Abstract
Prognosis is generally poor for patients with pancreatic ductal adenocarcinoma. However, patients with germline BRCA1 or BRCA2 mutations (gBRCAm) may benefit from first-line platinum-based chemotherapy and maintenance therapy with the poly(adenosine diphosphate-ribose) polymerase inhibitor olaparib following at least 16 weeks of first-line platinum-based chemotherapy without disease progression. Germline breast cancer gene (BRCA) testing is therefore important to ensure that patients receive the most effective treatment. In addition, testing for other DNA damage response gene mutations beyond gBRCAm may also guide treatment decisions. However, clinical pathways for genetic testing are often suboptimal, leading to delays in treatment initiation or missed opportunities for personalized therapy. Barriers to testing include low rates of referral and uptake, delays to referral and slow result turnaround times, cost, and biopsy and assay limitations if somatic testing is performed, leading to the requirement for subsequent dedicated germline testing. Low rates of referral may result from lack of awareness among physicians of the clinical value of testing, coupled with low confidence in interpreting test results and poor availability of genetic counseling services. Among patients, barriers to uptake may include similar lack of awareness of the clinical value of testing, anxiety regarding the implications of test results, lack of insurance coverage, fear of negative insurance implications, and socioeconomic factors. Potential solutions include innovative approaches to testing pathways, including ‘mainstreaming’ of testing in which BRCA tests are routinely arranged by the treating oncologist, with the involvement of genetic counselors if a patient is found to have a gBRCAm. More recently, the utility of multigene panel analyses has also been explored. Access to genetic counseling may also be improved through initiatives such as having a genetic counseling appointment for all new patient visits and telemedicine approaches, including the use of telephone consultations or DVD-assisted counseling. Educational programs will also be beneficial, and cost effectiveness is likely to improve as the number of targeted treatments increases and when the earlier detection of tumors in family members following cascade testing is considered.
Keywords
Introduction
Patients with pancreatic ductal adenocarcinoma (PDAC) face a particularly poor prognosis, with 5-year relative survival of 12.5% 1 and 5-year actual survival of only 4.2%. 2 However, for those with metastatic disease and a germline (i.e. heritable) BRCA1 or BRCA2 mutation (gBRCAm), first-line platinum-based chemotherapy followed by maintenance therapy with olaparib – a poly(adenosine diphosphate-ribose) polymerase (PARP) inhibitor – can significantly prolong progression-free survival (PFS).3,4 Thus, it is vital that patients with these mutations are identified, ideally at the time of diagnosis, to enable prompt receipt of the most effective treatment. In addition, testing for other aberrations in DNA damage response genes beyond breast cancer gene (BRCA), such as PALB2, may also guide treatment decisions, because patients who harbor these mutations may also be responsive to first-line platinum-based chemotherapies or PARP inhibitors. Initial evidence was seen in a phase-2 trial in which olaparib was beneficial in patients with breast cancer with mutated PALB2 5 ; further studies are warranted to evaluate this in the PDAC setting. Several recent studies and reviews support the need for gBRCAm testing and suggest that testing should be extended beyond gBRCAm to identify patients with PDAC who harbor other DNA damage response gene mutations.6–11 Although eligibility criteria for gBRCAm testing in patients with PDAC have been broadened,4,12 referral for genetic counseling and testing is still suboptimal and challenging, often resulting in significant delays to treatment initiation or missed personalized therapeutic opportunities. In this review, we outline the rationale for gBRCAm testing in patients with PDAC and highlight some of the obstacles to achieving this in a timely manner. Using examples from our own experience of pancreatic and other heritable cancers, we also discuss potential ways of overcoming these barriers, with a view to improving testing rates and reducing turnaround times (TATs).
