Abstract
Objective
To investigate the clinicopathological features, diagnosis and differential diagnosis of patients with primary ovarian carcinoid tumours arising in mature cystic teratomas.
Methods
This retrospective case series analysed the data from patients with primary ovarian carcinoid tumours arising in mature cystic teratomas.
Results
The study enrolled four patients. Histopathological analysis of the tumours identified the following subtypes: insular (n = 1), trabecular (n = 1) and strumal (n = 2). All four primary ovarian carcinoid tumours originated from a mature teratoma. The morphology of the primary ovarian carcinoids was similar to other neuroendocrine tumours. Strumal carcinoids were composed of different proportions of thyroid tissue intimately admixed with carcinoid tumour. Tumour tissue was arranged in insular and/or trabecular patterns. The nucleus of tumour cells displayed exquisite chromatin without obvious mitotic figures. Tumour tissues were positively stained for neuroendocrine markers chromogranin A, synaptophysin and CD56 to varying degrees. Strumal carcinoid tumours were cytokeratin 19 positive and thyroid transcription factor 1 negative. No recurrence or metastasis occurred during follow-up (12–71 months).
Conclusion
Primary ovarian carcinoid tumours arising in mature cystic teratomas are rare. Diagnosis and differential diagnosis should be confirmed by clinical features, histopathological characteristics and specific immunophenotyping.
Introduction
Primary ovarian carcinoid tumours within a mature cystic teratoma are relatively rare tumours that resemble well-differentiated neuroendocrine tumours of the gastrointestinal tract. 1 They account for 0.5% of all carcinoid tumours and 0.1% of all ovarian neoplasms. 2 Primary ovarian carcinoid tumours are composed of a single-layer of germ cells, mostly associated with teratoma. 3 They originate from neuroendocrine cells in a mature ovarian cystic teratoma arising from the epithelium of the middle respiratory tract and gastrointestinal tract.2,4,5 They are classified into four subtypes based on their histological morphology: (i) insular; (ii) trabecular; (iii) strumal; and (iv) mucinous.2,4,5
Carcinoids are most frequently identified in the ileum and appendix. 6 Although mature cystic teratomas are benign, ovarian carcinoids are generally considered to be malignant, being occasionally associated with metastases. 7 This current study aimed to evaluate the clinicopathological features, histological origin, clinical manifestations, treatment and prognosis of ovarian carcinoid tumours arising in mature cystic teratomas.
Patients and methods
Study population
This retrospective case series enrolled consecutive patients diagnosed with primary ovarian carcinoid tumours between January 2011 and December 2019 in the Department of Gynaecology, Beijing Shijitan Hospital, Capital Medical University, Beijing, China. Demographic and clinical data on age, symptoms, major complaints, imaging manifestations, surgical methods, intraoperative conditions and histopathological diagnosis were recorded. The inclusion criteria were as follows: (i) patients with a primary ovarian carcinoid tumour originating from a mature cystic teratoma; (ii) no history of carcinoid and/or malignant tumours of other organs. The exclusion criteria were as follows: (i) patients with secondary or metastatic ovarian carcinoid tumours; (ii) puncture specimens.
This study was approved by the Ethics Committee of Beijing Shijitan Hospital and was conducted in accordance with requirements in the Declaration of Helsinki. Patients provided written or verbal informed consent. The reporting of this study conforms with the CARE guidelines. 8
Histopathological and immunohistochemical analyses
All tumour specimens were fixed in 10% buffered neutral formalin and processed into paraffin-embedded biopsy tissue blocks, then continuously sliced into 3-μm sections. Immunohistochemical staining was performed on 3-μm sections of formalin-fixed paraffin-embedded tissue and processed in an automatic immunohistochemical staining machine using standard protocols (intelliPATH FLX® system; Biocare Medical, Pacheco, CA, USA; EnVision staining). Primary antibodies were used against the following antigens (all from OriGene China, Beijing, China unless stated otherwise): cytokeratin (CK) (clone AE1); cytokeratin 19 (CK19) (clone RCK108); thyroglobulin (TG) (clone 2H11/6E1); thyroid transcription factor 1 (TTF-1) (clone 8G7G3/1; synaptophysin (Syn) (clone SP11); chromogranin A (CgA) (clone SP12); cluster of differentiation 56 (CD56) (clone 123C3); oestrogen receptor (ER) (clone SP1); progesterone receptor (PR) (clone 16); mismatch repair protein (MLH1) (clone ES05); mismatch repair protein 2 (MSH2) (clone UMAB259); MutS protein homolog 6 (MSH6) (clone UMAB258); postmeiotic segregation increased 2 (PMS2) (clone MOR4G); Ki67 (clone UMAB107) and p53 (clone DO7). The positive control was established according to the manufacture’s instruction and 10 mM phosphate-buffered saline solution (pH 7.2–7.4) was used as negative control.
