Abstract
Background:
Nearly 30% of neuroendocrine tumors (NETs) have evidence of immunohistochemical (IHC) expression of estrogen (ER) and/or progesterone (PR) receptors. Therefore, targeting ER/PR may offer an effective NET-directed treatment to select patients.
Methods:
We conducted a multicenter Simon two-stage single-arm phase II trial of tamoxifen in patients with metastatic, progressive NETs. Eligible patients had positive IHC expression of ER and/or PR ⩾ 1%. Prior therapy with somatostatin analogs was required for progressing/functioning cases. Main exclusion criterion was aggressive disease requiring cytotoxic therapy. The primary end point was disease control rate (DCR) at week 24 by Response Evaluation Criteria in Solid Tumors version 1.1. We planned to enroll 23 patients in the first stage, to reach a DCR at week 24 of 70% (versus 50%); if ⩾12 patients reached the primary end point, a total of 37 would be included.
Results:
From February 2019 to February 2022, 23 out of 59 patients were eligible and enrolled: 15 (65%) were females; the most common sites were pancreas (11; 48%) and small bowel (6; 26%). In all, 13 patients (56.5%) had G2 NETs. At a median follow-up of 27 months, 13 patients (56.5%) had stable disease at week 24 and median progression-free survival (PFS) was 7.9 months [interquartile range (IQR): 3.7–12.1]. The best response was stable disease in 13 patients, with most patients experiencing minor tumor growth. Median PFS times were not significantly different according to ER/PR < or ⩾30% (p = 0.49) or ER versus PR expression (p = 0.19). One patient experienced grade 2 constipation.
Conclusion:
Tamoxifen for ER-/PR-positive NETs patients is safe but offers modest antitumor effects.
Trial registry name:
Study of Tamoxifen in Well Differentiated Neuroendocrine Tumors and Hormone Receptor Positive Expression (HORMONET).
URL:
https://clinicaltrials.gov/ct2/show/NCT03870399?term=03870399&draw=2&rank=1
Registration number:
NCT03870399
Introduction
Targeted therapies have significantly improved the outcomes of patients with metastatic well-differentiated neuroendocrine tumors (NETs). Somatostatin analogs (SSA) are typically prescribed as the first-line systemic therapy for patients with advanced gastroenteropancreatic (GEP) NETs, both for inhibition of tumor growth and control of hormonal syndromes. For patients with progressive disease, peptide receptor radionuclide therapy with 177Lutetium-Dotatate has demonstrated significant improvement in progression-free survival (PFS) in patients with advanced small bowel NETs when compared to high-dose octreotide. 1 Placebo-controlled randomized clinical trials of everolimus and sunitinib in GEP-NETs demonstrate median PFS durations of nearly 1 year.2–4 Despite these advances, new treatments are needed that offer more favorable benefit compared to toxicity.
Retrospective studies have identified that a significant proportion of NETs express estrogen (ER; alpha or beta) and/or progesterone receptors (PR) by immunohistochemistry (IHC). A retrospective study of 96 pancreatic NET samples found PR nuclear expression in 58% of cases, ranging from 5% to 100% of neoplastic cells, but no ER-alpha expression. 5 In the same study, investigators did not observe PR or ER-alpha expression in any gastrointestinal or lung NETs. In a series of 163 primary GEP NETs and 115 metastatic lesions, PR staining was detected in 32% of primary tumors and 18% of metastases. PR expression was most commonly observed in pancreatic NETs (77% of primaries and 46% of metastases). ER expression was seen in 27% of primary tumors and 18% of metastases, primarily in nonpancreatic NETs. 6 In a retrospective series of 96 NET samples conducted by our group, we observed ER positivity in 21% and PR positivity in 18% of patients. ER positivity was significantly associated with carcinoid syndrome. PR positivity was primarily observed in pancreatic NETs. Neither ER or PR IHC expression was associated with tumor grade. 7 The overall conclusion from these studies is that PR expression is primarily observed in pancreatic NETs and that ER expression is more commonly observed in nonpancreatic NETs.8–11