GBRCA testing in PDAC: general overview and clinical utility
Germline variants in BRCA genes constitute the most clinically relevant molecular aberrations in PDAC in terms of therapeutic actionability. 13 An estimated 5−8% of unselected patients with PDAC carry a gBRCAm, 13 although in some ancestry groups, such as Ashkenazi Jews and African Americans, the prevalence is significantly higher.14–16 gBRCAm are inherited and present from the time of conception in each cell, as opposed to somatic BRCA1 or BRCA2 mutations (sBRCAm), which are acquired during life and arise specifically in tumors. Identification of gBRCAm and sBRCAm is associated with distinct clinical pathways and diagnostic workups. The former can be identified through broad gene panel testing of tumor tissue using next-generation sequencing (NGS), the latter by testing blood or saliva samples (non-tumor). Germline variants in BRCA genes have relevant therapeutic implications in patients with PDAC. The BRCA1 and BRCA2 genes encode critical proteins involved in repairing double-strand DNA breaks via homologous recombination. Cancer cells with BRCAm thus have an impaired ability to repair double-strand DNA breaks, resulting in increased reliance on other DNA damage response pathways for survival, 13 making them highly sensitive to blockade of those pathways.13,17 In cancer patients with gBRCAm, both platinum-based chemotherapy, which causes double-strand breaks, 18 and PARP inhibition, which prevents repair of single-strand breaks and thereby generates double-strand breaks, 13 increase the likelihood of cancer cell death (Figure 1). Consequently, platinum-based chemotherapy is most effective in patients with PDAC who have a gBRCAm or a tumor that is homologous recombination repair deficient.19,20 In the POLO study, conducted in patients with deleterious or suspected deleterious gBRCAm with metastatic PDAC whose disease did not progress during first-line platinum-based chemotherapy, subsequent maintenance therapy with the PARP inhibitor olaparib significantly improved PFS versus placebo. 3 These results led to approval of olaparib in multiple countries as maintenance therapy in patients with metastatic gBRCAm PDAC whose disease has not progressed after at least 16 weeks of first-line platinum-based chemotherapy and highlighted the importance of universal gBRCA testing in identifying those who may benefit from targeted treatment. Treatment with PARP inhibitors is only appropriate for patients with bona fide pathogenic or likely pathogenic gBRCAm and is not appropriate for patients with BRCA1 and BRCA2 variants of unknown significance, because the majority of these will eventually be reclassified as non-pathogenic. 21

Rationale for using a PARP inhibitor in patients with gBRCAm.
Data further illustrating the importance of testing all patients with PDAC come from studies showing that 40–60% of patients with gBRCAm do not declare a family history of PDAC, emphasizing that relying on family history alone to guide genetic testing will result in under-detection of gBRCAm carriers and supporting a need for universal genetic testing of patients with PDAC.15,23 In addition, because pre-operative platinum-based chemotherapy is most effective in patients with gBRCAm,24,25 it is critically important to identify these patients early at the time of diagnosis. Given this evidence, BRCA genetic testing of all patients with PDAC, regardless of family history, age, and disease stage, is now recommended by the National Comprehensive Cancer Network 4 and the American Society for Clinical Oncology 12 (Table 1).
