Abstract
Tebentafusp is a first-in-class immunotherapy agent that comprises an engineered T-cell receptor targeting a gp100 epitope presented by human leukocyte antigen-A*02:01 cells, fused to an anti-CD3 single-chain variable fragment. Tebentafusp is both the first bispecific T-cell engager to show efficacy in the treatment of advanced solid cancer and the first anti-cancer treatment to demonstrate an overall survival benefit in patients with uveal melanoma (UM). This review article will focus on the clinical development of tebentafusp, the mechanism of action and resultant evolution of the management of advanced UM.
Introduction
Uveal melanoma (UM) is a rare cancer, with an approximate worldwide incidence of around 6500 cases per year, mainly affecting Caucasian patients.1,2 Treatment options for primary lesions include radiation (brachytherapy or proton beam therapy) or surgical enucleation. Approximately half of the patients will develop metastatic disease, 3 and patients remain at lifelong risk of relapse. Most commonly, relapse occurs in the liver (~90%) with less common sites for metastatic spread including the lung, bone and skin. 4 Risk of relapse is greatest at 1–5 years from initial diagnosis and is higher for patients over 50 years of age. 5 Risk factors for relapse include larger size of primary tumour, higher mitotic count and the presence of two genetic alterations: monosomy 3 and 8q amplification.6,7 Genomic analysis can assist in risk stratification for relapse, aiding in surveillance decisions.
Despite significant improvements in the management of metastatic cutaneous melanoma (CM) over the last decade, similar improvements for patients with UM have been less forthcoming, and until recently the median survival for metastatic UM remained less than 1 year. 8 No clinical trials of systemic agents have demonstrated significant survival advantages for patients to date (Table 1). Based on these trials, standard-of-care (SOC) treatment options have remained limited to the use of immune checkpoint inhibitors (ICPIs) which demonstrate only a very modest improvement in survival for most patients treated. When treatment responses do occur, they are often short-lived. Possible reasons for this include the low tumour mutational burden observed in UM, 9 low programmed death-ligand 1 (PD-L1) expression 10 and the immunosuppressive tumour microenvironment, 11 as summarised in Figure 1.
Summary of past clinical trials in UM.

Included studies were prospective in design and included a minimum of 10 patients with UM.
CI, confidence interval; HIA, hepatic intra-arterial; ICPIs, immune checkpoint inhibitors; m, months; mOS, median overall survival; mPFS, median progression-free survival; NR, not reached; UM, uveal melanoma.

Due to the generally poor outcomes for UM, the recommendation for patients with metastatic UM is to be offered clinical trials wherever possible. 31 International guidance also recommends consideration of locoregional management of liver disease including resection, ablation or stereotactic management in those patients for whom this is clinically and technically feasible.31,32 However, with the recent approval of tebentafusp, the treatment landscape of metastatic UM is finally set to change.
ImmTACs
Immune mobilising monoclonal T-cell receptors against cancer (ImmTACs) are a class of bispecific T-cell engagers. Tebentafusp is the first ImmTAC shown to demonstrate a survival benefit in any solid tumour. 33 ImmTACs combine a soluble T-cell receptor (TCR) domain fused to an effector activating domain. In the case of tebentafusp, this is an anti-CD3 single-chain variable fragment (scFv). The anti-CD3 scFv domain recruits and activates CD3+ T cells, while the TCR domain recognises peptides of interest presented by human leukocyte antigen (HLA) complexes. The two domains are linked by a disulphide bond (Figure 2(a)). As is the case with all T-cell therapeutic platforms, chosen epitopes should be expressed at high level by the tumour but minimally expressed elsewhere to minimise on-target, off-tumour toxicity. Furthermore, in contrast to antibody-based therapies, ImmTACs have the advantage of being able to target intracellular epitopes, where 90% of the neo-antigen pool are thought to be derived. While natural TCRs tend to possess low antigen affinity, ImmTAC TCRs are affinity enhanced to increase TCR binding to target. The T cells activated in the tumour microenvironment are not necessarily tumour specific but are directed to the tumour cell target by the ImmTAC. ImmTACs can therefore direct tumour-specific toxicity of non-tumour-specific T cells. T-cell activation induces the release of pro-inflammatory cytokines and cytolytic agents which ultimately leads to cell lysis. In turn, cell lysis may encourage ‘epitope spreading’ which can further stimulate T-cell activation, 34 as well as PD-L1 upregulation within the tumour microenvironment. 35

(a) Structure of ImmTAC – a soluble TCR domain and an anti-CD3 scFv domain joined by a disulphide linker. (b) Schematic demonstrating the mechanism of action of tebentafusp: The TCR domain binds HLA-A*02:01-positive melanoma cells presenting a melanoma-associated antigen gp100-derived peptide, and the anti-CD3 scFv recruits T cells.
