Abstract
Neuroendocrine neoplasms (NENs) are initially monoclonal neoplasms that progressively become polyclonal, with very different genotypic and phenotypic characteristics leading to biological differences, including the Ki-67 proliferation index, morphology, or sensitivity to treatments. Whereas inter-patient heterogeneity has been well described, intra-tumor heterogeneity has been little studied. However, NENs present a high degree of heterogeneity, both spatially within the same location or between different lesions, and through time. This can be explained by the emergence of tumor subclones with different behaviors. These subpopulations can be distinguished by the Ki-67 index, but also by the expression of hormonal markers or by differences in the intensity of uptake on metabolic imaging, such as 68Ga-somatostatin receptor and Fluorine-18 fluorodeoxyglucose positron emission tomography. As these features are directly related to prognosis, it seems mandatory to move toward a standardized, improved selection of the tumor areas to be studied to be as predictive as possible. The temporal evolution of NENs frequently leads to changes in tumor grade over time, with impact on prognosis and therapeutic decision-making. However, there is no recommendation regarding systematic biopsy of NEN recurrence or progression, and which lesion to sample. This review aims to summarize the current state of knowledge, the main hypotheses, and the main implications regarding intra-tumor spatial and temporal heterogeneity in digestive NENs.
Introduction: NENs are heterogeneous neoplasms
Neuroendocrine neoplasms (NENs) are rare malignancies arising from neuroendocrine cells organized as glandular organs (e.g. pancreas) or as the diffuse endocrine system (e.g. in the digestive and bronchial tracts). 1 NENs can present very variable histopathological characteristics, functioning profile, aspect on imaging and molecular abnormalities. These differences are important as they can impact prognosis and influence therapeutic decision-making.
NENs encompass well-differentiated neuroendocrine tumors (NETs) and poorly differentiated neuroendocrine carcinomas (NECs).2,3 These entities are very different in molecular landscape, prognosis, and treatment. Beyond cell differentiation, NENs can have heterogeneous histopathological features, notably variable architecture including nested, trabecular, and/or pseudo-glandular patterns in NETs, and organoid or diffuse patterns in NECs. 4 In addition, NENs show very variable degree of proliferation, measured by the Ki-67 index and mitotic count, which constitutes the basis of their histoprognostic classification.2,3
NENs have variable functioning profile, corresponding to their ability to produce, store, and secrete hormones and peptides, such as chromogranin A which is clinically inert, and serotonin, insulin, glucagon, and gastrin, whose hypersecretion can lead to functioning syndromes. Hormone production can be primarily observed using specific immunostaining, while hormone secretion can be assessed by plasmatic or urinary measurements. Most NET cells and a subset of NEC cells specifically express somatostatin (SST) receptors on their surface, which can be visualized by immunohistochemistry or nuclear imaging (68Ga-somatostatin receptor PET). Finally, different metabolic pattern can be measured by assessing their degree of glucose avidity using Fluorine-18 fluorodeoxyglucose positron emission tomography (18F-FDG PET).
The multiple molecular studies conducted during the two last decades have led to decipher the genomic landscape of NENs and identified significant heterogeneity, either between NETs arising from different organs or from the same origin but presenting different biological aggressiveness, notably for pancreatic NETs (PanNETs),5,6 small-intestine NETs (siNETs)7,8, and lung NETs.9,10
Why study intra-tumor heterogeneity of NENs?
Similar to all solid tumors, NENs are initially monoclonal neoplasms that progressively become polyclonal in relation to selection pressures due to metabolic (e.g. hypoxia), environmental (e.g. proximity to vessels, immune response, stroma), and therapeutic conditions. Nowell’s (1986) 11 theory on tumor heterogeneity relies on the fact that despite a common precursor cell, any genomic event conferring a survival advantage may promote new tumor clones with unique biological characteristics. These emerging clonal populations may coexist or replace preexisting clones, resulting in intra-tumor heterogeneity (Figure 1).12,13 Thus, NENs can be composed of different cell subclonal populations with very different genotypic and phenotypic characteristics leading to biological differences, including the Ki-67 proliferation index, morphology, or sensitivity to treatments.
