Abstract
Background:
There is no recognized optimal second-line treatment option for advanced hormone receptor-positive (HR+) breast cancer patients with CDK4/6 inhibitor (CDK4/6i) resistance.
Objectives:
This work aims to identify the optimal treatment option by evaluating the efficacy of various second-line treatment options in CDK4/6i-pretreated HR+ advanced breast cancer. Subgroup analyses aim to discuss how different genetic backgrounds and clinical characteristics influence the efficacy.
Design:
A systematic review and network meta-analysis (NMA) was designed.
Data sources and methods:
A comprehensive search was conducted in Medline, Embase, and Cochrane Library for randomized controlled trials (RCTs). The primary outcome was progression-free survival (PFS), with subgroup analysis based on visceral metastasis and ESR1 mutations. Secondary outcomes were overall survival (OS), overall response rate (ORR), and clinical benefit rate (CBR). Bayesian NMA was conducted using GeMTC in R, with hazard ratios and 95% confidence intervals as effect measures.
Results:
Our analysis included 19 clinical trials involving 13 different treatment regimens (n = 6621), with 14 studies (n = 3876) reporting subgroup results for CDK4/6i-pretreated patients. Results showed that the combination of CDK4/6i and fulvestrant (Ful) was the most effective regimen for improving PFS in CDK4/6i-pretreated HR+ advanced breast cancer patients. Further subgroup analyses of visceral metastasis and ESR1 mutations consistently confirmed this finding. For OS, the combination of Bcl-2 inhibitor (Bcl-2i) and Ful was most favorable. In terms of ORR and CBR, selective estrogen receptor degraders (SERD) and CDK4/6i + ET were the most beneficial, respectively, with no significant differences among regimens in direct comparisons.
Conclusion:
Our analysis reveals CDK4/6i + Ful is the most effective treatment option for CDK4/6i-pretreated HR+ advanced breast cancer, particularly in patients with visceral metastases or ESR1 mutations. In addition, Bcl-2i + Ful and SERD may be potential second-line strategy options.
Trial registration:
This meta-analysis was registered in PROSPERO (CRD42024518926).
Plain language summary
Hormone receptor-positive (HR+) breast cancer is a common type of breast cancer that often relies on hormonal signals to grow. CDK4/6 inhibitors, combined with endocrine therapy, are a standard first-line treatment for advanced HR+ breast cancer. These therapies work by stopping cancer cells from dividing and growing. However, some patients experience disease progression despite this treatment. This study explores the next steps in treatment after patients develop resistance to CDK4/6 inhibitors. Using a network meta-analysis, a method that compares multiple treatments by analyzing data from various clinical trials, we evaluated the effectiveness of several treatment options. These include switching to a different CDK4/6 inhibitor, continuing the same CDK4/6 inhibitor with a new hormonal therapy, or using newer drugs like selective estrogen receptor degraders (SERDs), BCL-2 inhibitors, ADC and so on. Our findings highlight that switching to another CDK4/6 inhibitor and combining it with fulvestrant can provide significant benefits, particularly in patients with visceral metastases or ESR1 mutations. Additionally, novel treatments, like the latest SERDs and Bcl-2 inhibitor, show promise and are under investigation in ongoing clinical trials. This study emphasizes the importance of tailoring treatment based on individual patient needs and the mechanisms driving resistance. The results provide valuable insights to guide clinical decisions and offer hope for better outcomes for patients with advanced HR+ breast cancer.