The main limitation of ImmTACs is the HLA restricted antigen presentation of the TCR target peptide. Given that differing HLA haplotypes will bind and present different peptides to TCRs, HLA restricted ImmTAcs will only be effective in a population expressing the relevant HLA haplotype. However, it has been argued that this is not unlike the restricted use of drugs targeting specific genetic alterations. 36 In the case of tebentafusp, patients must have the HLA-A*02:01 allele. There is substantial heterogeneity in the representation of all HLA-A*02 subtypes worldwide, with prevalence higher in European and North American Caucasian populations (~30%), and lower in Black and South Asian populations (~20–25%). HLA-A*02:01 is the most common HLA-A*02 variant seen in Caucasian patients (the population most frequently affected by UM) but is significantly less prevalent in some ethnic groups including South Asians.37,38 Despite this, HLA restriction leaves a significant proportion of patients ineligible for treatment. A second possible drawback of these treatments is potential variability in target epitope expression. Identification of tumour-associated peptide antigen targets which are highly expressed on the tumour population is therefore desirable. The main advantages and limitations of ImmTACs are summarised in Table 2.
Summary of the advantages and disadvantages of ImmTACs over other immunotherapy treatment modalities.

HLA, human leukocyte antigen; ImmTAC, immune mobilising monoclonal T-cell receptors against cancer.
Tebentafusp
Tebentafusp redirects T cells towards HLA-A*02:01-positive UM cells presenting a melanoma-associated antigen glycoprotein 100 (gp100)-derived peptide (antigen gp100280–288) 39 (Figure 2(b)). Gp-100 is a melanocyte-specific molecule involved in the maturation of melanosomes; organelles responsible for transporting melanin. Gp100 has been investigated in early phase trials as a therapeutic target for the treatment of both cutaneous and UM using approaches including peptide vaccines and adoptive cell transfer. Although results from these trials are generally encouraging, tebentafusp is the first gp100-targeted treatment to gain regulatory approval, 40 following the publication of positive phase I–III trial data (Table 3).
Summary of trials utilising tebentafusp.

CI, confidence interval; CM, cutaneous melanoma; ImmTAC, immune mobilising monoclonal T-cell receptors against cancer; m, months; mOS, median overall survival; mPFS, median progression-free survival; NA, not available; SOC, standard of care; UM, uveal melanoma.
IMCgp-100-01 was the first-in-human, phase I trial of tebentafusp, involving 84 patients with CM and 18 UM patients. 41 Of these 18 patients, 3 patients (16.7%) had a partial response to treatment, with a further 8 patients (44.4%) demonstrating stable disease. These results demonstrated promising clinical activity of tebentafusp, and the study also established a recommended phase II dose. The toxicity profile of tebentafusp was in line with the postulated mechanism of action.
Following this, the IMCgp100-102 single-arm phase I–II trials recruited 127 patients who had previously undergone treatment with at least one prior line of therapy for metastatic UM.
43
For the whole cohort, while median progression-free survival (PFS) was just 2.8 months, median overall survival (OS) was 16.8 months. The overall response rate (ORR) by RECIST 1.1
44
was 5%, but 44% of those patients with measurable disease (
A larger phase III study, IMCgp100-202
33
was then conducted with a primary endpoint of OS rather than ORR. A further co-primary endpoint was overall survival in patients who developed a rash. In this randomised controlled trial, treatment-naïve UM patients were randomised 2:1 to receive tebentafusp as a first-line therapy (
Eligible patients required at least one measurable lesion and a performance status of 0 or 1. Treatment with tebentafusp could continue beyond initial radiological disease progression in the absence of clinical progression, clinical deterioration and dose-limiting toxicity. However, patients were not allowed to continue treatment beyond the second response scan if this also demonstrated continued progressive disease (PD). The rationale for this was based on established evidence that patients may derive benefit from immunotherapy agents even after initial radiological appearances of PD (pseudoprogression). Similarly, patients in the trial receiving pembrolizumab or ipilimumab as SOC were allowed to continue beyond initial progression if they met the aforementioned criteria.