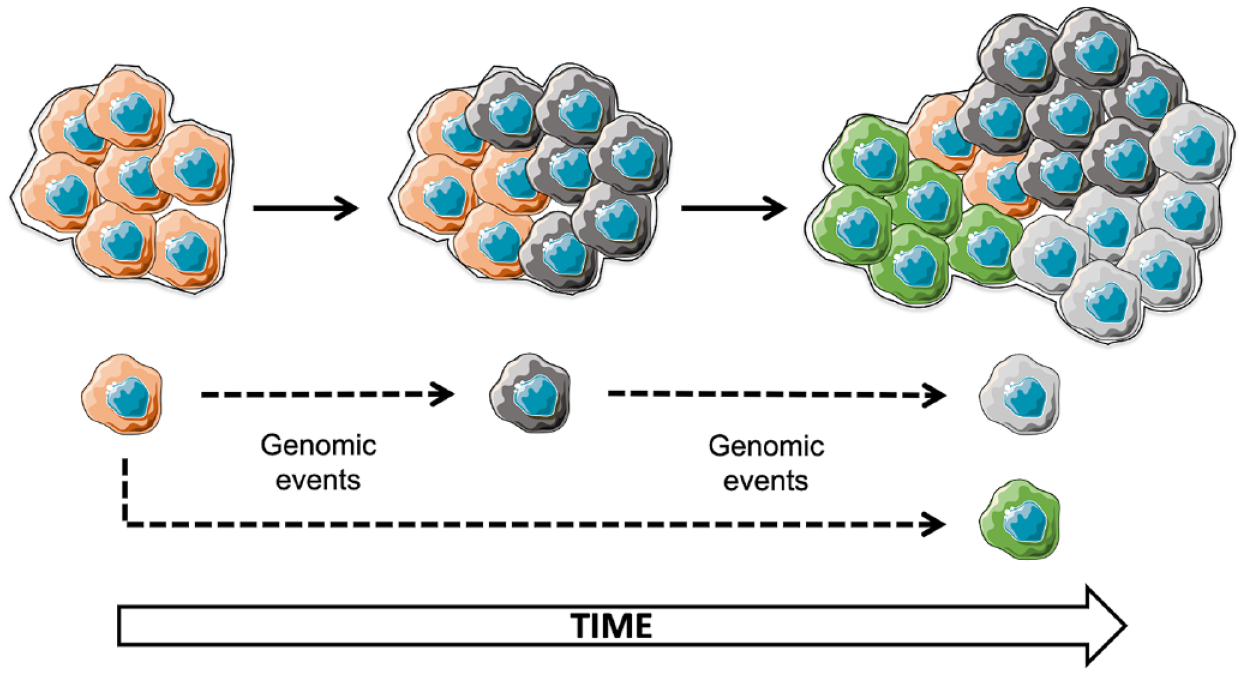
Polyclonality and spatial/temporal heterogeneity of solid tumors, including neuroendocrine neoplasms.
The intra-tumor heterogeneity of NENs, that is, the biological differences between subpopulations of tumor cells that successively evolve in a tumor mass constituting a patient’s disease, has been little studied. Intra-tumor heterogeneity is visible at the morphological level with different cellular aspects and architecture cohabiting within the same NEN and at the proliferative level with spatial and temporal heterogeneity of the Ki-67 index. Cell proliferation is one of the most important biomarkers for assessing the biological aggressiveness of digestive NENs and is a central component of the WHO histoprognostic classification.2,3 Measurement of the Ki-67 proliferation index is more accurate and reproducible than the mitotic index count and is the only feasible counting method on biopsies.14–16 However, the measurement of the Ki-67 index is subject to sampling bias.
The most important practical use of the study of tumor heterogeneity lies in its therapeutic implications. Treatments generally fail for three reasons: (1) intrinsic primary resistance, in which the cell does not respond to a treatment due to its biological properties (such as activation of xenobiotic metabolism by over-expression of P-glycoprotein or DNA repair actors), (2) environment-mediated resistance, in which local factors such as hypoxia or ischemia reduce drug levels or treatment efficacy in otherwise susceptible cells, and/or (3) acquired secondary resistance associated with therapeutic resistance, often in connection with treatment-induced neo-mutagenesis.