Guideline recommendations for genetic testing in patients with PDAC.
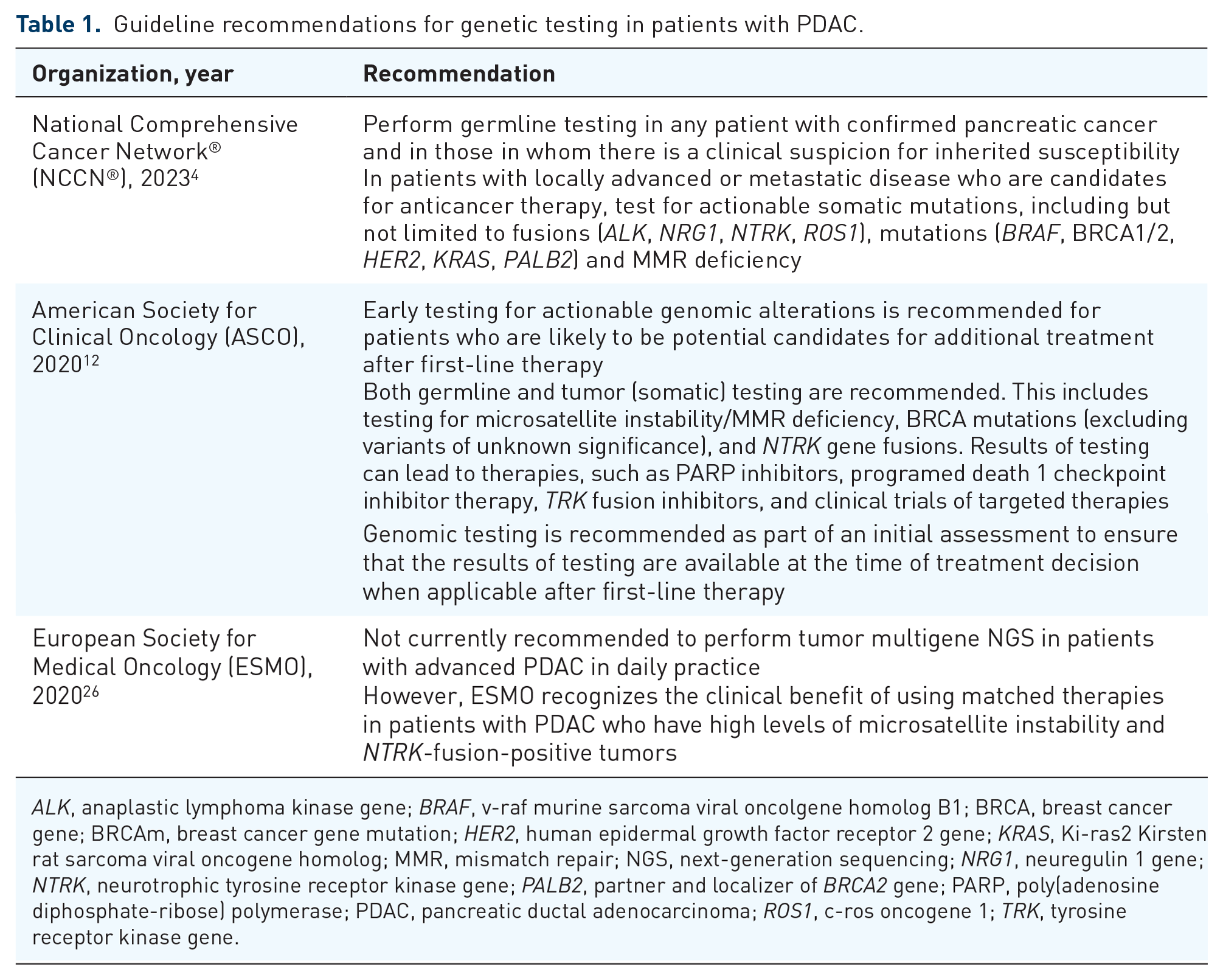
ALK, anaplastic lymphoma kinase gene; BRAF, v-raf murine sarcoma viral oncolgene homolog B1; BRCA, breast cancer gene; BRCAm, breast cancer gene mutation; HER2, human epidermal growth factor receptor 2 gene; KRAS, Ki-ras2 Kirsten rat sarcoma viral oncogene homolog; MMR, mismatch repair; NGS, next-generation sequencing; NRG1, neuregulin 1 gene; NTRK, neurotrophic tyrosine receptor kinase gene; PALB2, partner and localizer of BRCA2 gene; PARP, poly(adenosine diphosphate-ribose) polymerase; PDAC, pancreatic ductal adenocarcinoma; ROS1, c-ros oncogene 1; TRK, tyrosine receptor kinase gene.
Genetic testing of patients with PDAC is also critical to the strategy of ‘cascade testing’, in which family members of an affected individual are tested, in a stepwise manner, until all at-risk relatives have been screened. 27 Healthy individuals carrying gBRCAm may be offered tailored cancer screening or risk-reducing surgery aimed at early detection of cancerous lesions or prevention of cancer altogether.28,29
Challenges associated with gBRCA testing in PDAC
Despite the clear advantages of BRCA testing, many challenges at patient, provider, technical, and financial level limit its clinical implementation and utility in patients with PDAC.
Referral
Lack of patient referral is the first barrier to BRCA testing. In other tumor types, referral rates for patients eligible for genetic testing are far from optimal, ranging from 24% in breast cancer to 35% in colorectal cancer.30,31 Although published data on referral rates in PDAC are limited, these seem to be even lower owing to the multitude of symptomatic, endoscopic, and therapeutic demands inherent to managing the disease 32 and the relatively recent inclusion of genetic testing in treatment guidelines.
At a single center in the USA from 2015 to 2017, only 32% of patients with PDAC were referred for genetic counseling, with 19% completing germline testing. 33 In a 2020 survey of 67 Italian gastrointestinal oncologists, 28% reported that they did not perform any kind of gBRCA screening of patients with PDAC, 27% screened patients in an unselected manner, and 45% screened patients based only on family history, age, or eligibility for platinum-based therapies. 34
Owing to the high variability of guidelines on BRCA testing eligibility and reimbursement policy, referral rates may vary significantly across countries. In Israel, gBRCA testing has been covered by health insurance since 2019 for all patients with PDAC, suggesting that referral rates may be much higher in this country, although no objective data are currently available. Rates may vary by institution owing to institution-specific initiatives and barriers affecting access to testing. For instance, at the Fox Chase Cancer Center in Philadelphia, PA, USA, it is estimated that approximately 80% of patients with PDAC undergo germline genetic testing, primarily through matched tumor–normal testing and/or referral to the Department of Clinical Genetics for germline genetic testing.
