Abstract
Uveal melanoma (UM) is the most common primary intraocular malignancy in adults. Although UM and cutaneous melanoma are derived from melanocytes, UM differs clinically and biologically from its more common skin counterparts. More than half of primary UMs metastasize. However, there is currently no effective treatment for metastatic UM. Therefore, studying mutations related to the metastasis, growth, proliferation, and survival of UM can help researchers understand its pathogenesis and metastatic mechanism, thereby leading to a more effective treatment. In addition, we provide an overview of the recent basic and clinical studies to provide a strong foundation for developing novel anti-carcinogenesis targets for future interventions.
Introduction
Uveal melanoma (UM) is a common primary malignant eye tumor. If diagnosed early and treated locally before tumor metastasis, the survival rate can reach up to 90%. 1 Unfortunately, approximately 50% of patients develop systemic metastases at the time of diagnosis.2–4 The distant metastasis organs are most often the liver, followed by the lungs and bones. The average survival time of these patients is reported to be approximately 2–15 months.5,6 Traditional treatments include eyeball removal, local tumor resection, plaque brachytherapy, and laser photocoagulation.7,8 Eyeball removal causes visual deprivation and affects appearance. Metastasis rates are relatively high in tumor resection. Laser photocoagulation is not effective and is clinically less used. However, plaque brachytherapy is an effective treatment for UMs, including large melanomas or those near the optic nerve head. Plaque brachytherapy has been shown to provide good clinical outcomes with respect to long-term local control and prevention of distant metastasis. Recent improvements in plaque design not only allow higher effective radiation delivery rate, but also reduce radiation-induced damage to normal tissues. Therefore, lower radiation doses—less than the traditional radiation dose of 85 Gy—may be effective. Further, endoresection of melanomas combined with plaque brachytherapy can also be employed safely for primary local tumor control. 