Tumour marker determination
Serum for tumour marker assessment was separated by centrifugation immediately after clotting and stored at –20°C until it was used in these assays. The following serum cancer markers were measured using chemiluminescence immunoassay kits according to the manufacturer’s instructions (Abbott Laboratories, Abbott Park, IL, USA): alpha fetoprotein (AFP) (ng/ml), cancer antigen (CA) 19-9 (U/ml), CA15-3, CA125 (U/ml) and carcinoembryonic antigen (CEA) (ng/ml). The cut-off levels were recommended by the manufacturer. Internal quality control was performed for all the tumour markers before testing.
Results
This study collected data from four patients with primary carcinoid tumours of the ovary, with a median age of 52 years (range 32–75 years). One patient had a tumour on the left ovary and three patients had a tumour on the right ovary (Table 1). All of the carcinoid tumours were accompanied with a mature cystic teratoma. No obvious clinical symptoms occurred in two patients, which were identified accidentally. The other two patients were admitted to the hospital due to abdominal distension and massive abdominal mass, without fever, constipation, abdominal pain or diarrhoea. Patient 1 had elevated serum levels of alpha fetoprotein (AFP) (2853 ng/ml), slightly elevated cancer antigen (CA) 19-9 and CA15-3, normal CA125 and carcinoembryonic antigen (CEA); whereas all serum cancer indicators were within normal limits after the operation. In the other three patients, AFP, CA19-9, CA125, CA15-3 and CEA were within normal limits. Imaging identified fat-containing cystic masses with multiple septa and internal calcification. As the radiological findings suggested that these ovarian tumours were mature teratomas the patients underwent ovarian cystectomy.
Demographic and clinical characteristics of the four patients with a primary ovarian carcinoid tumour originating from a mature cystic teratoma that were analysed in this study.

TNM, tumour, nodes, metastases staging system; CK, cytokeratin; Syn, synaptophysin; CgA, chromogranin A; CD56, cluster of differentiation 56; CK19, cytokeratin 19; TTF-1, thyroid transcription factor 1; TG, thyroglobulin; ER, oestrogen receptor 1; PR, progesterone receptor.
Macroscopic examination revealed multicystic masses containing viscous sebaceous material and hairs, with a volume ranging from 7.5 × 6.5 × 5.0 cm to 32.0 × 24.0 × 9.5 cm (more clustering on the right ovary). Mature squamous and respiratory epitheliums, as well as mesenchymal tissue including cartilage, smooth muscle and adipose tissue were observed. An area consisting of nests and trabeculae was notable in the cyst wall, with a volume ranging from 0.5 × 0.3 × 0.2 cm to 0.7 × 0.6 × 0.5 cm. Tumour tissues were arranged in island, nest, girdle-rope or cord-like patterns. The neoplastic cells were polygonal or oval, with abundant eosinophilic cytoplasm and small round nuclei without atypia or mitotic activity. Histopathological analysis of the four patients demonstrated that the tumour subtypes were as follows: insular (n = 1), trabecular (n = 1) and strumal (n = 2).
Immunohistochemical staining displayed positivity of CK, Syn, CgA and CD56 to varying degrees (Figures 1–3), indicating the neuroendocrine origin of these tumours. The ovarian strumal subtype in two patients displayed intertwined thyroid and carcinoid components under morphological examination, but with clear distinction in the immunophenotypes between the two areas (Figures 2). Carcinoid cells were strongly positive for CK19, Syn, CgA and CD56, whereas they were negative for TTF-1 (Figures 1–3). Goitre region cells were positive for TTF-1, whereas they were negative for neuroendocrine markers. The Ki67 index was classified as low grade (1–2%; Figure 3A). The mismatch repair proteins were present in all patients.

