Abstract
According to the neuroendocrine tumour (NET) characteristics, 3 to 7 different treatment options are available, corresponding to 6 to 5,040 theoretical different sequences. Even though each patient is unique and despite a large heterogeneity in NET characteristics, the present review aims to discuss the main sequences and addresses how one can propose the best sequence to treat metastatic NET (mNET) on a case-by-case basis. Each treatment must be discussed during dedicated multi-disciplinary meetings, and inclusions in clinical trials should be favoured. After a thorough characterization of patients and their mNET, and taking into account the availability of drugs, the first-line treatment should be chosen according to the treatment aim. The latter is determined based on three main topics (efficacy, safety, and patient preferences) that do not necessarily converge and must be defined a priori. At baseline, physicians should design an a priori full therapeutic sequence, which may evolve at each step depending on the response to previous treatment, the occurrence of chronic toxicities, and the patients’ perception of the prior treatment. To improve knowledge in terms of effectiveness and risk of cumulative toxicities regarding the different sequences, real-world data using long follow-up durations are necessary; such issues will not be resolved by randomized clinical trials.
Introduction
The WHO classification enables the categorization of well-differentiated neuroendocrine tumours (NET) into three groups 1 according to the tumour grade, which takes into account the mitotic count/2 mm² and the proliferation index Ki67: NET-G1 (Ki67 < 3% and mitotic count < 2), NET-G2 (Ki67 3–20% or mitotic count 3–20), and NET-G3 (Ki67 > 20% or mitotic count > 20). In case of metastatic NET (mNET), several treatments, either systemic or locoregional, are available. When choosing the best therapeutic sequence, the goal should be to prolong overall survival (OS) while maintaining a good quality of life (QoL).
NET represent 80–90% of neuroendocrine neoplasms (NEN). 2 In the United States, the incidence of NEN was 6.98 per 100,000 persons in 2012. Among 53,465 NET of known stage, 14,657 (27.4%) were metastatic at diagnosis and the median OS of patients was longer than 5 years.2,3 Among the 10,102 patients who were mainly included in the Centres of Excellence of the European Neuroendocrine Tumor Society (ENETS-CoE), thus inducing a selection bias with a higher prevalence of mNET, the main primary tumour locations were the pancreas (p), the small intestine (si), the appendix, the lung (l), and the stomach. 4 NET in the appendix and stomach were rarely metastatic while metastases were present in 60% of siNET, 50% of pNET, and 36% of thoracic NET. Hence, the pNET, siNET, and lNET account for the major part of mNET prevalence; the available data and therapeutic sequences will thus be discussed in more detail for these three organs. Among mNEN, 38% of pNEN were G1, 50% G2, and 12% G3; while siNEN were mainly G1 (59%) or G2 (39%), few were G3 (2%). 4
The aim of this review was to describe the criteria used in clinical practice to choose the best therapeutic sequence for a particular patient, according to the characteristics of the patient, the mNET, and the healthcare system considered. At baseline, physicians should design an a priori full therapeutic sequence, each step of which should be re-discussed based on the outcomes of the previous treatment.
Treatment options
The available weapons
The available therapies as well as their actions, their results, and their use against mNET are detailed in several recent published studies.2,5,6 There are a rather large number of weapons currently available, and these can be categorized as either systemic or locoregional treatments. The latter include the surgical resection of the primary tumour and/or metastases (curative or palliative), the ablative therapies (including radiofrequency ablation), the liver embolization (either alone or associated with chemotherapy), and the selective internal radiation therapy of the liver (not reimbursed in some countries). Among the approved systemic therapies for mNET, two types of treatments targeting somatostatin receptors (SSTRs) are available, including two somatostatin analogues (SSA, lanreotide 7 and octreotide 8 ) and one peptide receptor radionuclide therapy (PRRT, 177Lu-edotreotide 9 ), two targeted therapies including one mTOR inhibitor (everolimus, the only drug approved for all NET10,11) and two tyrosine kinase inhibitors (TKIs, sunitinib approved for pNET, 12 and surufatinib for all NET in China13,14), and two types of cytotoxic chemotherapies including an alkylating agent [such as streptozotocin, the only chemotherapy approved for pNET, 15 and temozolomide (TEM)16,17 or dacarbazine 18 ] and oxaliplatin-based chemotherapy. 19 Telotristat 20 and interferon 21 (currently less used) are administered for their anti-secretory effect for the control of a carcinoid syndrome.
