Abstract
Background:
Research on the correlation between circulating tumor cells (CTCs) and gastric cancer (GC) has increased rapidly in recent years. However, whether CTCs are associated with GC patient prognosis is highly controversial.
Objective:
This study aims to evaluate the value of CTCs to predict the prognosis of GC patients.
Design:
A meta-analysis.
Data Sources and Methods:
We searched the PubMed, Embase, and Cochrane Library databases for studies that reported the prognostic value of CTCs in GC patients before October 2022. The association between CTCs and overall survival (OS) and disease-free survival (DFS)/recurrence-free survival (RFS) and progression-free survival (PFS) of GC patients was assessed. Subgroup analyses were stratified by sampling times (pre-treatment and post-treatment), detection targets, detection method, treatment method, tumor stage, region, and HR (Hazard Ratio) extraction methods. Sensitivity analysis was performed by removing individual studies to assess the stability of the results. Publication bias was evaluated using funnel plots, Egger’s test, and Begg’s test.
Results:
We initially screened 2000 studies, of which 28 were available for further analysis, involving 2383 GC patients. The pooled analysis concluded that the detection of CTCs was associated with poor OS (HR = 1.933, 95% CI 1.657–2.256, p < 0.001), DFS/RFS (HR = 3.228, 95% CI 2.475–4.211, p < 0.001), and PFS (HR = 3.272, 95% CI 1.970–5.435, p < 0.001). Furthermore, the subgroup analysis stratified by tumor stage (p < 0.01), HR extraction methods (p < 0.001), detection targets (p < 0.001), detection method (p < 0.001), sampling times (p < 0.001), and treatment method (p < 0.001) all showed that CTC detection was associated with poor OS and DFS/RFS for GC patients. Furthermore, the study showed that CTCs were associated with the poor DFS/RFS of GC when CTCs were detected for patients from Asian or No-Asian regions (p < 0.05). In addition, higher CTCs predicted poorer OS for GC patients who are from Asian regions (p < 0.001), but without statistical difference for GC patients from No-Asian regions (p = 0.490).
Conclusion:
CTC detection in peripheral blood was associated with poor OS, DFS/RFS, and PFS in patients with GC.
Highlights
The association between CTCs and GC is still controversial.
The subgroup analysis stratified by sampling times, detection targets, detection method, treatment method, tumor stage, and HR extraction methods was conducted to explore the source of heterogeneity.
The study showed that the detection of CTCs was associated with a poor prognosis of GC.
Introduction
Gastric cancer (GC) remains the fifth most prevalent tumor worldwide and is the fourth leading cause of oncological death.1,2 Despite the advanced progress achieved in the treatment of GC by various methods, such as laparoscopic surgery, 3 robotic surgery, 4 and perioperative chemotherapy, 5 its prognosis remains poor due to late detection, the aggressiveness of tumor cells, and high recurrence/metastasis rates. 6 Recurrence and metastasis may involve a range of cells, including circulating tumor cells (CTCs), which have been progressively recognized to play an important role in distant metastasis, according to the ‘seed and soil theory’. 7
CTCs are tumor cells that originate from primary or metastatic tumors and travel through the systemic circulation to distant organs, where they can initiate metastatic lesions. 8 CTCs can be isolated from a patient’s blood sample via different technologies that take advantage of their physical and biological properties. Recent studies have shown that CTCs can be found in many solid tumor types, such as breast, prostate, lung, and colorectal cancers,9–12 potentially enabling early detection of metastatic progression, treatment response, and tumor recurrence. 13 CTC analyses are considered a real-time ‘liquid biopsy’ for patients with cancer, and several studies have also demonstrated the clinical significance of isolated CTCs in cancer prognosis, treatment response, and recurrence,14,15 which enables minimally invasive and repetitive sampling and is clinically useful in early diagnosis, monitoring of therapeutic efficacy, and prognosis.