Timing
Even if all eligible patients were referred for genetic testing, a key challenge is the urgency with which results are needed. Each patient needs to be tested promptly, ideally at diagnosis, given the poor prognosis and urgency to administer the most effective treatments early and to optimize outcomes. However, the process of referring patients for genetic counseling and securing their consent to undergo testing can cause significant delays. Deciding when to broach the topic with patients may also be challenging. At diagnosis, patients may be experiencing information ‘overload’, and their focus is likely to be on other issues, such as prognosis, symptom management, and end-of-life planning. 35 Patients may also be hesitant to talk with their relatives about the hereditary implications of undergoing genetic testing. Data from a survey carried out at an academic medical center in the USA between 2016 and 2017 indicate that patients’ attitudes to genetic testing in PDAC are similar to those in breast and ovarian cancer, despite poor prognosis with PDAC diagnoses. 36
Turnaround time
Long testing TATs are a further barrier to genetic testing. Data indicate that TATs for genetic testing could range between 1.7 and 21 weeks, depending on the test type, whether it was a single-gene, small panel, or large multigene panel analysis, and the region in which the testing laboratory was based.37,38 Promisingly, however, our own insights suggest that TATs in non-clinical trial settings are becoming shorter, with mean TATs of 2–4 weeks depending on the type of test used. Another limiting factor may be that, in some cases, traditional genetic services models are still set up to determine hereditary risk in individuals with a strong family history of cancer, for which results are not needed as urgently as when tests are required to determine eligibility for targeted treatment.
Uptake
Submaximal uptake of genetic testing is an issue seen across all cancers, and rates vary markedly between and within countries, at least in part because of differences in healthcare systems, both nationally and locally. In a systematic review of 18 breast cancer studies, mean uptake of BRCA testing was 59%, but values varied considerably across populations and settings. 39 While similar rates (58%) were observed in a prospective Canadian study of patients with PDAC, 40 uptake of 98% was reported in a retrospective audit of New Zealand patients with ovarian cancer. 41 Low uptake has also been reported in family members of affected individuals. In a Canadian study, only 31% of informed at-risk relatives of individuals with PDAC and pathogenic germline alterations underwent genetic testing. 42
Other factors underlying suboptimal uptake can be broadly split into structural physician-, insurer-, or patient-related barriers (Table 2). A lack of biomarker-paired therapies is perhaps the simplest structural barrier, with results from a recent real-world study showing that gBRCAm testing rates in patients with HER2− advanced breast cancer increased significantly from 2015 to 2019/2020, coinciding with the availability of PARP inhibitors. 43 Poor awareness among physicians about the clinical value and availability of genetic testing and confidence in interpreting test results is another major concern, with a large survey of US physicians finding that, although the vast majority (97.6%) acknowledged that genetic variations can influence drug response, only 10.3% felt adequately informed about pharmacogenetic testing and interpretation. 44 Lack of provider referral is also a common barrier, 45 as is a shortage of counselors with genetics expertise. A good illustration is in the US state of Florida, which has a population of 21 million, yet trains only four genetic counselors per year. Various insurance restrictions can act as barriers to uptake, such as in the USA, where the single largest insurer (Medicare) does not reimburse genetic counseling visits, and in Italy, where there are substantial regional differences in reimbursement availability for genetic testing. Some US insurers also require genetic counseling to be conducted alongside genetic testing because this is an important practice to protect against patient misinterpretation of genetic testing results.
Potential barriers to uptake of BRCA testing in PDAC and proposed solutions to overcome them.

BRCA, breast cancer gene; gBRCA, germline BRCA; gBRCAm, germline BRCA mutation; PARP, poly(adenosine diphosphate-ribose) polymerase; PDAC, pancreatic ductal adenocarcinoma; TAT, turnaround time.