Positive IHC expression of ER and/or PR has also been associated with better outcomes in patients with NETs. In a retrospective cohort of 39 pancreatic NETs, high positive ER-beta gene expression was associated with small primary tumor size (p = 0.02), G1 tumors (p = 0.02), and earlier stage (p = 0.006). 12 Likewise, loss of PR IHC expression has been associated with shorter disease-free survival in patients with resected stage I and II pancreatic NETs, even after adjusting for tumor size, grade, and stage. 12 In another retrospective series of 277 pancreatic NET samples, loss of PR expression was associated with larger tumors, higher grade, perineural invasion, and lymph node metastases. 13
Based on these studies, we hypothesized that ER/PR expression influenced tumor behavior and could, therefore, constitute a therapeutic target for NETs. Notably, case reports have documented marked improvement in carcinoid syndrome and reduction in urine 5-hydroxyindoleacetic acid (5HIAA) levels, and regression of retroperitoneal fibrosis with tamoxifen.14,15 Yet, the only clinical trial that evaluated the efficacy of tamoxifen in 16 patients with advanced NETs, published in 1984, reported disappointing results. 16 However, tumor expression of ER and/or PR was not an inclusion criterion nor was it evaluated in post hoc analyses.
Our objective was to investigate the efficacy of tamoxifen as a targeted therapy in patients with metastatic NETs and positive tumor expression of ER and/or PR.
Methods
Study design and end points
HORMONET was an open-label, single-arm, Simon two-stage phase II multicentre clinical trial aimed to evaluate the efficacy of tamoxifen for patients with NETs with positive expression of hormone receptors. Recruitment took place at A.C.Camargo Cancer Center (São Paulo, Brazil) and the Moffitt Cancer Center (FL, USA). The study was conducted in accordance with Good Clinical Practice and ethical principles of the Declaration of Helsinki. The protocol was approved by the Ethics Committee or the Institutional Review Board at each participating center. All patients provided written informed consent. The study was registered in the clinical.trials.gov under the registration number NCT03870399.
The primary end point was disease control rate (DCR) at 24 weeks after initiation of tamoxifen, defined by the absence of radiological progression in conventional imaging tests by the Response Evaluation Criteria in Solid Tumors (RECIST v1.1). Secondary end points were PFS, objective radiological response rate, and adverse events. A waterfall plot was constructed to report the best percentage change from baseline in the size of target lesions for each patient. We planned an exploratory evaluation of DCR at 24 weeks according to percentages of ER-/PR-positive neoplastic cells by NETs, by type of hormone receptor expressed and to describe the characteristics of patients who achieved DCR beyond 12 months.
Patients
Eligible patients were 18 years of age or older, had an Eastern Cooperative Oncology Group (ECOG) performance status of 0–2, and histologically confirmed locally advanced or metastatic unresectable well-differentiated NET of GEP or lung origins of any grade, according to the World Health Organization (WHO) classification, 17 IHC expression of 1% or greater of ER-alpha and/or PR (see description of methodology below), measurable disease by RECIST v1.1 18 with radiological progression of at least 10% of tumor volume within 12 months before study entry, adequate organ function (serum aspartate aminotransferase, serum alanine aminotransferase ⩽ 2.5 times the upper limit of local laboratory normality [ULN-LL]), total serum bilirubin ⩽2.0 × ULN-LL, absolute neutrophil count ⩾ 1500/mm³, platelet count ⩾ 80,000/mm³, hemoglobin ⩾ 9.0 g/dL, estimated creatinine clearance as per the MDRD (formula ⩾ 30 mL/min), albumin ⩾ 3.5 g/dL, and INR ⩽ 1.5. Tamoxifen could be utilized in any line of therapy after SSA; however, patients with nonfunctioning NETs and low metastatic burden who were unwilling to receive SSA injections could be enrolled into this trial. At Moffitt Cancer Center, prior treatment requirements included SSA plus at least one more systemic therapy if somatostatin receptor positive, and at least one prior systemic therapy if somatostatin receptor negative. Patients with functioning NETs were maintained on SSA. Exclusion criteria were prior exposure to tamoxifen, aggressive disease requiring cytotoxic therapy or locoregional therapies, known synchronous neoplasm that demanded systemic treatment, post-menopausal patients with vaginal bleeding, pregnant or breastfeeding patients, concurrent anticoagulation, and history of deep vein thrombosis or pulmonary embolism in the last 12 months. All oncological therapy had to be completed ⩾3 weeks, and major surgeries ⩾4 weeks, before enrollment.
