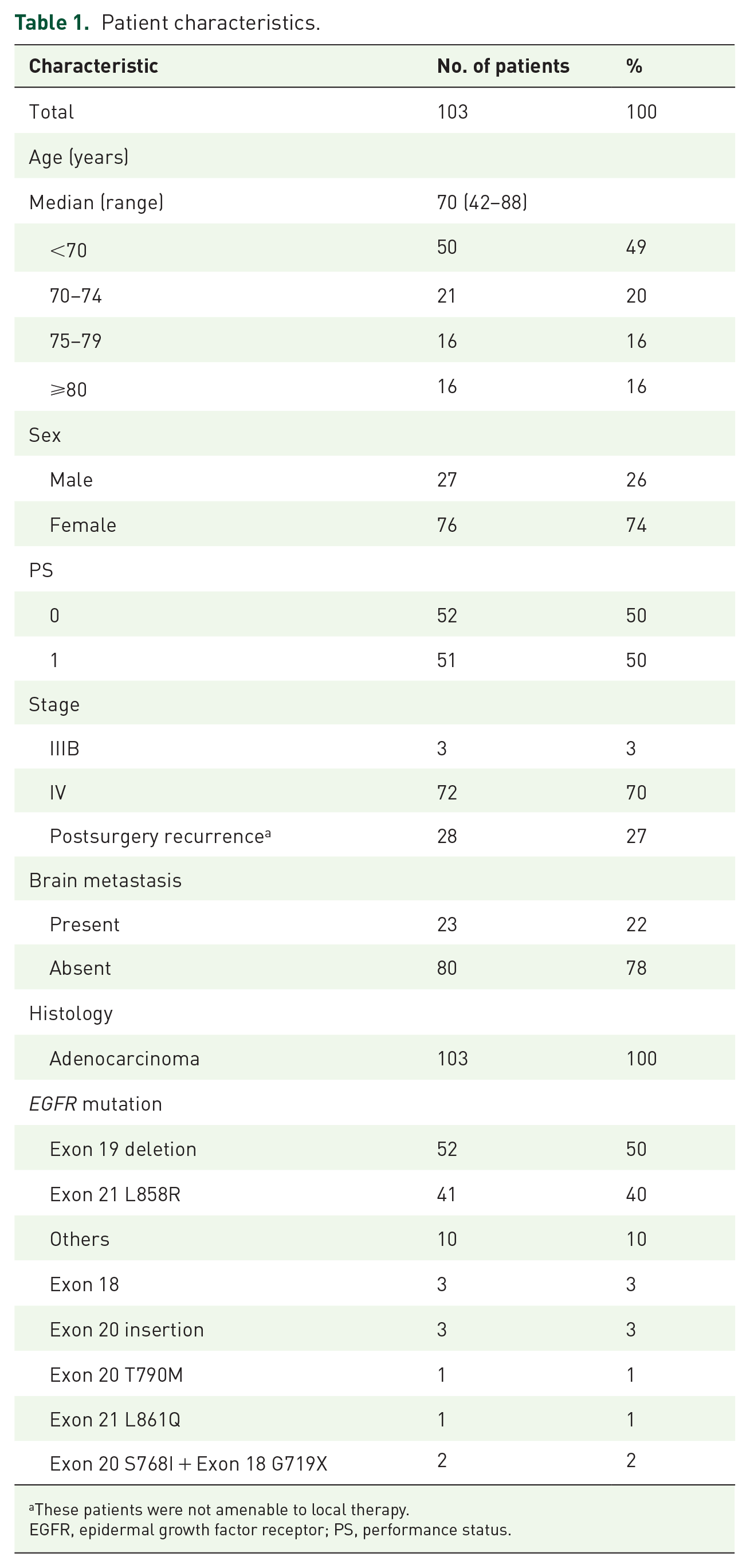
These patients were not amenable to local therapy.
EGFR, epidermal growth factor receptor; PS, performance status.