Introduction
Breast cancer is the leading cancer in the world, with more than 2.26 million new cases every year. 1 Approximately 4%–6% of patients are initially diagnosed with advanced breast cancer, and approximately one-third of patients with early-stage disease experience recurrence or metastasis after adjuvant therapy. Unfortunately, the 5-year survival rate for advanced breast cancer patients is only 20%. The hormone receptor-positive, HER2-negative (HR+/HER2−) breast cancer subtype accounts for 70% of all breast cancer cases. 2 Therefore, a therapeutic breakthrough in such advanced patients is highly important. The advent of CDK4/6i has brought new hope to advanced HR+ breast cancer patients. CDK4/6i blocks the transition of tumor cells from the G1 to the S phase by selectively inhibiting the function of CDK4/6, thereby exerting antitumor effects. 3 Multiple phase III clinical trials have confirmed the role of CDK4/6i combined with endocrine therapy (ET) in the first-line treatment of HR+ advanced breast cancer. For example, PALOMA-2 (a late-stage, first-line, phase III clinical trial) showed that the combination of palbociclib and aromatase inhibitor (AI) improved progression-free survival (PFS) by 10.3 months (hazard ratio (HR) = 0.6, 95% confidence interval (CI): 0.5–0.7, p < 0.01). 4 The findings of the MONALEESA-3 clinical trial underscore the efficacy of CDK4/6i + fulvestrant (Ful) as a first-line treatment for advanced HR+ breast cancer, with a median PFS (mPFS) of 33.6 months and median overall survival (mOS) of 67.6 months. 5 In light of this evidence, both the 2024 edition of the National Comprehensive Cancer Network guidelines 6 and the international consensus guidelines for the management of advanced breast cancer (6th and 7th) 7 unequivocally endorse CDK4/6i + ET as the standard first-line treatment for HR+ advanced breast cancer. However, almost all advanced breast cancer patients eventually develop resistance to CDK4/6i + ET, resulting in disease progression. Second-line treatment involves various options, making it a key point of research in the breast cancer field.
Clinical trials related to treatment strategies in CDK4/6i-pretreat are in progress. Activation of the phosphoinositide 3-kinase (PI3K) pathway is an important factor in endocrine resistance. The results for SOLAR-1, 8 BYLieve, 9 FAKTION, 10 and CapitELLA-291 11 confirmed that PI3K inhibitors, as well as AKT inhibitors, are important options for HR+ advanced breast cancer patients with CDK4/6i resistance. In addition, the post-MONARCH and MAINTAIN study showed that patients with CDK4/6i resistance could still achieve significant improvement in PFS with another CDK4/6i as a second-line treatment. Antibody–drug conjugates (ADCs) have also been explored in HR+ breast cancer patients with CDK4/6i resistance. 12 The DESTINY-Breast04 13 and TROPiCS-02 14 trials showed the favorable efficacy of trastuzumab deruxtecan (T-Dxd) and sacituzumab govitecan (SG) in these patients. Furthermore, the EMERALD study demonstrated a significant improvement in PFS with oral selective estrogen receptor degraders (SERDs) as the second-line treatment. 15 In addition, chemotherapy remains a key therapeutic option, with studies indicating that metronomic chemotherapy enhances PFS in populations resistant to CDK4/6 inhibitors. These results provide valuable therapeutic options for HR+ breast cancer patients with CDK4/6i resistance, but no unified conclusion has been reached. 16
This work is the first and most comprehensive network meta-analysis (NMA) targeting CDK4/6i-pretreated HR+ advanced breast cancer. Our analysis highlights that CDK4/6i + Ful is the most effective second-line treatment option for CDK4/6i-pretreated HR+ advanced breast cancer, particularly in patients with visceral metastases or ESR1 mutations. In addition, Bcl-2 inhibitor (Bcl-2i) + Ful and SERD may be potential second-line strategy options (Figure 1).

Options after progression on first-line CDK4/6 inhibitors in advanced breast cancer patients.
Materials and methods
Search strategy and study identification
We searched PubMed, EMBASE, Cochrane, and abstracts from recent international conferences (mainly from the American Society of Clinical Oncology) and the European Society for Medical Oncology (ESMO), with no clear time or language restrictions. A comprehensive and systematic search for “CDK4/6 inhibitor” and “advanced breast cancer” was independently conducted by two authors, and discrepancies were resolved by discussion with the third author. The full electronic search strategy for the PubMed database, including any limits and field tags, is provided as Supplemental Material 1. The protocol was submitted to the PROSPERO database (CRD42024518926, available from https://www.crd.york.ac.uk/PROSPERO/display_record.php?RecordID=518926). The systematic review and NMA were designed and conducted in accordance with the Cochrane Handbook for Systematic Reviews of Interventions. 17 This study was conducted and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines for Network Meta-Analyses (PRISMA-NMA). 18 The completed PRISMA-NMA checklist is provided as Supplemental Material 2.