In recent years, CTCs have been approved by the FDA as a prognostic biomarker for monitoring patients with breast, prostate, lung, hepatocellular, and colorectal cancer.16–18 Several studies have shown that the detection of CTCs in GC patients might be used for staging and assessing treatment outcomes.19,20 However, the prognostic value of CTC in GC is still controversial, because the previous studies were almost retrospective studies, with a small sample size and high heterogeneity. Furthermore, two meta-analyses evaluated the clinical significance of CTCs in GC.21,22 However, samples of one meta-analysis were drawn from peripheral blood and bone marrow, with few studies. 21 Another meta-analysis may have been inaccurate because the included studies utilized different detection assays; moreover, it selectively reported their positive results with high heterogeneity, but without further subgroup analysis stratified by detection method, detection target, and treatment, which might be a source of heterogeneity, and led to unstable outcomes; therefore, we conducted subgroup analysis stratified by detection method, detection target, and treatment to avoid the heterogeneity due to different detection assays, targets, and treatment. 22 In addition, with many novel studies emerging, it was necessary to review the value of CTCs in predicting the prognosis of GC. Thus, this meta-analysis aims to further discuss the association between CTCs and the prognosis of GC.
Methods
We used MOOSE (Meta-analysis Of Observational Studies in Epidemiology) 23 and PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) 24 as guidelines to conduct the systematic review. A comprehensive search of relevant studies (English language) published from inception to October 2022 was retrieved in the following databases: PubMed, EMBASE, and the Cochrane Library. By using the terms ‘Circulating tumor cells’, ‘CTCs’, ‘Stomach neoplasm’, and ‘GC’, we also checked the bibliographies of all relevant papers for further qualified studies. The detailed search strategy for the electronic database is shown in Supplemental Material 1.
Eligibility criteria
Inclusion criteria
(1) Patients with pathologically confirmed GC including stage I–IV;
(2) Patients accepting surgery or perioperative chemotherapy were all included;
(3) Samples collected at baseline or intra/post-treatment were all included;
(4) Reverse transcription-polymerase chain reaction (RT-PCR), immunohistochemistry, immunofluorescence, flow cytometry, immunostaining, fluorescence in situ hybridization (FISH) staining, and CellSearch detection methods were used to detect tumor-specific genes/antigens.
(5) Sufficient GC patient survival data with CTC status;
(6) The most recent study or the study with the most subjects was chosen if overlapping research studies were found.
Exclusion criteria
(1) Samples were not drawn from peripheral blood (e.g. from urine or bone marrow);
(2) The histology type of the included GC patients was neuroendocrine carcinoma or other types of GC;
(3) Patients with other tumors;
(4) The outcomes of interest were not reported, and it was impossible to calculate outcomes from the originally published data;
Data extraction and assessment of study quality
Two authors independently reviewed the qualified studies and extracted the following information: author, region, publication year, study design, number of patients, gender ratio, age, cancer stage, treatment status, sample volume and time, follow-up, cutoff, HR estimate, outcomes (OS, DFS/RFS, PFS), target, and detection method. The risk of bias for individual studies was assessed using the Newcastle–Ottawa scale (NOS). 25 Disagreements were resolved by a joint review of the manuscript to reach a consensus.
Data synthesis and analysis
The HRs were used to evaluate the prognostic effect (OS, DFS/RFS, and PFS), which were extracted from multivariate Cox regression. Generally, an HR > 1 indicated a worse outcome of GC patients with positive detection of CTCs. If the HR and its variance were not reported directly in the original study, these values were calculated from available reported data using software designed by Tierney et al. 26 All statistical values were combined with a 95% confidence interval (95% CI). Pooled HRs and corresponding 95% CIs were used to evaluate the relationship between the level of CTCs and the prognosis of patients with GC by lnHR, selnHR. p < 0.05 was considered statistically significant. Heterogeneity between studies was assessed using Cochran’s Q-test and Higgins I-squared statistics to measure the extent of variation not due to chance alone. If I2 ⩽ 25%, it indicates low heterogeneity; if 25% <I2 ⩽ 50%, it indicates moderate heterogeneity; and if 50% <I2 ⩽ 75%, it indicates substantial heterogeneity. If I2 ⩾ 75%, it indicates considerable heterogeneity. If p < 0.05, a random-effects model was used; otherwise, the pooled analysis was performed with the fixed-effects model. In the presence of heterogeneity, subgroup analyses were performed to assess the prognostic value of CTCs based on characteristics such as sampling times, detection markers, CTC detection methods, treatment methods, tumor stage, region, and HR (Hazard Ratio) extraction methods (data were calculated or provided by the article). Sensitivity analysis was also performed by sequential omission of individual studies to assess the stability of the results. Publication bias was evaluated using funnel plots, Egger’s test, and Begg’s test. All statistical analyses were performed using STATA version 15.0. (College Station, TX 77845, USA).
