Abstract
Background:
Gastroenteropancreatic (GEP) neuroendocrine neoplasms with Ki-67 > 20% were subdivided in the most recent 2019 World Health Organization histopathological classification into grade 3 (G3) neuroendocrine tumors (NETs), described as well-differentiated tumors, and neuroendocrine carcinomas, which are described as poorly differentiated tumors. This classification met the demand noted for different prognoses between these subgroups, prompting the need for treatment recommendations for well-differentiated G3 tumors.
Methods:
We systematically searched medical literature databases and oncology conferences for studies on G3 GEP NET to describe epidemiology, diagnosis, molecular features, and treatments used. We excluded studies that did not discriminate G3 NET data. Data were tabulated and described, and a quality analysis of the reports was performed.
Results:
We found 23 published studies and six abstracts; 89.7% of studies were retrospective, six were composed exclusively of G3 NETs. Among 761 patients, the median number of patients per study was 15, most were male and older than 60 years, and functional imaging tests were positive in more than 80% of cases. Overall, the scientific evidence supporting the treatment of G3 GEP NETs is limited. For localized disease, resection remains the standard treatment but there is no evidence to support neoadjuvant or adjuvant therapy. For advanced disease, capecitabine and temozolomide seems to be the most effective option, with a response rate, median progression-free survival, and median overall survival up to 37.9%, 20.6 months, and 41.2 months, respectively.
Conclusion:
The latest available data on the epidemiology, diagnosis, molecular changes, and treatment of G3 GEP NET are described. Yet, the level of evidence for treatment recommendations is low, as most studies are retrospective. A treatment algorithm for G3 GEP NET is proposed.
Introduction
Neuroendocrine neoplasms (NENs) are heterogeneous neoplasms, and those of gastroenteropancreatic (GEP) origin can be subdivided into neuroendocrine tumors (NETs) and neuroendocrine carcinomas (NECs). NETs have a well-differentiated morphology and are sub-classified as grade 1 (Ki-67 < 3%), grade 2 (Ki-67: 3–20%), or grade 3 (G3; Ki-67 > 20%). NECs have a poorly differentiated morphology and are composed of small or large cells.1,2 While the median Ki-67 index value for G3 NET is approximately 35%, it is generally 70% or higher for NEC. 3 NET and NEC occurring in the same organ are molecularly different, a fact that reinforces this sub-classification. 4 G3 pancreatic NETs are molecularly similar to G2 NETs, with 44% and 25–36% harboring pathogenic somatic mutations in MEN1 and DAXX/ATRX, respectively. 5 In contrast, pancreatic NECs often present mutations in TP53, RB1, and SMAD4, resembling their adenocarcinoma counterpart.6,7
Patients with metastatic G3 NET have longer overall survival (OS) than those with advanced NEC (32–40 versus 11–12 months), with a prognosis resembling that of G2 NET.5,8 Therefore, the treatment administered to patients with G3 NET is usually extrapolated from treatments used for G2 NET.
Because the G3 NET classification was only published in 2017 and 2019,2,9 this subgroup has not been examined in many epidemiological studies or clinical trials. Therefore, the incidence of G3 NET is probably underestimated, although retrospective studies have reported frequencies between 9% and 16%, with primary tumors localized mainly in the pancreas, stomach, or colon. 3 In a study conducted by our group, 23% of cases diagnosed initially as NEC were reclassified as G3 NET after pathology review based on the 2017 World Health Organization (WHO) classification. 10 Consequently, treatments for patients with G3 NET have not yet been established, and this population has been excluded from most clinical studies.
To inform the current knowledge about G3 NET, we performed a systematic review of the epidemiology, diagnosis, molecular aspects, and treatments for patients with G3 GEP NET.
Methods
Search and selection criteria
We performed a systematic review of studies published in scientific journals or presented in the form of abstracts that reported on patients diagnosed with G3 NET from GEP origins in 2017, when the WHO pathology classification of the pancreatic subtype was published, 4 until the end of September 2022.
The search for full publications was conducted using the Cochrane Central Register of Controlled Trials, PubMed, and EMBASE databases. No language restrictions were imposed. We complemented the search with abstracts and virtual meeting presentations from the American Society of Clinical Oncology conferences (ASCO), the European Society of Medical Oncology (ESMO), the North American Neuroendocrine Tumor Society (NANETS), and the European Society of Neuroendocrine Tumors (ENETS).
First, we searched for systematic reviews in the Cochrane Central Register of Controlled Trials database. For PubMed, the search was conducted using the following MeSH descriptors: ‘neuroendocrine tumors’ OR ‘neuroendocrine neoplasms’ AND ‘grade 3’ OR ‘high grade’, limiting the search from July 2017 to September 2022. At EMBASE, we used the terms ‘gastroenteropancreatic neuroendocrine tumor’ OR ‘gastroenteropancreatic neuroendocrine neoplasms’ AND ‘grade 3’ OR ‘high grade’, during the same period mentioned above. For ASCO, ESMO, ENETS, and NANETS abstracts, we used the words ‘neuroendocrine tumors’ OR ‘neuroendocrine neoplasms’ AND ‘grade 3’ OR ‘high grade’ located in the abstract body and/or title of studies.