9 At present, new therapeutic strategies, such as targeted therapy, immunotherapy, and radiation therapy, face challenges in improving the clinical survival rate with good functional outcomes.
Clinical feature of uveal melanoma
Ocular melanoma can originate from the uvea (83%), conjunctiva (5%), or other sites (10%) (Figure 1). UM may involve the iris, ciliary body, or choroid, which are collectively referred to as the uvea. The most common site of UM is the choroid, accounting for about 90%. 10 Choroid melanoma is the most frequently found UM, and presents as dome- or mushroom-shape due to a rupture of the Bruch’s membrane. The mean size of the basal diameter is 11.3 mm, with a thickness of 5.5 mm. 10 The lesions present as pigmented, non-pigmented, or mixed. Pigmented color melanoma should be differentiated from benign nevi. In iris melanoma, a majority of the tumor was round, and less was diffused.11,12 Changes in iris color (heterochromia), distortion of pupil margin, corectopia, and hyphema were found upon clinical examinations. 13 In addition, secondary glaucoma was noted, especially due to tumor compression into the angle. 14 Ciliary body melanoma is usually diagnosed late, after the lesion is too large to be hidden by the iris. Most patients express metamorphopsia or painless vision loss. Other symptoms include floater, photopsia, visual field loss, or pain. 15 In general, clinical features predicting growth include increased tumor thickness, retinal detachment, or the presence of subretinal fluid. Features indicating non-neoplastic tumors include the presence of drusen over the tumor surface, retinal pigment epithelial atrophy, and intraretinal migration of the pigment epithelium.16,17
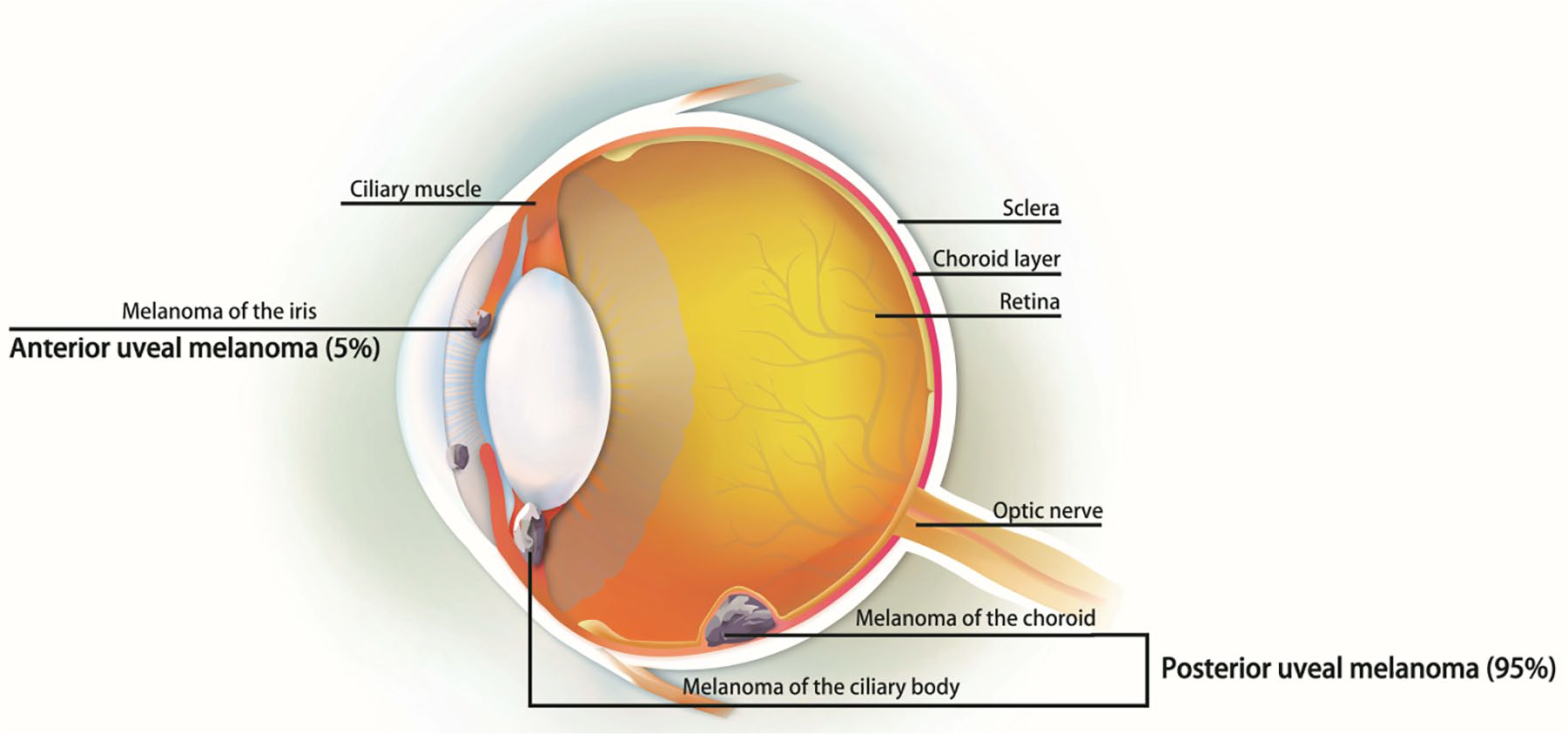
The ocular melanoma originated from the iris, choroid, and ciliary body, which divided into anterior UM (5%) and posterior UM (95%).
