Abstract
Background:
The combination of anti–programmed cell death-1 antibody with human epidermal growth factor receptor 2 (HER2)–targeted therapy and chemotherapy is widely used in the United States and Europe for HER2-positive advanced gastric cancer (AGC). Molecular profiles that predict the efficacy of this dual-target therapy are unclear.
Objectives:
To explore the clinical utility of circulating tumor DNA (ctDNA) as a predictive marker of the efficacy of standard chemotherapy plus HER2 and programmed death-ligand 1 dual-targeted therapy in patients with HER2-positive AGC.
Design:
Collaborative study of the Ni-High phase Ib clinical trial.
Methods:
A total of 21 patients with tissue-confirmed HER2-positive AGC who received chemotherapy with dual-targeted therapy (capecitabine/S-1, oxaliplatin, trastuzumab, and nivolumab) in a phase Ib clinical trial (UMIN000034222) were enrolled. The association of genomic profiles in plasma ctDNA with tissue HER2 amplification status and their correlation with clinical outcomes was investigated.
Results:
Among the 21 patients studied, 20 (95.2%) showed somatic alterations in ctDNA. ERBB2 amplifications and single-nucleotide variants (SNVs)/indels were found in 12 (57.1%) and 3 (14.3%) patients, respectively. Significant associations between maximum mutant allele frequency (mMAF) and tumor size and between ctDNA and tissue ERBB2 copy numbers were found. Patients without ERBB2 SNV/indels showed longer median progression-free survival (PFS) and overall survival (OS) than those with these alterations. Patients with focal ERBB2 amplification in ctDNA showed better outcomes than those with aneuploidy (median PFS: 20.8 vs 8.4 months, hazard ratio (HR) = 0.08; median OS: NA vs 14.8 months, HR = 0.077). Lower mMAF at cycle 2 was associated with a better response to chemotherapy with dual-targeted therapy.
Conclusion:
ERBB2 genetic status and mMAF changes in ctDNA may, respectively, predict and reflect the efficacy of chemotherapy with dual-targeted therapy in HER2-positive AGC.
Trial registration:
UMIN000034222
Keywords
Introduction
Human epidermal growth factor receptor (HER) 2 is a 185 kDa glycoprotein and a transmembrane receptor-type tyrosine kinase. 1 HER2 has no endogenous ligands but forms homodimers with HER2 and heterodimers with epidermal growth factor receptor (EGFR), HER3, and HER4, which results in its phosphorylation. 2 The gene encoding HER2 (also known as ERBB2) is located on the long arm of chromosome 17, and genetic alterations, such as amplification, result in HER2 protein overexpression on the plasma membrane, which plays an important role in the growth and progression of HER2-positive various cancers. 3 ERBB2 gene amplification and protein overexpression are observed in 10%–20% of breast and gastric cancer cases. 4
In the global phase III clinical trial ToGA, which evaluated the survival benefit of adding trastuzumab to chemotherapy with fluoropyrimidine (capecitabine or intravenous 5-fluorouracil) and cisplatin as first-line treatment for patients with HER2-positive advanced gastric cancer (AGC), the overall survival (OS) in the chemotherapy plus trastuzumab arm was significantly longer than that of the chemotherapy alone arm (median = 13.8 vs 11.1 months, hazard ratio (HR) = 0.74, 95% confidence interval (CI) = 0.60–0.91, p = 0.0046). 5 Consequently, trastuzumab plus chemotherapy is the standard of care as the first-line treatment for patients with HER2-positive AGC. Several phase II trials on chemotherapy containing trastuzumab combined with S-1 and cisplatin/oxaliplatin have been conducted in Japan; their results showed that the median survival time was approximately 16 months, and the response rates were 61.0%–82.1%.6 –8
The expression of programmed death-ligand 1 (PD-L1), a candidate biomarker for the anti–programmed cell death-1 (PD-1) antibody, was found in over 80% of patients with HER2-positive AGC. 9 Several preclinical studies in mice have demonstrated the synergistic effects of anti-HER2 and anti-PD-1 antibodies. 10 Therefore, combining trastuzumab, which has ADCC activity, and nivolumab, which is an anti-PD-1 antibody, may enhance the therapeutic effect of trastuzumab combination chemotherapy in patients with HER2-positive AGC. Based on the aforementioned data, a phase III trial (KEYNOTE-811) was conducted in which the addition of an anti-PD-1 antibody showed a significantly increased response rate and longer progression-free survival (PFS) and OS than the standard of care. 11
Although the effectiveness of treatment is increasing, the cost of medical care is also rising, causing a significant burden on the healthcare economy. To date, considerable efforts have been devoted to the exploration of biomarkers for trastuzumab combination chemotherapy, and several candidates have been proposed12 –18; however, data on chemotherapy using HER2 and PD-1 dual-targeted therapy are limited.17,18 For the benefit of patients and the medical economy, the chemotherapeutic efficacy and resistance to chemotherapy with HER2 and PD-1 dual-targeted therapy should be detected early.