Studies were eligible if they met the following inclusion criteria: retrospective or prospective studies, or case series (containing at least five cases); human studies; clinical studies that described clinical, epidemiological, and molecular characteristics or treatments administered; and their respective outcomes in patients with G3 NET of GEP origin (including NEN of unknown origin predominantly with abdominal metastases). Studies on NEN from the breast, lung, prostate, thyroid, kidney, head and neck, urinary tract, uterus, mediastinum, orbit or cervical site, and Merkel were excluded. Studies that did not discriminate G3 NET data or non-original studies, such as reviews, editorials, preclinical or pediatric studies, and trials in progress, were also excluded. For case reports, we excluded those with fewer than five patients, except if they were scientifically relevant for treatment or diagnostic purposes.
Duplicate publications were sorted out; relevant publications listed in article references and not retrieved by the primary search were included. If an abstract resulted in a full publication, the latter was selected.
For future perspectives on treatments, we searched ClinicalTrials.gov for studies currently recruiting or about to start recruitment of patients with GEP NET and NEN, which described G3 or high grade as an inclusion criterion.
Data extraction
In each eligible study, the information on the following variables was collected by two independent investigators: study name; name of the journal/conference and year of publication; type of publication (abstract or article); study design; study objective according to abstract or title description of each study (epidemiologic, when only clinical features were described; diagnostic, when the study evaluated pathological and/or molecular features only; prognostic, when there were associations between tumor and/or clinical characteristics and survival endpoints; or treatment, when outcomes from therapies were described); total sample size; sample composition of G3 NET; gender; age; known genetic syndrome; Eastern Cooperative Oncology Group (ECOG) status; functional tests [gallium-68 positron emission tomography (PET Ga-68) or Octreoscan and 18F-fluorine-deoxy-2-glucose (PET-FDG)]; primary location; WHO classification used (2017 or 2019); pathology review; Ki-67 index; immunohistochemical or molecular evaluation of markers, including p53/Rb, DAXX/ATRX and MEN1; stage; treatments (surgery, curative or palliative systemic treatments); and follow-up time. The variables of interest for studies reporting treatment were disease-free survival (DFS), response rate (RR), disease control rate (DCR), progression-free survival (PFS), time to treatment failure, and OS (median and annual rates). The data collected were related to patients with G3 NET only; otherwise, they were considered not to be reported. For some studies that provided survival curve figures, the digitizing software DigitizeIt 2.5.9 was used to extract unreported values.
Quality evaluation
The quality evaluation of the studies included was assessed by applying the Strengthening of the Report of Observational Studies in Epidemiology (STROBE) form. 11 The STROBE form is a checklist of 22 items considered essential for good study reporting. The items evaluate the title and abstract of the article (Item 1), Introduction (Items 2 and 3), Methods (Items 4–12), Results (Items 13–17), Discussion (Items 18–21), and other information (Item 22, on financing). For each selected study, one point was assigned for each item, and a final score was produced by summing all points. Studies were classified from 0 to 22 points, with higher scores reflecting higher methodological quality. We are aware that the STROBE evaluation applies to full publications but not abstracts. Therefore, we applied this quality assessment exclusively for fully published studies.
Two investigators (MDSD and ABCB) independently searched for studies, collected the data, and applied the STROBE form. A third investigator (RPR) was consulted when divergence occurred; and a consensus was achieved for all data.
Analysis and synthesis
The collected information was entered into a structured collection form. Finally, for quantitative synthesis, the data were analyzed using SPSS software version 24.0.
Results
Results of the search strategy
The search yielded 311 entries, of which 26 duplicate records were removed. Of the remaining 285 studies, 256 were excluded, as shown in Figure 1. In all, 29 studies met the predefined inclusion criteria, totaling 2955 NET patients, of which 25.8% (761 patients; range: 5–74) had G3 GEP or unknown primary G3 NET origins.4,12–39

Flow chart for search and eligible studies.
Characteristics of eligible studies
The characteristics of the 29 eligible studies are summarized in Table 1. The median number of patients per study was 15 (range: 5–74). Median follow-up periods ranged from 16 to 66.7 months. In total, 23 studies were published articles, and six (20.7%) were abstracts. In all, 26 (89.7%) were retrospective studies, and 13 were unicentric. Twelve studies evaluated treatments. Only six studies (20.7%) were composed exclusively of G3 NET patients.
Characteristics of the 29 eligible studies.