For patients, the main barriers seem to be poor awareness of the value of testing, concerns about costs, anxiety/psychosocial issues, and fear of negative insurance implications. 45 Patient preferences after genetic counseling, technical challenges, and insurance non-coverage are also limitations. In the US REACH study, cost barriers were shown to be a strong negative predictor of uptake of BRCA testing among breast and ovarian cancer survivors. 51 Recently, these barriers may have been largely removed in the USA as a result of competition between genetic testing laboratories, with most facilities guaranteeing a maximum out-of-pocket expense of $250, and the majority of patients ultimately paying nothing out of pocket. However, insurance implications remain a concern in other regions. Data from the Victorian Colorectal Cancer Family Study in Canada showed that half of individuals who were informed about potential implications of germline testing for insurance eligibility decided not to undergo testing, compared with only one-fifth of those who did not receive this advice. 57 Income, education level, and race/ethnicity may also affect uptake,53,54 and, even when traditional barriers are minimized, non-White patients are less likely than White patients to receive recommended follow-up for cancer risk counseling. 58 Finally, some patients may simply opt to forego testing in order to begin treatment sooner.
In summary, although the value of performing genetic testing in PDAC is clear, it is not known how often this service is offered or the proportion of patients who subsequently undergo testing. However, from our own insights and experience in other cancers, we know that this proportion is not 100%. Studies investigating actual rates of referral and uptake in PDAC are therefore needed to determine the extent of the issue and shed light on potential interventions that may improve these rates.
Biopsy and assay limitations
Both germline and tumor testing are increasingly used in PDAC, yet the indications for testing and the utility of each type of testing differ. 59 The current standard of care with regard to BRCA testing for PDAC is germline testing from blood samples.4,12 Paired germline testing of blood with somatic testing of tumors is an emerging practice in a few large centers. 16
Detection of somatic mutations can inform treatment choice, as well as identify risk factors and molecular markers for disease monitoring.59,60 DNA sequencing of PDAC biopsy tissue to identify somatic mutations is thus becoming increasingly common. However, current tumor sequencing assays cannot reliably distinguish between germline or somatic origin.59,60 Thus, if a BRCAm is detected, the mutation source must be verified using a germline assay (because targeted treatments are indicated only for patients with gBRCAm, not sBRCAm). This additional testing further delays results and treatment initiation. Furthermore, there are concerns that techniques that can distinguish between somatic and germline mutations, such as paired tumor–normal DNA sequencing, 61 may miss certain rare or more complex BRCAm, preventing utilization of targeted therapy. The question remains about whether a negative BRCAm result from such tests should be viewed in the same way as a negative result from a dedicated germline test.
Other challenges associated with tumor testing include the fact that not all patients with PDAC have available tissue. However, the situation is changing, and, increasingly, biopsies are being recommended for all patients with PDAC except those with early-stage disease scheduled for upfront surgery, in whom resected tissue could be tested instead. On the other hand, in France, even patients with early-stage PDAC are increasingly being recommended to undergo a biopsy to enable neoadjuvant treatment to be offered, including as part of a clinical trial.
Biopsy samples are usually collected during endoscopic ultrasound-guided fine needle aspiration (EUS-FNA), 62 but its accuracy varies widely. Thus, when PDAC is strongly suspected but the EUS-FNA result is negative, a second EUS-FNA is required. However, there are ethical considerations associated with performing repeat biopsies 63 because the process may incur additional risk and delay the start of chemotherapy, which may affect prognosis and treatment success.62,64 Furthermore, obtaining samples from the pancreas is anatomically challenging, 63 pancreatic tumors display a high degree of intratumor cellular heterogeneity, with multiple mutationally distinct subclones potentially existing in a primary tumor, 65 EUS-FNA samples are relatively small and may not be suitable for DNA analyses, 63 and only a limited number of analyses can be performed. 63 Another major barrier is the lack of specialist tertiary cancer centers and experts to analyze and interpret the complex data generated from tumor sequencing. 63