Selection criteria
To be considered for inclusion in this NMA, studies had to meet all the following inclusion criteria: (1) Advanced HR+ breast cancer patients who experienced treatment failure in first-line therapy with CDK4/6i; (2) patients who received subsequent drug treatments after CDK4/6i; (3) studies reporting treatment-related endpoint data; and (4) publications written in English. The exclusion criteria were as follows: (1) Patients with early-stage or adjuvant breast cancer. (2) Studies that did not report detailed treatment regimens or endpoint data. (3) Animal studies, phase I clinical trials, case reports, and non-clinical research.
Data extraction
Based on the inclusion and exclusion criteria, X.J. and Y.L. searched separately for the following information: author/trial name, year of publication, country, stages, outcomes, treatment arms, and number of patients. Disagreements were addressed via consensus. Data were extracted and reported as a single trial when several publications for the same trial were located.
Assessment of bias risk
Two authors independently assessed the risk of bias for each included study using the Cochrane Collaboration tool. Disagreements were resolved with the assistance of the third author. We evaluated the following using the Cochrane risk of bias tool: (1) random sequence generation (selection bias); (2) allocation concealment (selection bias); (3) blinding of participants, personnel, and outcome assessment (performance bias and detection bias); (4) incomplete outcome data (attrition bias); and (5) selective reporting data (reporting bias).
Statistical analysis
The primary endpoint of the study was PFS, and the secondary endpoints included OS, overall response rate (ORR), and clinical benefit rate (CBR). The Stata 16.0 and R 4.4.2 systems were used to analyze the extracted data. Relative Risk (RR), Hazard Ratio (HR), and 95% CI were estimated as effect size measures. The Bayesian NMA using the GeMTC package in R was performed to indirectly compare. The Higgins I2 index was calculated to validate homogeneity. If I2 exceeded 25%, the random-effects model (DerSimonian and Laird technique) was adopted; otherwise, the fixed-effects model (Mantel–Haenszel method) was applied. The probability rank command was used to rank the drug regimens, and the statistical significance of two-sided differences was tested (α < 0.05). Stata software was used to generate network plots, where circles represent different drug regimens and connecting lines represent direct comparisons between drug regimens. The size of the circles represents the sample size of the protocol group, and the line thickness represents the number of studies. The intervention hierarchy was summarized and reported as P score, which is considered a frequentist analog of the surface under the cumulative ranking curve (SUCRA). The P score ranges from 0 to 1, where 1 indicates the best treatment and 0 indicates the worst treatment. Sensitivity analysis is performed by perform_improved_sensitivity function. In the league table of the NMA, each row and column represents a different treatment regimen, and each cell shows the HR and 95% CI between the two treatment regimens. If the HR is less than 1, it indicates that the treatment regimen is more effective, and a narrower CI suggests more reliable results.
Results
Screening process and study characteristics
In this work, we screened a total of 764 references on treatment strategies for HR+ advanced breast cancer. After removing duplicate entries, irrelevant topics, review articles, and methodological studies, 446 articles remained for further analysis. Only 141 papers remained after screening the titles and abstracts of the full-text review. After full-text analysis, 19 studies with a total of 6621 patients were finally included for statistical analysis.8,11,15,19–34 Most were phase III randomized controlled trials (RCTs) and 14 studies (n = 3876) reported subgroup results for CDK4/6i-pretreated patients. Figure 2 illustrates the search process and Table 1 summarizes the study characteristics. Based on this, we conducted an NMA of the PFS result for both overall and CDK4/6i-pretreated patients, with NMA diagrams shown in Figure 3(a) and (c), respectively. Then, we designed NMAs for OS, CBR, and ORR in CDK4/6i-pretreated patients to comprehensively evaluate the efficacy of 10 regimens. NMA figure is shown in Supplemental Figure 1.

Flow diagram of the selection strategy.
Study characteristics of trials included in the network meta-analysis.

The PFS outcome is presented as the HR with a 95% CI.
Patients after the application of CDK4/6 inhibitors.
Standard-of-care.