Patient flow
Patient flow is summarized in Figure 1. Among all 103 enrolled patients, treatment was discontinued in 81 patients after a median follow-up of 35.0 months (range: 0.5–44.4). The reasons for discontinuation were: PD in 70 patients (68%); AEs in nine patients (9%); and physician’s decision in two patients (2%), including cognitive impairment in one patient and new onset of thyroid cancer in one patient. A total of 22 patients (21%) finally remained on afatinib treatment.
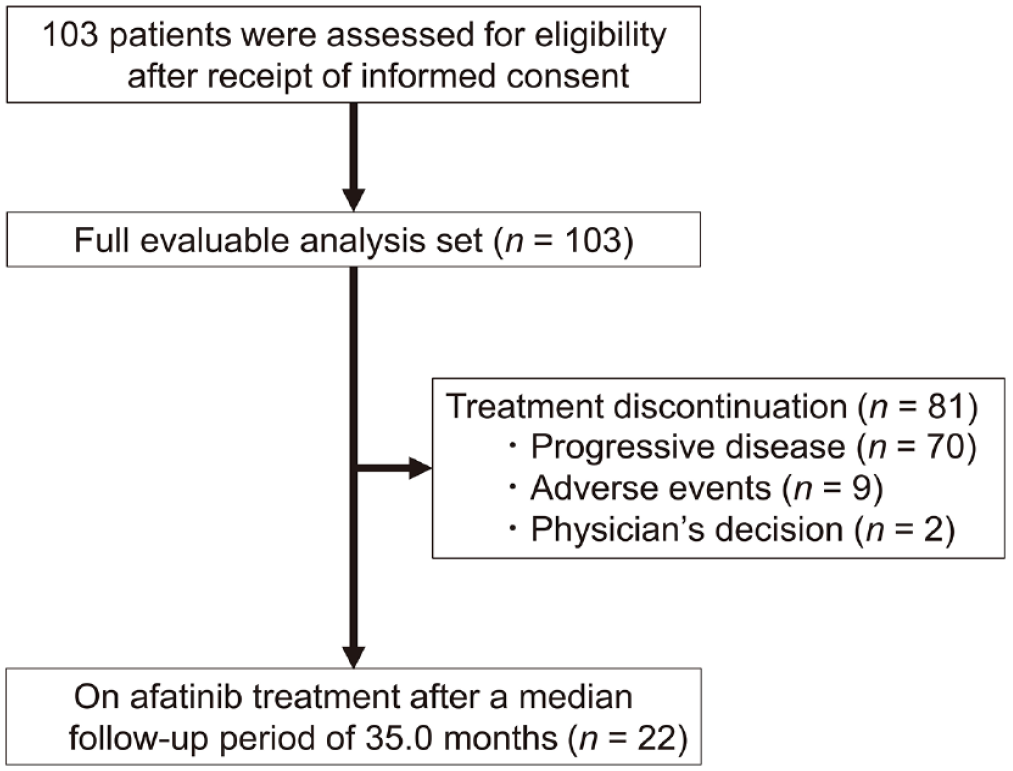
Patient flow in the EXTRA study.
Treatment efficacy
Tumor responses are summarized in Supplemental Table S1. The ORR and DCR were 60.2% (95% CI: 50.1–69.7) and 87.4% (95% CI: 79.4–93.1), respectively.
The Kaplan–Meier curve of PFS and a forest plot of median PFS are presented in Figure 2(a) and (b), respectively. The median PFS was 18.4 months (95% CI: 13.8–22.1), with a 3-year PFS rate of 23.3%. Subgroup analyses of median PFS indicated trends toward a longer PFS in patients with PS 0 (25.0 months, 95% CI: 18.8–28.4) versus 1 (13.6 months, 95% CI: 9.3–16.4), patients with postsurgery recurrence [27.7 months, 95% CI: 18.8—not calculable (NC)] versus stage IIIB/IV (15.4 months, 95% CI: 12.2–20.2), patients without brain metastasis (20.6 months, 95% CI: 15.4–24.7) versus those with brain metastasis (13.8 months, 95% CI: 8.4–18.0), and patients with EGFR exon 19 deletion mutation (21.2 months, 95% CI: 15.4–24.8) versus uncommon EGFR mutations (14.3 months, 95% CI: 0.3–31.0).