Results
Search results and basic characteristics
In all, 28 eligible studies19,20,27–52 containing 2383 patients were enrolled for analysis (Figure 1). Table 1 shows the basic characteristics and experimental design variables of all included studies. All studies were performed using various cellular and molecular methods, including RT-PCR (n = 8), CellSearch (n = 8), immunohistochemistry (n = 2), immunofluorescent staining (n = 3), FISH (n = 2), chromosomal microarray analysis (CMA; n = 1), flow cytometry (n = 2), RNA in situ hybridization (RNA-ISH; n = 1), and ligand-targeted enzyme-linked polymerase chain reaction (LT-PCR; n = 1). The most common detection target was EpCAM+, CK+, CD45−, and DAPI+ by CellSearch, followed by CK, CEA, and other targets. Three studies were conducted in Western populations, and 25 studies were conducted in Asian populations.

Flow chart of selection studies and specific reasons for exclusion.
The basic characteristic and experimental design variables of included studies of meta-analysis.

CA, covariates adjusted; Chemo, chemotherapy; DFS, disease-free survival; ECOG PS, Eastern Cooperative Oncology Group performance status; FC, HR calculate by figure; HC: healthy control; M/F, male/female; ML, multivariate Cox regression; NR, not report; P, provided by article; PFS, progression-free survival; PM, peritoneal metastasis; PPMM, peripheral blood micrometastasis; Pre, preoperative; Post, postoperative; OS, overall survival; RFS, recurrence-free survival; ROC, receiver operating characteristic curve; RT-PCR, reverse transcription-polymerase chain reaction.
Quality evaluation of the included literature
The assessment of the risk of bias for individual studies showed 20 studies assessed with a score ⩾6 and the remaining eight studies were evaluated with a score <6. The quality evaluation of the included literature is shown in Table 2.
The quality assessment of included studies of meta-analysis.

AE, ascertainment of exposure; AF, additional factors; AO, assessment of outcome; AFU, adequacy of follow-up; DO, outcome not present at the start of the study; FU, length of follow-up; REC, representativeness of exposed cohort; SNEC, selection of nonexposed cohort; SC, control for important factors.
Association between CTCs and prognosis of GC
There were 22 articles (17 retrospective studies and five prospective studies) covering HR, and the pooled HR showed a significant association between CTCs and the OS of GC (HR = 1.933, 95% CI 1.657–2.256, p < 0.001), with low heterogeneity. (Figure 2). Furthermore, 11 studies (eight retrospective studies and three prospective studies) reported DFS/RFS, and the pooled HR revealed that CTCs were related to the DFS/RFS of GC patients (HR = 3.228, 95% CI 2.475–4.211, p < 0.001) with low heterogeneity. (Figure 3). Furthermore, the outcomes of five prospective studies showed that CTC detection was associated with poor OS (HR = 2.655 95% CI 1.831–3.850, p < 0.001) and three prospective studies showed that DFS/RFS (HR = 2.853, 95% CI 1.702–4.782, p < 0.001), with low heterogeneity. There were six articles reporting PFS, with a statistically significant pooled HR (HR = 3.272, 95% CI 1.970–5.435, p < 0.001) and low heterogeneity.

The meta-analysis forest plot of pooled OS.

The meta-analysis forest plot of pooled DFS/RFS.