AI, aromatase inhibitor; CI, confidence interval; ET, endocrine therapy; Ful, fulvestrant; HR, hazard ratio; OFS, ovarian function suppression; PFS, progression-free survival; T-Dxd, trastuzumab deruxtecan.

Network plot of and forest plot of PFS. (a) Network plot in total patients. (b) The forest plot in total patients. (c) Network plot in CDK4/6i-pretreated patients. (d) Forest plot in CDK4/6i-pretreated patients.
Network meta-analysis
PFS was used as the primary endpoint in all included trials. PFS was defined as the time interval from the initiation of treatment to disease progression or death. All values are reported as HR and 95% CI. First, we pooled the PFS results of total patients in 19 studies for direct comparison, and the results are summarized in the forest plot (Figure 3(b)). An indirect comparison of the SUCRA results revealed that CDK4/6i + Ful was the most effective regimen (Figure 4(a) and (b)). As shown in Table 2, PFS benefit of CDK4/6i + Ful was more favorable than that of SERD (0.45 (0.10, 0.79)), CDK4/6i + ET (0.58 (0.06, 1.15)), Akti + Ful (0.51 (0.09, 0.93)), ADCs (0.51 (0.09, 0.93)), PI3Ki + Ful (0.62 (0.29, 0.97)), mTORi + Ful (0.84 (0.19, 1.50)), and HADCi + ET (0.74 (0.14, 1.37)) in the point-to-point comparison. In addition, ET was more favorable than mTORi + Ful (0.55 (0.06, 1.03)), HADCi + ET (0.44 (0.02, 0.88)). Subsequently, our results of subgroup analysis indicated that CDK4/6i + Ful was consistently the most effective treatment regimen in patients with visceral metastasis or ESR1 mutations (Supplemental Figure 2). Unfortunately, due to the inability to combine data, we were unable to conduct a network analysis in the subgroups of patients with PI3K mutations, CDK4/6i treatment duration >12 or ⩽12 months. Instead, we have summarized the PFS results for these subgroups, which are presented in Supplemental Tables 1 and 2.
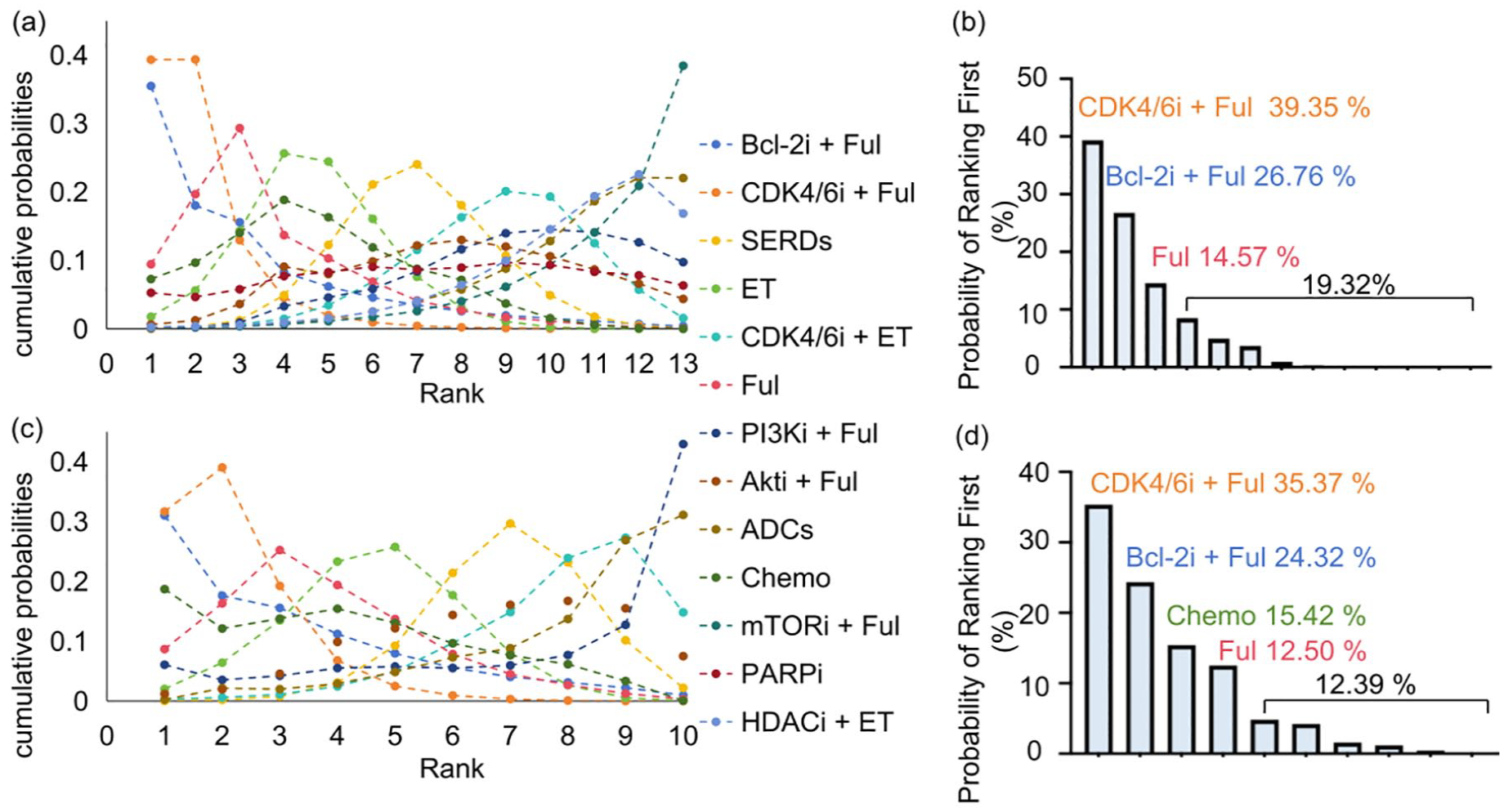