Intervention
Tamoxifen (20 mg) was administered orally, once daily, continuously until intolerance, disease progression, or consent withdrawal. Toxicities were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. 19 In case of Grade⩾ 3 adverse events, tamoxifen could be interrupted for up to 3 weeks and restarted at a dose of 10 mg PO daily. No further dose reductions were allowed and patients requiring more than 3 weeks to recover from an adverse event were permanently discontinued. Adherence was captured by counting the drug blisters at every medical visit.
Assessments
Clinical and laboratory (complete blood count, hepatic and renal functions) evaluations were performed every 6 weeks throughout the trial and in 30 days after the end of the study. Symptoms related to NET hormone secretion were collected by asking directly patients, documenting details of frequency and severity.
Radiological assessment using RECIST 1.1 by computed tomography or magnetic resonance imaging scans were performed by local radiologists blinded to patient characteristics within 8 weeks of enrollment and every 12 weeks until progression.
IHC was used to evaluate the expression of ER-alpha and PR. Sections of 5 μm in thickness were cut from archival paraffin-embedded tumor specimens of accrued patients and subjected to an IHC staining protocol using the Ventana Autostainer (Ventana Medical Systems, Tucson, AZ, USA). Primary antibodies clones for ER and PR were SP1 and 1E2, respectively. Immunoreactivity was expressed as previously reported, with cases in which 1% or more of the neoplastic cells showing nuclear staining for these markers being considered positive, and when less than 1% of neoplastic cells stained for ER-alpha and/or PR or the specimen lacked nuclear staining, the expression was deemed negative.20–22
Statistical plan and sample size
Descriptive statistics were used to report means, medians (range), and frequencies of all patients and by exploratory subgroups. The Kaplan–Meier method was used to estimate all time-to-event data [with respective interquartile ranges (IQR)] and the log-rank test was used to compare PFS times by level of ER/PR expression (⩾30% versus <30%) and by type of hormone receptors expression (ER positive versus PR positive); tumors with both ER and PR expression were classified by the highest hormone receptor expression. Two-sided p values <0.05 were considered significant. All statistical analysis was carried out using the STATA IC/16.0 software (StataCorp, College Station, TX, USA).
The sample size calculation was based on the two-stage Minimax Simon phase II design. 23 The null hypothesis was a DCR at 6 months of 50% (placebo arm of phase III studies),3,24 and the alternative hypothesis, a DCR at 6 months of 70%. Considering a dropout rate of 30%, alpha and beta errors of 5% (two-sided) and 20%, we planned to enroll 23 in the first stage; if ⩾12 patients reached the primary end point, we would enroll a total of 37 patients. If 23 out of 37 patients presented DCR at week 24, the trial would be deemed positive.
Results
From February 2019 to February 2022, 59 patients were consented and screened for eligibility. In all, 23 (38.9%) had ER/PR positively expressed NETs and were enrolled.
Ten out of eleven hormone receptor-positive pancreatic NETs had PR-positive staining, with three also expressing ER: one pancreatic NET had PR expression in 20% of tumor cells and ER expression in 5%; another had PR expression in 90% and ER expression in 10%; and the other case had PR expression in 10% and ER, in 15%. Among nine nonpancreatic gastrointestinal NETs (small bowel = 6; unknown primary = 2; ampulla = 1), seven cases had ER-positive tumor staining [an ampulla NET had both ER (30%) and PR (90%) expressions] and two cases had exclusive PR-positive expression. Among three lung NETs, two had a PR-positive and one had an ER-positive tumor. PR expression was associated with pancreatic NETs and ER expression with gastrointestinal NETs (chi-square; p = 0.007).