Cost and cost effectiveness
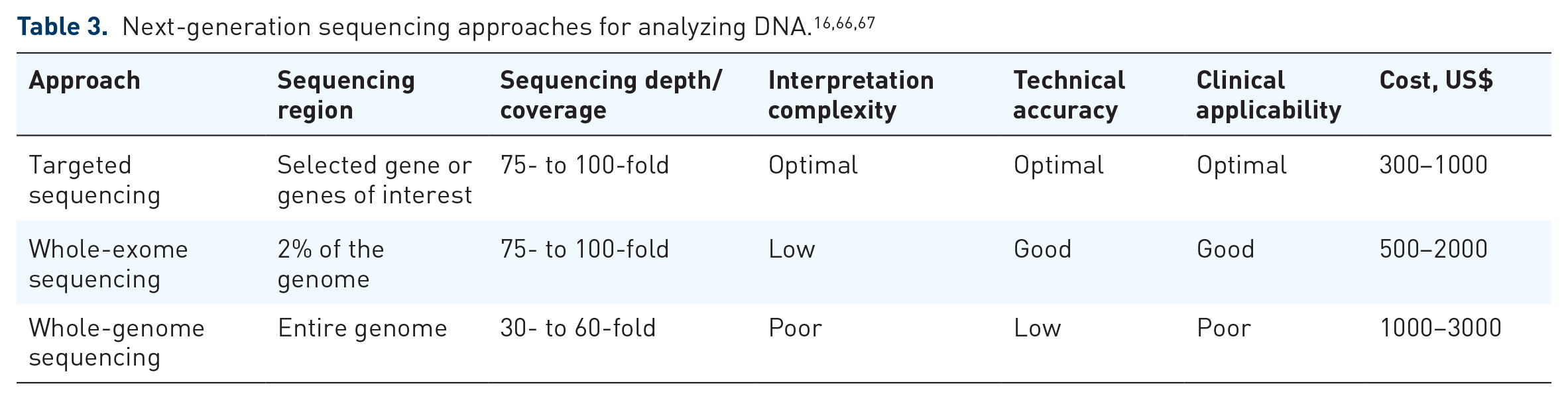
The vast majority of laboratories performing genetic testing use NGS to examine BRCAm. 37 The three main NGS approaches used for somatic or germline DNA analysis are targeted sequencing, whole-exome sequencing, and whole-genome sequencing (WGS) (Table 3).66,67 Targeted sequencing is the cheapest, most accurate, and easiest test to interpret and requires the least amount of sample (important for somatic testing, when biopsy samples may be limited).66,67 WGS is the most expensive and time consuming and requires large tissue volumes.66,67 However, early results from the observational COMPASS study, in which WGS of tumor samples from patients with PDAC was undertaken, have provided useful translational evidence, and the final results are eagerly anticipated. 68 With significant variation in the price of a gBRCA test, testing cost may act as a significant barrier in some regions of the world.
Potential solutions
Timing, turnaround, and uptake
Potential solutions are detailed in Table 2 and Figures 2 and 3. Numerous studies have demonstrated that innovative genetic services models can reduce genetic counseling/wait times, increase the proportion of patients undergoing germline testing within 7 days of initial oncology evaluation, reduce times to consent, testing, disclosure of results, and treatment initiation, and improve uptake levels. Successful approaches include DVD-assisted genetic counseling, 69 embedded genetic counseling models,48,50,70,71 models that incorporate an appointment with a genetic counselor at the ‘new patient’ visit,49,72 direct genetic testing, 73 universal genetic testing,42,48 automatic referrals, 49 and ‘mainstreaming’ programs.38,46,47


Barriers to gBRCA testing in PDAC and proposed solutions for overcoming them.
Perhaps the most useful, practical, and frequently adopted approach is ‘mainstreaming’, in which the testing process is streamlined, with genetic testing arranged by specialties more closely involved in patient care than the genetics services team. Typically, the treating oncologist or cancer team orders the genetic test, with genetics services only becoming involved if a patient tests positive (and for subsequent cascade testing of at-risk relatives).38,46,47,50 There are several successful examples in ovarian cancer, in which ‘mainstreaming’ in UK clinics has been able to reduce the time to disclosure of BRCA test results by up to fivefold (3−4 weeks with ‘mainstreaming’ versus up to 20 weeks via a traditional pathway).38,46 ‘Mainstreaming’ also resulted in a substantial increase in uptake, from 14% at baseline to 95% after implementation. 38 In an Italian study of women with ovarian cancer, implementation of gynecologic-oncologist-initiated testing significantly reduced median time from diagnosis to disclosure of BRCA test result from 27 weeks using the traditional model to 8 weeks with ‘mainstreaming’. 47 Testing uptake also increased, from 50% with previous methods to 86% with ‘mainstreaming’. 47
An oncology clinic ‘genetic testing station’ initiative at the University of California, San Francisco, CA, USA, which systematically incorporated an appointment with a genetic counselor at the ‘new patient’ visit to the gastrointestinal oncology department, demonstrated that, compared with a traditional referral-based genetic counseling pathway, uptake of germline testing increased from 19 to 71%. 72 The rate of germline pathogenic variant detection increased from 20 to 33%. 72 Low uptake of genetic testing due to poor access to genetics services is an issue that especially affects marginalized populations. Setting up dedicated testing clinics and services for these populations is an approach that has proven successful at reducing health disparities in Italy, resulting in improved access to cancer diagnoses and cancer care for historically underserved patients.