SUCRA ranking of treatment regimens based on PFS. (a) SUCRA ranking of each treatment regimen in the overall population. (b) The probability of each treatment regimen being ranked first in the overall population. (c) SUCRA ranking of each treatment regimen in CDK4/6i-pretreated population. (d) Probability of each treatment regimen being ranked first in the CDK4/6i-pretreated population.
Network meta-analysis for PFS in advanced HR+ breast cancer patients.

ADC, antibody–drug conjugates; Akti, Akt inhibitor; Bcl-2i, Bcl-2 inhibitor; ET, endocrine therapy; Ful, fulvestrant; HDACi, Histone Deacetylase inhibitor; HR+, hormone receptor-positive; mTOR, Mechanistic Target of Rapamycin; PARPi, Poly(ADP-Ribose) Polymerase inhibitor; PFS, progression-free survival; PI3K, phosphoinositide 3-kinase SERD, selective estrogen receptor degrader.
Bold entries in Table 2 represent statistically significant results.
Then, we performed an NMA of 14 studies which reported survival results in CDK4/6i-pretreated advanced HR+ breast cancer patients. A direct comparison of PFS was summarized in a forest plot (Figure 3(d)). The SUCRA ranking was consistent with that in the overall population, with CDK4/6i + Ful remaining the most effective regimen (Figure 4(c) and (d)). As shown in Table 3, in the point-to-point comparison, the PFS benefit of CDK4/6i + Ful was more favorable than that of SERDs (0.50 (0.11, 0.92)), CDK4/6i + ET (0.61 (0.02, 1.31)), Chemo was more favorable than that of ADCs (−0.49 (−0.84, −0.13)).
Network meta-analysis for PFS in CDK4/6i-pretreated advanced HR+ breast cancer patients.

AADC, antibody–drug conjugates; Akti, Akt inhibitor; Bcl-2i, Bcl-2 inhibitor; ET, endocrine therapy; Ful, fulvestrant; HDACi, Histone Deacetylase inhibitor; HR+, hormone receptor-positive; PFS, progression-free survival; PI3K, phosphoinositide 3-kinase; SERD, selective estrogen receptor degrader.
Bold entries in Table 3 represent statistically significant results.