Gene mutation in uveal melanoma
GNAQ and GNA11
Mutations of oncogenes
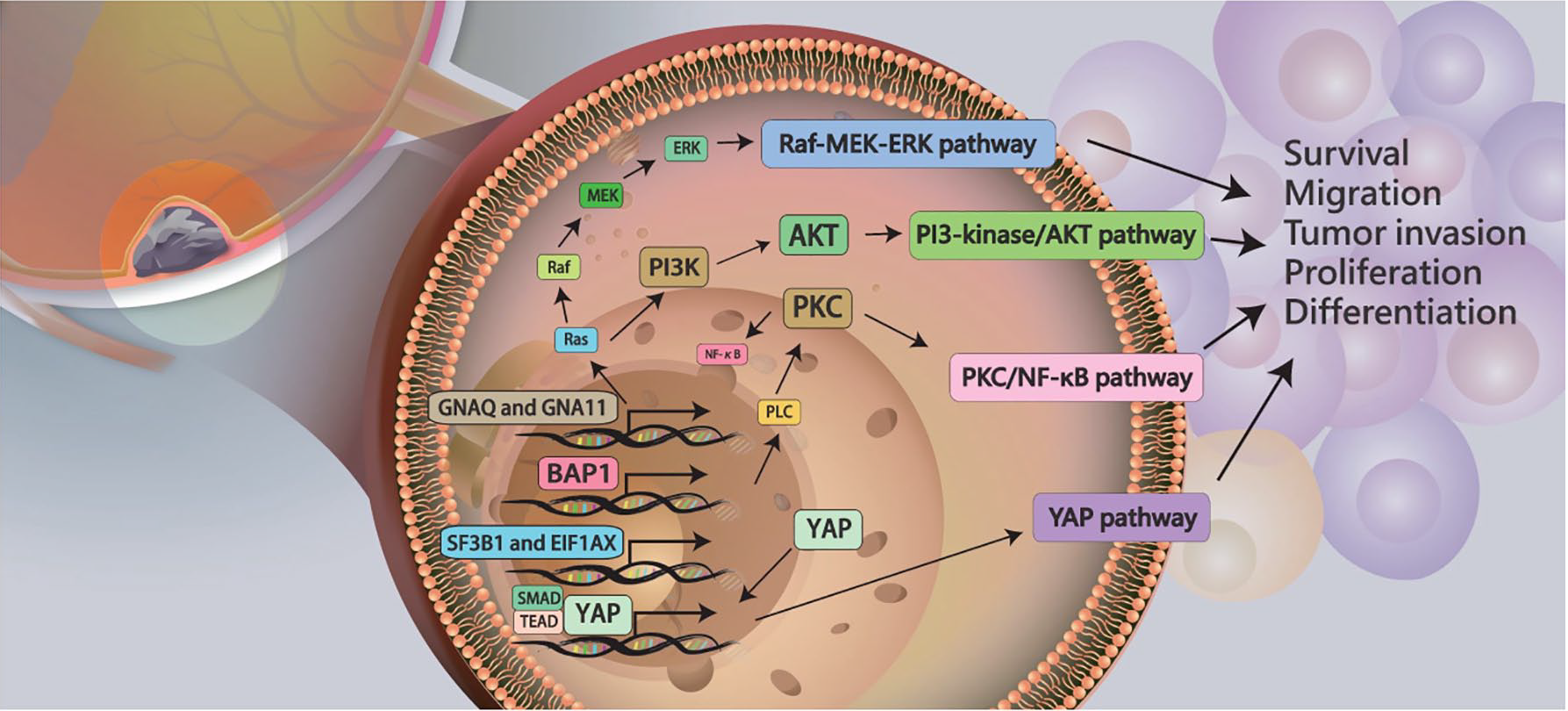
Several mutations of oncogenes, including GNAQ, GNA11, BAP1, SF3B1, and EIF1AX, may induced cell survival, migration, invasion, proliferation, and differentiation in UM
BAP1
Comparative analysis of genes on chromosome 3 in class 1 and class 2 tumors revealed that 85% of the class 2 tumors had mutations in BAP1 [breast cancer susceptibility gene 1 (BRCA1)-associated protein 1], while no mutations were detected in class 1 tumors.
24
BAP1 is located at 3p21.1, and class 2 tumor cells have only one copy of the BAP1 gene on chromosome 3. BAP1 plays a role as a tumor suppressor gene in UM, and its loss makes tumor cells more prone to metastasis. The BAP1 molecule is a deubiquitinating enzyme that regulates the function of target proteins through the removal of ubiquitin molecules. For example, BAP1 can remove ubiquitin molecules on histone H2A, thereby altering the expression of downstream genes that are regulated by histone H2A. BAP1-regulated genes play an important role in melanocyte differentiation. Further, BAP1 deletion de-differentiates UM cells, exhibiting stem cell-like morphology and promoting tumor metastasis.