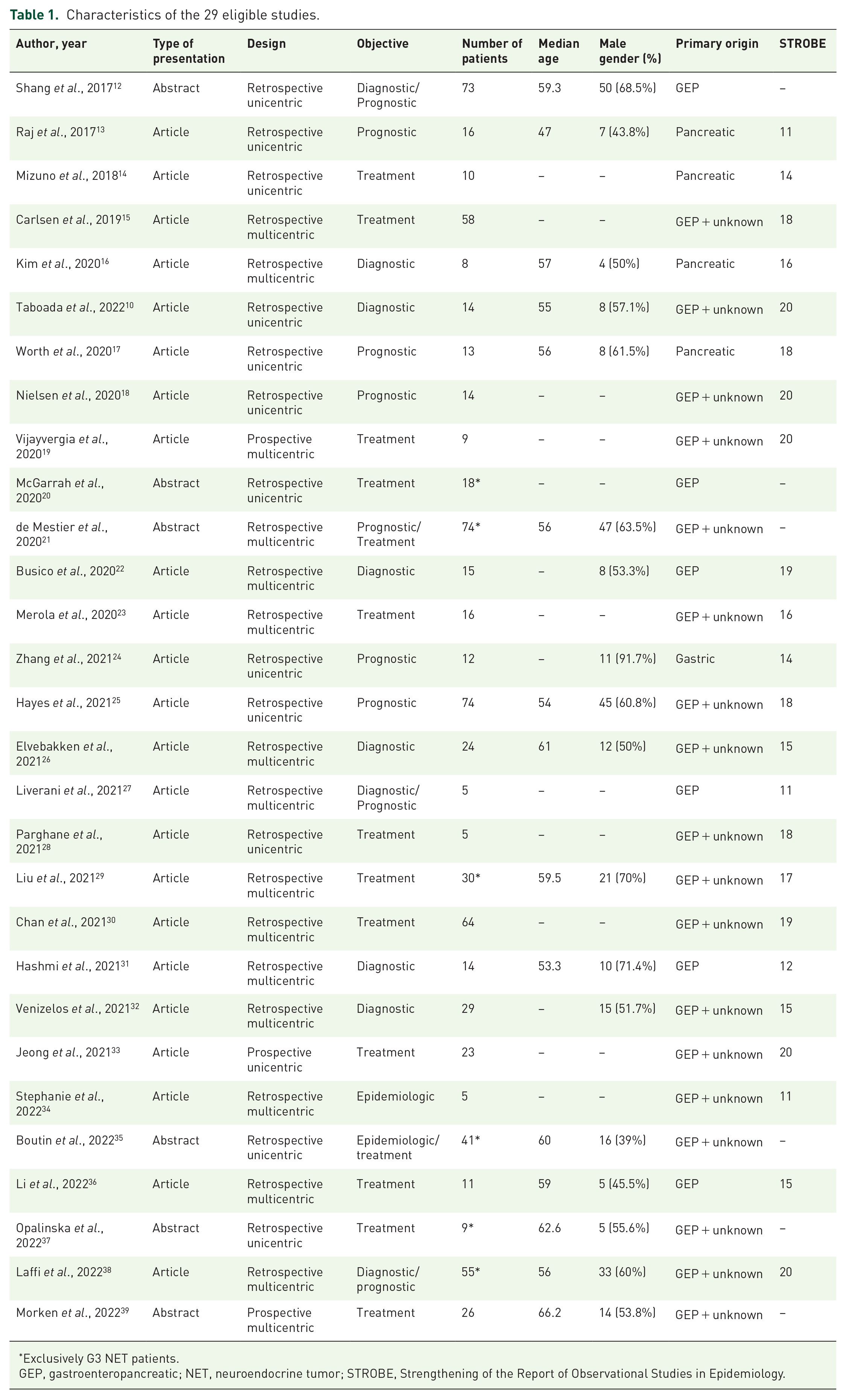
Exclusively G3 NET patients.
GEP, gastroenteropancreatic; NET, neuroendocrine tumor; STROBE, Strengthening of the Report of Observational Studies in Epidemiology.
Quality assessment using STROBE was performed for the 23 published studies, with a median score of 17 (range: 11–20). Table 1 lists the scores obtained from each study.
Clinical and pathological characteristics of patients
Sex was described in 18 studies, totaling 538 patients, with the male sex corresponding to 59.9% of cases (n = 322). The median age of patients ranged from 47 to 66.2 years. ECOG status was described in six studies, with 95.6% reporting an ECOG score of 0 or 1. In all, 13 studies reported the use of PET-Ga68 or Octreoscan/scintigraphy, totaling 476 patients, of which 402 patients (84.5%, range: 44.4–100%) had tumor positive uptake. In terms of PET-FDG, 137 out of 171 patients (80.1%, range: 59–100%) had positive NET uptake, as shown in Table 2.
Functional test results (PET-Ga68 or Octreoscan/scintigraphy and PET-FDG).

Octreoscan/scintigraphy.
Positive uptake on functional test was an inclusion criterion.
PET-FDG, 18F-fluorine-deoxy-2-glucose positron emission tomography; PET-Ga68, gallium-68 positron emission tomography.
Pathology review was performed in 23 studies (79.3%).10,12–14,16,18,20–27,29,31–36,38,39 As shown in Table 3, the Ki-67 index was described in 15 studies, and the median values ranged from 23% to 61%; p53 immunohistochemistry (IHC) expression and/or TP53 mutation status was evaluated in six studies, with mutation or hyperexpression present in 0% to 21%; DAXX/ATRX and MEN1 mutation status were described in three studies, and were found in 0–25% and 0–21% of the patients, respectively, mostly in pancreatic NET.