Representative photomicrographs showing the histology and immunophenotype of an insular subtype carcinoid tumour: (a) neoplastic cells were uniform with small round nuclei, without atypia or mitotic activity (haematoxylin and eosin, scale bar 100 µm); (b–d) diffuse positive staining of cytokeratin, chromogranin A and synaptophysin, respectively (EnVision staining, scale bar 100 µm). The colour version of this figure is available at: http://imr.sagepub.com.
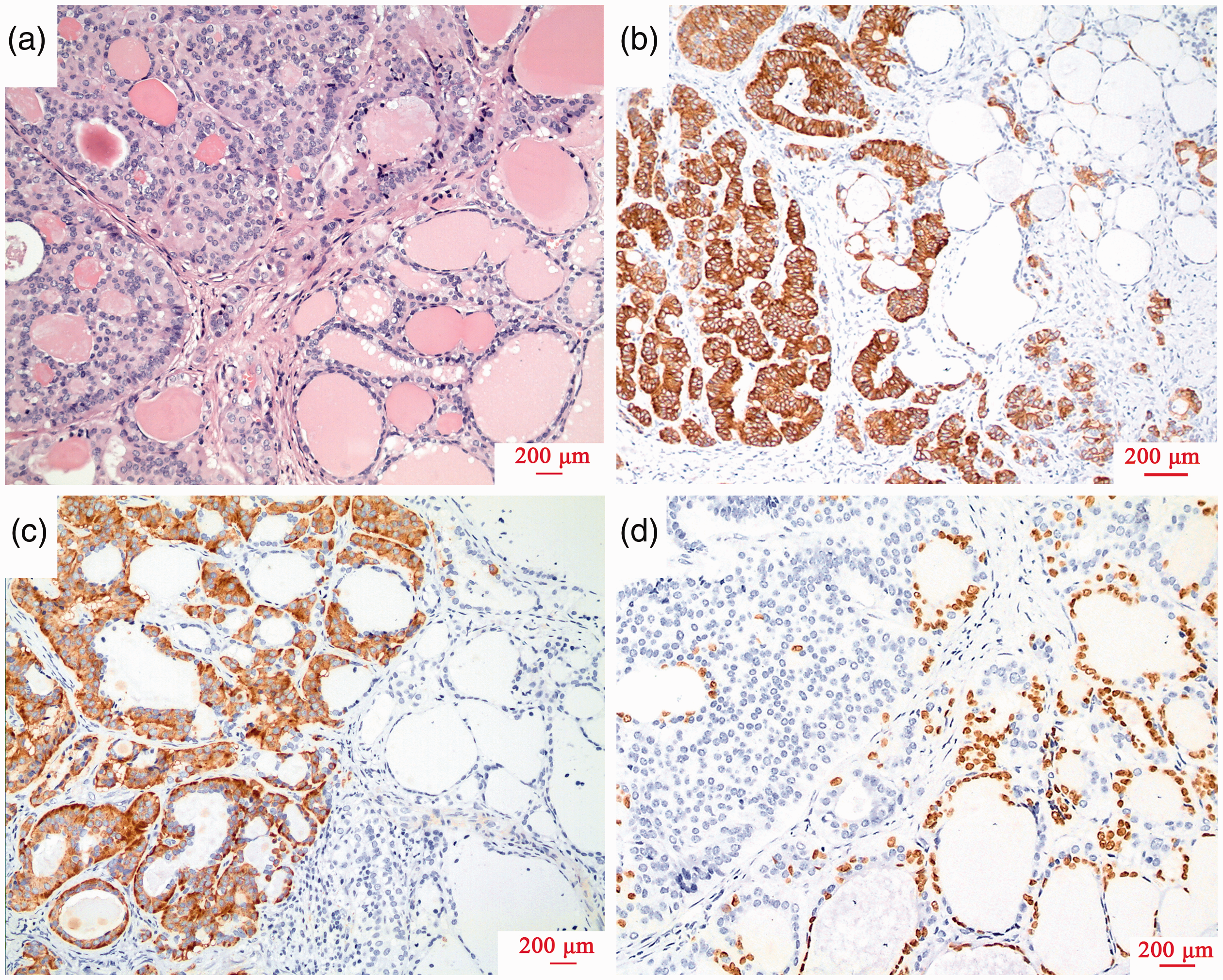
Representative photomicrographs showing the histology and immunophenotype of a strumal subtype carcinoid tumour: (a) carcinoid and thyroid regions are intertwined with eosinophilic secretions in the cavity (haematoxylin and eosin, scale bar 200 µm); (b and c) positive staining of cytokeratin 19 and synaptophysin, respectively, in carcinoid regions (EnVision staining, scale bar 200 µm); (d) negative staining of thyroid transcription factor 1 in carcinoid regions but positive in thyroid follicles (EnVision staining, scale bar 200 µm). The colour version of this figure is available at: http://imr.sagepub.com.