25
In a retrospective cohort study by Gupta
SF3B1 and EIFlAX
SF3B1 (the splicing factor 3b1) is involved in pre-mRNA splicing. A mutation in
Pathogenesis of uveal melanoma
Multiple downstream signaling pathways, such as MEK, PI3K/AKT, and protein kinase, have been investigated in UM. MEK/MAPK and P13K/AKT signaling pathways are activated in UM.30,31 High activation of the P13K/AKT signaling pathway is attributed to
The downstream signaling of mutant Gαq and Gα11 was investigated, especially in RAF-MEK1/2-ERK1/2 signaling. MEK1/2 small molecule inhibitors with trametinib or selumetinib inhibit the growth of a variety of UM cells. In metastatic UM patients, the resistance to MEK inhibitors was commonly reported and several studies found that hepatocyte growth factor (HGF) signaling may elicit resistance to MEK inhibitors.38,39 HGF expressed by the liver at the secondary site plays an important role for UM to preferentially metastasize to the liver. After binding HGF, activation of c-MET triggered several cellular signaling pathways involved in proliferation, migration, and invasion. 40 In UM population, the expression of c-Met is significantly associated with mortality. 41 As such, hepatic metastases are more likely to develop when the primary UM expresses high levels of c-Met. 42 Some researchers have compared c-Met expression in UM primary tumors and metastases, and detected c-Met expression in both primary tumors and metastases; however, c-Met expression was higher in metastatic tumors than in primary tumors. 43 Another growth factor secreted by the liver is insulin-like growth factor 1 (IGF-1). IGF-1 regulates cell proliferation, survival, and migration by binding to its receptor, IGF-1R, which is expressed in primary UM tissues. 41 Multivariate analysis of immunohistochemistry showed that IGF-1R expression was associated with UM lethality. Immunohistochemical analysis of tissue samples from 24 patients with UM liver metastases revealed exogenous and endogenous IGF-1 and their interaction with IGF-1R in hepatic metastasis of UM. 44 In addition, previous studies have demonstrated that hepatic stellate cells provide innate resistance to MEK inhibitors in metastatic UM through HGF-cMET signaling. After inhibition of HGF signaling, the cMET-targeting agent in UM cells overcomes the resistance to MEK inhibitors mediated by stellate cells or exogenous hepatocyte growth factors. The mechanism affecting MEK, in turn, includes growth factors NRG1 and HGF-induced metastatic UM cells.38,45 However, the molecular mechanisms by which HGF/c-Met and IGF-1/IGF-1R promote UM liver metastasis have not been elucidated.
Targeted therapy in uveal melanoma
Targeted therapy refers to the use of standardized biomarkers to identify the presence of a disease-specific gene or gene profile that controls tumor growth and to determine a treatment for a specific target. The main target sites discovered in UM include:
BAP1 gene targeted therapy
BAP1 is a tumor suppressor gene that encodes a histone H2A ubiquitin hydrolase that regulates cell differentiation, cell cycle arrest, and DNA repair.48–50 Inactivation or gene mutations in
GNAQ gene targeted therapy
c-Kit gene targeted therapy
The c-Kit gene is a class III receptor tyrosine kinase transmembrane receptor family member and plays a role in differentiation, proliferation, and programmed cell death. Further, it is involved in intracellular signaling and plays a key role in hematopoiesis. It also shows overexpression in UM and is a potential carcinogenic driver in this tumor type.59,60 c-Kit protein is overexpressed in non-metastatic tumors, yet its expression is significantly reduced after metastasis
61
; however, the specific reasons are still unknown. Imatinib is a specific tyrosine kinase inhibitor targeting the
Role of IGF-1
Liver metastasis often occurs in UM, and the cause of its occurrence is still unknown. In recent years, many studies have demonstrated that growth hormone secretion by the liver facilitates tumor metastasis to the liver.5,41,69,70 One of the growth factors is IGF-1, which promotes tumor cell proliferation and tumorigenesis in UM and many other malignant tumors.71,72 A combination of IGF-1 and epidermal growth factor (EGF) signaling induces UM cell migration and invasion, thereby increasing the risk of metastasis.69,73 In addition, differences in expression levels of the IGF-1 receptor, IGF-1R, have been found in many UM tumors. It has become an important clinical parameter for UM assessment of prognosis.41,69 Studies on human UM stem cells have found that the IGF-1R inhibitor, cyclo-lignan picropodophyllin (PPP), can inhibit cell survival, growth, invasion, and migration.70,74 In an UM mouse model study, the use of PPP in mice subjected to xenograft liver transplantation effectively induced tumor regression and reduced liver metastasis.