Immunohistochemical and molecular features.
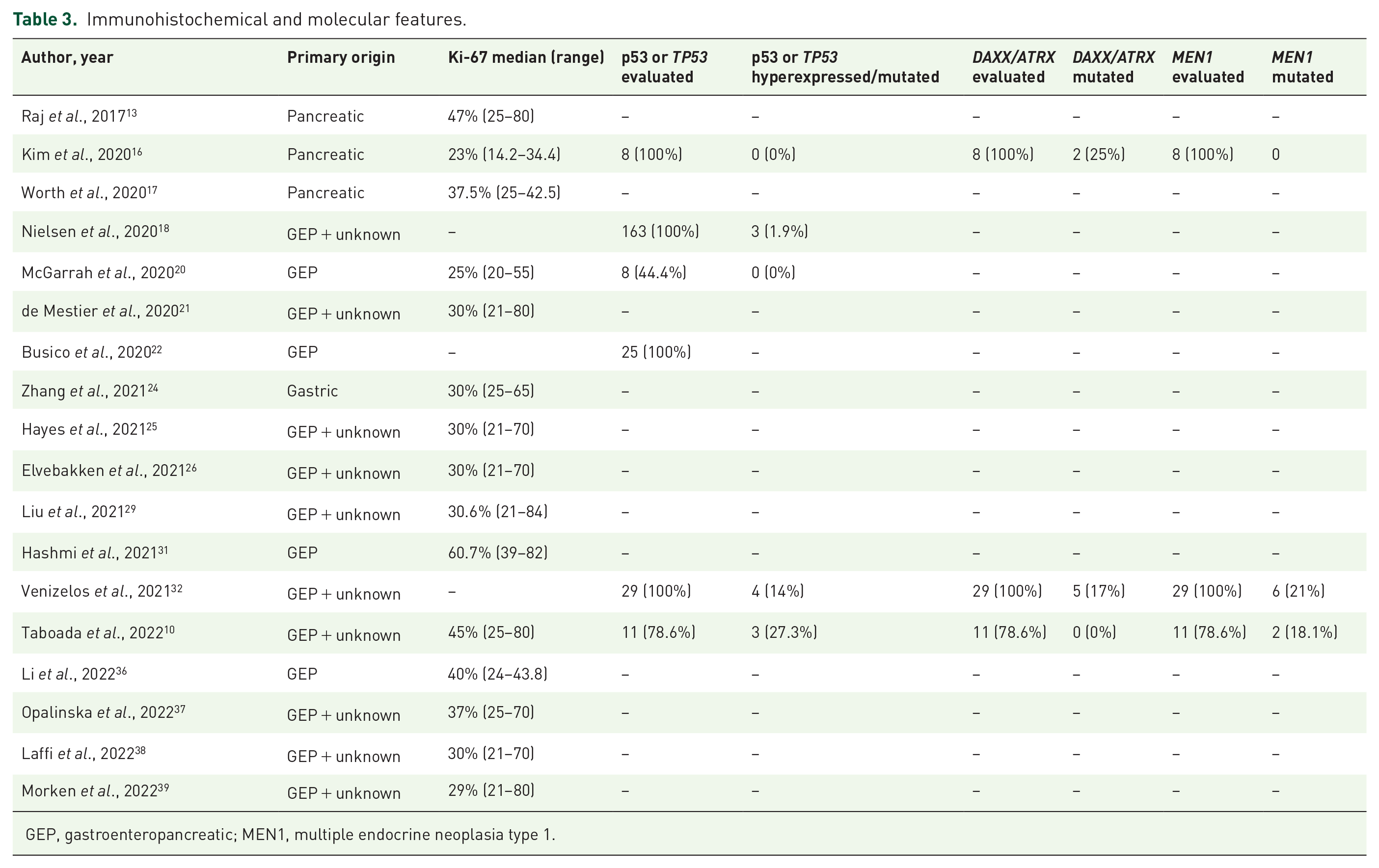
GEP, gastroenteropancreatic; MEN1, multiple endocrine neoplasia type 1.
Outcomes definitions and assessment
We evaluated outcome definitions for each time-to-event variable of interest in each study. OS and PFS were defined as death from any cause and death or disease progression, respectively, from surgery, diagnosis, presence of metastasis, and initiation of palliative therapy in four,16,17,23,24 six18,22,25,32,36,38 one, 26 and nine studies,10,13–15,21,28–30,33 respectively. Nine studies did not describe the definitions used.12,19,20,27,31,34,36,37,39 The RECIST 1.1 assessment was reported in 13 out of 15 studies with descriptions of assessment methods (86.7%).13–15,19,21,22,25,26,28,29,32,33,38 In all, 14 studies did not describe disease response assessment methodology.12,16–18,23,24,27,31–37,39
Treatments and outcomes
In all, 14 studies (48.3%) included only patients with metastatic disease,10,15,19–21,25–30,33,35,39 six studies reported only patients with localized disease (20.7%),16,17,23,24,31,34 and nine contained both (31%).12–14,18,22,32,36–38
Localized disease
In total, 15 studies described performing surgery of the primary tumor,13,16,17,21–24,26,29,31,32,35,36,38,39 and neoadjuvant or adjuvant treatment were cited by six studies but they lacked information on the types of chemotherapy regimens used.16–18,22,23,36 The median DFS times ranged from 9 to 33.9 months (Table 4).