The outcomes of subgroup analysis with OS
The subgroup analysis on the tumor stage (I + II/III + IV means a proportion of the total number of people in stage I and II to stage III and IV) of studies in the study revealed that higher CTCs were associated with shorter DFS/RFS (I + II/III + IV > 1: HR = 1.549, 95% CI: 1.206–1.991, p = 0.004; I + II/III + IV < 1: HR = 2.212; 95% CI: 1.819–2.691; p < 0.001; Tables 3 and 4). Furthermore, the subgroup analysis on the HR extraction method of studies in the study revealed that higher CTCs were associated with shorter OS (Figure calculation: HR = 1.717, 95% CI: 1.317–2.238, p < 0.001; extracted by multivariate Cox regression HR = 2.091, 95% CI: 1.736–2.520, p < 0.001). The subgroup analysis of CTC detection with different methods still showed a poor prognostic effect for GC patients. Higher CTCs predicted poorer OS of GC patients where CTCs were detected by CellSearch (HR = 2.299; 95% CI 1.764–2.996; p < 0.001), RT-PCR (HR = 1.586; 95% CI: 1.215–2.070; p = 0.001), and other methods (HR = 1.979; 95% CI 1.510–2.594; p < 0.001). In addition, the subgroup analysis of different detection targets showed a poor prognostic effect for GC patients of CTCs expressing CK (HR = 1.378; 95% CI: 0.990–1.943; p = 0.057); EpCAM+, CK+, CD45−, and DAPI+ (HR = 2.299; 95% CI: 1.764–2.996; p < 0.001); and EpCAM (HR = 2.881; 95% CI: 1.538–5.396; p = 0.001). The subgroup analysis on the sampling times of GC patients in the study revealed that higher CTCs significantly occurred with shorter OS both in pre-treatment (HR = 1.639 95% CI 1.288–2.085, p < 0.001), post-chemotherapy (HR = 3.382 95% CI 2.386–4.795, p < 0.001), post-surgery (HR = 1.591 95% CI 1.184–2.139, p = 0.002), and post-chemotherapy and surgery (HR = 2.122 95% CI 1.364–3.301, p = 0.001). The subgroup analysis on CTC detection showed a poor prognostic effect for GC patients from different regions. Higher CTCs predicted poorer OS for GC patients who are from Asian regions (HR = 1.986; 95% CI 1.694 to 2.329; p < 0.001), but without significant statistical difference for patients from No-Asian regions (HR = 1.251; 95% CI 0.662–2.365; p < 0.001). Furthermore, no significant differences were observed in the test for subgroup analysis (p > 0.05; Supplemental Table A).
The outcome of pooled OS of meta-analysis.

Chemo, chemotherapy; FC, HR calculate by figure; N, number of studies; P, provided by article; Pre, preoperative; Post, postoperative; OS, overall survival; RT-PCR, reverse transcription-polymerase chain reaction.