Table 1 summarizes the main characteristics of patients. The median age was 56 (range: 33–87) years, 15 (65%) were females, and the most common primary sites were pancreas (11; 47.8%) and small bowel (6; 26%). Most (N = 16; 69.5%) patients had nonfunctioning NETs, and liver metastases (N = 22; 95.6%). The median Ki67 index was 6% (1%–40%), and 8 (34.8%), 13 (56.5%), and 2(8.7%) had G1, G2, G3 NETs, respectively. The large majority had positive tumor uptake on 68Gallium-Dotatate PET-CT scan. Nearly two-thirds had received two or more prior NET-directed therapies, with the most common one being SSA. The median time since diagnosis of metastatic disease to the first dose of tamoxifen was 43 months (2–306).
Characteristics of patients.

One patient with unknown Ki67 index; **ACTH: adrenocorticotropic hormone; &capecitabine and temozolomide; #5-fluorouracil and oxaliplatin.
Sum is not 100% as some patients had more than one metastatic site.
ECOG, Eastern Cooperative Oncology Group; ER, expression of estrogen; PR, progesterone.
At a median follow-up of 27 months, as of July 2022, 20 (87%) patients experienced progression: documented radiologically (N = 18) or clear clinical progression only (N = 2), two patients withdrew consent despite radiological stable disease and one patient was not evaluable for response. In all, 13 patients (56.5%) had disease stability at week 24. The best radiological response (thus