This review aims to summarize the current state of knowledge, the main hypotheses, and the main implications regarding intra-tumor spatial and temporal heterogeneity in NENs. To provide a comprehensive and up-to-date review, we searched PubMed and Web of Science and electronic databases for relevant articles with no time or language restriction, using the following keywords: ‘neuroendocrine neoplasm’, ‘neuroendocrine tumor’, ‘neuroendocrine carcinoma’, and ‘heterogeneity’, ‘spatial heterogeneity’, ‘temporal heterogeneity’, ‘clonal selection’, or ‘evolution’.
Spatial heterogeneity in NENs
Inter-tumor heterogeneity (between different locations within one patient)
Tumor heterogeneity is frequent between the primary tumor and the metastases and between different metastatic locations in the same patient. In several studies, including patients with metastatic PanNETs or siNETs, the Ki-67 index was reported higher in metastases compared to primary tumors.14,17–19 Consequently, the histoprognostic grade of metastases was reported higher (grade 2, G2) than that of the primary tumor (grade 1, G1) in 13–35% of cases. This increase in grade was reported to be more frequent in distant metastases compared to locoregional metastases.18,19 In one study, the size of metastases >4 cm was associated with the existence of a significant increase in the Ki-67 index compared to primary tumors. 17 Finally, such tumor heterogeneity was associated with a significant adverse prognostic impact. Thus, the authors of these studies recommend systematically biopsying NET metastases, especially those in the liver and/or measuring more than 4 cm. A similar degree of heterogeneity was reported between different synchronous metastatic locations.20,21 In a previous study performed in our institution on 27 patients, the intraclass correlation coefficient of Ki-67 between different liver metastases was 0.69, indicating 31% of heterogeneity. 20 Again, Ki-67 heterogeneity between lesions was significantly higher when assessed on large metastases (p = 0.04). 21 It is currently unknown whether Ki-67 index is more frequently higher in some specific metastatic site (e.g. liver, bone, lymph nodes) in comparison with others.
This inter-tumor heterogeneity is the result of molecular alterations. The genomic landscape of digestive NETs is generally uneventful, suggesting that progression does not rely on classical strong oncogenic drivers but rather on subtle epigenetic mechanisms. This was particularly well described in PanNETs, whose genomic heterogeneity partially overlaps with the proposed epigenomic and transcriptomic classification of PanNETs, suggesting a probable causal link.22,23 Integrative multiomic analyses clustered PanNETs into three subgroups : (i) a beta-like subtype with no mutations in MEN1/DAXX/ATRX and an epigenomic profile resembling that of normal beta cells, (ii) an alpha-like subtype with mutations in MEN1 and epigenomic profile resembling normal alpha cells, (iii) an intermediate subtype with mutations in MEN1 and DAXX or ATRX, hypoxic transcriptomic signatures, higher proliferation index, higher metastatic capability, and a poorer prognosis.5,6,24 Clinically, this molecular profiling of NET heterogeneity, that can be partially summarized by DAXX/ATRX immunostaining, might have prognostic implication and might notably help deciding between surveillance and surgery for small PanNETs. 25
Inter-tumor spatial heterogeneity can also be identified by differential expression of hormones and SST receptors, whose expression exists in >90% of NETs and predicts efficacy of peptide-radionuclide receptor therapy (PRRT) using 177Lu-DOTATATE. 26 In a Japanese study, the concordance rate of gastrin, insulin, glucagon, and SST expression was 50% between primary NETs and lymph-node metastases and 11% between primary NETs and liver metastases. 27 The progression of NETs is often accompanied by changes in morphology with more atypia and stroma remodeling and decreased expression of SST receptors. Discordant expression of type 2 SST receptors has been reported in 31% of synchronous metastases and 21% of metachronous metastases. 20 Concordantly, NET progression is frequently associated with a decrease in uptake on 68Ga-somatostatin receptor PET (using DOTANOC, DOTATOC, or DOTATATE).