The four types of features that should be taken into account when choosing a treatment
Designing the best therapeutic sequence for a given patient requires a comprehensive knowledge of the available treatments.2,22,23 Of course, at each step of the sequence, information regarding the response, toxicity, and QoL must be collected to eventually adapt the next steps of the sequence according to these outcomes. In addition, before initiating a new treatment, the possible inclusion of the patient in a clinical trial should be carefully considered; clinical trials enable to improve the level of evidence and sometimes offer the possibility of administering a new drug or a drug that would not be reimbursed otherwise. In the context of rare diseases, it is essential to discuss and decide the appropriate course of treatment in dedicated NET multi-disciplinary tumour boards (MTBs), if possible in expert centres (such as the ENETS or European reference network on Rare adult Cancers (EURACAN) centres of excellence). National dedicated networks conducting pathological review and expert MTB are also helpful to improve the care of patients with mNET. 24 The choice of treatment depends, at each step, on the four main factors described below: the NET characteristics, the patient characteristics, the healthcare system characteristics, and the aims of the treatment. In this context, several questions must be discussed (Figure 1), the main ones being (a) whether the prognosis and symptoms are related to the primary tumour or to the metastases, to the tumour burden, and/or to hormone secretion, and (b) whether a local treatment (interventional radiology or surgery) can be considered. However, one has to keep in mind that no specific therapeutic sequence has been validated and that predictive factors of therapy efficacy are lacking, thereby highlighting the importance of the dedicated MTB when choosing the best treatment option.

General considerations that should be taken into account for each new treatment within a therapeutic sequence in patients with metastatic neuroendocrine tumours (mNET).
NET characteristics
A thorough characterization of the mNET is required before designing the therapeutic sequence (Table 1). First, knowing the origin of the mNET is important as some drugs are only reimbursed and/or available for pNET for instance (sunitinib or streptozotocin), and similarly knowing whether some clinical trials are open for this mNET helps in treatment selection. In addition, one must define whether the primary NET is still present, and whether there is a locoregional risk of complications (e.g. icterus induced by a pNET within the pancreatic head, occlusion from an siNET with mesenteric fibrosis, haemoptysis from a proximal lNET). Second, the histological analysis remains the main prognostic factor and must provide information regarding differentiation, tumour grading, and Ki67 level. Other possible prognostic factors might be useful in the future, if confirmed by prospective studies [e.g. O6-methylguanine DNA methyltransferase (MGMT) status to define the best chemotherapy regimen in pNET].16,25 In case of multiple Ki67 levels, the highest one should be used to decide the best treatment. Third, hormone-related syndromes should be diagnosed, as some of them are life-threatening (insulinoma, cushing, VIPoma), and because their treatment may be more urgently needed. Carcinoid syndrome is the most frequent functional NET syndrome (between 20% and 38% of metastatic siNET and lNET2,23,26,27) and is treated by SSA, 28 as are the rare glucagonoma and VIPoma. 29 Zollinger-Ellison syndrome and insulinoma are the most frequent hormone-related syndromes for pNET; cushing syndrome may occur in few (<5%) lNET and pNET cases. 29 The anti-secretory treatment is usually maintained during the entire therapeutic sequence and adapted according to the level of secretion; sometimes, an anti-tumour treatment is needed even in the absence of progressive disease according to the RECIST criteria in order to better control the anti-secretory syndrome. The dedicated cardiologist team must be involved in the NET MTB for regular screening, follow-up, and treatment of the carcinoid heart disease. 30 Fourth, a somatostatin receptor imaging (SRI), preferably 68Ga-DOTA-SSA PET/CT, must be performed for all mNET in order to evaluate the tumour spread, the prognosis (the stronger the SSTR expression, the better the prognosis), and to assess whether PRRT could be an option. 9 Fifth, the tumour burden of the mNET (liver tumour volume, location and amount of extrahepatic disease, symptoms related to tumour burden, and level of the surrogate marker chromogranin A) should be evaluated.2,27 Sixth, the aggressiveness of the mNET constitutes one of the most powerful prognostic factors; it is defined by the tumour slope/tumour growth rate (TGR), 31 or through a surrogate marker of this tumour slope such as the grading/Ki67 index or the intensity of uptake on 18F-fluorodeoxyglucose positron emission tomography. 32 Lastly, the presence of a genetic disorder could influence the choice of treatment for metastatic pNET in case of von Hippel-Lindau (VHL) syndrome (belzutifan is FDA approved for VHL disease based on a phase II trial 33 ).
Minimal characterization of the metastatic neuroendocrine tumour (mNET) required before choosing the therapeutic sequence.

FDG-PET, 18F-fluorodeoxyglucose positron emission tomography; l, lung; MEN1, multiple endocrine neoplasia type 1; MGMT, O6-methylguanine DNA methyltransferase; NET, neuroendocrine tumour; NSE, neuron-specific enolase; p, pancreas; PRRT, peptide receptor radionuclide therapy; si, small intestinal; SRI, somatostatin receptor imaging, preferably 68Ga-DOTA-SSA PET/CT; SSTR, somatostatin receptor; TGR, tumour growth rate; VHL, von Hippel-Lindau.