Representative photomicrographs showing the histology and immunophenotype of a trabecular subtype carcinoid tumour: (a) Ki67 staining in a primary ovarian carcinoid (EnVision staining, scale bar 200 µm); (b) trabecular subtype (black arrow) showing transition with bronchial epithelium (red arrow) (haematoxylin and eosin, scale bar 200 µm); (c) trabecular subtype showing tumour cells arranged in a beam and cable pattern, with a mucinous cystadenoma component at the right lower field (red arrow) (haematoxylin and eosin, scale bar 200 µm); (d) ovarian borderline mucinous cystadenoma (haematoxylin and eosin, scale bar 200 µm). The colour version of this figure is available at: http://imr.sagepub.com.
Morphological and immunohistochemical findings indicated that the carcinoid tumours presented with coexisting mature cystic teratomas. Mature squamous and respiratory epitheliums, as well as mesenchymal tissue including cartilage, smooth muscle and adipose tissue were identified. In one patient with the insular subtype, the carcinoid tumour was located adjacent to respiratory epithelium (Figure 3B). In one of the patients with a strumal subtype carcinoid tumour displayed a mucinous cystadenoma area (Figure 3C). The patient with a trabecular subtype carcinoid tumour exhibited a borderline mucinous cystadenoma area (Figure 3D).
All four patients received no further treatment and no recurrence or metastasis was observed after 12–71 months of follow-up.
Discussion
Mature teratoma of the ovary is known to be the most common ovarian tumour, representing 20% of all ovarian neoplasms. 2 Malignant transformation of teratomas has been described, but they are rare. 9 The most common form of malignant transformation from ovarian mature cystic teratomas are squamous cell carcinomas, followed by adenocarcinomas then carcinoid tumours.10,11 Carcinoid tumours most frequently occur in the gastrointestinal tract, followed by the lung.12,13 Ovarian carcinoid tumours can be primary or metastatic. 14 Most primary tumours are unilateral masses. 15 However, the contralateral ovary displayed a cystic teratoma or mucinous neoplasms in 16% of patients. 15 The age of onset varied greatly, mostly (more than 2/3) at intra- or postmenopause (>40 years old). 16 Literature reports that these tumours can originate from neoplastic hyperplasia of gastrointestinal or respiratory tract epithelium, which mimics amine precursor uptake decarboxylation (APUD) cells, or directly within teratomas from primitive neuroectodermal or amorphous endodermal epithelium with potential to differentiate to APUD cells.17,18 Based on the coexistence of teratoma components, this type of carcinoid tumour might belong to teratomas with a high specificity in a single layer of germ cell origin. 2 In the 4th World Health Organization classification of female reproductive system tumours, carcinoid tumours were classified as monoblast teratomas and somatic tumours originating from epithelioid cyst, and both components were considered as the endodermal origin of teratomas. 2 Most patients with carcinoid tumours have no specific symptoms or displayed adnexal masses identified during physical examination. 19 Many carcinoids resulted in clinical syndromes upon autocrine secretion of vasoactive substances. Some patients’ main symptom was severe constipation. 20 The polypeptide YY hormone produced by carcinoids was related to severe constipation. 21 Carcinoid symptoms included steroid hormone elevation, hyperinsulinaemia, skin flushing, abdominal pain and diarrhoea. 17 When the diameter of the tumour cell island was greater than 10 cm, the occurrence of symptoms significantly increased. 22
A previous study reported that the rate of positive expression of serum CA19-9, CA125, AFP, CEA and CA153 in patients with ovarian mature cystic teratoma was 45.83%, 6.09%, 0.00%, 0.64%, and 2.78%, respectively. 23 In this current study, the serum levels of AFP, CA125 and CA19-9 varied greatly: in patient 1, the preoperative AFP level was high, but it returned to normal after the operation. It is possible that the high levels of AFP were related to a component of teratoma, but this had not been previously reported in the literature.