Resection results for localized disease.
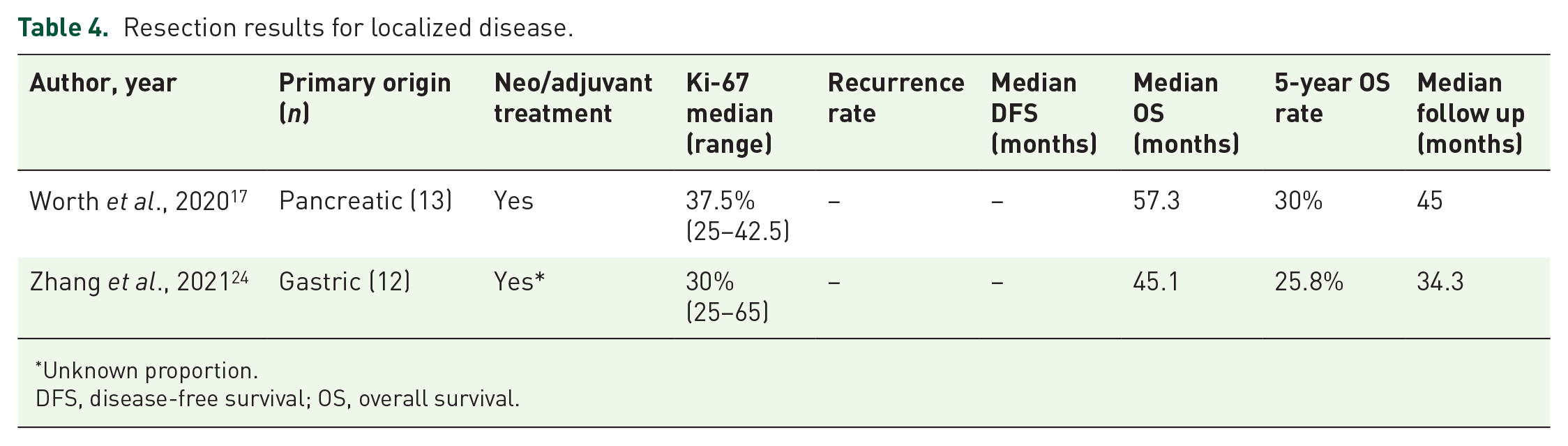
Unknown proportion.
DFS, disease-free survival; OS, overall survival.
Only two studies described baseline populations undergoing curative surgical treatment for localized disease. One study evaluated 13 patients with pancreatic tumors and mentioned that 3 and 9 patients received neoadjuvant or adjuvant therapy, respectively, but did not detail the chemotherapy regimens used. 17 The surgical techniques varied from open surgery to minimally invasive surgical enucleation. 17 At a median follow-up to 45 months, median OS was 57.3 months and 5-year OS rate was 30%. There was no significant difference in OS between 13 patients with G3 NETs and 15 patients with NECs. 17 The other study was a retrospective series of Chinese patients with gastric high-grade NEN (N = 12 G3 NETs) who underwent surgical resection of the primary tumor. The OS rates at 5 years were 81.6% for stage I, 57.5% for stage II, 45% for stage III, and 9.1% for those with metastatic disease. 24 The authors report that nearly half (80 out of 151) of patients received adjuvant chemotherapy (37 received platinum-based regimens, including etoposide and cisplatin and oxaliplatin combined with a fluoropyrimidine) but neither the type of regimen nor the proportion of patients with G3 gastric NETs who received adjuvant treatment were described. 24 The median OS of patients with G3 tumors was 45.1 months and their 5-year OS rate was 25.8%; there were no statistically significant differences in OS or DFS between G3 NETs or NECs in multivariable analyses. In the overall population, adjuvant chemotherapy did not impact on OS or DFS. 24
Advanced/metastatic disease
In all, 17 studies described palliative treatments used in the first, second, or later lines.12–15,19–21,25,26,28–30,33,35,37–39 The regimens used were platin doublets or triplets, capecitabine and temozolomide (CAPTEM), folinic acid + fluorouracil + oxaliplatin (FOLFOX), and somatostatin analog (SSA), among others. Table 5 summarizes the outcomes of systemic chemotherapies, respective Ki-67 indices, and lines of treatment. Platinum-based regimens and CAPTEM were the most commonly used regimens. After a median follow-up that ranged from 16 to 66.7 months, the RR, DCR, median PFS, median OS, and 5-year OS rates for platinum-based regimens were up to 42%, 82.4%, 7.2 months, 39.6 months, and 36.1%, respectively. For CAPTEM, after a median follow-up ranging from 16.8 to 66.7 months, the results of RR, DCR, median PFS, median OS, and 5-year OS rate were up to, respectively, 37.9%, 87%, 20.6 months, 41.2 months (not reached with a follow-up time of 19.2 months in one study), and 68%.