The outcome of pooled DFS/RFS of meta-analysis.
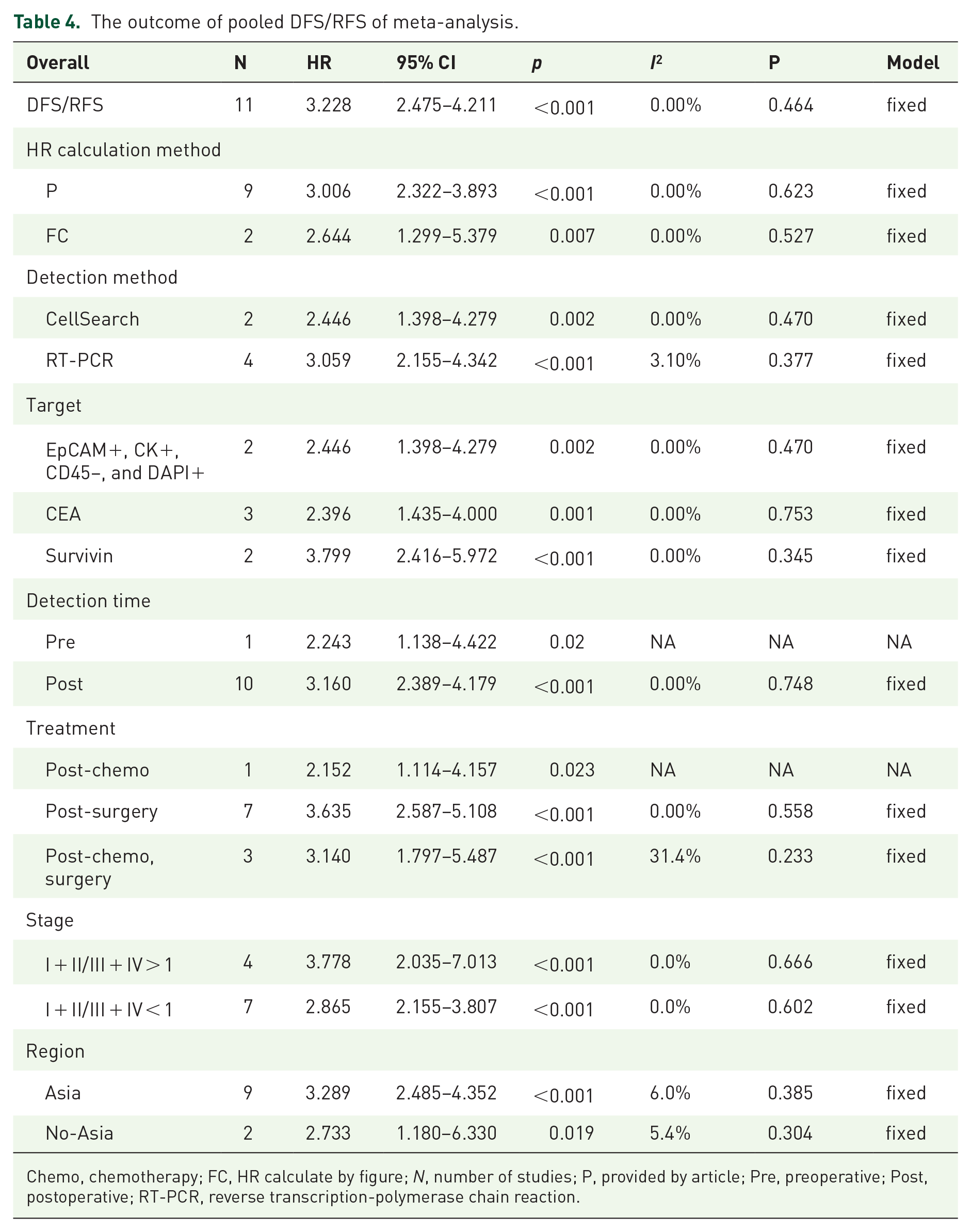
Chemo, chemotherapy; FC, HR calculate by figure; N, number of studies; P, provided by article; Pre, preoperative; Post, postoperative; RT-PCR, reverse transcription-polymerase chain reaction.
The outcomes of subgroup analysis with DFS/RFS
The subgroup analysis on the tumor stage of studies in the study revealed that higher CTCs were related to shorter DFS/RFS (I + II/III + IV > 1: HR = 3.778; 95% CI: 2.035–7.013, p < 0.001; I + II/III + IV < 1: HR = 2.865, 95% CI: 2.155–3.807, p < 0.001). Furthermore, the subgroup analysis on the HR extraction method of studies in the study revealed that higher CTCs were associated with shorter DFS/RFS (figure calculation: HR = 2.644, 95% CI: 1.299–5.379, p < 0.001; extracted by multivariate Cox regression: HR = 3.066, 95% CI: 2.322–3.893, p < 0.001). The subgroup analysis of CTC detection with different methods still showed a poor prognostic effect for GC patients. Higher CTCs predicted poorer DFS/RFS of GC patients where CTCs were detected by Cellserach (HR = 2.446; 95% CI 1.398–4.279; p = 0.002) and RT-PCR (HR = 3.059; 95% CI: 2.155–4.342; p < 0.001). In addition, the subgroup analysis of different detection targets showed a poor prognostic effect for GC patients of CTCs expressing CEA (HR = 2.396; 95% CI: 1.435–4.000; p = 0.001); EpCAM+, CK+, CD45−, and DAPI+ (HR = 2.446; 95% CI: 1.398–4.279; p = 0.002); Survivin (HR = 3.799; 95% CI: 2.416–5.972; p < 0.001). The subgroup analysis on the sampling times of GC patients in the study revealed that higher CTCs significantly occurred with shorter DFS/RFS (posttreatment; HR = 3.160 95% CI 2.389–4.179, p < 0.001). The subgroup analysis on CTC detection showed a poor prognostic effect for GC patients receiving different treatments. Higher CTCs predicted poorer DFS/RFS of GC patients when CTCs were detected post-surgery (HR = 3.635; 95% CI 2.587–5.108; p < 0.001), post-chemotherapy (HR = 2.152; 95% CI: 1.114–4.157; p = 0.001), and post-chemotherapy and surgery (HR = 3.140; 95% CI 1.797–5.487; p = 0.001). Higher CTCs predicted poorer DFS/RFS for GC patients who are from Asian regions (HR = 3.289; 95% CI 2.485–4.352; p < 0.001), and from No-Asian regions (HR = 2.733; 95% CI 1.180–6.330; p < 0.001). Furthermore, no significant differences were observed in the test for subgroup analysis (p > 0.05; Supplemental Table B).