AGC is a heterogeneous tumor, thus making re-biopsy difficult and hindering the assessment of changes in the molecular status of the tumor. Recent advances in liquid biopsy (LB), particularly for circulating tumor DNA (ctDNA), may overcome this problem. 19 LB, which is simpler, less invasive, and more repeatable than tissue biopsy, reflects the genetic information of the entire tumor, including heterogeneity. 19 Furthermore, artificial intelligence (AI) has the potential to greatly improve the accuracy and speed of disease detection and prognosis by utilizing these cutting-edge technologies. 20 However, few reports have evaluated the factors related to treatment efficacy and resistance by collecting samples over time, particularly in patients with HER2-positive AGC.21 –24 The current study aimed to explore the clinical utility of ctDNA using the Guardant360® assay in patients with HER2-positive AGC who were treated with standard chemotherapy plus HER2 and PD-1 dual-targeted therapy.
Methods
Patients
This is a collaborative study of a phase Ib trial of chemotherapy combined with anti-HER2 and PD-1 dual-targeted therapy (UMIN000034222, Ni-High). 25 Among the 42 patients with chemotherapy-naïve, histopathologically confirmed HER2-positive AGC who were enrolled in this trial between March 2019 and September 2020, 27 patients provided informed consent to participate in this biomarker study. Six patients were excluded because ctDNA could not be collected due to the coronavirus disease 2019 pandemic. Finally, the ctDNA of 21 patients was evaluated. Although excluded patients had similar baseline demographics, disease stage, and HER2 status compared to included patients (Supplemental Table 1), this represents a subset analysis of consenting participants. This study received approval from the Institutional Review Board of the Japanese Foundation for Cancer Research (2018-GA-1137), Tokyo, Japan.
Tumor IHC
The HER2 status of tumors was evaluated using IHC (HercepTest®, Dako, Denmark) and fluorescence in situ hybridization (FISH) (HER2 FISH pharmDx®, Dako). Patients were considered HER2 positive if the tumor was scored as 3+ on IHC or had a centromere enumeration probe 17 ratio of ⩾2 by FISH in case of a HER2 score of 2+. 26
Blood samples, ctDNA isolation, and sequencing
Blood samples (20 ml × 2) were collected in a cell-free DNA blood collection tube (Streck) just before the initiation of cycles 1 and 2 and at progressive disease (PD). The samples were then sent to the central laboratory. The next-generation sequencing of ctDNA was performed using Guardant360® (Guardant Health), in which 5–30 ng of cell-free DNA isolated from plasma was labeled with non-redundant oligonucleotides (“molecular barcoding”), enriched using targeted hybridization capture, and sequenced on the NextSeq 550 platform (Illumina). 27 This assay detects single-nucleotide variants (SNVs), insertions–deletions (indels), fusions, and copy number alterations in 74 genes 27 and can distinguish between focal amplification and aneuploidy. Focal amplification was defined as the amplification of a specific gene without the amplification of nearby genes (copy number > 4.0). Aneuploidy amplification was defined as genes amplified in the context of a larger chromosomal amplification event. 28 Base call files generated using RTA software version 2.12 (Illumina) were demultiplexed using bcl2fastq version 2.19 and then processed using a custom pipeline for molecular barcode detection, sequencing adapter trimming, and base quality trimming. Processed reads were aligned to hg19 using the Burrows–Wheeler aligner maximal exact match algorithm (arXiv:1303.3997v2). Finally, the ctDNA fraction was determined from the maximum allelic variant fraction.
Evaluation and statistical analysis
The response was evaluated according to the RECIST guidelines (v1.1). The objective response rate (ORR) was calculated as the proportion of patients showing complete response (CR) or partial response (PR) among those with measurable disease, and the disease control rate (DCR) was that of patients showing CR, PR, or stable disease (SD). PFS was defined as the time from initiation of the first-line chemotherapy to either disease progression or death from any cause, and OS was defined as the time from initiation of the first-line chemotherapy to death from any cause. Continuous variables were analyzed using the Mann–Whitney U test, and between-group comparisons were conducted using the χ2 test and Fisher’s exact test. Statistical tests provided two-sided p values, with a significance threshold set at p < 0.05. Statistical analyses were performed using EZR (Easy R), which is based on R and R commander (R 4.3.1 and R commander 2.9-1). 29
Results
Patient characteristics
The characteristics of the 21 patients in the study are summarized in Table 1. The median age was 67.5 years (range: 38–81 years), and 18 patients (85.7%) were male. Lymph nodes were the most frequent sites of metastasis (81.0%), followed by the liver (38.1%), lungs (33.3%), and peritoneum (19.0%). Eighteen (90.5%) patients had differentiated adenocarcinoma, and 15 patients had a HER2 status of IHC 3+ (71.4%; Table 1).
Patient demographics and clinical characteristics.

CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; ECOG PS, Eastern Cooperative Oncology Group Performance Status; FISH, fluorescence in situ hybridization; IHC, immunohistochemistry.