At initial interim analysis after a median of 14.1 months of follow-up, a significant improvement in OS was observed for patients receiving tebentafusp; 21.7 months [confidence interval (CI): 18.6–28.6] compared to 16.0 months (CI: 9.7–18.4) for patients receiving SOC [hazard ratio (HR): 0.51,
Although radiological responses did not correlate well with survival benefit, emerging data on reduction in ctDNA in patients on the 102 expanded phase II study showed linear correlation between the degree of ctDNA reduction and improvement of overall survival. 43 More than 90% of patients being treated in the second-line and third-line settings had detectable ctDNA which correlated with tumour burden. The extent of reduction in ctDNA levels in patients treated with tebentafusp was closely correlated with OS benefit. This observation has led to the suggestion that serial measurement of ctDNA may represent a more clinically accurate marker of response than scan assessment in patients receiving ImmTACs, 47 especially in the first few months of treatment.
One criticism of the IMCgp100-202 study design has been that patients in the SOC arm were not permitted treatment with doublet immunotherapy (ipilimumab/nivolumab). Two single-arm phase II studies have reported outcomes in UM patients treated with ipilimumab/nivolumab.28,48 One-year landmark OS in these studies was not however superior to that seen in the SOC arm of IMCgp100-202 (52% and 56%
Pharmacokinetics and pharmacodynamics
Pharmacodynamic studies of tebentafusp have demonstrated increased levels of pro-inflammatory cytokines including CXCL10, CXCL11, interleukin (IL)-6, interferon gamma (IFN-γ), tumour necrosis factor alpha (TNFα) after treatment. Serum levels of IFN-γ, TNFα, IL-2 and Il-10 are seen to increase to up to 10-fold that of pre-treatment levels in the 8–24 h after drug administration. 35 Levels then fall to almost pre-treatment levels prior to the next dose administration. The amplitude of serum cytokine elevation is also observed to be smaller after three doses have been administered. 35
Tumour tissue studies show increases in CD3+, CD4+ and CD8+ lymphocyte infiltration and increased expression of the cell death marker cleaved caspase 3, 35 suggesting clear evidence of an increase in tumor-infiltrating lymphocytes (TILs) following treatment, with associated tumour cell death. In a sub-study within the phase I clinical trial, pre- and post-treatment tumour biopsies from 11 patients (8 CM, 1 UM, 1 acral melanoma and 1 lentiginous melanoma) were compared. An at least twofold increase in the number of intratumoral T cells following treatment was observed in 8 out of the 11 patients (73%). In the single UM patient included in the sub-study, a low level of intratumoural T cells was observed pre-treatment, which increased noticeably following treatment with tebentafusp. 35
Pharmacokinetic studies demonstrate that tebentafusp has a half-life of around 7.5 h (FDA, 2022). Studies suggest that body weight, age, gender, renal or liver dysfunction do not significantly impact this. To date however, patients with severe renal function (creatinine clearance < 30 ml/min) or moderate to severe liver dysfunction (total bilirubin > 1.5 × ULN) have been excluded from studies. Roughly, one-third of patients in the reported clinical trials are noted to develop anti-tebentafusp antibodies. 33 To date, there is no evidence that outcomes are worse for patients who developed antibodies. The use of tebentafusp has not been found to cause reduced tumoral gp100 expression. 35
Dosing considerations
In the phase I trial, there were two treatment arms. In arm 1, patients received weekly treatment with a dose escalation from 20 µg (cycle 1 day 1) to 30 µg (cycle 1 day 8) to 68 µg (cycle 1 day 15). This approach was adopted to reduce the frequency of higher-grade cytokine release syndrome (CRS) during therapy induction. In arm 2, patients received daily treatment for 4 days every 3 weeks with doses ranging from 10 to 50 µg between participants. The recommended phase II dose was 68 µg.
Acute toxicity tends to peak around 4–6 h after drug administration and for this reason, treatment delivery should ideally be early in the day, in a clinician-supported treatment delivery facility, with an extended monitoring period available for patients receiving the first few doses of treatment.
Adverse events and toxicity management
Adverse events (AEs) can be grouped into two main categories (Table 4): cytokine-mediated toxicity and skin toxicity. Cytokine-release-mediated AEs include pyrexia, chills and hypotension, usually occurring some hours after drug administration and are generally short-lived. In contrast to this, skin toxicities relate to the on-target off-tumour effect of gp100 expression in normal melanocytes and take somewhat longer to resolve – usually days. Most toxicities occur during the first cycle of treatment. In the phase III trial, Grade 3 and 4 AEs were seen in 44% of patients in the tebentafusp arm
AEs observed following tebentafusp administration. 33

AEs, adverse events; AST, aspartate aminotransferase; CRS, cytokine release syndrome.
Cytokine-mediated AEs
CRS is defined by the presence of hypotension, hypoxia and pyrexia. Almost 90% of patients treated with tebentafusp experience CRS of any grade, usually within a few hours of drug administration. 99% of CRS observed was Grade 1 or 2 (G1-2).