excluding two patients who were not evaluable for radiological response) was stable disease achieved by 13 patients, and eight patients had early radiological progression at week 12. The waterfall plot (Figure 1) depicts the best percentage change from baseline in the size of target lesions for each patient. In most cases, tumors continued to grow throughout the trial.

Waterfall plot.
Median PFS was 7.9 months (IQR: 3.7–12.1). Median PFS was 9.4 months for patients with ER-positive NETs (IQR: 3.5–22.2) and 3.9 months for PR-positive cases (IQR: 3.7–8.4; Figure 2; log-rank p = 0.19). According to ER and/or PR IHC expression < or ⩾ 0%, the median PFS was 6.2 months (IQR: 3.5–8.4) versus 9.4 months (IQR: 3.9–14.6), respectively (Figure 3 supplement; log-rank p = 0.49). Table 2 summarizes the tumor characteristics of patients with ER/PR IHC expression ⩾30% and their respective week of PD. Table 3 describes characteristics of patients without disease progression at 12 months.
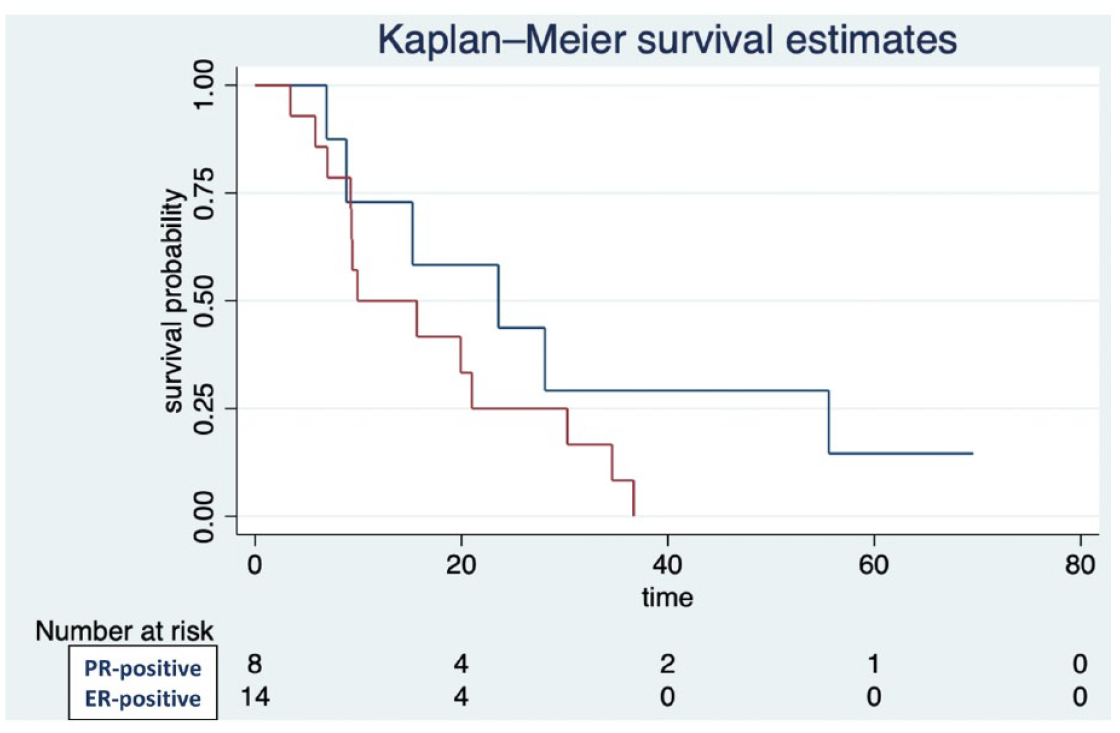
Progression free survival by type of hormone receptor IHC expression.

Progression-free survival by ER and/or PR NETs IHC expression < or ⩾ 30%.
Characteristics of NETs with ER/PR ⩾ 30% IHC expression.

ER, expression of estrogen; PR, progesterone.
Characteristics of patients without disease progression at 12 months.
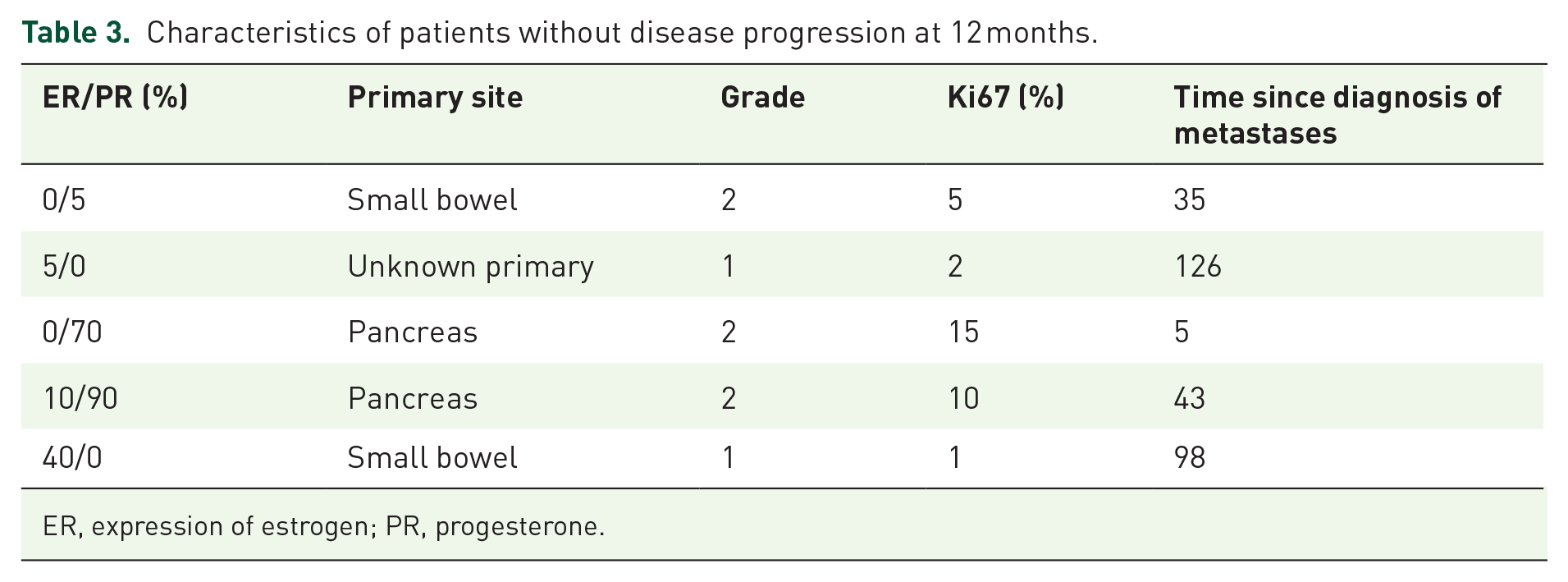
ER, expression of estrogen; PR, progesterone.