Detection of somatic alteration in plasma
Somatic alterations were detected in the ctDNA of 20 out of 21 (95.2%) patients: SNV or indels in TP53, APC, ERBB2, and CDK12 detected in 17 (81.0%), 5 (23.8%), 3 (14.3%), and 3 (14.3%) patients, respectively, whereas those in KRAS (9.5%), RAF1 (9.5%), ARAF (9.5%), ARID1A (9.5%), BRCA1 (4.8%), SMAD4 (4.8%), RHOA (4.8%), PIK3CA (4.8%), NTRK1 (4.8%), MAPK1 (4.8%), HRAS (4.8%), and KIT (4.8%) were less common (Figure 1(a)). Copy number variations (CNVs) in ERBB2, CCNE1, MYC, and EGFR were detected in 12 (57.1%), 7 (33.3%), 5 (23.8%), and 4 (19.0%) patients, respectively, whereas those in MET (9.5%), BRAF (4.8%), CCND2 (4.8%), CDK6 (4.8%), ESR1 (4.8%), and FGFR1 (4.8%) were less common (Figure 1(b)). Alterations that were newly detected at the time of PD are listed in Table 2. The copy numbers of ERBB2 in the tissue and those in ctDNA were significantly correlated (r = 0.66, p = 0.019), but TMBs were not (r = 0.27, p = 0.39; Supplemental Figure 1).

(a) Frequencies of mutated genes in circulating tumor DNA (ctDNA) of 20 patients with advanced gastric cancer (AGC). (b) Types of CNV and (C) copy number distribution.
New alterations acquired during progressive disease.
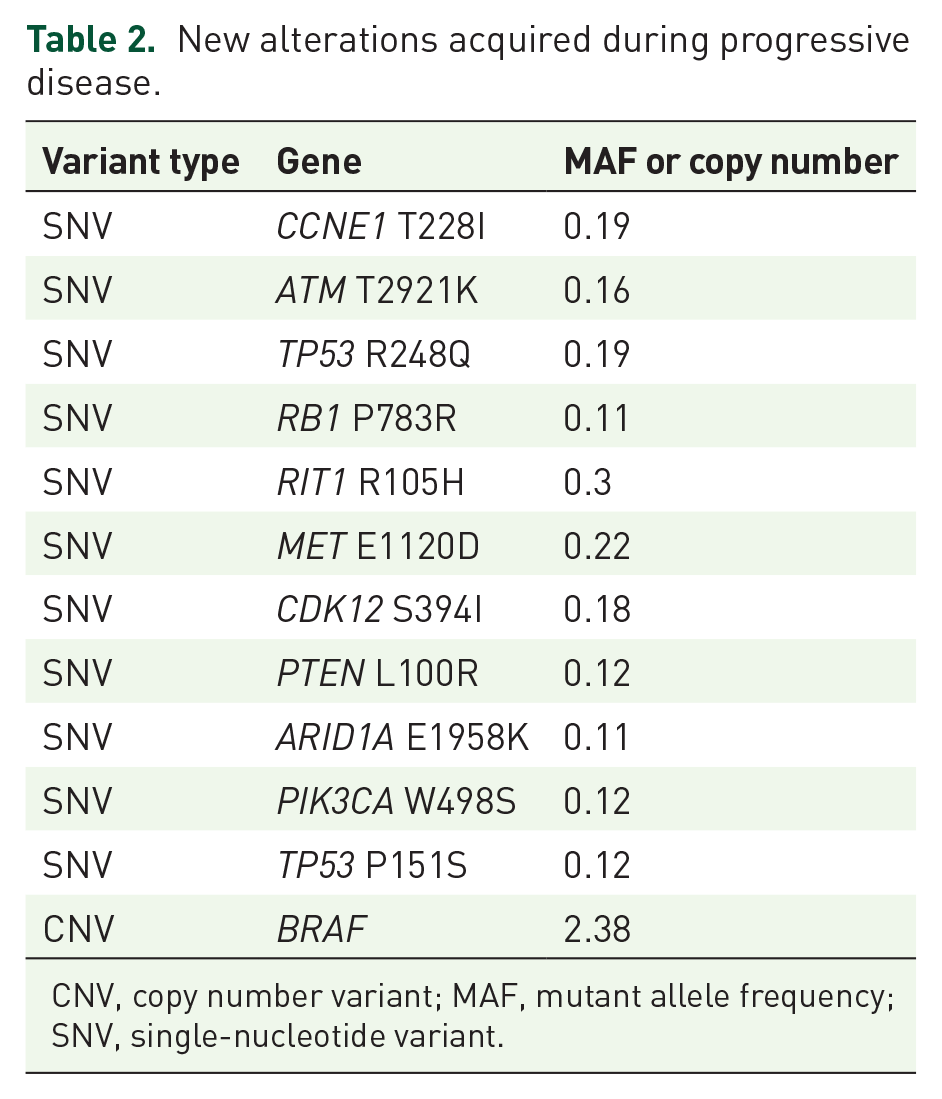
CNV, copy number variant; MAF, mutant allele frequency; SNV, single-nucleotide variant.
Association of clinical factors with ctDNA in patients with AGC
Patients with liver metastasis tended to exhibit a higher maximum mutant allele frequency (mMAF) than those without liver metastasis (p = 0.08; Table 3). The sum of tumor diameters was significantly correlated with mMAF (p = 0.00002; Table 3). However, no association was found between ERBB2 copy numbers in ctDNA and any clinical factors.
Association of clinical factors with maximum MAF and ERBB2 copy number in plasma.
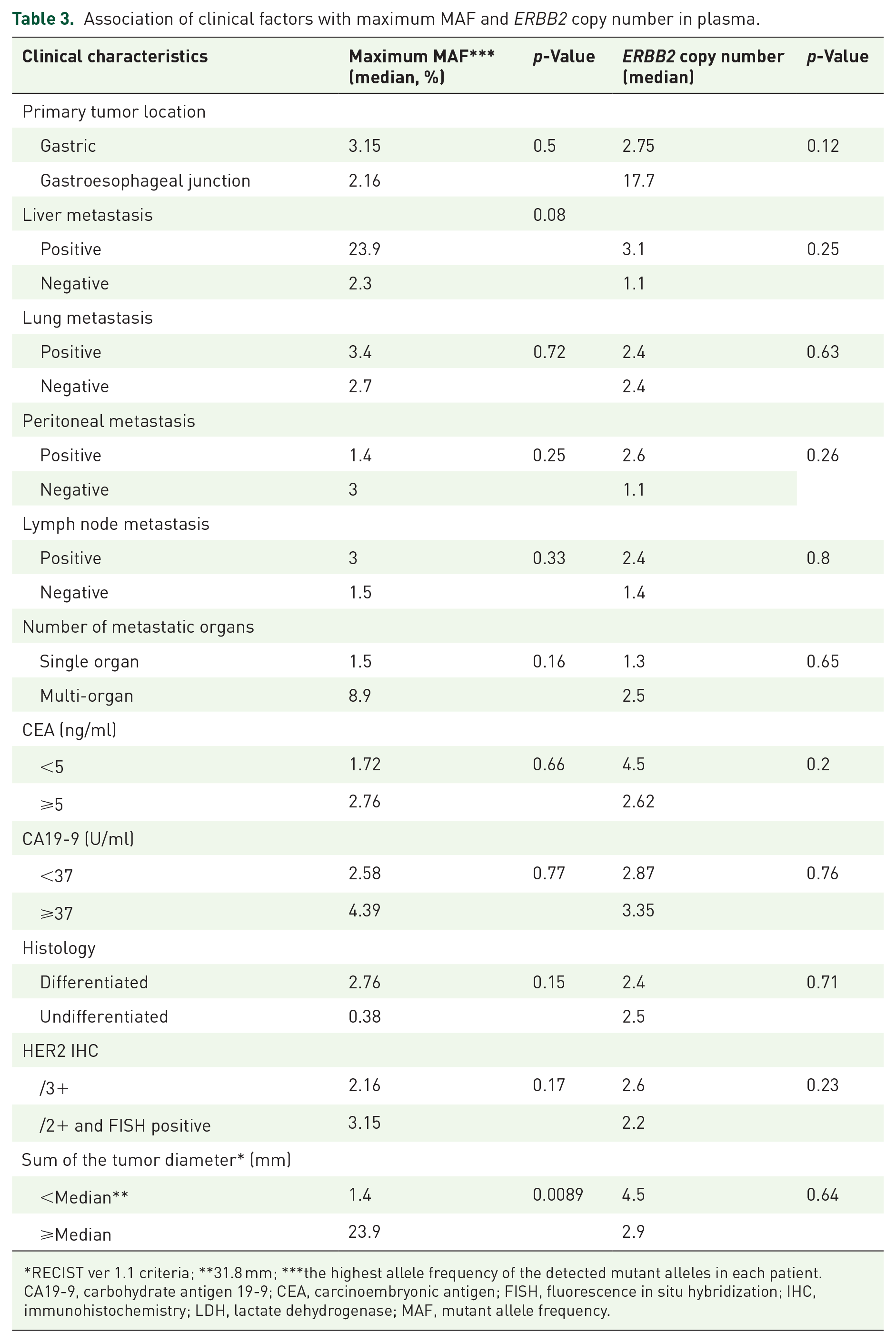
RECIST ver 1.1 criteria; **31.8 mm; ***the highest allele frequency of the detected mutant alleles in each patient.
CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; FISH, fluorescence in situ hybridization; IHC, immunohistochemistry; LDH, lactate dehydrogenase; MAF, mutant allele frequency.
Association of molecular factors with PFS and OS in plasma
The median PFS was 13.75 months (2.6–38.9), and the median OS was 20.5 months (6.1–40.0; Table 1).
Patients without ERBB2 mutations (SNV or indels; n = 17) showed a significantly longer PFS (median PFS: 20.8 months vs 8.4 months, p = 0.028, HR = 0.24) and OS (median OS: NA vs 14.0 months, p = 0.037, HR = 0.18) than those with ERBB2 mutations (n = 3; Figures 2(a) and (b)). Moreover, among patients with baseline ERBB2 CNV in ctDNA (n = 12), those with focal ERBB2 gene amplification (n = 9) showed a significantly longer PFS (median PFS: 20.8 months vs 8.4 months, p = 0.007, HR = 0.08) and OS (median OS: NA vs 14.8 months, p = 0.0046, HR = 0.077) than those with ERBB2 aneuploidy (n = 3; Figures 2(c) and (d)). Patients without APC SNV or indels (n = 16) showed a significantly longer PFS than those with APC SNV or indels (n = 4) (median PFS: 20.8 months vs 10.3 months, P = 0.049, HR = 0.30), but OS did not differ significantly between the two groups (median OS: NA vs 21 months, p = 0.69, HR = 0.71; Supplemental Figure 2). Moreover, the effect on PFS and OS of other alterations detected in three or more patients (MYC, CCNE1, TP53, CDK12, and EGFR) is shown in Supplemental Figures 2–4.
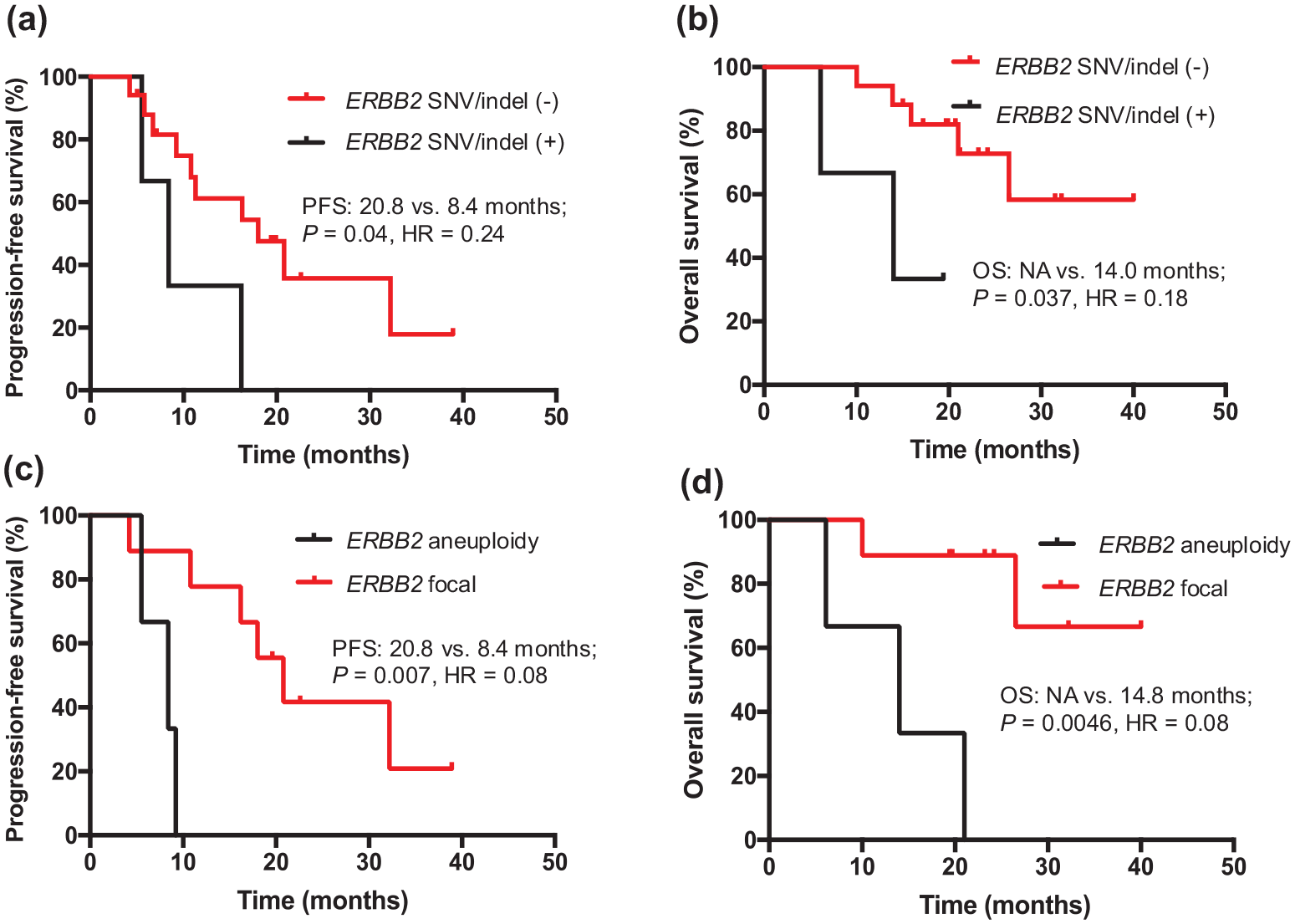
Genomic status of ERBB2 (a,b : with SNV/Indel or without, c,d: with focal amplification or with aneuploidy) in pretreatment ctDNA and progression-free survival (PFS)/overall survival (OS).