Other innovative approaches that can improve uptake include the universal genetic testing pathway implemented by an oncology clinic at McGill University Health Center, Montreal, Canada. Here, the in-clinic offer of germline testing to all patients with PDAC demonstrated improved testing uptake and TAT, as well as a higher detection rate of pathogenic germline alterations, compared with a traditional medical genetics referral model. 42 Uptake increased from 59 to 97%, and median testing report TAT reduced from 42 to 13 days, after implementation of universal testing. 42 This approach also increased the pathogenic germline alteration detection rate from 6.6 to 17.2%. 42 At Dana-Farber Cancer Institute, Boston, MA, USA, implementation of an automatic referral system for patients with PDAC also increased uptake of genetic counseling and multigene germline testing compared with traditional oncologist referral (16.5% versus 38.0%), including uptake rates within 7 days of initial oncology evaluation (14.7% versus 60.3%). 49
However, some novel approaches may reduce testing rates compared with traditional models. For example, BRCA testing uptake in US patients with breast or ovarian cancer was 6–10% points lower in individuals who received telephone genetic counseling than in those who received traditional genetic counseling.74–76 Furthermore, although many patients may be better served by models that increase access to genetic testing or genetic counseling (e.g. by reducing the number of in-person appointments or clinical wait times), providers should recognize that alternative models may not be appropriate for all patients or clinical situations, and certain populations may be better served by traditional models. 50 Alternative models may also not suit certain geographic regions. The US ‘fee for service’ model of care does not lend itself to innovative programs such as telemedicine, artificial intelligence, and some of the models previously outlined, because payers do not reimburse for these services. Outside of subsidized programs at academic medical centers, each patient would instead need to be seen and evaluated individually for reimbursement of the work needed to order the test, track the results, and return the results. Thus, there is no ‘one size fits all’ counseling model that will be suitable for adoption in every setting, and when non-traditional models are used, it will be important to ensure that standard counseling is also available as an alternative.
Additional solutions beyond adoption of novel genetic services models include providing training to mid-level healthcare professionals (such as advanced registered nurse practitioners) to enable them to order and follow up on genetic testing as an alternative to the treating oncologist, a practice that has been initiated in Florida. Providing educational materials to patients is also another potential solution. In a study of patients with ovarian or breast cancer, the main reasons reported by those who agreed to undergo genetic counseling were a desire to learn information for their family members and a desire to aid their own health, whereas one of the main reasons for declining was a perceived lack of benefit of testing. 77 Initiatives aimed at encouraging the former and reducing the latter may therefore be helpful.
Biopsy and assay limitations
Targeted treatments in PDAC are indicated only for patients with gBRCAm. However, in ovarian and prostate cancer, these therapies are effective (and indicated) in patients with sBRCAm. The same may therefore be true in PDAC, with indications expanded in future to cover gBRCAm or sBRCAm. Indeed, a phase-2 study showed that maintenance rucaparib treatment was efficacious in patients with platinum-sensitive, advanced prostate cancer who harbored gBRCAm, as well as those with gPALB2m and sBRCAm, suggesting that targeted treatments may be beneficial beyond gBRCAm. 78 Another phase-2 clinical trial evaluating niraparib and the immune checkpoint inhibitor, dostarlimab, in patients with pancreatic cancer with germline or somatic mutations in BRCA, PALB2, BARD1, RAD51C, or RAD51D is under way. 79 In addition, other studies are ongoing to evaluate novel combination therapies in PDAC patients with gBRCAm, such as the phase-2 trial of olaparib and pembrolizumab (NCT04548752), as well as studies in those with somatic and germline mutations in DNA damage repair genes, including a phase-2 trial of olaparib plus durvalumab (NCT05659914).
In lung cancer, the scarcity of biopsy tissue prompted a broader, less-specific approach to NGS testing – a lesson that could also be applied to PDAC. Likewise, it is important to ensure that molecular profiling can ‘piggyback’ on any tissue samples obtained during routine clinical care to avoid the need for repeat biopsies. 64 Another consideration is the use of EUS-FNB, which yields larger tissue cores than EUS-FNA with preserved architecture. 64 Indeed, studies have shown that, compared with EUS-FNA, use of EUS-FNB results in a significantly higher proportion of samples that are sufficient for genomic testing, both overall and in samples taken from tumors at least 3 cm in size or from the pancreatic head/neck. 80
Molecular profiling tests of any tumor tissue should also be optimized for formalin-fixed paraffin-embedded tissues that can be processed using standard, less-time-sensitive protocols than real-time tissue processing. 64 Alternative tissue sampling techniques, such as liquid biopsy, may also offer advantages over current methods, being much quicker, easier, and less invasive to obtain than other biopsies. 81 In PDAC, use of liquid biopsy has mainly been investigated for diagnostic and prognostic purposes, 62 but several studies have demonstrated its feasibility, accuracy, sensitivity, and utility for targeted NGS analysis.82,83 Adoption of novel techniques that can distinguish between somatic and germline mutations, without the need for matched ‘normal’ tissue controls, may also be useful (e.g. the somatic–germline–zygosity algorithm developed by Foundation Medicine, Inc. 84 and its subsequent ‘afdis’ modification 85 ). However, these techniques are experimental only and are not recommended in current clinical guidelines.