In summary, at this time, treatment choice is mainly based on prognostic factors rather than on predictive biomarkers. NET patients can be classified into two main groups: (i) mNET at low risk of progression for which a watch-and-wait strategy or SSA are the main options, and (ii) mNET with a high risk of fast progression requiring a more active treatment (chemotherapy for pNET or PPRT for siNET).2,22,23
Patient characteristics
As for any disease, patient characteristics (such as age, performance status, comorbidities, and contraindication to some treatments) will first influence and determine the feasibility of a treatment. In addition, the occurrence of toxicity and the QoL under prior treatments have to be taken into account for the following steps of the therapeutic sequence. It is therefore important to know the patients well beyond their mNET: do they continue to work or not, what are their favourite activities, what are their short-term and medium-term projects? This knowledge can, for instance, lead to delaying a treatment sequence that would require many appointments or would induce adverse events (AE) in order to avoid decreasing the QoL in the short term. As described below, patient preferences are becoming increasingly important in the decision-making process.
Healthcare system characteristics
The two main cost drivers during the management of patients with mNET are drugs and hospital stays (75% of the overall cost 6 ), the latter depending on the type of administration [pills can be administered to outpatients, whereas chemotherapy or PRRT have to be administered intravenously (IV) to hospitalized patients]. The treatment cost estimated from the societal perspective (per patient per month) is highest for PRRT, followed by targeted therapies, chemotherapy, and then SSA. However, this must be interpreted according to the duration of each treatment (very long for SSA, whereas usually less than 6 months for PPRT, chemotherapy, and targeted therapy) and more importantly according to its effectiveness (Tables 2–4). However, because of their costs, not all FDA/ European Medicines Agency (EMA) approved drugs are reimbursed in all countries. The first step in defining the therapeutic sequence is therefore to identify which therapies are reimbursed. Another way of proposing new/recent drugs to patients is to include them in clinical trials, which should be preferred when feasible because treatments are therefore reimbursed and these trials help improve knowledge.
Randomized studies evaluating systemic treatments in metastatic pancreatic neuroendocrine tumours.

Beva, bevacizumab; CAPTEM, capecitabine temozolomide; HR, hazard ratio; L, line of treatment; MGMT, O6-methylguanine DNA methyltransferase; mPFS, median progression-free survival; NA, not available; Oct, octreotide; OR, objective response; ORR, objective response rate; PRRT, peptide receptor radionuclide therapy; SSA, somatostatin analogues; STZ, streptozotocin; TEM, temozolomide.
Randomized studies evaluating systemic treatments in metastatic jejuno-ileum neuroendocrine tumours.
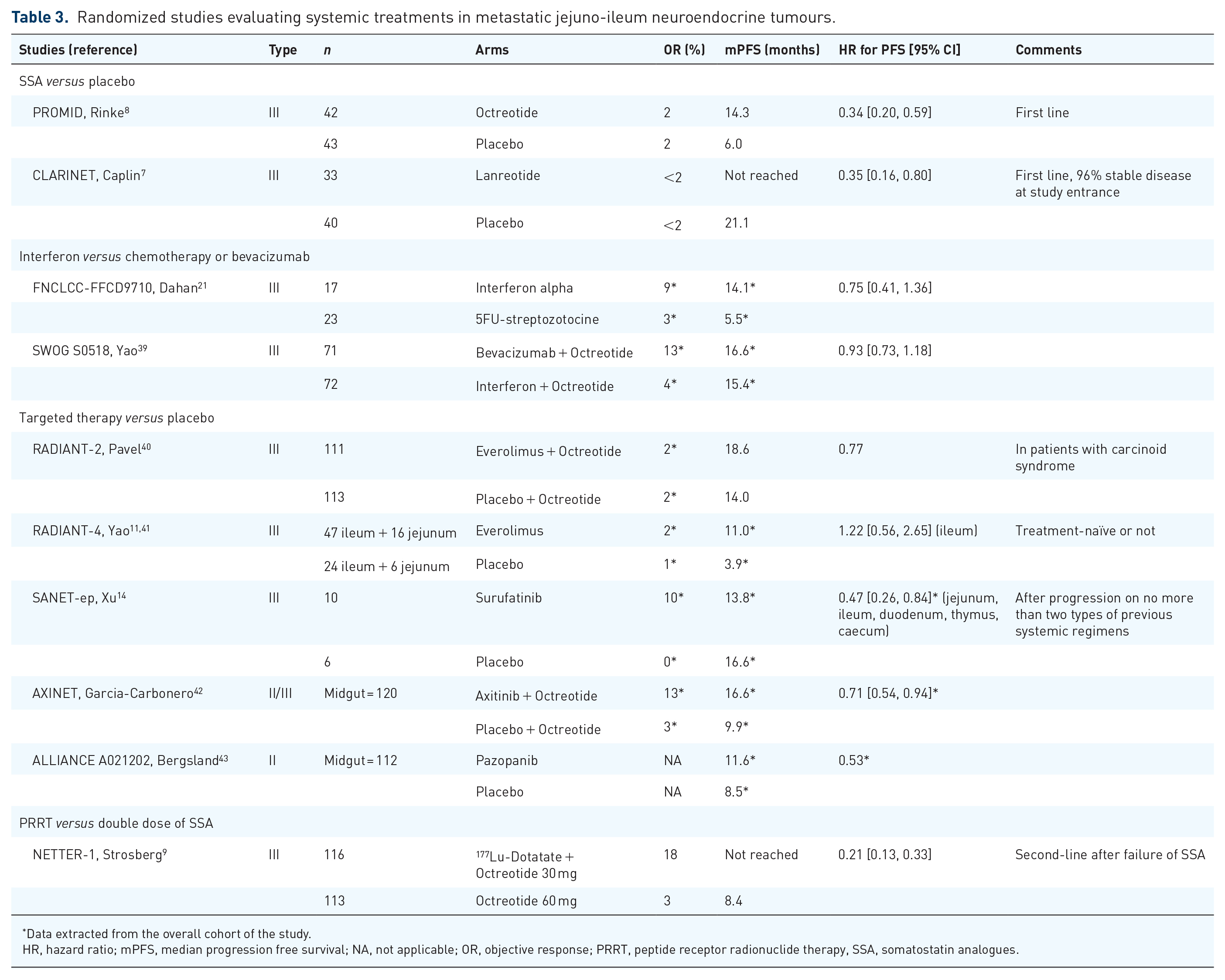
Data extracted from the overall cohort of the study.
HR, hazard ratio; mPFS, median progression free survival; NA, not applicable; OR, objective response; PRRT, peptide receptor radionuclide therapy, SSA, somatostatin analogues.
Randomized studies evaluating systemic treatments in metastatic lung neuroendocrine tumours.

Data extracted from the overall cohort of the study.
HR, hazard ratio; mPFS, median progression free survival; OR, objective response; SSA, somatostatin analogues.
Treatment aims
The aim of each treatment must be clearly defined. An anti-secretory treatment, such as SSA or telotristat, could be sufficient in some cases, or associated with an anti-tumour treatment if necessary. A curative treatment using a surgical approach is indicated in very selected patients affected by mNET who show exclusive or predominant liver disease, after evaluation of the tumour grading, distribution of liver metastases, and primary site. 2 The need to quickly reduce the tumour burden (because of tumour-related symptoms and risk of local complications) or stop a fast progression will be more often met by a locoregional treatment when feasible (surgery or ablative radiological treatment, vascular approach such as liver embolization), by chemotherapy in pNET, and by PRRT for all mNET with high SSTR expression. In contrast, if the aim of the treatment is to prolong the OS and/or prolong the QoL of an asymptomatic patient with good prognostic factors, one should target treatments with the best safety profile such as SSA for instance, which can be administered upfront7,8 or as a maintenance treatment. 34 Sometimes, the initial evaluation of an unresectable tumour requires a neoadjuvant treatment aiming at reducing the tumour load before a curative surgery, and usually requires the highest objective response rate (ORR, Tables 2–4). Regarding the treatments able to affect a fast tumour slope (≈ high TGR), there is currently no available data from randomized studies including patients with mNET progressing within 3–6 months; in this setting, data on the efficacy of SSA, targeted therapies, and PRRT should become available with the Netter-2 (NCT03972488) and Compose (NCT04919226) studies.
The sequence options
When considering only the systemic treatments available, there are at least six options for pNET (SSA, TEM- or streptozotocin-based chemotherapy, everolimus, sunitinib, PRRT, and oxaliplatin-based chemotherapy), three for siNET (SSA, PRRT, and everolimus), and three for lNET (SSA, everolimus, and PRRT). Moreover, many more options become available when locoregional treatments are considered feasible, and in case of re-challenge using a second-generation TKI (lenvatinib, cabozantinib, surufatinib. . .), PRRT, or another type of chemotherapy. In theory, 3 therapeutic options correspond to 6 possible sequences, 4 therapeutic options to 24 sequences, 5 therapeutic options to 120 sequences, etc. Fortunately, several guidelines propose some sequences, mainly according to the primary origin, the tumour grade, and the expression of SSTR.2,5,22 Moreover, the analysis of real-world data is informative as all patients will not be able to receive all the options. 48
What is done in real-life settings?
Although patients may benefit from multiple available therapeutic options, in most cases they are not able to receive all available therapies before mNET-related death or due to permanent related-toxicity. Several retrospective studies (described below) conducted in real-life settings provide practical information on the performed sequential treatment regimens, long-term outcomes, cumulative toxicity, and its impact on subsequent treatments. However, due to the design of such studies, there is often a gap between what was done and analysed several years ago and what is currently done. The current practices are indeed impacted by the results of randomized clinical trials (RCT) and/or the availability of new drugs; PRRT for instance has been made available in the United States only recently.
Pancreatic NET
In the study by Berdelou et al., 48 the most frequent first-line treatments for patients with advanced pNET were cytotoxic chemotherapy (60%) followed by SSA (25%). All lines combined, SSA, cytotoxic chemotherapy, trans-arterial chemoembolization, and everolimus were used in nearly half of pNET patients. However, some available treatment options such as sunitinib (26%) and PRRT (28%) were less often used in second-line and further treatments. In the LyREMeNET study 6 including 102 consecutive patients with metastatic pNET, patients were often treated with cytotoxic chemotherapy (72%), followed by SSA (56%), targeted therapies (33%), PRRT (11%), and liver embolization (9%). If the same type of study was to be performed after 2020, that is a period with a greater availability of PRRT, it would probably find a higher use of PRRT earlier within the sequence.
Small intestinal NET (jejuno-ileum NET, midgut)
A recent retrospective study conducted by Kulke et al. 49 assessed treatment patterns and outcomes of 273 patients with advanced siNETs in the United States. The first-line treatments used were SSA alone (88%) or in combination (2%), liver-directed therapy (8%), and cytotoxic chemotherapy or interferon (2%). Then, 57% of siNET patients received second-line therapy, among whom 93% continued SSA, most often with the addition of a second treatment (3% combined with PRRT). The median time to first progression following treatment initiation was 27.9 months. The median OS was 12.7 years for siNET patients after initiating the first-line therapy. In the LyREMeNET study including 153 consecutive metastatic siNET patients, almost all patients received SSA (93.5%) for either their anti-secretory effect and/or anti-tumour effect, followed by liver embolization (18%), everolimus (17%), and PRRT (7%) which was poorly available at this time in France. Although 86% of siNET patients had synchronous metastases, 72.5% of patients underwent a resection of their primary tumour. 6
Lung NET
Dasari et al. 50 assessed the treatment patterns and outcomes of 83 patients with advanced lNET in the United States. The first-line treatments were SSA alone (56%) or in combination (6%), cytotoxic chemotherapy (20%), external radiation therapy (9%), liver-directed therapy (4%), and everolimus/other (5%). Then, 72% of patients with lNET received second-line therapy: SSA alone (18%) or in combination (40%), cytotoxic chemotherapy (17%), everolimus (12%), liver-directed therapy (7%), external radiation therapy (3%), and others (3%). The median time to first progression following treatment initiation was 12.4 months. The median OS was 5.5 years after initiation of the first-line therapy. Another study 27 included 162 patients with metastatic lNET from two European expert centres. Patients received a median of one locoregional and three systemic treatments over the course of the disease. The most frequent first-line systemic treatment was SSA. All lines combined, 438 systemic treatments were analysed, and the 4 main systemic treatments received were cytotoxic chemotherapy (88%), SSA (81%), everolimus (42%), and PRRT (14%). The highest ORR (27%) and the longest median progression-free survival (PFS; 9.5 months) were reported with PRRT. 27
The three main factors influencing the choice of the best therapeutic sequence
During the dedicated MTB, the best sequence is chosen according to the criteria described above and according to three main different objectives: aiming for the highest treatment efficacy, aiming for the best safety profile, or following the patient preferences. Since none of these objectives is better than the others per se, the particular context of each situation must be considered.
Aiming for the highest efficacy
Unfortunately, there is no RCT assessing the question of therapeutic sequence. It was the initial primary-end-point of the Seqtor study [time to treatment failure by adding PFS1 + PFS2 + time between both treatments (chemotherapy and everolimus)], but the study aim was changed to a direct comparison in first-line (PFS1 after chemotherapy or everolimus). 35 Hence, the impact of a prior treatment on the efficacy of a subsequent one is unknown, so is the risk of selecting a more aggressive NET under therapeutic pressure.