Kaplan–Meier curve of PFS (a), forest plot of median PFS (b), and Kaplan–Meier curve of OS (c) in patients treated with afatinib.
The Kaplan–Meier curve of OS is presented in Figure 2(c). The median OS was not reached (95% CI: 34.9—NC), with a 3-year OS rate of 58.5%. Subgroup analyses of median OS could, therefore, only be calculated for patients aged ⩾75 years (42.4 months, 95% CI: 24.8—NC), patients with PS 1 (31.6 months, 95% CI: 24.8–42.4), stage IIIB/IV (35.4 months, 95% CI: 31.6—NC), brain metastasis (32.2 months, 95% CI: 17.1—NC), and patients with uncommon EGFR mutations (34.9 months, 95% CI: 9.8—NC).
Toxicity analysis
All AEs are summarized in Table 2. Among 103 patients, grade 3, grade 4, and grade 5 AEs occurred in 21 (20%), 2 (2%), and 1 (1%) patients, respectively. The most frequent grade ⩾3 AEs were diarrhea in 12 patients (12%), anorexia in 8 patients (8%), and rash acneiform in 6 patients (6%). In addition, pneumonitis was observed in three patients (3%), comprising one case each of grade 2 (1%), grade 3 (1%), and grade 5 (1%).
Summary of all AEs.

AE, adverse event; ALT, alanine aminotransferase; AST, aspartate transaminase.
Nine patients discontinued afatinib treatment because of AEs, comprising three discontinuations due to pneumonitis (one each grade 2, grade 3, and grade 5), rash acneiform in three patients (grade 2 in two patients and grade 4 in one patient), diarrhea in two patients (grade 1 in each patient), and anorexia in one patient (grade 4).
Treatment duration according to final dose
The treatment duration according to the final dose of afatinib is presented in Figure 3. The median afatinib treatment durations in patients with final doses of 40 (n = 27), 30 (n = 23), and 20 mg/day (n = 35), and 20 mg every other day (n = 18) were 13.4, 15.4, 18.8, and 18.3 months, respectively.

Treatment duration by the final reduced dosage of afatinib.
Regarding the nine patients who discontinued treatment because of AEs, five patients were in the group with a final dose of 40 mg/day, and no patient was in the group with a final dose of 20 mg every other day. In contrast, the 22 patients who remained on afatinib treatment were distributed equally among the four dosage groups.
Poststudy treatment
Eighty-one patients discontinued treatment with afatinib during the follow-up period (Figure 1), including 62 patients who received poststudy treatment (77%) (Supplemental Table S2) and 53 patients who underwent re-biopsy (65%) before second-line treatment, resulting in the detection of EGFR T790M mutation in 16/53 patients (30%).
A total of 25 of the 81 patients (31%) received osimertinib as poststudy treatment. Osimertinib was administered as second-line therapy in 19 patients (23%), comprising 11 patients with EGFR T790M mutation-positive status and 8 patients with EGFR T790M mutation-unknown status. Similarly, osimertinib was administered after second-line therapy in six patients (7%), comprising three patients with EGFR T790M mutation-positive status and three patients with EGFR T790M mutation-unknown status.
Impact of osimertinib on OS
The Kaplan–Meier curves of OS for patients who did (n = 25) and did not (n = 78) use osimertinib during the entire course of treatment are presented in Figure 4. The median OS was 42.4 months (95% CI: 30.1—NC) and not reached (95% CI: 34.2—NC), respectively, resulting in no significant difference between the groups (log-rank test, p = 0.654).

Kaplan–Meier curves of OS in patients who did and did not receive osimertinib during the course of treatment.
The median treatment durations of osimertinib in all 25 patients, 14 patients with EGFR T790M mutation-positive status, and 11 patients with EGFR T790M mutation-unknown status were 7.9 months (95% CI: 5.3–10.5), 8.0 months (95% CI: 5.1–10.9), and 7.8 months (95% CI: 2.9–12.7), respectively.