Finally, we conducted an NMA focusing on other efficacy indexes in CDK4/6i-pretreated advanced HR+ breast cancer patients. OS was defined as the interval from the initiation of treatment to the date of death. Our OS network included four studies, with five treatment regimens. The SUCRA results showed that the combination of the Bcl-2i and Ful appeared to be the most favorable drug regimen for improving OS (Figure 5(a)). Forest plots for direct comparison of OS results are presented in Supplemental Figure 3. As shown in Supplemental Table 3, the point-to-point comparison showed that five regimens performed similarly in terms of OS benefit, with no significant difference. The ORR refers to the proportion of patients who achieved complete or partial response after treatment, and the CBR refers to the proportion of patients who achieved disease control or partial response after treatment. The ORR and CBR were reported in both the PACE, MORPHEUS HR+BC, and PALMIRA studies, and the CBR was also reported in VERONICA. Direct comparisons of the ORR and CBR were made via forest plots, and the results are summarized in Supplemental Figures 4 and 5, respectively. An indirect comparison of SUCRA results showed that SERD and CDK4/6i + ET were the most favorable drug regimens for ORR and CBR, respectively (Figure 5(b) and (c)). As shown in Supplemental Table 4, point-to-point comparisons revealed that SERD was superior to Ful (20.30 (2.35, 59.59)) and CDK4/6i + ET (19.5 (1.69, 58.79)). Point-to-point comparisons of CBR benefits demonstrated similar performance among regimens, with no significant differences (Supplemental Table 5). Supplemental Table 6 summarizes ongoing and completed umbrella clinical trials related to second-line treatments for HR+/HER2− advanced breast cancer.

SUCRA ranking of treatment regimens based on (left) OS, (middle) ORR, and (right) CBR.
Bias risk assessment and sensitivity analysis
Supplemental Figure 6 shows the bias risk assessment for included studies, and our study has no substantial bias. We performed a sensitivity analysis by excluding each study individually. The results demonstrated strong stability, as the pooled effect estimates remained consistent regardless of which study was excluded, with highly overlapping CIs (Supplemental Figure 7).
Discussion
CDK4/6i can arrest the cell cycle transition from G1 to S phase by selectively inhibiting CDK4/6 kinase, thereby inhibiting tumor proliferation. At the same time, CDK4/6i can inhibit the expression of the estrogen receptor (ER) signaling pathway, which can be synergistic with ET. Pivotal phase III clinical studies have confirmed the absence of cross-resistance to CDK4/6i. 35 Therefore, researchers have proposed that when patients progress on CDK4/6i-based therapy, switching to another CDK4/6i is one of the main ideas for second-line treatment. The MAINTAIN study showed that the mPFS was significantly improved with ribociclib (5.2 vs 2.7 m) in patients with HR+ advanced breast cancer after progression on CDK4/6i (87% palbociclib) + ET therapy. A multi-center retrospective study showed that patients with HR+ advanced breast cancer who progressed after palbociclib/ribociclib-containing regimens still benefited from the administration of abemaciclib alone or in combination with ET (e.g., Ful), with an mPFS of 5.4 months. 36 Second, keeping the original CDK4/6i unchanged and switching to other endocrine drugs is another idea. The PALMIRA study suggested that the mPFS was 4.9 months (control group: 3.6 months) for patients with HR+ advanced breast cancer who crossed over to palbociclib plus another ET as second-line treatment after progression to palbociclib + ET. 25 Another single-center retrospective study also suggested that the mPFS was 11.8 months with continued use of CDK4/6i + ET (changing the type of CDK4/6i or ET drug). 37 However, it remains unknown which endocrine agent is most effective when combined with CDK4/6i. Ful, a commonly used SERD in the clinic, can competitively bind to the ER and induce ER degradation to antagonize the effect of estrogen. The efficacy of Ful has been confirmed in China CONFIRM, 38 PrECOG0102, 39 and other clinical trials. The results of the PARSIFAL study showed no difference in the efficacy of CDK4/6i combined with Ful or letrozole. 40 This NMA compared the efficacy of various treatment regimens in both the overall population and CDK4/6i-pretreated HR+ advanced breast cancer, with PFS as the primary outcome. Our analysis consistently indicated that CDK4/6i + Ful represents the optimal treatment option. Further subgroup analyses demonstrated that CDK4/6i + Ful exhibited favorable efficacy both in patients with visceral metastasis or ESR1 mutations. Unfortunately, network analysis could not be performed in the PI3K mutation and CDK4/6i treatment duration subgroups. However, we observed that in the PI3K mutation population, the INAVO120 study confirmed that CDK4/6i + Ful showed a PFS benefit compared to Ful alone (0.43 (0.32–0.59)). In the post-MONARCH study, CDK4/6i + Ful also achieved significant PFS benefits in the duration of the prior CDK4/6i >12 months subgroup. This study confirmed the efficacy of CDK4/6i cross-line therapy in HR+/HER2− advanced breast cancer patients, even for high-risk groups such as those with symptomatic visceral metastasis, visceral crises, or ESR1 mutations. We look forward to further research to explore predictive biomarkers for CDK4/6i crossover therapy, aiming to optimize treatment strategies and improve patient survival and quality of life. Future clinical research should focus on elucidating individualized resistance mechanisms and developing precision treatment strategies.