Besides, the biological and clinical progression of NETs is also associated with an increase in glycolytic metabolism, leading to an increase in tumor uptake on 18F-FDG PET. Therefore, the combined use of 68Ga-somatostatin receptor PET and 18F-FDG PET enables visualizing several types of NEN lesions, synchronous or metachronous, with different degrees of glycolysis and SST receptor concentration. 12 Uptake on 18F-FDG PET correlates with histoprognostic grade and Ki-67 index, thus allowing for non-invasive prognostic assessment.28,29 Furthermore, because PET is a whole-body imaging, it may overcome the limitation of sampling bias related to histopathological assessment.20,30 Ezzidin et al. 30 previously reported the relevance of a metabolic grading system using the median tumor-to-liver ratio of maximal standardized uptake value (SUVmax T/L) on 18F-FDG PET. This study and others reported that the metabolic grade better was a better prognostic tool than the pathological grade assessed by the Ki-67 index.30,31 However, these studies compared SUVmax determined on whole-body FDG-PET with the Ki-67 index of the most easily accessible lesion that was generally sampled long ago and was not necessarily the lesion with the highest uptake, leading to potential discrepancies. To better assess this relationship, we previously reported a significant lesion-by-lesion correlation between the preoperative SUVmax T/L ratio and the Ki-67 index analyzed on 36 surgically resected specimens of PanNETs. 32 Uptake on 18F-FDG PET could predict the pathological grade of NET lesions and supplement pathological evaluation of biological aggressiveness or guide the choice of the most relevant lesions to biopsy in case of discrepancy (Figure 2).32,33

Correlation of the uptake intensity on 18F-FDG PET/CT and Ki-67 proliferation index in neuroendocrine neoplasms.
The clinical consequences of intra-tumor heterogeneity rely on the ‘scale invariance of entropy’ hypothesis, which states that there is a consistent correlation between heterogeneity at the genetic and cellular levels (observed using molecular and histopathological techniques) and heterogeneity at the tissue level, such as vascular density, blood flow, and molecular metabolism (measured by morphological or nuclear imaging). 12 As a corollary, tumors with the highest degrees of heterogeneity in density and enhancement on computed tomography (CT), intensity and diffusion restriction on MRI, and/or uptake on PET imaging would be those with the highest degrees of heterogeneity in microvascular density, cellularity, proliferation, and molecular abnormalities at the cellular and genetic levels. Consequently, the most heterogeneous NETs are those with the highest risk of therapeutic inefficiency, especially heterogeneity of expression of a theranostic marker. The best example reported so far concerns PRRT. Indeed, it has been reported that heterogeneity of 68Ga-somatostatin receptor uptake on PET/CT is associated with lower efficacy of 177Lu-DOTATATE.34–36
Intra-tumor heterogeneity (within the same location)
Spatial intra-tumor heterogeneity can lead to an inexact assessment of histopathological diagnostic and prognostic biomarkers. Notably, the prognostic impact of the Ki-67 index is higher when determined in hotspots rather than measured as a mean on a NEN specimen.14,37 Spatial intra-tumor heterogeneity was reported in 37% of metastases measuring >2 cm in size. 20 Interestingly, the amount of inter- and intra-tumor spatial heterogeneity was similar. This may indicate that the optimized identification of Ki-67 index hotspots within one tumor location may allow estimating the Ki-67 index at other locations. Intra-tumor heterogeneity increases with higher mean Ki-67 values37,38 and larger tumor size. 21 Notably, a study that included NET lesions with a Ki-67 index >10% reported intra-tumor heterogeneity in 10% of lesions <1 cm, compared with 77% of lesions >1 cm (for each additional cm, odds ratio 2.55; p < 0.001). 21
Accordingly, the sampling bias induced by intra-tumor heterogeneity is all the more important that the size of the biopsy material is reduced. Previous studies have consistently shown discordances between the Ki-67 index determined on endoscopic ultrasonography-guided biopsy and PanNET surgical specimens.38–40 Conversely, the concordance rate increased with the surface area of the biopsy sample and/or with increasing numbers of cells used for Ki-67 counting. Another study reported higher intra-tumor heterogeneity (coexistence of G1 and G2 components) when the Ki-67 index was determined on whole slides (47%), in comparison to when it was determined on three distinct areas (16%) – virtually representing three biopsies. 37 Again, heterogeneity was more frequent in NET specimens in which the mean Ki-67 index corresponded to G2 than G1. The histoprognostic grade had a greater prognostic impact when it was determined on hotspots rather than on the mean of the whole slide; and when it was determined on three virtual biopsies rather than on a single randomly selected virtual biopsy. 37
The Memorial Sloan Kettering group 41 described a series of heterogeneous PanNETs characterized by the coexistence of areas classified as G1, G2, and G3. This heterogeneity was observed both in primary tumors and in metastasis specimens, mainly located in the liver. High-grade components generally showed features suggestive of ‘dedifferentiation’ (necrosis, stroma, cellular organization) without clear histopathological nor molecular features of NEC. Compared to G1/G2 PanNETs and ‘pure’ pancreatic NECs, heterogeneous PanNETs had intermediate biological aggressiveness and prognosis but were closer to pure NETs. 41 Mixed neuroendocrine-non-neuroendocrine neoplasms (MiNEN) are a hallmark illustration of spatial heterogeneity and a good opportunity for the study of its mechanisms.42,43 These rare NENs combine a neuroendocrine component (usually a NEC) and a non-neuroendocrine component (usually an adenocarcinoma), both accounting for at least 30% of the neoplasm.2,3,44 They can be morphologically classified into collision (juxtaposition of two components that remain topographically separate), composite (two morphologically distinct components, intermingled or with one predominant component and a focal area of another one), and amphicrine (single-cell population that displays the phenotypes of two neoplasms) MiNEN.43,44 Their classification (high, intermediate, or low grade) and prognosis depend on the metastatic potential of each of its components (e.g. NET or NEC; adenoma or adenocarcinoma). Molecular studies consistently reported that whatever the organ of origin, when the neuroendocrine component is a NEC, it shares genomic abnormalities (mostly RB1, TP53, or APC) with the non-neuroendocrine component, which supports a common origin. Of note, while each component of a MiNEN must account for at least 30% of the whole neoplasm,2,3 a minor (i.e. <30%) high-grade component can impair prognosis.42,45 In addition, this cutoff value only applies after examination of the entire resected specimen and particular caution should be taken when evaluating biopsies, due to the abovementioned sampling bias.
Overall, the histoprognostic grade of NETs is likely to be underestimated due to the significant intra-tumor heterogeneity of NETs, if it is performed on random tumor zones and/or on limited tumor areas, all the more so in NETs with a high mean proliferation index. Hence, the assessment of the Ki-67 index should be performed on at least 2000 cells (or 500 cells in case of biopsies),2,16 and it seems that the larger the sample size analyzed, the more accurate the assessment of Ki-67 and intra-tumor heterogeneity will be.
Spatial heterogeneity has been mostly explored through tumor expression of proliferative markers. Nevertheless, heterogeneity has also been described regarding hormone expression, as multiple hormone expression was reported in up to 30% of PanNETs, including some clinically nonfunctioning tumors. 27 There is unfortunately very little data on molecular spatial heterogeneity of digestive NENs. In lung NENs, several clones coexisted in tumors with important heterogeneity in the proliferation index, with a gradual acquisition of driver mutations from a common ancestor. 46 In PanNETs, Di Domenico et al. 24 proposed a stepwise progression model from normal beta cells on one hand (unknown driver) and from alpha cells on the other hand with early genomic drivers (MEN1/DAXX or ATRX) and late environmental driver (hypoxia, oxidative stress). The stroma of NENs has been poorly studied so far but might greatly contribute to intratumor spatial heterogeneity of NENs. The immune infiltration seems variable but is usually low, which is consistent with the low mutational burden of NETs. 47 Reports are conflicting regarding the prognostic value of T-cell infiltration, but it seems to be overcome by the amount of immunosuppressive macrophages.47,48 Yet, it seems that the immune microenvironment may evolve, since metastatic PanNETs have a more immune-tolerant infiltrate that could represent a potential target. 49 In addition to the immune infiltrate, the fibrotic stroma and the vasculature are also remodeled though NET progression with an increase in extracellular matrix deposition and a decrease in vessel density associated with NEN aggressivity, which may be assessed by imaging.50,51
Non-invasive assessment of intratumor heterogeneity is challenging but could be performed using radiomics approaches. Canellas et al. 52 showed that tumor heterogeneity on CT (assessed by evaluating entropy) was predictive of tumor grade, as G2 and G3 NEN had significantly higher entropy than G1 NETs. Our group used CT histogram analysis and showed that NEC were more heterogeneous on portal venous phase images than G3 NET. 50 Finally, Atkinson et al. 53 showed that measures of heterogeneity on coarse-texture scale CT and unfiltered 68Ga-DOTATATE PET-CT images predicted shorter progression-free survival (PFS) and shorter overall survival (OS). Multivariate analysis identified that CT texture analysis (coarse kurtosis) independently predicted PFS, and PET texture analysis (unfiltered skewness) independently predicted OS.