When considering available RCT data, which now use PFS as the primary endpoint, and ORR as secondary endpoint (Tables 2–4), these data indicate that, in terms of efficacy:
- SSA was associated with improved PFS compared to a watch-and-wait strategy (placebo) for pNET, 7 siNET,7,8 and probably lNET even though it was not formally demonstrated in the Spinet study because of its early interruption due to patient recruitment issues. 44
- Doublet chemotherapy (CAPTEM) was associated with improved PFS and ORR compared to TEM in pNET, even though the results were already quite good for TEM monotherapy in patients who do not tolerate capecitabine (CAP). 16
- Doublet chemotherapy (5FU-Streptozotocin) was associated with similar PFS as everolimus in pNET, but the ORR was higher using chemotherapy. 35
- Everolimus was associated with improved PFS compared to placebo in all NET,10,11 even though a doubt remains for siNET (not significantly different in the subgroup of Radiant-2, 40 and the hazard ratio was 1.22 – in favour of the placebo – in Radiant-4 for the 71 ileum NET patients 41 ).
- Sunitinib was associated with improved PFS compared to placebo in pNET 12 ; surufatinib was also associated with improved PFS compared to placebo in all NET in a Chinese population13,14; second-generation TKI, such as axitinib 42 and pazopanib, 43 were also associated with a better PFS than placebo in non-pNET.
- PRRT (177Lu) was associated with improved PFS and ORR compared to a double dose of SSA in siNET, 9 and even though the RCT was non-comparative, the PFS was numerically longer compared to sunitinib in pNET. 38 In addition, recent retrospective studies have suggested that upfront PRRT after progression under SSA significantly improved PFS compared with upfront chemotherapy or targeted therapy in all NET, positioning PRRT earlier in the therapeutic sequence.51,52 Other ongoing RCT such as Compete (NCT03049189) or Compose (NCT04919226), comparing PRRT with everolimus or chemotherapy, should help confirm these data.
The indirect comparison is not methodologically valuable, especially in mNET, because of the heterogeneity of the disease (primary, grading, SSTR expression. . .), thus their inherent prognoses are very variable. In addition, most studies consider different treatment lines and different populations, even though the two studies focusing on pNET and evaluating targeted therapies (sunitinib or everolimus, both versus placebo) were similar on these points.10,12 It should be noted that none of these RCT have proven an OS gain in patients with mNET.
Aiming for safety
The data related to AE available from the above-mentioned RCT should evidently be taken into account, but are not detailed herein as they are beyond the scope of the present review. However, when presenting the different treatment options to patients, physicians must state the differences between:
The short-term toxicity, that is tolerance under treatment, which is estimated by the frequency and grade of AE. Some AE could be very prevalent, but of low grade, and still have an impact on the patient’s QoL. This aspect should be considered while taking into account the duration of the treatment (e.g. high frequency of AE for liver embolization or IV chemotherapy, but administered for a short period of time). The frequency of symptomatic AE, their grade, and the duration of treatment will impact the QoL/preference of patients (see below).
The risk of long-term toxicity, that is sequelae, is usually rarer but of importance as it may preclude the administration of further treatments.
Regarding the systemic treatment options, SSA is the only drug for which there is almost no cumulative risk, no need for laboratory follow-up, and for which very long-term administration can be performed without sequelae risk.7,8,34 The risk of biliary lithiasis and pancreatic exocrine insufficiency are easily manageable in routine practice. In contrast, chemotherapies, targeted therapies (everolimus and sunitinib), or PRRT require caution and regular clinical and laboratory follow-up because AE are more frequent under treatment, sometimes life-threatening (of grade 3–4), and/or are associated with a risk of sequelae such as renal deficiency (streptozotocin, sunitinib, everolimus, PRRT), cardiotoxicity (sunitinib), and haematological toxicity (all four treatments).10–12,15,53 The choice between a doublet chemotherapy (CAPTEM) compared to a monotherapy with TEM in pNET must take into account the cumulative short-term and long-term toxicities (doubling of grade 3–4 toxicity rates in CAPTEM compared to TEM arm, 45% versus 22%, p = 0.005), counterbalanced with the cumulative efficacy due to the addition of CAP. 16 Of note, this specific study reported the secondary occurrence of seven primary cancers, three in the TEM arm, and four in the CAPTEM arm, including one case of myelodysplastic syndrome in the CAPTEM arm. The choice between everolimus or sunitinib, which appear to have a relatively similar efficacy in pNET, will be mainly driven by their respective contra-indication and toxicity profiles, which are more metabolic for everolimus and cardiovascular for sunitinib. Another very important point, that is not investigated in RCT, is the cumulative risk of administering these different treatments in association (rarely) or sequentially. Berdelou et al. 48 reported that over a median OS duration of 5 years, the frequency of chronic toxic events increased with the number of lines received, from 4% after the first line to 24% after five lines. Two classic examples of such chronic toxicities concern the severe chronic renal deficiency that may appear over 3–5 years due to the cumulative nephrotoxicity of each treatment (steptozotocin, everolimus, sunitinib, PRRT, iode because of multiple CT scans or during liver embolization) and the risk of myelodysplasia syndrome/acute leukaemia, which may be increased by associating alkylating agents and PRRT.54,55 This illustrates the importance of real-world data for the evaluation of the cumulative risk. Lastly, the surgical removal of the primary tumour, sometimes discussed even at a metastatic stage, is risky in itself, post-operatively of course, but also in terms of long-term toxic effects such as diarrhoea and short bowel syndrome after siNET resection or exocrine pancreatic insufficiency after duodenopancreatic (Whipple) resection. Biliary anastomoses after Whipple resection will also induce a relative contra-indication for further liver embolizations.