Association between mMAF and ERBB2 copy numbers and clinical response before and after chemotherapy
The ORR and DCR were 76.2% and 100% (CR = 1, PR = 15, SD = 5), respectively. Both the mMAF (18.4% vs 5.25, p = 0.068) and ERBB2 (9.33 vs 1.45, p = 0.075) copy number at baseline tended to be lower than those before the start of cycle 2 (Figures 3(a) and (b)). By contrast, the mMAF at PD increased compared with that prior to the start of cycle 2 (5.25% vs 12.16, p = 0.28; Figure 3(a)), whereas the ERBB2 copy number at PD remained low compared with that before the start of cycle 2 (1.45 vs 1.25, p = 0.83: Figure 3(b)). The mMAF at each point according to the response is shown in Figure 4(a). Although no significant difference was observed in the mMAF, at baseline (p = 0.30), and PD (p = 0.97) between the patients showing CR/PR and SD, patients achieving CR/PR showed significantly lower mMAF at two cycles (p = 0.0087). No significant differences were observed in ERBB2 copy numbers between the patients showing CR/PR and SD at any point (baseline: p = 0.30; cycle 2: p = 0.58; PD: p = 0.43; Figure 4(b)).

Changes in the maximum mutant allele frequency (mMAF) of SNV (a) and ERBB2 copy number (b) in ctDNA during treatment.

The mMAF of SNV (a) and ERBB2 copy number (b) in ctDNA before and after chemotherapy and clinical response.
Discussion
In the context of the limited data on ctDNA in HER2-positive gastric cancer, we obtained several findings from this study. In patients with HER2-positive AGC, ctDNA was detected in over 90% of patients, and ERBB2 amplification was detected in 52%–57% of patients with HER2-positive tissue at diagnosis.30,31 Similar to the results of previous reports, we also observed a highly sensitive detection of ctDNA in plasma (95.2%) and a comparable frequency of ERBB2 amplification (57.1%), thus suggesting that this assay for LB may be feasible in patients with HER2-positive AGC and that the results of the present analysis are valid.
Patients with focal ERBB2 gene amplification detected in ctDNA (n = 9) had significantly longer PFS and OS than those with ERBB2 aneuploidy (n = 3). Previous studies on breast cancer have demonstrated that IHC scores of 2+ and 3+, which exhibit discordance between IHC and FISH outcomes, are attributable to chromosome (chr) 17 aneuploidy. This chromosomal anomaly is prevalent in cases showing equivocal HER2 protein expression and gene amplification and in those with conflicting HER2 protein and gene copy numbers. 32 Tumors featuring chr 17 aneuploidy without ERBB2 amplification more closely resemble HER2-negative tumors than HER2-positive ones. 32 HER2 overexpression by IHC in the absence of gene amplification may be due to chromosome 17 aneuploidy, various molecular epigenetic, and post-transcriptional events. 33
Treatment response was poor in IHC 3+ and aneuploid cases (best response: SD; PFS = 8.4 months; OS = 14 months). ERBB2 aneuploidy was associated with worse clinical outcomes because the ERBB2 copy numbers in the group with aneuploidy were lower than those in the focal amplification group (median: 2.37 (2.19–2.62) vs 4.50 (2.51–47.74), p = 0.033). In general, ERBB2 copy number correlates with treatment response to anti-HER2 mAbs,34 –36 and similar results have been observed in HER2-positive metastatic colorectal cancer treated with trastuzumab derxutecan. 34 By contrast, focal ERBB2 gene amplification showed a significantly longer PFS and OS in this study. According to analyses in The Cancer Genome Atlas of 5225 tumor samples from 12 cancer types, tumors with high levels of CNVs showed an elevated expression of cell cycle and cell proliferation markers (cell cycle signature) and a reduced expression of markers for cytotoxic immune cell infiltrates (immune signature). Notably, the increased expression level of the cell cycle signature was primarily predicted by focal amplification. 35 These findings underscore the importance of considering not only HER2 IHC status but also ERBB2 amplification status and copy number to predict the treatment response to chemotherapy with dual-targeted therapy against HER2 and PD-1.
In this study, patients without ERBB2 mutations (SNV or indels; n = 17) also had significantly longer PFS and OS than those with ERBB2 mutations (n = 3). Activating mutations in HER2 in multiple solid cancers play an oncogenic role similar to that of ERBB2 amplification. 36 An overall ERBB2 mutation frequency of 3% was reported in a pan-cancer study (MSK-IMPACT) involving >10,000 tumor samples. 37 ERBB2 amplification and mutations are mutually exclusive, thus indicating that the majority of ERBB2 mutations in several solid cancers have an oncogenic nature, thereby making them actionable targets. 38 ERBB2 mutation may also serve as an independent biomarker for the poor treatment response to study therapy in some solid tumors. 39 The ERBB2 mutations detected in this study were V314F, V910V, and G776delinsVC. Previous reports have shown three resistance mechanisms have been shown, namely, secondary mutations in the ERBB2 kinase domain, overexpression and heterodimerization of the other receptor, EGFR, ERBB3, and activation of the PIK3CA/AKT/mTOR pathway. An ERBB2 mutation caused resistance to anti-estrogen treatment in estrogen receptor-positive breast cancer cells, which could be reversed by neratinib treatment.35,36 In addition, the co-occurrence of ERBB2 mutation and amplification was associated with poor response to trastuzumab or lapatinib treatment. 40 Targeting ERBB2 mutations in solid tumors remains an ongoing challenge. The low prevalence of ERBB2 mutations, differentiating drivers from passenger alterations, and identifying appropriate treatments (monoclonal antibody vs tyrosine kinase inhibitors, antibody–drug conjugate, or a combination) are significant challenges in effectively targeting ERBB2 mutations.