Cost effectiveness
Experience in other familial cancers has shown that NGS is most likely to be cost effective if used for multigene panels rather than single-gene testing. For example, panel sequencing to detect mutations in 7–12 genes in patients with metastatic or advanced non-small-cell lung cancer has moderate cost effectiveness or is more cost effective versus single-marker genetic testing.86–88 Likewise, in Israel, NGS is cost effective in patients with non-small-cell lung cancer when screening for multiple mutations at a time. In PDAC, only three genetic alterations are biologically targetable: gBRCAm (with olaparib – tumor-specific approval); neurotrophic tyrosine receptor kinase gene fusions (entrectinib or larotrectinib – tumor-agnostic approval); and defective DNA mismatch repair/high microsatellite instability (pembrolizumab – tumor-agnostic approval). Fortunately, the cost of NGS assays is reducing; therefore, screening for just three alterations may soon become cost effective in PDAC. The number of targetable mutations is also likely to increase, further improving cost effectiveness. Indeed, although rare in PDAC, 89 the KRASG12C mutation is potentially targetable with the KRAS inhibitor AMG 510, 90 and initial results from the Know Your Tumor program found actionable genomic alterations in 50% of patients with PDAC, covering 30 different genetic markers. 91 A study in the USA (from April 2018 to March 2020), implementing universal testing with an 83-gene NGS panel, also revealed that one in six patients with PDAC had pathogenic germline alterations (11% of patients had alterations other than gBRCAm in 15 different cancer predisposition genes). 92 Finally, as previously noted, it is not yet known whether the PDAC treatments that are effective in patients with gBRCAm are also effective in patients with sBRCAm (approximately half of whom do not have gBRCA) or in patients who have mutations in homologous recombination genes other than BRCA1 and BRCA2. Notably, almost 70% of pathological germline alterations may be in homologous-recombination-repair-deficient genes. 92 This is an important area for future research, and, if the treatments are found to be effective, multigene panel testing may be expanded to include these mutations.
Cost effectiveness of BRCA1/2 testing is also likely to become more favorable when unaffected carriers are identified through cascade testing,93–95 because they may be offered interventions aimed at reducing their risk of developing cancer or detecting cancer earlier, with subsequently improved outcomes in cases when cancer does still develop.93,94 Indeed, models have shown that adopting this type of long-term management approach in gBRCAm carriers would be cost effective in Australia, Canada, and the USA.93–95 The growing role of cascade testing in at-risk relatives of patients with PDAC further supports the rationale of helping patients to understand that the benefits of BRCA testing extend beyond improving their own treatment outcomes, also providing opportunities for cancer prevention and earlier cancer detection for their family members.
Conclusions
By identifying the subgroup of patients who may benefit from targeted treatment, the value of performing genetic testing in patients with PDAC is clear. Notably, genetic testing in patients not only provides theranostic value, to inform therapeutic decisions through identification of actionable biomarkers, but also diagnostic value, to permit risk reduction in family members who also carry a gBRCAm. However, as seen in other heritable cancers, referral rates and subsequent uptake of genetic testing remain suboptimal. By broadening the eligibility criteria for gBRCAm testing and recommending that genetic testing is performed in all patients with PDAC, the recent updates to clinical guidelines will go some way toward improving uptake rates. However, a range of structural physician-, insurer-, and patient-related barriers remain. Fortunately, actionable solutions are available to address many of these challenges, helping to increase uptake rates, reduce testing TATs, and improve cost effectiveness. Further research to identify additional barriers and solutions is warranted, along with real-world studies investigating the extent to which the latest testing guidelines are being followed.
Footnotes
Acknowledgements
Medical writing support was provided by Abigail Woollard and Charlie Foster, both of Oxford PharmaGenesis, Oxford, UK, and was funded by AstraZeneca and Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA.