Abstract
Background:
In the FLIPPER trial, palbociclib/fulvestrant significantly improved progression-free survival (PFS) compared with placebo/fulvestrant in postmenopausal women with HR+/HER2− advanced breast cancer (ABC).
Objective:
We assessed health-related quality of life (QoL) using patient-reported outcomes (PROs).
Design and methods:
In this phase II double-blinded study, PROs were assessed at baseline after every three cycles and at the end of the treatment using the European Organisation for Research and Treatment of Cancer QLQ-C30 and QLQ-BR23. Time to deterioration (TTD) in global health status (GHS)/QoL was defined as a decrease of ⩾10 points. Changes from baseline (CFB) and TTD were analysed using linear mixed-effect and Cox regression models, respectively.
Results:
Of the 189 randomised (1:1) patients, 178 (94%) completed ⩾1 post-baseline assessment; 50% received ⩾22 cycles of study treatment, with a questionnaire compliance >90%. Mean baseline scores were comparable between arms. GHS/QoL scores were maintained throughout the palbociclib/fulvestrant treatment. CFB showed significant differences for GHS/QoL, appetite loss, constipation and systemic therapy side effect scores favouring placebo/fulvestrant. TTD in GHS/QoL was delayed in placebo/fulvestrant versus palbociclib/fulvestrant [30.3 versus 11.1 months; adjusted hazard ratio (aHR): 1.57, 95% CI: 1.03–2.39, p = 0.036]; this difference was not significant in patients with progressive disease (aHR: 1.2, 95% CI: 0.6–2.2, p = 0.658). No statistically significant differences in TTD were found for the other QLQ-C30 and QLQ-BR23 scales.
Conclusions:
Although TTD in GHS/QoL was prolonged with placebo/fulvestrant, no differences were observed on other functional or symptom scales. This finding and the improvement in PFS support the combination of palbociclib/fulvestrant as a beneficial therapeutic option for HR+/HER2− ABC.
Trial registration number:
Sponsor Study Code: GEICAM/2014-12
EudraCT Number: 2015-002437-21
ClinTrials.gov reference: NCT02690480
Keywords
Introduction
In the FLIPPER trial (ClinTrials.gov number, NCT02690480), the efficacy and safety of the CDK4/6 inhibitor palbociclib plus fulvestrant was compared with placebo plus fulvestrant for the treatment of postmenopausal woman with endocrine-sensitive hormone receptor-positive (HR+) and human epidermal growth factor receptor 2-negative (HER2−) advanced breast cancer (ABC). Primary results demonstrated a statistically significant improvement for a number of metrics with palbociclib/fulvestrant as compared to placebo/fulvestrant. Specifically, 1-year progression-free survival (PFS) rate [83.5% versus 71.9%; hazard ratio (HR): 0.55, 80% confidence interval (CI): 0.36–0.83, p = 0.064], PFS [median 31.8 versus 22.0 months; adjusted HR (aHR): 0.48; 80% CI: 0.37–0.64, p = 0.001)] and overall response rate (68.3% versus 42.2%; odds ratio: 2.9; 80% CI: 1.8–4.6, p = 0.004) with a manageable safety profile. Results on overall survival (OS) are still immature 1
The addition of CDK4/6 inhibitors to endocrine therapy (ET) as first-line therapy for postmenopausal woman with HR+/HER2− ABC leads to a statistically and clinically significant improvement in PFS when compared with endocrine monotherapy.2–4 However, combination treatments can expose patients to additional treatment-related toxicities, which may negatively affect their quality of life (QoL). Recommendations from medical oncology societies experts and regulators highlight the need for integration of patient-reported outcomes (PROs) into the discussion of efficacy and safety drug profile to better define their clinical benefit.5–7 Here, we report validated cancer-related and breast cancer-specific PRO results from the FLIPPER trial.
Material and methods
Study design and patients
The FLIPPER is a phase II, randomised, international, multicentre, double-blinded, placebo-controlled trial comparing the efficacy of palbociclib/fulvestrant versus placebo/fulvestrant in postmenopausal women with HR+/HER2− ABC. Patients had either de novo metastatic disease or remained disease free for >12 months after completing at least 5 years of adjuvant ET. Patients were assigned in a 1:1 ratio to receive palbociclib (125 mg/day, 28-day cycle; 3 weeks on, 1 week off) or matched placebo, and both arms received fulvestrant (500 mg on day 1 of each 28-day cycle, with an additional dose on day 15 of cycle 1). Treatment continued until objective progressive disease (PD) according to the Response Evaluation Criteria in Solid Tumours version 1.1, 8 symptomatic deterioration, unacceptable toxicity, death or withdrawal of consent, according to which occurred first. The detailed study design and characteristics of patients have been reported previously. 1
PRO assessments
The comparison of the health-related QoL (HRQoL) between treatment arms as reported by patients was a secondary objective of the trial using as secondary endpoints the changes from baseline (CFB) and time to deterioration (TTD) PRO measures of HRQoL were assessed using the European Organisation for Research and Treatment of Cancer (EORTC) QoL questionnaire-Core 30 (QLQ-C30) V3.0 9 and its breast cancer-specific module (QLQ-BR23; v1.0). 10
Patients were asked to complete each questionnaire at baseline, every three cycles until the end of treatment, and at the post-treatment visit (performed 30 days after the last study treatment dose). Questionnaires were completed by patients in the clinic environment prior to any testing or discussion with healthcare personnel at the site.
The EORTC QLQ-C30 is a 30-item questionnaire composed of a global health status (GHS)/QoL multi-item scale, five multi-item functional scales (physical, role, emotional, cognitive and social), three multi-item symptom scales (fatigue, nausea/vomiting and pain) and six single-item symptom scales (dyspnoea, insomnia, appetite loss, constipation, diarrhoea and financial difficulties) 9 (Table 1). The EORTC QLQ-BR23 questionnaire is a 23-item companion module consisting of two multi-item functional scales (body image and sexual functioning) and three multi-item symptom scales (systemic therapy side effects, arm and breast symptoms) and single item functional and symptom scales covering sexual enjoyment, future perspective and upset due to hair loss 10 (Table 1).
Item numbers and definition of the MID as CFB values by scale in the EORTC QLQ-C30 and QLQ-BR23 instruments.

A deterioration event is an increase of ⩾MID from baseline for the symptom scales and a decrease of ⩾MID from baseline for the functional scales and GHS/QoL. A clinically meaningful is a decrease of ⩾MID from baseline for the symptom scales and an increase of ⩾MID from baseline for the functional scales and GHS/QoL.
The GHS/QoL items were rated on a seven-point Likert scales with responses from ‘very poor’ to ‘excellent’ and the other QLQ-C30 items and all QLQ-BR23 items were rated on a four-point Likert scale from ‘not at all’ to ‘very much’.
CFB, change from baseline; EORTC QLQ-BR-23, European organisation for research and treatment of cancer breast-specific questionnaire; EORTC QLQ-C30, European organisation for research and treatment of cancer core questionnaire; GHS/QoL, global health status/QoL; MID, minimally important difference.
Two seven-point Likert scales for GHS/QoL with responses from ‘very poor’ to ‘excellent’ and four-point Likert scales to assess functioning and symptoms with responses from ‘not at all’ to ‘very much’ were provided.
Responses to all item measures were converted into linear scales ranging from 0 to 100 using a standard scoring algorithm. 11 For the GHS/QoL and functional scales, a higher score represents a better level of QoL/functioning and an increase from baseline indicates an improvement in QoL. By contrast, for symptomatic scales, a higher numerical score represents greater or worse symptom severity.
Statistical analysis
PROs analyses were performed in cases with baseline and at least one post-baseline assessment (QoL population). Completion rates were summarised by visit in the intention-to-treat (ITT) population; a questionnaire was considered received if at least one question was answered. For partially completed multi-item scales, missing scores were equal to the average of the completed items if at least half of the items of that scale were answered, but were not included in the analysis if less than that were completed.
Descriptive statistics, including 95% CI for the means of actual values and CFB were tabulated at the scheduled time points for each scale of the EORTC QLQ-C30 and EORTC QLQ-BR23 questionnaires. The means and 95% CIs of CFB, as well as comparisons between treatment arms with their respective p-values, were analysed using a linear mixed model. The model factors were treatment arms, time points, treatment–time interaction terms, and stratification criteria, and the covariates were the baseline scores. A random-intercept only model with a first-order autoregressive covariance structure was used. Baseline scores were compared between treatment arms using t-test.
TTD investigated both in the entire study QoL population and in subgroups, according to therapy response (with/without PFS event), was defined as the time from the date of randomisation to the date of first detection of a deterioration event. The minimally important difference (MID) is the change in score of a PRO that is important from the patient’s or clinician’s perspective and would warrant a change in the patient’s management. The MID values used were between 2 and 10 points according to data previously published.12,13 A deterioration event was defined as an increase of ⩾MID from baseline for the EORTC QLQ-C30 and QLQ-BR23 symptom scales and a decrease of ⩾MID from baseline for the EORTC QLQ-C30 and QLQ-BR23 functional and GHS scales. Results were defined as clinically meaningful based on MIDs for EORTC QLQ-C30 and QLQ-BR23 (Table 1). Patients with no definitive deterioration event were censored at their last available QoL assessment. In patients with no post-baseline assessment, TTD was censored on day 1.
The Kaplan–Meier method was used to estimate the distribution of TTD for each treatment arm and according to PFS event. A log-rank test was performed to compare the TTD between treatment arms. HRs were adjusted by stratification factors: disease site (visceral versus non-visceral) and (recurrent versus de novo metastatic disease). Adjusted HR (aHRs) and two-sided 95% CIs were estimated using Cox regression model for the comparison of treatment arms.
Results
Patient characteristics
Between February 2016 and January 2018, 189 patients were recruited across 32 institutions in two countries [177 patients in Spain (GEICAM) and 12 in Ireland (Clinical Trials Ireland)]. The QoL population comprised 178 (94.2%) patients with completed baseline and ⩾1 post-baseline PROs. Of these, 88 were included in the palbociclib/fulvestrant arm and 90 in the placebo/fulvestrant arm (Supplemental Figure S1).
Baseline demographic and disease characteristics were well balanced between arms (Supplemental Table S1). Median age was 64 years, 97% of patients had an Eastern Cooperative Oncology Group performance status of zero or one, 45.5% had de novo metastatic disease and among those having non-de novo metastatic disease, 39.7% had a treatment-free interval after adjuvant ET greater than 36 months. Most patients (60.3%) had visceral involvement; overall, 33.9% patients had lung involvement, 15.9% liver involvement and 15.9% bone-only disease.
Completion rates
All PRO analyses were based on a cut-off date of 11 January 2020, at a median follow-up time of 28.6 months (range, from 1.5 to 44.8). At this point, 50% of patients had received the first 22 cycles of study treatment and the overall questionnaires compliance was high in both treatment arms, with a completion rate ⩾96% at baseline, ⩾92% on treatment and ⩾84% at post-treatment visit (Supplemental Table S2).
The compliance rates of each EORTC QLQ-C30, EORTC QLQ-BR23 scale were like the global questionnaire completion rates, except for sexual enjoyment and, upset due to hair loss, where sample sizes were smaller versus other scales (Table 2).
Baseline mean scores of EORTC QLQ-C30 and EORTC QLQ-BR23 scales by treatment arm.

Reference baseline value for recurrent and metastatic breast cancer patients across all lines of treatment. 12
The patients were asked to answer this question only if they responded that they were sexually active in a previous question.
Only patients with alopecia were asked to answer this question.
EORTC QLQ-BR-23, European organisation for research and treatment of cancer breast-specific questionnaire; EORTC QLQ-C30, European organisation for research and treatment of cancer core questionnaire; QoL, quality of life; SD, standard deviation.
EORTC QLQ-C30 and QLQ-BR23 baseline scores
Mean baseline scores were similar in all dimensions among the treatment arms, except for the body image, with better level of functioning in placebo/fulvestrant arm (Table 2). Globally, all baseline scores were in line with the EORTC QLQ-C30, QLQ-BR23 reference values expected for ABC. 14
CFB per treatment arm
Palbociclib/fulvestrant
The combination of palbociclib/fulvestrant showed a GHS/QoL mean scores maintained numerically up to cycle 22, and a decrease occurred at the post-treatment visit (Figure 1). When analysing the mean CFB by visit, significant improvement at different time points was found for emotional functioning, social functioning, body image, pain, dyspnoea, diarrhoea, breast symptom and arm symptom (Supplemental Table S3). Meanwhile, significant impairment was found for future perspective, constipation, financial difficulties and systemic therapy side effects when the analysis of mean CFB by visit was performed (Supplemental Table S4). No significant differences were observed for other functional, symptom or GHS/QoL scales.

GHS/QoL mean scores in the EORTC QLQ-C30 scales by visit.
Placebo/fulvestrant
The GHS/QoL mean scores were numerically improved up to post-treatment visit (Figure 1). Statistically significant increases in the mean CFB during all study treatment visits were found for GHS/QoL (Table S3).
Significant improvements were found at different time points in the mean CFB by visit for role functioning, emotional functioning and appetite loss (Table S3). Significant impairment was found for cognitive functioning, future perspective and systemic therapy side effects (Supplemental Table S4). Most scales worsened in both study arms at the post-treatment visit since the reason for terminating the treatment was PD in 82.4% of the cases (Supplemental Table S5).
Clinically meaningful improvements
Clinically meaningful improvements (based on MID values) were observed in both treatment arms at different time points for emotional functioning, pain, appetite loss and breast symptoms; while such improvements developed for arm symptoms in the palbociclib/fulvestrant arm, in the placebo/fulvestrant arm improvements developed for role functioning and dyspnoea (Figure 2).

Clinically meaningful improvements from baseline in the EORTC QLQ-C30 and QLQ-BR23 scales by treatment arm.
Comparison of CFB between treatment arms
The overall CFB for all scales of the QLQ-C30 and QLQ-BR23 questionnaires are presented in Figure 3. Significant improvements were observed for GHS/QoL [6.8 (95% CI: 3.5–10.0) versus 0.2 (95% CI: −3.2–3.6); p = 0.005], and appetite loss [−7.3 (95% CI: −10.4–4.3) versus −0.6 (95% CI: −5.7–4.5); p = 0.021], and significantly less deterioration for constipation [2.5 (95% CI: −2.0–6.9) versus 10.5 (95% CI: 5.0–15.9); p = 0.027] and for systematic therapy side effects [2.7 (95% CI: 0.2–5.1) versus 8.3 (95% CI: 5.2–11.4); p = 0.004] favouring the control arm with placebo/fulvestrant.

Overall CFB in EORTC QLQ-C30 and BR23 scales.
According to the linear mixed model analysis, the change of GHS/QoL score from baseline to cycle 7 was 1.5 (95% CI: −2.5–5.5) for palbociclib/fulvestrant versus 7.9 (95% CI: 3.4–12.4) for placebo/fulvestrant. The mean difference was −6.4 (95% CI: −12.4–0.5; p = 0.035), maintained up to cycle 19, showing a higher improvement in the placebo/fulvestrant arm. No significant differences were observed between treatment arms at other time points (Supplemental Figure S2).
Time to deterioration
The median TTD in GHS/QoL, using a MID = 10, was 11.1 months with palbociclib/fulvestrant versus 30.3 months with placebo/fulvestrant (aHR: 1.57, 95% CI: 1.03–2.39; p = 0.036) ((Figure 4(a)). The analysis performed only in patients without a PFS event showed similar results, 11.1 months with palbociclib/fulvestrant versus 30.3 months with placebo/fulvestrant (aHR 2.04: 95% CI: 1.11–3.77; p = 0.023). No statistically significant difference was seen in TTD in the analysis performed in patients with a PFS event (aHR: 1.15, 95% CI: 0.61–2.18; p = 0.658) (Figures 4(b) and (c)).

Kaplan–Meier estimates for TTD in GHS/QoL based on the EORTC QLQ-C30 questionnaire data.
No significant differences were seen in that respect among patients with or without a PFS event in both arms, palbociclib/fulvestrant (aHR: 1.21; 95% CI: 0.69–2.11, p = 0.504) and placebo/fulvestrant (aHR: 0.55; 95% CI: 0.26–1.13, p = 0.104) (Supplemental Figure S3).
No significant differences were seen for the risk of deterioration between the study arms of the median TTD for the other QLQ-C30 and QLQ-BR23 scales, except for arm symptoms, using a MID = 5, with a median TTD of 20.4 months with palbociclib/fulvestrant versus 8.3 months with placebo/fulvestrant (aHR: 0.59, 95% CI: 0.39–0.88; p = 0.011). In the Forest-plot with the QLQ-C30 and QLQ-BR23 scales for TTD, a trend towards one of the treatment arms cannot be observed, the distribution from the unit towards both sides is heterogenous (Figure 5).
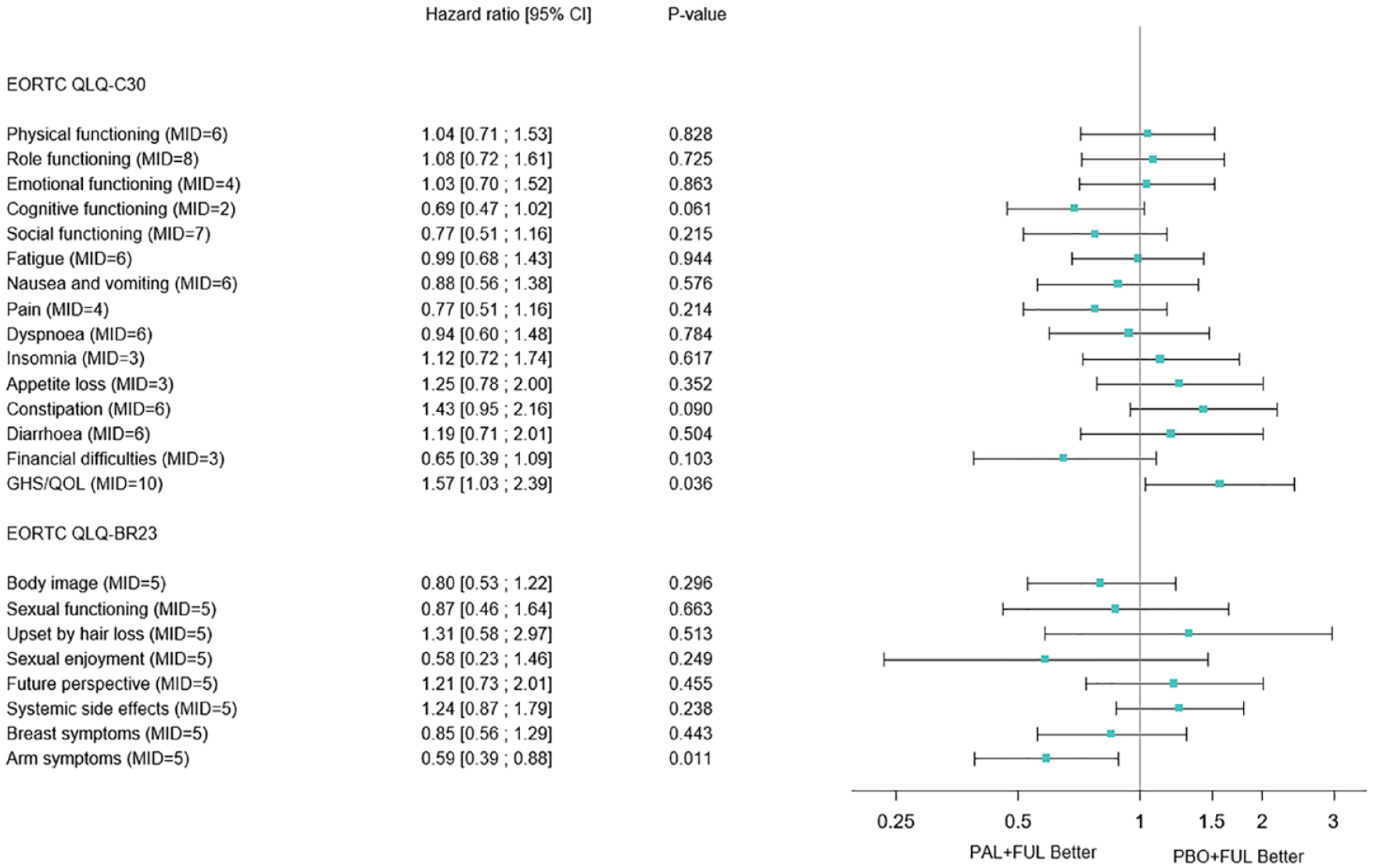
Forest plot TTD in the various scales of the EORTC QLQ-C30 and EORTC QLQ_BR23 questionnaires.