It is worth noting that all of the studies included in this analysis involved advanced patients who had received second- or third-line treatments, with no efficacy analysis available for patients with more severe conditions. The DB-04 and TROPiCS-02 studies provided data on the PFS benefit of ADC in the treatment of HR+ advanced breast cancer in later lines, showing that compared with chemotherapy, T-Dxd and SG resulted in PFS gains of 10.1 and 5.5 months, respectively. ADC41,42 are targeted biologic agents that link cytotoxic drugs to monoclonal antibodies via a linker, allowing the retention of antitumor properties while minimizing off-target effects of small molecule chemotherapy, thus improving the efficacy-to-risk ratio of anticancer treatment. Our analysis included two studies comparing ADC with chemotherapy (TROPION-Breast01, DB-06), which suggested that ADCs also offer significant survival benefits for CDK4/6i-treated HR+ advanced breast cancer patients in the second-line setting. Therefore, as new ADCs continue to emerge and their combinations and sequential therapies are further explored, ADCs are expected to play an increasingly important role in breast cancer treatment, bringing new hope to patients at different treatment stages.
Moreover, the genetic background of patients plays a crucial role in influencing the efficacy of second-line treatments for breast cancer. For example, in the PI3K mutation population, the SOLAR-1 study showed a significant PFS benefit with the addition of alpelisib (0.65 (0.50–0.85)). The BOLERO-2 study indicated that for patients with confirmed PI3K/AKT/mTOR pathway mutations, everolimus may be considered for later-line therapy. In addition, both the OlympiAD and LUCY 43 trials demonstrated that earlier use of PARPi provides greater survival benefits for advanced breast cancer patients with gBRCA mutations. A prognostic analysis 44 presented at the 2024 ESMO conference on HR+ metastatic breast cancer patients with pathogenic mutations in BRCA1, BRCA2, or PALB2 showed that sequential treatment with CDK4/6 inhibitors followed by PARPi significantly extended OS in HR+/HER2− advanced breast cancer patients. These results highlight the critical role of biomarkers in optimizing treatment strategies for HR+/HER2− advanced breast cancer. As more biomarkers are discovered,45,46 future research should prioritize the identification and validation of predictive markers through genomic sequencing, liquid biopsy, and immunohistochemistry, aiming to facilitate personalized treatment and enhance patient survival and quality of life.
BCL-2i has been mainly studied in hematologic malignancies to inhibit tumor progression by inducing apoptosis. For solid tumors, Xu et al. 47 demonstrated that Venetoclax plus ET had acceptable efficacy and a manageable safety profile in ER+ breast cancer patients. VERONICA (a randomized, phase II study of HR+ breast cancer) showed that Venetoclax + Ful did not significantly improve PFS or the CBR compared with Ful alone. Our study revealed the opposite results: First, the PFS network analysis results showed that Bcl-2i + Ful is the second-ranked treatment option in both the overall and CDK4/6i-pretreated population. Second, the OS network analysis results demonstrated that Bcl-2i + Ful provides the greatest OS benefit for CDK4/6i-pretreated HR+ advanced breast cancer patients. We attribute this discrepancy to the ability of NMA to integrate more clinical data. Combining PFS and OS results from different studies and utilizing a larger sample size enhance the robustness and reliability of these findings, revealing the potential benefits of Bcl-2i on PFS and OS. Thus, we provide a preliminary basis for Bcl-2i in CDK4/6i-pretreated HR+ advanced breast cancer. Currently, PALVEN (phase Ib) is ongoing, and future research should focus on identifying effective synergistic treatment options or potential biological targets.