Temporal heterogeneity in NETs
Most patients with PanNET have a stable clinical phenotype over time, at least in the subclinical phase of carcinogenesis, as evidenced by the fact that most functioning syndromes are diagnosed at the time of the initial diagnosis of PanNET, mainly because they are a classic circumstance of discovery. However, in rare cases, a hormonal syndrome may appear during the course of the NET disease. These metachronous hormonal syndromes were reported in approximately 4% of patients with PanNET in two studies.54,55 They mainly occurred in patients with heavily pretreated, progressive metastatic PanNET after a mean duration of 4–5 years after the initial diagnosis. Notably, most patients had previously received chemotherapy with notable efficacy. In the French study, the median Ki-67 index was higher on new samples taken at the time of the onset of metachronous hormone syndrome than on the samples taken at diagnosis (18% versus 7%, respectively). 54 In both studies, the occurrence of a metachronous hormonal syndrome marked a turning point in the course of the disease with a pejorative prognostic impact, especially in the case of metachronous insulin secretion.
From a histopathological point of view, it is challenging to dissociate spatial from temporal heterogeneity, as the same lesion cannot be resected more than once and is rarely biopsied repeatedly over time. Initially, several studies, including a limited number of patients with NET from various origins, compared the Ki-67 index of metachronous metastases with primary tumors and/or synchronous metastases, and reported a mismatch in tumor grade (an increase in the majority of cases) in about 35% of cases.20,56,57 Inter-tumor heterogeneity was generally higher in metachronous than synchronous metastases.19,58 An increase in the histoprognostic grade between initial and metachronous samples was more frequently observed in PanNETs compared to NETs from other origins when the time elapsed between the two samples was longer and when the second sample was taken in a context of disease progression. 57 Merola et al. 59 reported an increase in Ki-67 index and/or in tumor grade for 66% and 34% of patients (p < 0.01), between curative-intent surgical resection and biopsy-proven recurrence of digestive NETs (median interval of 40 months). Median recurrence-free survival was shorter in patients with increasing Ki-67 index over time (36 months versus 61 months, p = 0.02).
Overall, these studies advocate for the relevance of systematic sampling of metachronous NET metastases in order not to miss an increase in histoprognostic grade. As observed with spatial heterogeneity, temporal heterogeneity increases with higher values of the initial Ki-67 index, and G3 NETs are more frequently observed in metastases, especially when sampling is performed after chemotherapy. This suggests that digestive NETs may evolve from G1/G2 to G3, especially after chemotherapy, with an expected negative prognostic impact.
This hypothesis was explored in a cohort of 46 patients with PanNETs in which multiple samples were performed over time (median interval: 44 months). 60 The median difference in the Ki-67 index between the two specimens was +14%, with a grade increase in 58% of cases. The new specimens also had more frequent cytonuclear atypia and necrosis. In this study, patients whose PanNET had an increase in the Ki-67 index over time had reduced survival. Especially, progression from a G1/G2 to a G3 PanNET was an independent poor prognostic factor at multivariable analysis. The risk of progression to a G3 PanNET was increased in patients with initially higher Ki-67, and/or who received a high number of treatments, particularly in the case of alkylating chemotherapy. 60 This suggests an important impact of taking new tumor biopsies in patients with heavily pretreated advanced PanNETs experiencing progression. In order to be as informative as possible, new tumor sampling could target lesions that are increasing in size on morphological imaging and/or have high 18F-FDG uptake on PET/CT as it correlates with higher Ki-67 values. 32 Of note, pre-treatment with temozolomide has been associated with increasing biological aggressiveness in other malignancies, such as colorectal adenocarcinoma and glioblastoma.61,62 In these models, alkylating chemotherapy induced the emergence of a hypermutator phenotype with increased tumor mutational burden and mismatch repair deficiency through chemo-induced mutagenicity and/or clonal selection. Interestingly, acquiring these abnormalities might favor the acquisition of sensitivity to immune checkpoint inhibitors, 63 a hypothesis worth exploring in patients with PanNETs progressive after alkylating agents.