74
In addition, inhibition using a monoclonal antibody, IMC-A12 (cixutumumab), reduced IGF-1 activity and migration of UM cells. However, the result of the clinical study using IMC-A12 for metastatic UM was negative. A phase II trial by Patel
VEGF treatment
Although current systemic anti-angiogenic therapy does not produce significant clinical response in UM patients,75,76 VEGF blockers have produced partial positive results in animal models. Anti-VEGF monoclonal antibody, ranibizumab, has been investigated as a single drug for phase II clinical trials of UM patients.
77
In the Neoadjuvant IntraviTreal Ranibizumab treatment in high risk Ocular melanoma patients trial (NITRO Trial, EudraCT Number: 2011-000961-10), a two-stage, single-center, Phase II single-arm study, progressive disease was found in all treated patients (
Immunotherapy
Immunotherapy is the application of immunological principles and methods to enhance the patient’s active or passive immunity through exogenous injection of tumor vaccines, monoclonal antibodies, adoptive immune cells, and effector molecules, thereby stimulating the body’s anti-tumor response. 81 The immune response ultimately kills tumor cells and inhibits tumor growth. 82 Immunotherapy with checkpoint inhibition, including anti-CTLA-4 and PD- 1/PD-L1 blockade, in patients with advanced UM has garnered great interest. Other agents may include IMCgp100, glembatumumab, and MEK inhibitors. Because of better responses and fewer treatment-related deaths, checkpoint inhibitors serve as second-line therapy for locally advanced or metastatic UM. UM is generally insensitive to traditional chemotherapeutic drugs; therefore, researchers have adopted an emerging treatment, immunotherapy, for UM clinical trials.
Interferon in UM
interferon (IFN) is a cytokine with a wide range of biological activities. It can exert anti-tumor effects through various mechanisms, including inhibition of tumor cell proliferation and tumor angiogenesis, and apoptotic induction, to enhance the body’s immune function to kill tumor cells and inhibit the expression of multiple oncogenes.
83
At present, IFN is used widely as a clinical treatment for various tumors, especially for tumors caused by some infectious agents. In UM patients, IFN served as an adjuvant treatment for primary UM, but not for treatment of metastatic UM. However, IFN has shown a poorer therapeutic effect in UM than in other tumors. Treatment with IFN-α-2b does not improve patient survival, with a 5-year mortality rate of 15–17%.
84
In a study in which 39 UM patients were injected subcutaneously with the same dose of IFN-α-2b, 46% of the patients had to reduce the therapeutic dose due to severe side effects, including decreased white blood cell counts, thrombocytopenia, abnormal heart and liver function, etc. Further, IFN did not extend survival for UM patients, even though IFN showed a positive effect in the treatment of cutaneous melanoma. A phase II trial by Nathan
TGF-β in UM
The eyes of UM patients are in an unusual microenvironment for immune remission. UM escapes immune system surveillance, which promotes tumor growth, increasing cell survival and metastasis. The mechanism whereby UM avoids the immune response is still unclear; however, TGF-β has shown immunosuppressive effects in UM. In clinical studies, TGF-β1 and TGF-β2 immunohistochemical examinations were performed on 13 surgically resected UM specimens. All tissue specimens showed positive results for TGF-β, and confirmed the immunosuppressive properties of TGF-β. It is also believed that TGF-β causes immunosuppression in the eye of UM patients, thereby inhibiting the host’s immune response against the tumor. An increase in the level of TGF-β may suggest an enhanced ability of the tumor to evade host immune surveillance. 90 Other studies have confirmed that TGF-β is negatively correlated with major histocompatibility complex I (MHC-I) in UM. Specifically, when cultured UM cells were treated with TGF-β, MHC-I expression decreased by about 30%, thereby increasing the sensitivity of UM cells to natural killer cell-mediated cell autolysis. 91
Immune checkpoint blockades
In recent years, immunological checkpoint inhibitors have achieved favorable results in skin management due to targeting of cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) ipilimumab and programmed cell death-1 (PD-1) pembrolizumab in the management of cutaneous melanoma. 92 Immune checkpoint inhibitors are the new revolutionary first-line treatment for the management of advanced or metastatic UM. 93 Immune checkpoint inhibitors target checkpoints to promote T cell activation, leading to tumor lysis and degradation. 93