In addition to traditional ET, novel ER-targeting SERDs are also important candidates for CDK4/6i resistance. The EMERALD trial showed that patients with SERD had a significantly improved 12-month PFS rate compared with patients with traditional ET (9.4% vs 22.3%). The phase II SERA-2 study suggested a clinically significant improvement in PFS with camizestrant compared with Ful, with an acceptable safety profile. The phase I SERENA 1 study revealed the combination of camizestrant and abemaciclib achieved an ORR of 26.3% and a CBR of 66.7%, with well tolerated. The results of the NMA for ORR indicated that the novel SERD ranked as the preferred drug regimen. Therefore, this new SERD has potential therapeutic value after CDK4/6i resistance, and the ongoing phase III study of SERA-4 (NCT04711252) will add a greater amount of evidence to the treatment of HR+ advanced breast cancer with the new SERD.
Conclusion
Our analysis highlights that CDK4/6i + Ful is the most effective second-line treatment option for CDK4/6i-pretreated HR+ advanced breast cancer, particularly in patients with visceral metastases or ESR1 mutations. In addition, Bcl-2i + Ful and SERD may be potential second-line strategy options. Future research should focus on individualized drug resistance and precise treatment. We look forward to more large-scale phase III clinical trials providing stronger evidence-based support for this topic.
Limitations
Our NMA has several limitations. First, although our study includes multiple clinical trials, some data sources were not publicly available or did not provide detailed patient characteristics, which may lead to potential bias. Second, the lack of long-term OS data affects the comprehensive evaluation of treatment efficacy. Finally, since most trials did not conduct detailed analyses of patients’ specific genetic backgrounds, the analysis of treatment effects in certain subgroups requires further validation.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251336623 – Supplemental material for Options after progression on first-line CDK4/6 inhibitors in advanced breast cancer patients
Supplemental material, sj-docx-1-tam-10.1177_17588359251336623 for Options after progression on first-line CDK4/6 inhibitors in advanced breast cancer patients by Xiaomeng Jia, Kainan Wang, Xueqing Wang, Yiyang Luo, Huixin Liu, Zuowei Zhao, Jinbo Zhao and Man Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251336623 – Supplemental material for Options after progression on first-line CDK4/6 inhibitors in advanced breast cancer patients
Supplemental material, sj-docx-2-tam-10.1177_17588359251336623 for Options after progression on first-line CDK4/6 inhibitors in advanced breast cancer patients by Xiaomeng Jia, Kainan Wang, Xueqing Wang, Yiyang Luo, Huixin Liu, Zuowei Zhao, Jinbo Zhao and Man Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359251336623 – Supplemental material for Options after progression on first-line CDK4/6 inhibitors in advanced breast cancer patients
Supplemental material, sj-docx-3-tam-10.1177_17588359251336623 for Options after progression on first-line CDK4/6 inhibitors in advanced breast cancer patients by Xiaomeng Jia, Kainan Wang, Xueqing Wang, Yiyang Luo, Huixin Liu, Zuowei Zhao, Jinbo Zhao and Man Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-4-tam-10.1177_17588359251336623 – Supplemental material for Options after progression on first-line CDK4/6 inhibitors in advanced breast cancer patients
Supplemental material, sj-docx-4-tam-10.1177_17588359251336623 for Options after progression on first-line CDK4/6 inhibitors in advanced breast cancer patients by Xiaomeng Jia, Kainan Wang, Xueqing Wang, Yiyang Luo, Huixin Liu, Zuowei Zhao, Jinbo Zhao and Man Li in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