Discussion
We are currently conducting genomic, proteomic, epigenomic, and metabolomic analyses of peripheral blood samples (free molecules in serum/plasma and exosome-packaged molecules) collected from patients before, during, and after treatment in the EXTRA study. Furthermore, a comprehensive association study based on the ORR, DCR, PFS, OS, and AEs reported here is also in progress. The results for these clinical indicators, based on a sufficient observation period of 35.0 months in a clinical study of patients with advanced NSCLC, are considered to reflect the latest real-world data for first-line afatinib, which has been used in Japan since 2014.
In the EXTRA study, the median PFS in patients receiving afatinib was 18.4 months (95% CI: 13.8–22.1), and subgroup analyses indicated trends toward longer PFS for patients with PS 0 (25.0 months, 95% CI: 18.8–28.4), postsurgery recurrence (27.7 months, 95% CI: 18.8—NC), without brain metastasis (20.6 months, 95% CI: 15.4–24.7), and with EGFR exon 19 deletion mutation (21.2 months, 95% CI: 15.4–24.8). Historically, the median PFS of 18.4 months (95% CI: 13.8–22.1) in the EXTRA study seemed to be better than the median PFS reported in the LUX-Lung 3 study [n = 230 ; 11.1 months (95% CI: unpublished)] and its Japanese subset [n = 54 ; 13.8 months (95% CI: 11.0–19.1)], the LUX-Lung 6 study [n = 242 ; 11.0 months (95% CI: 9.7–13.7)], and the LUX-Lung 7 study [n = 160 ; 11.0 months (95% CI: 10.6–12.9)].9–12 The potentially better PFS in the EXTRA study compared with these previous studies might be attributable to the inclusion of patients with postsurgery recurrence, while the LUX-Lung 3, 6, and 7 studies only included stage IIIB/IV patients. Indeed, about a quarter of patients (27%) in the EXTRA study had postsurgery recurrence and demonstrated a median PFS of 27.7 months (95% CI: 18.8—NC); however, even if the analysis was limited to stage IIIB/IV patients, the median PFS was 15.4 months (95% CI: 12.2–20.2), which still seemed better than in the previous studies. Given that there was little difference in other patient characteristics, including PS, brain metastasis, and EGFR mutation status, the potentially better PFS in stage IIIB/IV patients in the EXTRA study may be related to the difference in rates of treatment discontinuation due to AEs.9–12
Differences in the body-surface area (BSA) and/or liver metabolic functions mean that the incidence of AEs following administration of a fixed-dose EGFR–TKI is generally higher in Japanese compared with Western patients. 13 Notably, a BSA ⩽ 1.7 m2 was significantly associated with severe afatinib-related AEs, 14 although the strategy of using a fixed-dose of afatinib was decided based on the results of the phase I study, demonstrating only a weak correlation between total body clearance of afatinib and BSA (r2 = 0.06). 15 The rate of afatinib discontinuation due to AEs in the Japanese subset of the LUX-Lung 3 study was 19%, compared with 8%, 6%, and 6% in the global LUX-Lung 3, 6, and 7 studies, respectively.9–12 A similar trend was observed in the FLAURA study, with osimertinib discontinuation rates of 26% in Japanese patients (n = 65) versus 13% in global patients (n = 279), and discontinuation rates of first-generation EGFR–TKIs of 35% in Japanese patients (n = 55) versus 18% in global patients (n = 277).16,17 As a result, the actual median treatment durations in Japanese patients in the osimertinib (15.0 months) and first-generation EGFR–TKI groups (10.3 months) were shorter compared with the median PFS values in the two groups (19.1 and 13.8 months, respectively), whereas the actual median treatment durations in all patients in the osimertinib (20.7 months) and first-generation EGFR–TKI groups (11.5 months) were almost the same as the median PFS values in the respective groups (18.9 and 10.2 months, respectively).16–18 Therefore, if the rate of treatment discontinuation due to AEs in Japanese patients in the FLAURA study had been lower, their median PFS might have been much better. Given these results, the fact that the rate of treatment discontinuation due to AEs (9%) in the EXTRA study was about half of that (19%) in the Japanese subset of the LUX-Lung 3 study is presumed to be one factor responsible for the favorable PFS in the EXTRA study.