Subgroup analyses of TTD in GHS/QoL showed longer delayed deterioration with placebo/fulvestrant over palbociclib/fulvestrant (Supplemental Figure S4).
Discussion
Maintaining QoL is a key goal of the treatment of ABC patients. Although treatments can reduce symptoms, thereby slowing the impairment in QoL and prolonging time to PD, treatment-related toxicities affect the HRQoL. Here, we present detailed cancer-related and breast-cancer-specific PROs regarding the palbociclib/fulvestrant administration in the first-line treatment of endocrine-sensitive HR+/HER2− ABC patients. We demonstrated that the favourable efficacy achieved with palbociclib/fulvestrant is accompanied by maintained overall QoL throughout the study treatment period. Interestingly, baseline GHS/QoL and functioning subscale scores were maintained and similar between study arms, except for GHS/QoL, appetite loss, constipation and systemic therapy side effects, which statistically favoured the control over time. Appetite loss, constipation, and systemic therapy side effects, as treatment-related toxicities that can adversely affect the HRQoL, could have contributed to the reduction in GHS/QoL observed in our study. Indeed, this is a relative reduction compared to the placebo arm and not an absolute reduction, since the GHS/QoL scores in the experimental arm are maintained and numerically improved during treatment compared to baseline values. In addition, declines in these symptom scales were consistent with the safety profile observed with palbociclib/fulvestrant in the FLIPPER trial. 1 In the ITT population, most adverse events (AEs) observed in the experimental arm were of mild severity (except for neutropenia), with higher rates of Common Terminology Criteria for Adverse Events grade I-II anorexia reported in the palbociclib/fulvestrant group (16%) versus the placebo/fulvestrant arm (9.5%) and no differences in grade I-II constipation (19%). Permanent discontinuation of the whole study treatment due to AEs was similar in both arms and occurred in four patients (4.3%) receiving palbociclib/fulvestrant and 4 patients (4.2%) receiving placebo/fulvestrant. Overall, these results may reflect the importance of chronic low-grade toxicities in patient experience, and how in regulatory clinical trials, these are generally not captured well. 15
Although no statistical or clinically meaningful differences were observed in the key subdomains of the EORTC QLQ-C30 and QLQ-B23 questionnaires, patients treated with placebo/fulvestrant experienced a significantly greater delay in deterioration of GHS/QoL than patients treated with palbociclib/fulvestrant. In addition, patients treated with ET in monotherapy without PD experienced delayed TTD in GHS/QoL but not patients whose disease progressed, suggesting that negative impacts on functioning scores may be primarily due to the study treatment AEs.