Publication bias, sensitivity analysis
The funnel plots, Egger’s, and Begg’s tests were utilized to gauge publication bias for the studies related to OS (Egger’s p = 0.10 and Begg’s p = 0.05, respectively) and DFS/RFS (Egger’s p = 0.073 and Begg’s p = 0.731, respectively), which showed that there was no significant publication bias found in the HR of either OS (Figure 4(a)) or DFS/RFS (Figure 4(b)). Sensitivity analysis was performed by sequentially removing a study and then calculating its pooled HR and 95% CI. Furthermore, after excluding articles with NOS scores <6, the remaining studies concluded similar results. This demonstrates that after removing anyone, the HR of OS and DFS/RFS was always distributed within the original 95% CI, thus showing imperceptible variability between studies (Figure 5). This revealed that our outcomes were robust.

The meta-analysis funnel plot of OS and DFS/RFS: (a) the meta-analysis funnel plot of OS and (b) the meta-analysis funnel plot of DFS/RFS.

The sensitivity analysis of OS: (a) the sensitivity analysis of OS and (b) the sensitivity analysis of DFS/RFS.
Discussion
CTCs have recently been applied to provide further information to support clinical treatment response assessment for GC patients with poor prognosis despite R0 resection combined with perioperative chemotherapy. Our meta-analysis concluded that the detection of CTCs was associated with poorer OS and could act as a risk factor for relapse/metastasis in patients with GC during the follow-up period, with low heterogeneity and no publication bias. Furthermore, the meta-analysis of prospective studies reported similar results. CTCs cause tumor metastasis or recurrence because they flow with blood circulation to various tissues and organs throughout the body and then grow, causing distant metastasis of the tumor. 53 To better understand the relationship between CTCs and the prognosis of GC, subgroup analyses stratified by estimated HR, detection target, detection method, treatment, sample time, and stage were conducted. Our study included patients with stage I–IV, subgroup analysis showed that CTCs could predict both patients diagnosed at an early or advanced stages, which could assist the clinician to make appropriate treatment strategies for patients. Furthermore, we found that regardless of whether the data were calculated or provided by the article, the results of the association between CTCs and the OS and RFS/DFS of GC patients were consistent.