Following patient preferences: QoL and symptom tracker
The QoL data are now usually reported in all pivotal RCT that lead to approvals of systemic therapies. 56 The Netter-1 study was the only study to find a statistically significant improvement in overall QoL in several domains, 57 while the trial comparing sunitinib to placebo in pNET found a worsening of the QoL only in the diarrhoea domain. 58 Interestingly, all the RCT (for sunitinib, everolimus, and SSA)58–60 performed versus placebo showed no worsening in the overall QoL. This could be explained in two ways: (a) the AE are well counterbalanced by the higher efficacy of therapies and potentially fewer disease-related symptoms than those reported with placebo, (b) there is no adequate tool to assess small differences in QoL between treatments for mNET. 61 This highlights the importance of patient-reported outcome measures (PROM) as a key element in making appropriate treatment decisions. Specific symptom assessment using PROM aims to capture data more objectively while increasing the granularity level of these data. Interestingly, the EORTC-QLQ-C30 scale seems limited to reveal slight differences in flushing, which is, with diarrhoea, one of the two main symptoms of the carcinoid syndrome. In two randomized studies, better QoL was reported for the diarrhoea domain in favour of the experimental arm (PRRT 57 and telotristat 20 ), while there was no difference in flushing. However, using a symptom tracker more focused on the main symptoms, patients also noted a significant improvement in flushing episodes under PRRT 62 and telotristat 63 compared to the control arm. Therefore, by connecting a symptom tracker app to medical files, physicians can monitor patients more closely and better manage troublesome symptoms that affect patient QoL. 64
A survey including patients who had received three or more different treatments for mNET showed a poorer perceived tolerance for IV versus oral chemotherapy. The best perceptions among treatments were obtained for SSA and PRRT, while the worse were obtained for liver embolization and IV chemotherapy. 65 Because of AE and the higher frequency of appointments during treatment, patients may prefer short-term systemic treatments with a well-defined treatment period (PRRT, induction of chemotherapy) rather than a continuous systemic treatment. More and more, studies now investigate the aims of patient-centred care and how to keep patients at the centre of treatment decisions. 66 This is important since better patient involvement can enhance adherence and compliance to the treatment. 67
Proposition of therapeutic sequences
Pancreatic NET
Functioning pNET
The anti-secretory treatment of a functioning pNET is the priority (PPI for Zollinger-Ellison syndrome; diazoxide, pasireotide, and everolimus for their proglycaemic effect could help against metastatic insulinoma; SSA for VIPoma and glucagonoma). 68 For refractory functioning metastatic pNET, locoregional (interventional radiology or debulking surgery) or systemic treatments must be rapidly discussed to reduce the tumour volume. Against metastatic insulinoma, everolimus is a good first-line treatment option, not only because PFS under everolimus is similar to that under chemotherapy, 35 but because of its proglycaemic effect which helps in the control of hypoglycaemia. 69
Besides controlling the secretory syndrome, a biliary stent is sometimes required for a NET within the head of the pancreas, which is also the case for non-functioning pNET. Although the removal of the primary pNET is discussed on a case-by-case basis in order to treat or avoid locoregional complications, there is no proof of a survival benefit of removal in the context of pNET 70 (Figure 2).

First-line treatment (a) and therapeutic sequence (b) for metastatic pancreatic, jejuno-ileum, and lung neuroendocrine tumours (NET).