These PROs from the FLIPPER trial add to the growing body of evidence reported of CDK4/6 inhibitors trials. 16 For postmenopausal patients, the majority of trials report that HRQOL has been maintained in the first-line setting17–20 or improved in the second-line or later-line setting. 21 Our results are not fully consistent with those observed in the PALOMA-3 trial, where premenopausal and postmenopausal patients with endocrine-resistant HR+/HER2− metastatic breast cancer were randomised to palbociclib/fulvestrant versus placebo/fulvestrant. This trial demonstrated improved PFS and a non-statistically significant increase in OS 22 with a delay in the TTD of QoL and pain symptoms. 23 This inconsistency regarding QoL may reflect a younger, more heavily pre-treated, population with fewer comorbidities included into the PALOMA-3 trial. Conversely, the FLIPPER trial recruited only postmenopausal patients with ABC, older (median age was 64 in the FLIPPER versus 57 in the PALOMA-3 trial), sensitive to ET, less symptomatic and who were receiving the first-line therapy. Sensitivity was defined as de novo ABC or recurrent disease ⩾1 year after the completion of ⩾5 years of (neo) adjuvant ET, a patient population specifically excluded in the PALOMA-3 trial. Similar results were observed with palbociclib over placebo in PALOMA-2 trial, where the addition of palbociclib to letrozole significantly improved PFS while maintaining HRQOL in treatment-naïve postmenopausal women with ER+/HER2− MBC. 24 However, in PALOMA-2 study, HRQOL deterioration was delayed in patients with response and/or no progression. This inconsistency regarding HRQOL may be due to type of ET and less sensitivity to ET (22% of patients with a relapse ⩽12 months) into the PALOMA-2 trial. 2 Finally, when palbociclib in combination with ET has been compared versus chemotherapy (capecitabine) in a more heavily pre-treated, endocrine resistance population HRQOL was maintained regardless of menopausal setting,21,25 albeit in PEARL trial patients showed earlier HRQOL deterioration with chemotherapy. 21
Our results shed light on benefit–risk assessments to inform decision-making for palbociclib in postmenopausal patients with ABC. Maintaining HRQoL in patients with ABC is crucial, especially for drugs that improve PFS with no OS benefit. Efficacy data recently published 1 shown OS data were still immature at the time of this analysis with only a 15% of ITT population with an OS event. Ongoing follow-up will help for a better knowledge of the risk–benefit profile of palbociclib (expected date of publication 2025).
Potential limitations of FLIPPER study are as follows: (i) a small number of participants in a phase II study (ii) and the number of cycles included in the analysis. The cut-off of this analysis, not pre-planned, was cycle 22 because at this point 50% of patients had received the study treatment and the overall questionnaires compliance was high in both treatment arms. Therefore, delayed TTD were not captured and ⩾50% of patients were still on treatment at the analysis cut-off date. Further follow-up and updated data collection will help to mitigate this bias. Another potential limitation in the design of PRO analyses should be considered as data cannot be assumed missing at random because frail patients are likely to have lower compliance and bias could occur as a result. To address this limitation, the mixed-model approach was chosen as well as the high compliance rates of treated patients in all study visits minimised the impact of this limitation. Despite these, strength of FLIPPER study are (i) a randomised double-blinded design, (ii) high compliance rates ⩾90% and (iii) the study included multiple instruments with a dedicated questionnaire to identify breast-cancer-specific functions and symptoms and differentiated MID values for each multi-item scale of EORTC QLQ-C30 and EORTC QLQ-BR23 modules.
Conclusions
Our findings provide evidence that overall HRQoL in the FLIPPER trial was maintained in postmenopausal women with endocrine-sensitive HR+/HER2− ABC receiving palbociclib in combination with fulvestrant as first-line treatment. Taken together, the improved clinical efficacy, manageable safety profile and PRO results provide a meaningful assessment of the benefits, risks and tolerability of palbociclib/fulvestrant in this patient population.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221148921 – Supplemental material for Quality of life with palbociclib plus fulvestrant versus placebo plus fulvestrant in postmenopausal women with endocrine-sensitive hormone receptor-positive and HER2-negative advanced breast cancer: patient-reported outcomes from the FLIPPER trial
Supplemental material, sj-docx-1-tam-10.1177_17588359221148921 for Quality of life with palbociclib plus fulvestrant versus placebo plus fulvestrant in postmenopausal women with endocrine-sensitive hormone receptor-positive and HER2-negative advanced breast cancer: patient-reported outcomes from the FLIPPER trial by Ariadna Tibau, M. Teresa Martínez, Manuel Ramos, Luis De La Cruz-Merino, Ana Santaballa, Miriam O’Connor, Noelia Martínez-Jañez, Fernando Moreno, Isaura Fernández, Juan Antonio Virizuela, Jesús Alarcón, Juan de La Haba-Rodríguez, Pedro Sánchez-Rovira, Cinta Rosa Albacar, Coralia Bueno Muiño, Catherine Kelly, Maribel Casas, Susana Bezares, Libertad Rosell and Joan Albanell in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We acknowledge the patients who participated in the FLIPPER study and their families and all the participating investigators. We also acknowledge the support of the staff at each study site and at both GEICAM and Cancer Trials Ireland headquarters. We would like to thank Elsevier Language Editing Services for English language editing.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