Platinum-based and CAPTEM regimens results for advanced/metastatic disease.

Not reported RECIST 1.1 assessment.
Cisplatin or carboplatin + etoposide/fluorouracil + carboplatin + streptozocin/fluorouracil + cisplatin + streptozocin.
CAPTEM, capecitabine + temozolomide; DCR, disease control rate; GEP, gastroenteropancreatic; OS, overall survival; PFS, progression-free survival; RR, response rate; VP, etoposide.
The results of FOLFOX, temozolomide alone or in combination with everolimus, and pembrolizumab were described in only one study each. Outcomes from SSA, peptide receptor radionuclide therapy (PRRT), and sunitinib were described in four, three, and two studies, respectively, and are summarized in Table 6.
Results of other therapies for advanced/metastatic disease.

Not reported RECIST 1.1 assessment.
CAPOX, capecitabine + oxaliplatin; DCR, disease control rate; FOLFOX, folinic acid + fluorouracil + oxaliplatin; GEP, gastroenteropancreatic; OS, overall survival; PFS, progression-free survival; PRRT, peptide receptor radionuclide therapy; RR, response rate; SSA, somatostatin analog.
Discussion
In this systematic review, we describe the available data on the clinical and molecular features and treatment outcomes of 761 patients with GEP G3 NET. There was a predominance of male patients (nearly 60%) in their sixth decade of life, with tumors having a Ki-67 index of approximately 30% and PET-Ga68 or Octreoscan/scintigraphy and PET-FDG positive uptake in more than 80% of cases. Treatments varied; however, most patients were treated with cytotoxic chemotherapy with platinum-based regimens or CAPTEM. The outcomes of these therapies also varied, with RRs ranging from 10% to 25% with platin combinations, and 30% to 35% with CAPTEM, but with longer median PFS times being achieved with CAPTEM. Very few studies reported the results of targeted therapy. Of the studies that reported treatment of localized disease with surgery, with or without neoadjuvant and/or adjuvant therapy, median DFS times ranged from 9 to 33.9 months.
Combined somatostatin receptor imaging and PET-FDG have been shown to be complementary for lesion detection in patients with G3 GEP NET, and findings of FDG-positive NET on PET-CT indicate a worse prognosis.40,41 Patients with a homogeneous PET-Ga68 are reportedly associated with longer median PFS and OS than those with inhomogeneous/negative tests, and negative/inhomogeneous PET-FDG uptake is associated with longer OS than those with widely positive PET-FDG uptake. 38 Here, we observed that more than 80% of patients had both PET-FDG and PET-Ga68 positive uptake, suggesting that G3 NET patients should undergo both functional tests performed at baseline for proper staging, prognostic information, and to aid in treatment decisions.
A complete pathology report was not performed in most studies. Data on the Ki-67 index were not available for half of the patients. The median Ki-67 value ranged between 23% and 60.7%, with a mean of 35%, in accordance with historical data. The frequency of findings of p53 or TP53, DAXX/ATRX (described in pancreatic NET only), and MEN1 were in the range of 20%.3,7,42 We observed a low frequency of TP53 mutations when compared to NEC. Only three studies performed a molecular assessment of patients, and MEN1 mutations were highly present.
Outcomes data on localized disease were restricted to gastric and pancreatic primary sites. Five-year OS rates were 25%–30% and not significantly distinct between NEC and G3 NET. Yet, the number of patients in both studies was quite small to draw firm conclusions. In addition, both studies did not provide details about adjuvant or neoadjuvant therapies and their respective effects on survival outcomes. Despite the lack of evidence, the use of perioperative therapies in localized G3 GEP NET is common in clinical practice. We think that for highly selected fit patients with aggressive tumors and ki67 index higher than 50%, especially those with negative tumor uptake on PETGa68, or with resected oligometastases, neoadjuvant or adjuvant chemotherapy could be exceptionally recommended after deliberation in a multidisciplinary team board. Regardless of perioperative therapies, with reported median OS outcomes between 45.1 and 57.3 months, we consider that R0 surgical resection of G3 GEP NET should be performed.
For advanced disease schemes using alkylating agents (CAPTEM) showed more consistent results than platinum-based schemes in terms of RR, DCR, PFS, and OS values, especially for those with Ki-67 up to 30%. A retrospective study evaluating patients with advanced G3 NET found that alkylating-based chemotherapy achieved a higher RR (37.9%) compared to other treatments [multivariable odds ratio: 4.22, 95% confidence interval (CI): 1.5–12.2; p = 0.008], while the shortest PFS was observed with SSAs and etoposide–platinum (EP) chemotherapy. 43 Another retrospective analysis using German databases, which included predominantly G3 GEP NET treated with palliative first-line therapy, found RR of 35.1% for EP, 56.4% for FOLFOX, 27.3% for CAPTEM, and 45.0% for streptozotocin/5-fluorouracil. The median PFS, respectively, were 6.9, 6.9, 12, and 4.8 months. All non-EP patients combined showed a significantly prolonged median PFS compared to EP in univariate analysis (9.0 months; p = 0.049). 44 Although previous data showed that O-6-methylguanine-DNA methyltransferase (MGMT) low IHC expression predicted temozolomide efficacy, 45 a Korean study assessed 25 patients with methylation-specific polymerase chain reaction and found 96% of methylated patients, but neither MGMT protein expression nor MGMT promoter methylation status was found to be significantly associated with tumor response or survival outcomes in G3 GEP NET. 33
G3 NET have a better prognosis than NEC despite therapy. Considering pancreatic NEN, an unicentric retrospective study found a median OS of 52.2 months (95% CI: 19.3–86.9 months) in patients with G3 NET compared to a median OS in patients with NEC of 10.1 months (95% CI: 6.9 to 12.4 months; p = 0.0009). OS was still inferior for the NEC group (p = 0.02, HR 3, 95% CI: 1.2–7.5 months) even after adjusting for potential confounders such as the therapies received and ECOG performance status. 13 This difference was also observed in a recently presented phase II study of the combination of everolimus and temozolomide in first line which showed that, for NET G3 and NEC, respectively, RR was 27% versus 33.3%, median PFS was 12.6 versus 3.4 months (p = 0.133) and median OS 31.4 versus 7.8 months (p = 0.003). 39
Sunitinib was evaluated in a predominantly subsequent-line setting, in a series of 10 patients who achieved a RR of 60% (RECIST based) and disease control at 1 year of 40%. 14 These data align with those of Panzuto et al., who suggested that everolimus, another targeted therapy, provides a persistent rate of disease control, 40% for 1 year or more, in G3 pancreatic NEN with well-moderately differentiated morphology and Ki-67 < 55%. 46 Given the small number of patients and the outlier results in terms of RR, we think more data are needed to better evaluate the role of sunitinib and everolimus in G3 GEP NET.
Immunotherapy offers discouraging results with 3.4% RR, 2.0 months PFS, and 4.7 months OS. Pembrolizumab was examined in a small sample of patients with NEN G3, both G3 NET and NEC, previously treated with platinum-containing chemotherapy; however, the RR was disappointingly low at 5%. 47 Most recently, the open-label phase II NICE-NEC study enrolled patients with advanced G3 NEN of GEP or unknown origins treated with six cycles of nivolumab, carboplatin, and etoposide combination followed by nivolumab maintenance for up to 24 months. Most patients had poorly differentiated tumors with Ki-67 > 55%. The RR was 54.1%, DCR was 83.8% and median PFS was 5.7 months (95% CI: 5.1–9). The OS rate at 12 months was 58.3% versus 54.7% for well and poorly differentiated, respectively. 48 However, data were not presented separately for G3 NEN. Immunotherapy is being evaluated in combination with other treatments, such as PRRT. 49
PRRT has demonstrated promising activity in G3 NET, with RR, DCR, median PFS, and median OS up to 42%, 93%, 19 months, and 44 months, respectively. In one cohort study at 12 centers encompassing 149 G3 GEP NEN patients (60 well differentiated and 62 poorly differentiated), PRRT provided 41% partial response, 1% complete response, and 38% stable disease. The outcomes were better among those with well-differentiated G3 NEN than for those with NEC (median PFS 19 versus 8 months and median OS 44 versus 19 months; p < 0.001). 15 These data justify the current ongoing trials with PRRT for G3 GEP NET, alone or in combination, as shown in Table 7.
Recruiting prospective phase II and III clinical trials in advanced G3 GEP NET.
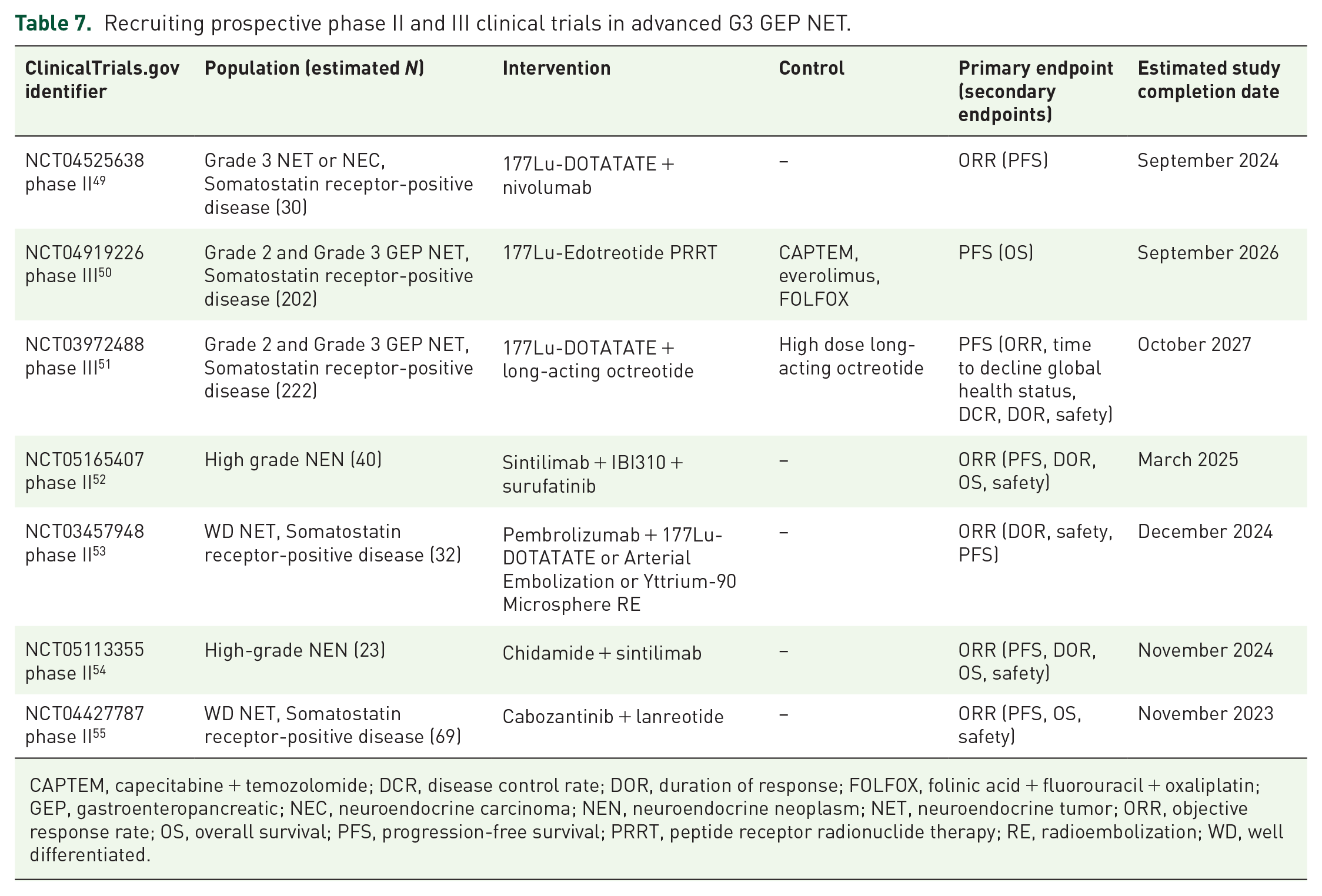
CAPTEM, capecitabine + temozolomide; DCR, disease control rate; DOR, duration of response; FOLFOX, folinic acid + fluorouracil + oxaliplatin; GEP, gastroenteropancreatic; NEC, neuroendocrine carcinoma; NEN, neuroendocrine neoplasm; NET, neuroendocrine tumor; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; PRRT, peptide receptor radionuclide therapy; RE, radioembolization; WD, well differentiated.
Finally, our quality analysis showed that no study covered all recommendations contained in the STROBE form. The median score was 17 (77.3% punctuation), which, together with the other factors already mentioned in this discussion, shows that the scientific evidence on G3 GEP NET lacks significant quality.
Our study has limitations. We lacked some clinical, pathological, treatment, and outcome data and most studies were not controlled. Several studies reported outcomes of different tumors jointly. There is a lack of standardized definitions for outcomes and assessment methods across studies, and we used published results instead of individual patient data. The use of the STROBE form for quality analysis should also be considered a limitation, as it was originally designed for observational studies. However, despite all limitations, we think our systematic review offers the best available evidence to help clinicians in the management of G3 GEP NET patients.
In conclusion, we describe the latest available data on the epidemiology, diagnosis, molecular changes, and treatments for patients with G3 GEP NET. Pathological review is of fundamental importance to better diagnose G3 GEP NET and both PET-Ga68 and PET-FDG should be routinely performed for staging, prognosis, and treatment planning. Although studies currently available are generally of limited quality, for localized G3 GEP NET, R0 resection remains standard but there is no evidence to support the indication of neoadjuvant or adjuvant therapy. For metastatic disease, chemotherapy with alkylating agents seems to be the best option. PRRT might improve outcomes for patients with lower Ki-67 and PET-Ga68 avid NET. We highly encourage clinicians to refer patients with G3 GEP NET for clinical trials. Here, we propose an algorithm for diagnosis, staging, and treatment based on the Ki-67 score and functional tests that may aid in the management of patients with G3 GEP NET (Figure 2).

Algorithm for diagnosis, staging, and treatment of G3 GEP NET.
Footnotes
Acknowledgements
None.
Authors’ note
All authors have made significant contributions to this manuscript, have seen and approved the final manuscript, and agree with its submission to the journal.
Warnings: The opinions expressed in the report are those of the authors and do not necessarily represent the official position of the institution to which they belong.