Three patients stopped their SSAs prior to starting the trial because they had no prior history of carcinoid syndrome. However, they developed flushing or diarrhea within 3 months of starting the trial and therefore resumed SSA with good symptomatic response. Three more patients started tamoxifen combined with an SSA because they had known functioning NETs. The median PFS of patients with functioning NETs and concurrent use of SSA versus those who received tamoxifen monotherapy were 6.1 versus 7.9 months, respectively (log-rank p = 0.14). Among the three patients with carcinoid syndrome, none experienced symptom improvement or decrease of 24 h urinary 5-hydroxyindoleacetic acid.
One patient, a 52-year-old female with metastatic small bowel PR-positive (10%) NET and refractory carcinoid syndrome, presented extensive mesenteric fibrosis and started tamoxifen as a fifth-line therapy combined with SSA. She did not experience benefit from her carcinoid syndrome or fibrosis and was discontinued from the trial at week 12 due to radiological progression.
Tamoxifen was well tolerated, with four (17.4%) patients experiencing treatment-related adverse events: three patients complained of G1 hot flashes and one patient reported G2 constipation.
Discussion
This multicentre phase II single-arm clinical trial in patients with metastatic and progressive ER or PR-positive NETs demonstrated that tamoxifen is safe but offers modest efficacy. Neither the intensity of ER/PR IHC staining, the concurrent administration of SSA nor the type of hormone receptor expressed seemed to significantly influence PFS.
With 13 patients achieving DCR at 24 weeks, the HORMONET study technically met criteria for continuation of enrollment beyond the initial phase. However, the trial was stopped for futility because of short median PFS, which is similar to the median of 5–6 months reported by placebo arms in randomized trials. In addition, the waterfall plot demonstrates that the large majority of patients developed radiological tumor growth throughout the treatment, and patients with functioning NETs did not experience clinical improvement of their symptoms. Disappointingly, even in a target-selected NET population, tamoxifen appeared to be ineffective.
The reasons why HORMONET was a negative study are unknown. Since tamoxifen is a strong inhibitor of ER/PR signaling, it is unlikely that the lack of efficacy observed here was caused by pharmacological aspects, or that other antiestrogen agents could be effective in this setting. It is possible that neither ER nor PR are key drivers of NET development and progression, and that blocking ER/PR signaling alone is not enough to induce tumor shrinkage and prolong tumor control.
The main limitation of our trial is its single-arm phase II design. A randomized clinical trial could better evaluate whether tamoxifen offers some degree of antiproliferative effects in ER-/PR-positive NETs. However, we think that even if there is any benefit, this would be of small magnitude. Another limitation is the definition of ER-/PR-positive staining on IHC, which was based on ER-alpha (the tamoxifen target) in a single paraffin-embedded tumor biopsy. In addition, intra-tumor heterogeneity might have led to false-negative cases or over-estimate of ER-/PR-positive cancer cells; and most PR+ NETs were of pancreatic origin, what may have contributed to a shorter PFS in comparison with ER+ tumors, which were mostly of small bowel origin. Lastly, our study had a heterogeneous sample composed of NETs of different origins, grades, and functionality – although we still did not observe signals of efficacy in any trial patient.
Future research on the physiology of estrogen and progesterone signaling in NET are encouraged. However, clinical trials of estrogen-directed therapies should be conducted only after more preclinical data enlighten the NET carcinogenic mechanisms of hormone receptors.
In conclusion, despite the appealing scientific rationale, tamoxifen monotherapy appears relatively ineffective for patients with ER-/PR-positive metastatic NETs.