The reason for the low rate of treatment discontinuation due to AEs in the EXTRA study, despite the Japanese ethnicity, might be that the minimum dose of afatinib specified in the LUX-Lung 3, 6, and 7 study protocols was 20 mg/day, compared with 20 mg every other day in the EXTRA study. A growing body of Japanese evidence supports this hypothesis. At least six phase II studies of first-line afatinib in patients with EGFR mutation-positive NSCLC have been conducted in Japan, including three studies (n = 30, 40, and 38, respectively) with a protocol-specified minimum dose of afatinib of 20 mg/day,19–21 and three studies (n = 53, 46, and 35, respectively) with 20 mg every other day.22–24 The rate of treatment discontinuation due to AEs was lower in the latter (8, 11, and 11%, respectively) compared with the former studies (17, 20, and 21%, respectively). Furthermore, the median PFS seemed better in the latter (12.6, 15.2, and 15.6 months, respectively) than in the former studies (11.8, 12.9, and 14.2 months, respectively), although the patients’ characteristics were not necessarily the same in all studies. These results suggest that a dose of 20 mg every other day may be more appropriate for some Japanese patients than 20 mg/day in terms of tolerability and preserved efficacy. In addition, plasma afatinib concentration was shown to be positively correlated with grade 3 AEs and negatively with BSA but not with treatment duration in Japanese patients. 24 We therefore agree with previous reports indicating that tolerability-guided dose reduction of afatinib had no impact on treatment duration.25,26 Most patients in the EXTRA study with a final dose of 20 mg every other day showed a durable response, with tolerability-guided dose reduction to 20 mg every other day within several months after initiating a dose of 20 mg/day.
Considering the use of EGFR–TKI monotherapy in terms of OS, the HR of osimertinib over first-generation EGFR–TKIs in the FLAURA study was 1.00 (95% CI: 0.75–1.32) in Asian and 1.39 (95% CI: 0.83–2.34) in Japanese patients; although the latter result was from an exploratory posthoc analysis, suggesting that EGFR–TKIs other than osimertinib may also be a treatment option, especially in Japanese patients.2,18 Notably, the Kaplan–Meier OS curve for first-generation EGFR–TKIs in the Japanese subset in the FLAURA study was initially inferior to that of osimertinib, crossing over at approximately month 27, after which the gap widened. 18 In contrast, the Kaplan–Meier OS curves in the EXTRA study and for afatinib in the Japanese subset of the LUX-Lung 3 study were almost identical to that for the first-generation EGFR–TKI in the Japanese subset of the FLAURA study.10,18 As a result, the 3-year OS rates were also similar across the studies: 59% in the EXTRA study, approximately 61% (estimated from Kaplan–Meier curve in the published literature) for afatinib in the Japanese subset of the LUX-Lung 3 study, and 63% for first-generation EGFR–TKI in the Japanese subset of the FLAURA study.10,18 Moreover, these results were based on patient characteristics with little overall difference between the three studies, especially the treatment rates with second-line osimertinib.10,18
The potentially better OS associated with first- and second-generation EGFR–TKIs compared with osimertinib in Japanese patients might be attributable to better postprogression survival (PPS) after first-line treatment with these EGFR–TKIs compared with osimertinib. In the Japanese subset of the FLAURA study, PPS after treatment with osimertinib and a first-generation EGFR–TKI was 20 months and NC, respectively, 18 while PPS after treatment with afatinib in the Japanese subset of the LUX-Lung 3 study and the EXTRA study was 33 months and NC, respectively. 