Based on the mechanism of action of monoclonal antibodies on tumor cells, currently used antibodies can be roughly classified into the following categories: direct anti-tumor monoclonal antibody and specific monoclonal antibody that can be directed to the surface of a tumor cell. Antigen binding, which blocks the transduction of certain signaling pathways in tumor cells, can inhibit tumor cell proliferation or induce apoptosis. Anti-tumor monoclonal antibody conjugates, including chemical compounds, radionuclides, and toxins, can be transported to tumor cells in a targeted manner by the specificity of monoclonal antibodies without killing normal cells, thereby improving efficacy and reducing side effects. There is also an immune-mediated tumor cell. Killing substances, such as monoclonal antibodies, injected into the human body can induce complement activation, activate antibody-dependent cytotoxicity, and participate in the regulation of T lymphocyte function, which enhances the autoimmune response to kill tumor cells. 94
Anti-CTLA-4 therapy
Ipilimumab is a specific CTLA-4 (cytotoxic T lymphocyte antigen 4) inhibitor that was approved by the Food and Drug Administration (FDA) in 2011 for the treatment of advanced malignant cutaneous melanoma. Its main mechanism of action is through enhanced T lymphocyte cell-mediated immune responses that exert an anti-tumor effect. CTLA-4 is a transmembrane receptor on the surface of T lymphocytes, which can compete with cluster of differentiation 28 (CD28) for binding to ligand B7 and negatively regulate the activity of T lymphocytes. Studies have found that CTLA-4 binds to B7 and can limit the signal transduction involved in CD28, resulting in the inability of T lymphocytes to respond to immune responses. T lymphocytes can also be produced by inhibiting the production of IL-2 (interleukin 2) and its receptors. Remaining in the G1 phase, inhibition of proliferation can occur and induce apoptosis of T lymphocytes. Injectable ipilimumab can specifically bind to CTLA-4, block CTLA-4 binding to B7, restore T lymphocyte activity, and enhance the body’s own immune response to kill cancer cells.
95
A study by Heppt
Anti-PD-1 therapy
Anti-PD-1 antibodies, pembrolizumab or nivolumab, have been shown to target PD-1 in tumor cells. Pembrolizumab (2 mg/kg), working as an anti-PD-1 immunotherapy, demonstrated ORRs of 30%. Nivolumab (3 mg/kg), a monoclonal antibody against PD-1, showed 6% ORRs. However, there are few studies investigating immune checkpoint inhibitors, especially in combination treatment. Therefore, additional randomized clinical trials are necessary to understand the efficacy of immune checkpoint inhibitors and provide strong evidence in UM.81,97 Regarding the RR of ipilimumab in metastatic UM, the immune-related best ORR was 5% (4 patients), and 29% (24 patients) had stable disease for more than 3 months with 34% disease control rate. The median OS and PFS were 6.0 months (1-year OS rate: 31%) and 3.6 months (1-year PFS rate: 11%).
98
Anti-PD-1 antibodies boost the host immune system as anti-tumorigenesis treatment. Cancer cells have been shown to over-express PD-L1 with immunosuppressive function to inhibit the interaction between PD-1 and PD-L1, leading to decreased T cell activity and antitumor function. In a study by Bender
Combination therapy with anti-PD-1 and anti-CTLA4 therapeutics has emerged as a novel anti-tumor treatment and has been reported to be more effective than single antibody treatment. Anti-CTLA4 antibodies may promote activity of the T cell immune response. Further, anti-PD-1 can induce reactivation of CD8+ T cells to lyse cancer cells. In a study by Algazi
Conclusion
This review presents treatment trends for UM, including genetic mutations, molecular prognosis, new advances in targeted therapies, and adjuvant immunotherapy. For patients, due to the high mortality rates and high frequency of metastasis associated with UM, there is an immediate requirement to explore new treatments that inhibit melanoma growth before metastasis to distant tissues. Established or new treatments including targeted therapy or immunotherapies provide better treatment choices and benefits at an early time-point in tumor development.
Footnotes
Acknowledgements
Giou-Teng Yiang, Po-Chen Lin, Chien-Sheng Chen and Andy Po-Yi Tsai participated in discussions, provided critical intellectual input.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The present study was funded by grants SRD-108014 and SRD-108015 from Show Chwan Memorial Hospital, Taiwan.