This current study described four patients that had developed unilateral carcinoid tumours in the ovary, with no abnormal gastrointestinal tract and no constipation or other carcinoid symptoms. Patient 1 had a pregnancy complicated by a mature cystic teratoma. She had increased levels of preoperative AFP, CA19-9 and CA15-3 to varying degrees. The increase in AFP might be related to her having a teratoma combined with a carcinoid tumour. The levels of serum tumour markers, including AFP, CA19-9, CA125, CA15-3 and CEA, in the other three cases were within the normal ranges as previously reported.23,24
Most primary ovarian carcinoids coexist with teratomas and mucinous cystadenomas. 25 Approximately 85% of primary ovarian carcinoids are accompanied by components of teratomas. 25 Occasionally, mucinous cystadenomas or Brenner-like transitional cell nests can be identified. 3 Only 10–15% of primary ovarian carcinoids are pure carcinoids. 3 The majority of primary ovarian carcinoids are diagnosed incidentally. 26 In this current study, all four patients had tumours that were complicated by a mature cystic teratoma of the ovary and two had tumours that coexisted with mucinous cystadenomas. Morphological and immunohistochemical findings indicated a carcinoid tumour arising in the epithelium of the mature cystic teratoma.
Generally, no typical greyish-yellow nodules were observed in the proximal cephalic region after extensive sampling in the current study. Sufficient tumour tissue sampling is necessary to identify very small lesions and to define unusual complications (such as immature teratoma components, primitive nerve tube, germ cell tumours or neuroendocrine tumours), so as to provide a better evaluation of prognosis and efficient clinical therapy. 27 Primary ovarian carcinoid tumours originate from neuroendocrine cells. 2 These slow-growing tumours are classified into four subtypes. 2 The insular subtype mainly consists of islands and nest structures, while the trabecular subtype contains wavy, striped or parallel trabecula, surrounded by dense fibrous matrix of connective tissue, which is similar to a carcinoid originating from the hindgut or foregut. 28 For the trabecular subtype, tumour cells were elongated columnar, with their long axes perpendicular to the striations. 29 The strumal subtype is mostly associated with ovarian teratoma and is mainly composed of thyroid gland mixed with insular carcinoid island. 30 The morphology of thyroid tissue was similar to normal thyroid gland. 30 Follicles are well differentiated and lined with flat, cubic or columnar epithelium. 31 The gland lumen contains abundant powdery glia. 31 Carcinoid cells have a similar morphology to that of the thyroid. 32 Carcinoid components can be mixed together or separated by a transitional zone in between. 30 In the strumal subtype, two components were closely mixed with a transitional zone, which was consistent with other research. 30 Thyroid tissue varied greatly in different patients, from well-formed thyroid follicles to nodular goitre and adenomatous hyperplasia. 33 No mucinous carcinoids were observed in this current study. Mucinous carcinoid has a higher proliferative activity and invasiveness, and its prognosis is slightly worse than the other subtypes (insular, trabecular and strumal subtypes). 34 In this current study, for patients 3 and 4, their ovarian carcinoid tumours were combined with a (borderline) mucinous cystadenoma arising in a mature cystic teratoma, which was extremely rare in primary ovarian tumours.
The immunophenotype of the tumours in the current four patients showed positivity for CK, Syn, CgA and CD56 to varying degrees, which was consistent with carcinoid tumours. In the ovarian strumal subtype, the carcinoid and thyroid regions were intertwined, with clear distinction in the immunophenotypes between the two areas. TTF-1 was positive in the nodular goitre region and negative in the carcinoid region. Neuroendocrine markers were negative in the goitre region and strongly positive in the carcinoid region. Both components were of an endodermal origin in the teratoma, instead of being differentiated from pluripotent stem cells. The incidence of ovarian strumal carcinoid is very low, 35 so neuroendocrine markers that are positive in the carcinoid region and negative in the thyroid region could help differentiate from primary malignant struma ovarii. In most of the primary ovarian carcinoid tumours reported in the literature,28,36 including in this current study, the neuroendocrine immunohistochemical markers were all positive to different degrees; meanwhile Syn had the highest sensitivity and specificity.