Non-functioning pNET
SSA is the recommended upfront treatment for slow-growing advanced G1-2 pNET. 7 In contrast, chemotherapy (streptozotocin-based or TEM-based) is considered as the standard systemic upfront treatment for patients with bulky disease, significant tumour growth, high Ki67 (>10%), and/or symptomatic pNETs.15,16 When the treatment aims to reduce the tumour burden, a bichemotherapy (TEM in combination with CAP 16 ) or PRRT are preferred. Regarding the choice between chemotherapies (alkylating agent versus oxaliplatin-based chemotherapy), a prospective randomized study is warranted, especially to evaluate whether MGMT status could help in this decision. 25 For patients in whom an objective response is not needed, a targeted therapy could be a first-line treatment option (as shown with everolimus in the Seqtor study). 35 In second-line, either after SSA or chemotherapy, everolimus and sunitinib are recommended in progressive G1-2 pNET,10,12 while belzutifan is the treatment of choice for patients with advanced VHL-related pNET. 33 Since the presentation of the first results of the Oclurandom study, 38 patients with SSTR-positive pNET can be treated with PRRT before the administration of targeted therapies (Table 2 and Figure 2(b)). In addition, a recent retrospective study in mNET supports the earlier use of PRRT, especially after progression under SSA. 51 In this setting, some ongoing RCT are aiming to assess whether PRRT could be an option, possibly upfront in first-line treatment, and in the subgroup of patients with high Ki67 [10–50% for the Netter-2 study (NCT03972488) and 15–55% for the Compose study (NCT04919226)] in whom a high expression of SSTR remains.
Small intestinal NET (also referred to as jejuno-ileum NET or midgut NET)
Functioning siNET (carcinoid syndrome)
SSA is almost always required for its anti-secretory effect due to the presence of a carcinoid syndrome in the majority of patients with metastatic siNET. 26 Telostristat can help control the carcinoid syndrome in addition to SSA, but it has no additional anti-proliferative effect. 20 Liver embolization may also play an important role in controlling the carcinoid syndrome when hepatic tumour burden is predominant. 71 Moreover, several recent studies suggest that PRRT may have an anti-secretory effect, but further confirmation is warranted. 72 In case of carcinoid syndrome and/or elevated 5HIAA, regular screening and treatment of the carcinoid heart disease must be performed (Figure 2(a)).30,73
Non-functioning siNET
Fewer options are available for the management of metastatic non-functioning siNET patients. SSA can also be used for its anti-tumour effect7,8 since most siNET are related to good prognostic factors (low grade, slow growth, Ki67 ⩽ 10%). Because of the long survival of these patients, even at a metastatic stage (>10 years), the surgical resection of the primary siNET, and of its mesenteric mass if present, is performed for most patients (with cholecystectomy in order to avoid possible complications under SSA or liver embolization).74,75 Liver embolization can also be used for anti-tumour purposes to control liver tumour growth, especially since few systemic options are available (everolimus or TKI are less effective against siNET than pNET).2,73 Since most siNET harbour high SSTR expression levels, PRRT is the standard systemic treatment right after SSA9,51; everolimus however, is less effective and TKI are not yet approved (Table 3 and Figure 2(b)).
Lung NET
Functioning lNET (carcinoid syndrome)
The management of carcinoid syndrome associated with lNET is similar to that described above for siNET. SSA is the recommended first-line anti-secretory treatment. 23 Screening and follow-up for carcinoid heart disease is also similar. A Cushing syndrome associated with lNET must be treated first and foremost, in close collaboration with an endocrinologist. 23
Non-functioning lNET
Even fewer published data regarding the best therapeutic sequence in the context of lNET are available (Table 4). Even though the Spinet study has been interrupted early because of a low inclusion rate, SSA is the recommended upfront treatment in slow-growing advanced G1-2 lNET. 23 Everolimus is the only approved drug for lNET and is therefore recommended in second-line after SSA, or upfront for more aggressive cases (Figure 2(a)).11,47 As for pNET with higher tumour grade and rapid growth (often without SSTR expression), chemotherapy could be used, even though this treatment has been less evaluated and is less effective than for pNET (Figure 2(b)). One RCT (NCT04665739) is comparing PRRT and everolimus in the context of lNET and aims to better determine the place of PRRT in the management of lNET with SSTR expression, which is less often encountered in the context of lNET compared to pNET and siNET. Following the results of current ongoing RCT, TKI will probably become available soon, as they already are for extra-pNET.14,42
Conclusion
Depending of the NET characteristics, 3 to 7 different treatment options are available, corresponding to 6–5,040 theoretical different sequences. Even though each patient is unique and the NET characteristics are heterogeneous, the present review discusses the main sequences and offers perspectives on how one can propose the best sequence to treat mNET on a case-by-case basis. Each treatment must be discussed within a dedicated MTB conference, and inclusions in clinical trials should be favoured. After a thorough characterization of the patients and their mNET, and taking into consideration the available drugs, the first-line treatment is chosen according to the treatment aim. This aim is based on three main topics (efficacy, safety, and patient preferences) that do not necessarily converge, and must be defined a priori. At baseline, physicians should design an a priori full therapeutic sequence, which may evolve at each step based on the response to previous treatment, the occurrence of chronic toxicities, and the patients’ perception of the prior treatment. To improve knowledge in terms of effectiveness and risk of cumulative toxicities regarding the different sequences, real-world data using long follow-up durations are necessary; such issues will not be resolved by RCT.