To date, several detection methods have been applied to detect CTCs; however, it remains challenging to identify CTC in whole blood due to their extremely low concentrations. Previous studies reported that a traditional method of isolating cells, such as flow cytometry, density gradient centrifugation, and immunocapture by magnetic beads, cannot detect rare CTCs.32,41,42 The CellSearch system has been approved by the US FDA for the detection of CTCs in patients with cancer and is the first and only system approved for these purposes with high specificity and reproducibility, but cannot capture cells that do not express EpCAM and cytokeratin on the cell membrane. 54 The use of real-time RT-PCR assays, based on mRNA expression, is an alternative to detect CTCs with high specificity and moderate sensitivity. Our study demonstrated that the two methods could both predict the prognosis of GC. However, there is still controversy about whether RT-PCR is superior to CellSearch.55,56 Furthermore, a novel CTC-biopsy detection system based on the principle of filtration and isolation of tumor cells according to cell size has been developed, with a 93% of CTC detection rate, which was more sensitive than CellSearch, several studies have reported the efficacy of filtration principle for CTC detection in breast cancer, 57 esophageal squamous cell carcinoma, 58 and renal cancer. 59 Chen et al. and his colleagues 60 used pre-established surface molecule-independent subtraction enrichment (SE)-iFISH strategy, maintained at high viability and suitable for primary tumor cell culture, to detect CTC for GC, with a detection rate of 33.3%. A previous study also reported that researchers used Cytelligen to detect CTCs. 52 In addition, few studies with small sample sizes reported other detection methods (CMA, flow cytometry, RNA-ISH, and LT-PCR). Thus, it was necessary to further validate the superiority of the detection methods. The most frequently detected targets of CTCs were EpCAM+, CK+, CD45−, and DAPI+ by CellSearch. Moreover, as cell surface markers for the detection of cancer cells, cytokeratins (CK8, CK18, and CK19) play an important role. Other detection targets, such as CEA, cMET, Survivin, and B7-H3, could be used to detect CTCs by RT-PCR. Our study showed that CTCs were associated with a poor prognosis of GC when CTCs were detected by EpCAM+, CK+, CD45−, and DAPI+, CEA and Survivin. Three articles reported the results of CK target, however, it showed no association between CTCs and prognosis of GC patients. As we know, the ToGA study showed that HER2-targeted therapy using trastuzumab has clinical benefits in patients with advanced HER2-positive GC. 61 Recently, one study demonstrated the potential clinical utility of trastuzumab combined with chemotherapy in patients with HER2-positive CTCs even if they are histologically HER2-negative. 62 Thus, the assessment of CTCs may be useful for monitoring real-time tumor response during HER2-targeted therapy, which required more high-quality studies to validate it. Our study showed that EpCAM was associated with poor OS. Furthermore, CEA, biomarkers in CellSearch and Survivin were associated with poor DFS/PFS. Miki et al. and colleagues 41 demonstrated that CEA-positive CTCs will be a clinically beneficial biomarker in patients with GC. Furthermore, a previous study 38 also reported detection of CTCs expressing Survivin mRNA could be a good clinical biomarker used to predict the prognosis of patients with curatively resected GC. There are relatively few studies on other targets, and more research is needed to explore the relationship between targets and the prognosis of GC. Thus, no matter what detection method and detection maker, our meta-analysis showed that higher CTCs were associated with poor OS and DFS/PFS for GC patients.
According to our study, there was a high risk of poor prognosis when CTCs were detected pre- or intra/during treatment. A previous study demonstrated that RFS and OS for patients with CTCs were significantly lower than those for patients without CTCs by preoperative peripheral blood sample. 39 A prospective study designed by Zhang et al. 40 evaluated preoperative and postoperative CTC changes in resectable GC and possible correlations with postoperative recurrence and found that preoperative CTCs were not correlated with the recurrence pattern. Patients with hematogenous metastases who had postoperative CTCs ⩾ 2/7.5 ml had markedly shorter DFS, which confirmed that postoperative CTCs may be a more direct marker of postoperative recurrence, especially in patients who experience hematogenous recurrence. Our study showed that postoperative HR has a better ability to predict OS (post; HR: 2.169 > pre; HR: 1.639), DFS/RFS (post; HR: 3.160> pre; HR: 2.243) than preoperative HR. Theoretically, a patient’s postoperative CTCs may be more responsive to the patient’s prognosis, as the surgical procedure may cause the spread of CTCs, thereby altering CTCs in the blood. Furthermore, the surgery itself can also change the survival of cancer patients. Thus, we suggest that the predictive ability of postoperative CTCs might be superior to that of preoperative CTCs. In addition, Liu et al. and colleagues reported that CTC count may be a good monitoring parameter for chemotherapy and an ideal prognostic parameter for palliative chemotherapy patients. 48 Neoadjuvant chemotherapy has been proven to reduce the tumor volume, reduce the tumor stage, and improve the R0 resection rate to improve the prognosis of patients.63,64 However, the relationship between the detection of CTCs in blood after neoadjuvant chemotherapy and tumor prognosis remains to be further verified due to the lack of sufficient research.