In patients who achieved a CR or PR (n = 16), the mMAF 3 weeks after the initiation of chemotherapy was significantly lower than that in patients who achieved SD (n = 4; p = 0.0087). Notably, MAF is strongly correlated with the initial tumor burden, as estimated from RECIST, thus suggesting that tumor volume is significantly related to ctDNA detection in the plasma. Several reports have demonstrated the effect of ctDNA changes after chemotherapy on the clinical outcomes of patients with not only mCRC but also other cancers.41 –43 A meta-analysis of the association between early dynamics in ctDNA levels and PFS/OS showed that the largest decrease in ctDNA levels was associated with a longer PFS and OS (PFS: pooled HR = 3.0, 95% CI = 2.2–4.2; OS: pooled HR = 2.8, 95% CI = 2.1–3.9). 44 In this study, changes in MAF were not associated with those in PFS/OS because of the small sample size, and early changes in MAF may serve as a biomarker to predict therapeutic efficacy and clinical outcomes in patients with AGC. Furthermore, if the cost of a single ctDNA measurement becomes low enough to allow multiple measurements, as is the case with tumor markers, it will be possible to determine the optimal measurement point, which is a future challenge.
ERBB2 copy number was not associated with tumor burden, and ERBB2 copy number dynamics were not associated with tumor response. In patients whose ERBB2 amplification could be measured before and after treatment, 5 out of 11 (45.5%) had undetectable ERBB2 amplifications in PD. These results indicate that the ERBB2 amplification clone may have been eliminated immediately after initiating trastuzumab and that resistance was driven by other non-ERBB2 amplification clones. 45 In addition, several studies have suggested that multiple genes such as FGFR, MET, MYC, STAT3, PI3K, ARID1A, AXL, ATM, mTOR/TSC2, and SRC may be involved in trastuzumab resistance. 45 Similar to these studies, the current study confirmed the resistance-related molecular alterations that occurred during treatment (Table 2). These molecular alterations could be responsible for resistance to trastuzumab therapy.
In AGC, shifts in HER2 status from HER2 positive to HER2 negative have been reported in approximately 40% of cases after first-line trastuzumab treatment.46 –48 Such changes in HER2 status could serve as a prognostic indicator for subsequent anti-HER2 therapy, underscoring the necessity to reassess HER2 status periodically.46,47 In the present study, there was no difference in treatment efficacy in the study treatment between the HER2-undetectable (n = 5) and HER2-detectable (n = 6) groups at PD. However, to date, the potential benefits of continued trastuzumab as a second-line therapy have been explored in the T-ACT study, which was a randomized phase II clinical trial conducted in Japan. 48 However, the definitive efficacy of prolonged trastuzumab therapy remains unproven. Moreover, a subgroup analysis has indicated a trend toward better outcomes with trastuzumab plus paclitaxel in patients with a trastuzumab-free interval (TFI) of ⩾30 days. 48 This trend is believed to result from a shift from HER2-negative back to positive, which can be attributable to the TFI; however, clear evidence for this phenomenon in gastric cancer is still lacking. In this context, a prospective observational study (jRCTs031240055, EN-MARK) is currently underway to assess the efficacy of trastuzumab deruxtecan (T-DXd) in the tertiary treatment of patients with advanced or recurrent HER2-positive gastric cancer and to investigate its correlation with biomarker shifts post-primary treatment. By gathering data on biomarkers after primary treatment, this study aims to elucidate the role of T-DXd in tertiary treatment. Based on these findings, it is important to assess HER2 status prior to each line of therapy to determine eligibility for anti-HER2 treatment.
Currently, reports on HER2-positive advanced cancer and AI indicate that IHC assessments by pathologists and AI are equivalent 49 ; however, there are few reports on the association with ctDNA. In the future, it may be possible to objectively determine HER2 positivity using AI and to predict the HER2 status following HER2 inhibitor administration based on information from pre-treatment samples. This could contribute to treatment strategies for HER2-positive AGC.
This study has a few limitations. First, although this study is a prospective multicenter study, the sample size is too small to make definitive conclusions. Therefore, further validation in a cohort with a larger number of cases is necessary. Second, given that this study was exploratory in nature without prior hypothesis, it cannot be denied that some of the findings were obtained by chance. Finally, considering that we do not have any data on trastuzumab-based chemotherapy, we cannot distinguish whether these results are specific to trastuzumab or nivolumab. Therefore, the factors that caused the differences in treatment outcomes, such as mMAF dynamics, ERBB2 mutations, in this study cannot be definitively identified as predictive factors; they may also have prognostic significance. Further validation in a larger cohort with a control arm will clarify whether these factors are predictive or prognostic. Nonetheless, the findings of this study could provide valuable insights into identifying biomarkers for combined anti-HER2 and anti-PD-1 antibody with chemotherapy in HER2-positive gastric cancer. Further research involving a larger cohort is necessary to confirm the hypothesis generated.
ctDNA is highly detectable in HER2-positive AGC and may be useful for early prediction of treatment efficacy. ERBB2 focal amplification in ctDNA is a prognostic marker for improved outcomes with anti-HER2 therapy, whereas ERBB2 mutations are associated with reduced efficacy.