The specific impact of treatments on the biology of NETs has been poorly described. In one study including G3 NENs, most samples taken after treatment (mainly platinum-based chemotherapy) showed at least a focal decrease in the Ki-67 index, and 30% were classified as G1/G2. 64 Half showed significant heterogeneity with a mean intra-tumor Ki-67 delta between 20% and 40%. These observations, already mentioned in an older publication, 65 may be explained by (1) transformations between NETs and NECs, but which are nevertheless exceptional or even nonexistent, as pointed out by the authors, 64 (2) the initial coexistence of the two components, 41 the high-grade contingent having decreased or disappeared after chemotherapy while an initially under-evaluated G1/G2 component would be resistant, and (3) initial classification issues, this hypothesis being made unlikely by the overall decrease in Ki-67 post-therapy.
Whether temporal heterogeneity and, more generally, the progression of PanNETs to high grade are associated with ‘NEC-like’ molecular alterations such as TP53 mutations or Rb loss has been scarcely explored. We previously reported that a small proportion of G3 NETs might show inappropriate expression of Rb (10%), p16 (20%), or p53 (30%). 66 Nevertheless, NETs and NECs have different pathways of oncogenesis, and it is unlikely that the dedifferentiation of NETs could result in a complete histopathological and molecular phenotype of NEC. 41 However, recent studies suggest that a subset of digestive and lung large-cell NECs might result from the evolution of preexisting well-differentiated NETs.46,67 Indeed, a clustering analysis of molecular abnormalities of a 10-gene panel identified that some G2 NETs presented some hallmarks of NECs (alterations in TP53, KRAS, BRAF, RB1, PIK3CA) and were associated with worse prognosis. Conversely, a subgroup of large-cell NECs did not have these hallmarks alterations, correlated with better prognosis, and appeared to coexist with well-differentiated components in particularly heterogeneous tumors. 67 Similarly, comparative spatial/temporal analyses of high-grade lung NETs with carcinoid morphology indicated that they were genetically heterogeneous and progressed from clones of lower aggressivity with accumulation of NEC-like genetic alterations such as TP53/RB1 alterations. 46 Finally, the analysis of post-chemotherapy pathological specimens of initially poorly differentiated NECs confirmed this hypothesis, showing significant heterogeneity and sometimes only a tumor remnant in the form of a slower proliferating, possibly original clone with features of well-differentiated NET. 64
Future prognostic biomarkers could include inflammatory features of the microenvironment and the tumor, with the G1/G2-to-G3 transition associated with changes in immune profile and resistance to immune surveillance.68,69 In particular, G3 NENs are characterized by increased expression of COX-2, β-catenin, and PD-L1, decreased expression of HLA-I, and increased expression of PD-1, PD-L1, and decreased expression of NGFR, pS6, HLA-I, and HLA-DR in the stroma. 69 In most cases, the abnormal expression of these biomarkers was gradual between G1 NETs, G2 NETs, G3 NETs, and maximal in NECs. Altogether, tumor inflammation, HLA molecule repression, and T-cell inhibition seem to be associated with the molecular progression of NENs and negatively impact the survival of patients.69,70 Activation of these mechanisms, therefore, represents a hypothesis that may explain the temporal heterogeneity of NETs represented by tumor biological and molecular progression.
Conclusion
NENs present a high degree of heterogeneity, both spatially within the same location or between different locations (between primary tumor and metastases and between metastases), and through time. This can be explained by the emergence of tumor subclones with different behaviors, and, therefore, different responses to treatment. The tumor microenvironment also seems to play an important role in selecting these clones. These subpopulations can be distinguished by the Ki-67 index, but also by the expression of hormonal markers or by differences in the intensity of uptake at metabolic imaging, such as 68Ga-somatostatin receptor PET and 18F-FDG PET. As these characteristics are directly linked with prognosis, it seems mandatory to move toward a standardized, improved selection of the tumor areas to be studied to be as predictive as possible (Table 1). Although it is well acknowledged that determining the Ki-67 index in the hotspot zones of a NET lesion best enables the prognosis evaluation and treatment decision, there is still no recommendation regarding systematic biopsy at the time of recurrence and which lesion to target. However, the temporal evolution of NETs frequently leads to changes in tumor grade over time, which would likely have a therapeutic impact should this evolution be evidenced.
Main issues related to heterogeneity of NENs, and proposals for taking it into account in patient management.

18F-FDG PET, Fluorine-18 fluorodeoxyglucose positron emission tomography; NENs, neuroendocrine neoplasms; SSTR, somatostatin receptor.