Recently, another liquid biopsy for GC, ctDNA, has been demonstrated to be associated with the diagnosis, treatment response, and prognosis of GC. 65 ctDNA and CTC may focus on different clinical applications due to their different sources, evaluation criteria, and biological characteristics. ctDNA has significant advantages in accurately reflecting tumor cell micro-metastases in the blood circulation and can effectively assess the state of tumor development, and therefore has a unique position in early screening, adjuvant diagnosis, recurrence monitoring, and efficacy assessment. In contrast, ctDNA is more focused on reflecting the variation of DNA within tumor cells, and thus it plays an important role in therapeutic decision-making (driver gene detection) and targeted drug resistance monitoring. CTC analysis provides complete genomic information of living cells, but ctDNA does not provide complete genomic and protein expression information of each cell. From a clinical perspective, CTC is mostly used for prognosis and recurrence monitoring; furthermore, the information obtained from simultaneous detection of CTC and ctDNA may be complementary. The coexistence of the two liquid biopsy technologies may be the future direction of development.65–67 Furthermore, the study showed that CTCs were associated with the poor DFS/RFS of GC when CTCs were detected for patients from Asian or No-Asian regions. In addition, higher CTCs predicted poorer OS for GC patients who are from Asian regions, but without significant statistical difference for patients from No-Asian regions, which might account for only three studies from No-Asian regions with a small sample size.
There were several limitations in our meta-analysis. First, the heterogeneity between our included studies with different detection methods, markers, and treatment had impact on our results. We did a careful subgroup analysis stratified by the above factors, and the results were consistent. Furthermore, to a certain extent, the effect value of pooled HR calculated by lnHR and selnHR could avoid some heterogeneity between different studies. Moreover, the whole heterogeneity of our research was slight according to the heterogeneity test. Furthermore, most included studies did not provide detailed data on stage I, II, III, IV separately, only providing mixed stages I–II, I–III, I–IV, II–IV, and so on, thus, we could not distinguish each stage of GC. In addition, the detection methods of CTCs have different sensitivities and specificities, which would cause bias in our research. For example, a panel of molecular markers can improve the sensitivity of CTC detection compared to the single markers being used. In addition, CTCs could be heterogeneous, and it remains unknown what subpopulations are best for predicting survival and monitoring treatment response. Future well-designed prospective studies with larger sample sizes should be conducted to answer these questions. Finally, we have tried our best and failed to register in PROSPERO. However, our meta-analysis was made in strict accordance with the PRISMA statement to ensure the quality of the article.
Conclusion
CTCs were associated with a poor prognosis in patients with GC. The use of CTCs in clinical practice is promising and could provide help for clinical decisions.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231183678 – Supplemental material for Association between circulating tumor cells in the peripheral blood and the prognosis of gastric cancer patients: a meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359231183678 for Association between circulating tumor cells in the peripheral blood and the prognosis of gastric cancer patients: a meta-analysis by Tao Jin, Pan-Ping Liang, Ze-Hua Chen, Feng-Jun He, Ze-Dong Li, Zheng-Wen Chen, Jian-Kun Hu and Kun Yang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359231183678 – Supplemental material for Association between circulating tumor cells in the peripheral blood and the prognosis of gastric cancer patients: a meta-analysis
Supplemental material, sj-docx-2-tam-10.1177_17588359231183678 for Association between circulating tumor cells in the peripheral blood and the prognosis of gastric cancer patients: a meta-analysis by Tao Jin, Pan-Ping Liang, Ze-Hua Chen, Feng-Jun He, Ze-Dong Li, Zheng-Wen Chen, Jian-Kun Hu and Kun Yang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