10 We hypothesized that the potentially better PPS of Japanese patients treated with first- or second-generation EGFR–TKIs compared with osimertinib might be because rechallenge therapy with EGFR–TKIs may be less effective after osimertinib. In contrast, rechallenge with osimertinib after first- or second-generation EGFR–TKIs will be effective in patients with EGFR T790M mutation, and rechallenge with EGFR–TKIs other than osimertinib at any treatment line may be effective even in patients without EGFR T790M mutation. In a phase II study (n = 12) of rechallenge therapy with dacomitinib after osimertinib, the median PFS was only 1.8 months, with limited results even in patients with second-site EGFR mutations (C797S or G724S). 27 The reason for this phenomenon is considered to be the frequent development of resistance to osimertinib with the co-occurrence of two or more mutations, making EGFR–TKI monotherapy less effective. In the Osiris study (n = 50), the co-mutation rate after osimertinib was 42%, and PFS was comparable between patients treated with cytotoxic chemotherapy and individualized treatment with molecularly targeted therapy. 28 However, two phase II studies of rechallenge therapy with first- or second-generation EGFR–TKIs other than osimertinib in patients with EGFR T790M mutation-negative status showed median PFS values of 4.2 months (n = 12) and 4.7 months (n = 32), respectively.29,30 Additionally, in a retrospective study (n = 1603) of rechallenge therapy with first- or second-generation EGFR–TKIs before osimertinib became available in Japan, rechallenge was performed once in 28% of patients and twice or more in 12% of patients. 31
We, therefore, inferred that, even if osimertinib is not available after first-line afatinib, multiple rechallenge therapy with EGFR–TKIs may still be effective. The EXTRA study found no significant difference in OS between patients treated with and without osimertinib throughout the treatment. However, a difference might eventually be observed because patients currently not receiving osimertinib may subsequently receive osimertinib if an EGFR T790M mutation is detected during long-term follow-up. Nonetheless, the lack of any difference in patients with advanced NSCLC after 35.0 months is clinically meaningful.
Notably, the current frequency of 30% for detecting EGFR T790M mutation in the EXTRA study does not seem to be satisfactory. However, several studies have shown that the EGFR T790M mutation-positivity rate increased with increasing treatment duration with first-line EGFR–TKI.32–34 Thus, there is a high probability that the 21% of patients still receiving treatment with first-line afatinib will become EGFR T790M mutation-positive in the future. In contrast, there may be some situations in the real-world setting where osimertinib is expected, taking into consideration the treatment duration with first-line EGFR–TKI, to patients with EGFR T790M mutation-unknown status because of difficulty in performing re-biopsy for various reasons. In fact, the EXTRA study, reflecting real-world clinical practice, included 11 patients who received osimertinib despite their EGFR T790M mutation-unknown status, resulting in a relatively favorable median treatment duration of 7.8 months. However, we have to take care that it is considered the current standard of care in Japan as well to offer chemotherapy to patients with EGFR T790M mutation-negative or mutation-unknown status If these patients are chemotherapy-naive.
To date, only one retrospective cohort study has directly compared afatinib and osimertinib in terms of OS in Japanese patients. 35 Consecutive patients were treated with afatinib (n = 224) or osimertinib (n = 326) as first-line therapy, resulting in median OS after propensity score matching of 36.2 and 25.1 months, respectively (HR 1.47, 95% CI: 1.07–2.02), and median PFS of 16.5 and 20.5 months, respectively (HR 1.02, 95% CI: 0.81–1.28). The median PPS values in the two groups were 19.7 and 4.6 months, respectively, indicating better PPS after first-line treatment with afatinib compared with osimertinib in Japanese patients in a real-world setting. However, further studies are needed to determine the optimal first-line EGFR–TKI.