Patients with G1-2 CRS may develop fever, hypotension, tachycardia and rigours. These are seen mostly in the first 2–12 h following administration, peaking 4–6 h post-dose. Patients on antihypertensive agents should omit these for 48 h prior to, and 24 h after, their first few of tebentafusp, with the timing of restarting antihypertensives dependent on their clinical condition. Early detection of hypotension is vital; a baseline blood pressure (BP) measurement should be taken prior to commencement of therapy based on at least three separate measurements taken over a period of at least several minutes. BP should then be checked at least every 2 h after treatment. Patients may be managed with delivery of antipyretic agents and intravenous (IV) fluids. In cases of hypotension refractory to IV fluid administration, IV glucocorticoid administration may be required, under the guidance of a senior clinician. A suggested management algorithm for cytokine-mediated hypotension is outlined in Figure 3(a).

(a) Management algorithm for cytokine-mediated hypotension following tebentafusp administration. Patients on antihypertensives should omit these for 48 h before, and 24 h after treatment with tebentafusp for the first few doses. (b) Stepwise management of pyrexia.
Due to the potential for life-threatening CRS to occur, it is important that centres ensure adequate support and training for the clinical team. Establishing a clear protocol of management for the described immune toxicity is vital. In centres with critical care links, it is important that these teams are educated on CRS management. Careful patient selection is an important consideration, with consideration paid to the renal and cardiac function of patients eligible for tebentafusp who may require aggressive fluid resuscitation. Given the role for corticosteroids in the management of fluid refractory CRS, patients with inadequate adrenal function, or on long-term corticosteroids should have their baseline steroid dose doubled prior to and immediately following drug administration. A proposed management algorithm for severe or fluid refractory CRS is outlined in Figure 3(b).
Skin toxicity
The most common skin toxicities include rash, pruritus and erythema, and most often occur within the first few days of drug administration. 35 The majority are low grade (65%) but higher grade or refractory cases have been seen (18%). In general, skin toxicity is easily managed with supportive measures. Non-pharmacological interventions to manage mild (G1) skin toxicity include fans and cold showers, while for persistent or higher-grade (>G2) toxicity, pharmacological interventions including antihistamines, emollients and topical steroids may be of use. In management of pruritus, calamine containing topical therapies, emollients and antihistamines may have utility (Figure 4).
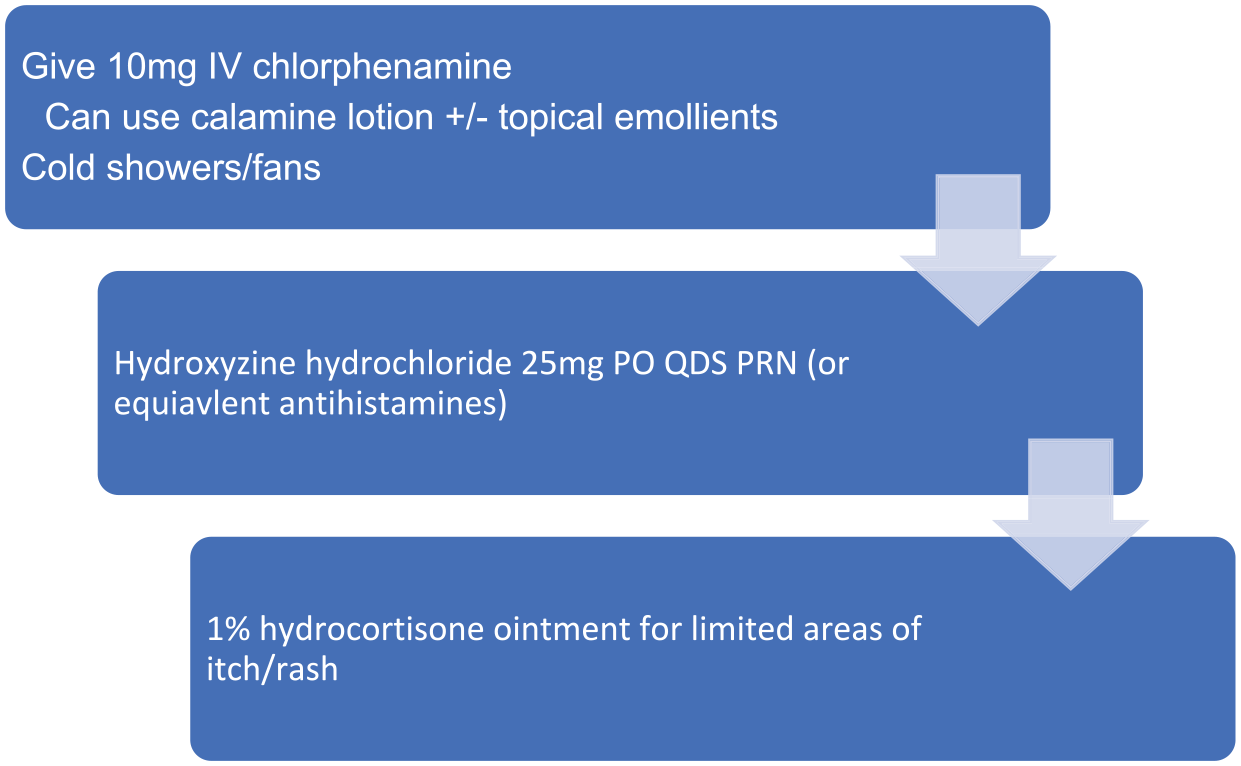
Management of skin toxicity (pruritus). IV = intravenous, PO = oral, QDS = four times daily, PRN = as needed.