Supplemental Material
sj-jpeg-2-tam-10.1177_17588359251367344 – Supplemental material for Clinical relevance of circulating tumor DNA in HER2-positive advanced gastric cancer: a collaborative study of a phase Ib trial of dual HER2 and PD-1 targeted therapy (Ni-High)
Supplemental material, sj-jpeg-2-tam-10.1177_17588359251367344 for Clinical relevance of circulating tumor DNA in HER2-positive advanced gastric cancer: a collaborative study of a phase Ib trial of dual HER2 and PD-1 targeted therapy (Ni-High) by Hiroki Osumi, Takeru Wakatsuki, Akira Ooki, Keisho Chin, Hirokazu Shoji, Mariko Ogura, Izuma Nakayama, Noriko Yamamoto, Hidekazu Hirano, Hiroki Hara, Keiko Minashi, Eiji Shinozaki, Ken Kato, Naoki Ishizuka, Shigehisa Kitano, Kengo Takeuchi, Narikazu Boku, Kensei Yamaguchi and Daisuke Takahari in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpeg-3-tam-10.1177_17588359251367344 – Supplemental material for Clinical relevance of circulating tumor DNA in HER2-positive advanced gastric cancer: a collaborative study of a phase Ib trial of dual HER2 and PD-1 targeted therapy (Ni-High)
Supplemental material, sj-jpeg-3-tam-10.1177_17588359251367344 for Clinical relevance of circulating tumor DNA in HER2-positive advanced gastric cancer: a collaborative study of a phase Ib trial of dual HER2 and PD-1 targeted therapy (Ni-High) by Hiroki Osumi, Takeru Wakatsuki, Akira Ooki, Keisho Chin, Hirokazu Shoji, Mariko Ogura, Izuma Nakayama, Noriko Yamamoto, Hidekazu Hirano, Hiroki Hara, Keiko Minashi, Eiji Shinozaki, Ken Kato, Naoki Ishizuka, Shigehisa Kitano, Kengo Takeuchi, Narikazu Boku, Kensei Yamaguchi and Daisuke Takahari in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpeg-4-tam-10.1177_17588359251367344 – Supplemental material for Clinical relevance of circulating tumor DNA in HER2-positive advanced gastric cancer: a collaborative study of a phase Ib trial of dual HER2 and PD-1 targeted therapy (Ni-High)
Supplemental material, sj-jpeg-4-tam-10.1177_17588359251367344 for Clinical relevance of circulating tumor DNA in HER2-positive advanced gastric cancer: a collaborative study of a phase Ib trial of dual HER2 and PD-1 targeted therapy (Ni-High) by Hiroki Osumi, Takeru Wakatsuki, Akira Ooki, Keisho Chin, Hirokazu Shoji, Mariko Ogura, Izuma Nakayama, Noriko Yamamoto, Hidekazu Hirano, Hiroki Hara, Keiko Minashi, Eiji Shinozaki, Ken Kato, Naoki Ishizuka, Shigehisa Kitano, Kengo Takeuchi, Narikazu Boku, Kensei Yamaguchi and Daisuke Takahari in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpeg-5-tam-10.1177_17588359251367344 – Supplemental material for Clinical relevance of circulating tumor DNA in HER2-positive advanced gastric cancer: a collaborative study of a phase Ib trial of dual HER2 and PD-1 targeted therapy (Ni-High)
Supplemental material, sj-jpeg-5-tam-10.1177_17588359251367344 for Clinical relevance of circulating tumor DNA in HER2-positive advanced gastric cancer: a collaborative study of a phase Ib trial of dual HER2 and PD-1 targeted therapy (Ni-High) by Hiroki Osumi, Takeru Wakatsuki, Akira Ooki, Keisho Chin, Hirokazu Shoji, Mariko Ogura, Izuma Nakayama, Noriko Yamamoto, Hidekazu Hirano, Hiroki Hara, Keiko Minashi, Eiji Shinozaki, Ken Kato, Naoki Ishizuka, Shigehisa Kitano, Kengo Takeuchi, Narikazu Boku, Kensei Yamaguchi and Daisuke Takahari in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-1-tam-10.1177_17588359251367344 – Supplemental material for Clinical relevance of circulating tumor DNA in HER2-positive advanced gastric cancer: a collaborative study of a phase Ib trial of dual HER2 and PD-1 targeted therapy (Ni-High)
Supplemental material, sj-pdf-1-tam-10.1177_17588359251367344 for Clinical relevance of circulating tumor DNA in HER2-positive advanced gastric cancer: a collaborative study of a phase Ib trial of dual HER2 and PD-1 targeted therapy (Ni-High) by Hiroki Osumi, Takeru Wakatsuki, Akira Ooki, Keisho Chin, Hirokazu Shoji, Mariko Ogura, Izuma Nakayama, Noriko Yamamoto, Hidekazu Hirano, Hiroki Hara, Keiko Minashi, Eiji Shinozaki, Ken Kato, Naoki Ishizuka, Shigehisa Kitano, Kengo Takeuchi, Narikazu Boku, Kensei Yamaguchi and Daisuke Takahari in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