There is also a need to differentiate carcinoid tumours from some other tumours. The major differential diagnoses were the metastatic mucinous carcinoid tumours. 37 Neuroendocrine tumours, accounting for 98.8% of all cases, mainly occur in the gastrointestinal tract (73.7%) and respiratory tract (25.1%). 4 Therefore, in order to be able to diagnose a primary ovarian carcinoid tumour it is essential to first exclude metastasis. A metastatic carcinoid contains bilateral multiple ovarian foci, accompanied by peritoneal metastases with no component of a teratoma. 38 The presence of positive CDX2 could assist with differentiating a primary ovarian carcinoid tumour from metastatic tumours. 39 A previous study reported that most primary ovarian carcinoid tumours originated from the foregut and hindgut, which can express CDX2. 40 Furthermore, careful examination during surgery and other postoperative investigations should not reveal any other primary neoplasms. Ovarian malignant goitre (especially goitre-type carcinoid) is usually composed of thyroid and carcinoid tissues, but if the goitre tissue becomes the dominant component, then small amounts of carcinoid tissue arising from the thyroid tissue could often become hidden. The diagnostic criteria of ovarian malignant goitre is the same as those for primary thyroid cancer, of which the most common type is papillary cancer. 30 Histological features including ground glass nucleus, grooves and intranuclear inclusion bodies have the most diagnostic value. 30 Other differential diagnoses include Krukenberg tumours, Sertoli cell tumours and granulocytomas. 41 Clinical history, histological features and immunohistochemical markers could be used for differential diagnosis.
At present, there is no optimal treatment for this type of low-grade malignant tumour. Almost all ovarian carcinoid patients are stage I (The International Federation of Gynecology and Obstetrics staging system) with an excellent prognosis. 38 Especially small lesions confined to the cystic wall of teratomas are mostly benign. 2 While surgery is the mainstay of treatment, complete surgical excision of unilateral appendages and close follow-up are highly recommended. 42 Mucinous carcinoid is more invasive than the other subtypes.11,43 For those with extraovarian dissemination, the lesions should be removed as completely as possible. Therefore, young patients with an intact tumour capsule, a normal contralateral ovary and without metastases should undergo unilateral ovariectomy and be followed-up closely. For elderly women without the need for fertility preservation, hysterectomy and bilateral adnexal excision combined with chemotherapy is an option.
Patients with carcinoid tumours where the disease is confined to one ovary show excellent survival. 33 However, if the disease is diagnosed at a more advanced TNM stage, then the outcome is poor. 44 A previous report described a rare case of goitre carcinoid metastasis to the contralateral ovary, myometrium and lung. 45 Another report described a case of primary ovarian goitre carcinoid with multiple bone and breast metastases that occurred within 3.5 years of the original surgery and chemotherapy was ineffective. 46 The prognosis of goitre carcinoid with a high expression of topoisomerase II alpha and Ki67 may be poor. 46 Further studies are required to determine the appropriate chemotherapy regimen. The four patients with primary ovarian carcinoid tumours included in this current study underwent unilateral adnexal excision without radiotherapy or chemotherapy, but remained clear of tumour recurrence or metastasis during follow-up.
In conclusion, primary ovarian carcinoid tumours arising in mature cystic teratomas are rare tumours. Diagnosis and differential diagnosis should be confirmed by clinical features, histopathological characteristics and specific immunophenotyping. It is important to sample any solid part within a mature teratoma extensively in order not to miss the possibility of small carcinoid tumours. Surgery with or without chemotherapy is still the main treatment for those with high proliferative activity or high TNM stage.
Footnotes
Acknowledgement
We would like to thank all the participants for their contribution to this research project.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the following: Beijing Municipal Administration of Hospitals’ Ascent Plan (no. DFL20180701); Special Fund for the Capital Characteristic Clinical Medicine Development Project (no. Z161100000516077); Beijing Municipal Grant for Medical Talents Group on Peritoneal Surface Oncology (no. 2017400003235J007); Key Discipline Development Fund of Beijing Shijitan Hospital affiliated to the Capital Medical University (no. 2016fmzlwk); Beijing Natural Science Foundation (no. 7172108); Youth Research Fund of Beijing Shijitan Hospital (no. 2020-q11).