Other toxicities
Fluctuations in liver function tests may be noted either due to inflammation related to drug action or clinical disease progression. In cases of Grade 3 liver dysfunction, tebentafusp should be held until results return to Grade 1 or normal ranges. Management of liver dysfunction should be in line with local management guidelines for drug-induced liver injury.
Conclusions and future directions
There is scope to build upon the initial success of tebentafusp with combination studies underway, investigating tebentafusp together with ICPIs. Current studies include tebentafusp alone
Future trials could also evaluate the role of tebentafusp in the adjuvant treatment of patients diagnosed with high-risk UM. 50 These studies, if successful, could dramatically change the treatment landscape for patients with UM. Despite this, just as is the case with adjuvant ICPI treatment, attention must be paid to the risks and benefits of such treatments given the potential quality of life impact on patients; not only because of toxicity risk, but also because of the relatively intense drug delivery schedule.
Liquid biopsies, including ctDNA, are finding an ever-expanding role in management of malignant disease.
51
The tebentafusp clinical trials published to date suggest that monitoring of ctDNA levels may yet become a more accurate method of response assessment that conventional imaging, especially in the months immediately following treatment initiation.
47
In a wider clinical context, ctDNA monitoring has the potential to become a commonly used tool to guide clinical decision-making in all melanoma patients, and future trials should include translational sub-studies to investigate the utility of this.
52
TebeMRD (NCT05315258) aims to establish whether treating uveal and CM patients who develop molecular relapsed disease after definitive treatment (assessed
A limitation to the use of tebentafusp is the requirement for high-intensity monitoring, This, coupled with the weekly dosing schedule may be difficult to replicate in healthcare settings where oncology services may be far from the population they serve or in poor-income countries. An area of further research/consideration is to assess whether CRS effects are predictable or preventable without loss of efficacy. This may reduce the need for initial admissions.
As well as a potentially expanded role for tebentafusp, other bispecific immunotherapy agents are now being developed for use in other solid tumours. An immTAC targeting a MAGE-4 peptide (IMC-C103C) is being investigated for use in malignancies such as upper gastrointestinal, and head and neck cancers, 53 and early phase studies are underway investigating IMC-F106C, an immTAC targeting PRAME, a tumour antigen overexpressed in tumours including squamous cell carcinoma, small-cell lung cancer, endometrial carcinoma as well as melanoma. 54 IMC-F106C is being studied both as a single agent and in combination with both ICPIs and tebentafusp (NCT04262466). Limitations of both these agents, like tebentafusp, is their restriction to HLA*0201-positive individuals. Strategies to overcome this limitation and account for population HLA diversity, such as employing non-polymorphic HLA molecules, may circumvent this problem in the future. 39 Outside of use in malignancies, other bispecifics are in development which may be utilised in the management of HIV or Hepatitis B.55,56 Finally, in addition to targeting gp100 in UM, ongoing research should look to identify other druggable targets in this rare disease. Emerging evidence suggests an evolving role for human endogenous retroviruses (HERV) in the molecular pathogenesis of UM. Specific HERV loci are identified in primary UM and may represent potential neoantigenic targets for future immunotherapeutics. 57
The recent approval of tebentafusp is an exciting step forward not just in the field of cancer immunotherapy but also in the treatment of UM; a rare, aggressive and notoriously treatment-resistant malignancy. The clinical applications for tebentafusp are likely to expand as combination and adjuvant studies are conducted and reported on. In the future, wider clinical applications for ImmTAC agents and other bispecific antibody platforms may further change the already varied treatment landscape for a wide range of solid tumour malignancies.