Our study had several limitations. First, this was a single-arm study with no comparison group. We are therefore now conducting a randomized phase II study comparing first-line afatinib and osimertinib in patients with EGFR mutation-positive NSCLC, with 3 year OS rate as the endpoint (the Heat on Beat study). 36 Patient accrual (n = 100) was completed on September 7, 2021, and the results will be published in the future. Second, we could not analyze the median treatment duration for sequential afatinib and osimertinib, like the Gio-Tag retrospective and Up-SwinG studies, nor the details of the rechallenge therapy with EGFR–TKI, because we did not schedule these items for analyses at the start of the study. We, therefore, aim to collect these data, together with new OS data, after a minimum follow-up period of 5 years. We also anticipate the results of the ongoing and prospective Gio-Tag Japan study (UMIN000037452). Third, we did not assess serum afatinib concentrations to monitor its pharmacokinetic profile, despite the importance of these data for validating tolerability-guided dose reduction. However, our comprehensive association study with multi-omics analyses, including metabolomics, will provide useful toxicity predictors for afatinib.
In conclusion, the EXTRA study is the largest prospective study reflecting current real-world data for the use of first-line afatinib in patients with EGFR mutation-positive NSCLC in Japan, in an era when osimertinib almost exclusively monopolizes first-line treatment. The results confirmed the favorable OS following first-line afatinib, possibly because of favorable PFS based on a low rate of treatment discontinuation due to AEs and favorable PPS independent of treatment with osimertinib. In the near future, the EXTRA study will identify novel predictive biomarkers for longer OS associated with first-line treatment with afatinib via a comprehensive association study using genomic, proteomic, epigenomic, and metabolomic analyses.
Supplemental Material
sj-docx-2-tam-10.1177_17588359231177021 – Supplemental material for Prospective exosome-focused translational research for afatinib (EXTRA) study of patients with nonsmall cell lung cancer harboring EGFR mutation: an observational clinical study
Supplemental material, sj-docx-2-tam-10.1177_17588359231177021 for Prospective exosome-focused translational research for afatinib (EXTRA) study of patients with nonsmall cell lung cancer harboring EGFR mutation: an observational clinical study by Saori Takata, Kei Morikawa, Hisashi Tanaka, Hidetoshi Itani, Masashi Ishihara, Kazuya Horiuchi, Yasuhiro Kato, Shinnosuke Ikemura, Hideyuki Nakagawa, Yoshiro Nakahara, Yoshitaka Seki, Akihiro Bessho, Nobumasa Takahashi, Kentaro Hayashi, Takeo Endo, Kiyoshi Takeyama, Toshiya Maekura, Nagio Takigawa, Akikazu Kawase, Makoto Endoh, Kenji Nemoto, Kazuma Kishi, Kenzo Soejima, Yusuke Okuma, Kenichi Yoshimura, Daisuke Saigusa, Yae Kanai, Koji Ueda, Akira Togashi, Noriyuki Matsutani and Nobuhiko Seki in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pptx-1-tam-10.1177_17588359231177021 – Supplemental material for Prospective exosome-focused translational research for afatinib (EXTRA) study of patients with nonsmall cell lung cancer harboring EGFR mutation: an observational clinical study
Supplemental material, sj-pptx-1-tam-10.1177_17588359231177021 for Prospective exosome-focused translational research for afatinib (EXTRA) study of patients with nonsmall cell lung cancer harboring EGFR mutation: an observational clinical study by Saori Takata, Kei Morikawa, Hisashi Tanaka, Hidetoshi Itani, Masashi Ishihara, Kazuya Horiuchi, Yasuhiro Kato, Shinnosuke Ikemura, Hideyuki Nakagawa, Yoshiro Nakahara, Yoshitaka Seki, Akihiro Bessho, Nobumasa Takahashi, Kentaro Hayashi, Takeo Endo, Kiyoshi Takeyama, Toshiya Maekura, Nagio Takigawa, Akikazu Kawase, Makoto Endoh, Kenji Nemoto, Kazuma Kishi, Kenzo Soejima, Yusuke Okuma, Kenichi Yoshimura, Daisuke Saigusa, Yae Kanai, Koji Ueda, Akira Togashi, Noriyuki Matsutani and Nobuhiko Seki in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors would like to thank all the patients who participated in this study and their families, as well as the investigators. The authors express their gratitude to ASCA Corporation for editing a draft of this article funded by Boehringer Ingelheim.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
