Abstract
Background:
Cyclin-dependent kinase 4/6 (CDK4/6) inhibitors have become a cornerstone in the treatment of HR+/HER2- advanced breast cancer. While their efficacy is well-established, emerging reports of nephrotoxicity warrant further investigation into its incidence, risk factors, and potential impact on survival outcomes.
Objectives:
This study aimed to evaluate the incidence and risk factors for nephrotoxicity in patients receiving CDK4/6 inhibitors (palbociclib or ribociclib) and to analyze its association with progression-free survival (PFS) and overall survival (OS).
Design:
This was a single-center, retrospective cohort study.
Methods:
We reviewed the medical records of 120 patients with advanced breast cancer treated with palbociclib or ribociclib between October 2018 and July 2024. Nephrotoxicity was defined as a ⩾20% decline in creatinine clearance (CKD-EPI 2021) from baseline. Statistical analyses included descriptive statistics, chi-square tests, t-tests, Kaplan–Meier survival analysis, and Cox regression models.
Results:
Nephrotoxicity occurred in 28 patients (23.3%). Older age (⩾65 years) and higher baseline urea and creatinine levels were significant risk factors (p < 0.001). Paradoxically, patients who developed nephrotoxicity showed a trend toward better survival outcomes: median PFS was 30 months versus 20 months (p = 0.188), and the 3-year OS rate was 77.9% versus 63.8% (p = 0.801), though these differences were not statistically significant. In multivariate Cox analysis, the development of nephrotoxicity showed a trend toward a 71% reduction in mortality risk (HR = 0.293, p = 0.078), but it was not statistically significant.
Conclusion:
Nephrotoxicity is relatively common in patients treated with CDK4/6 inhibitors, particularly in older individuals and those with elevated baseline renal parameters. Contrary to conventional expectations, its occurrence may be associated with a trend toward improved survival, possibly reflecting higher drug exposure or effective target inhibition. These findings highlight the need for careful renal monitoring and suggest that nephrotoxicity could serve as a potential surrogate marker for treatment efficacy, warranting validation in larger prospective studies.
Introduction
Breast cancer is the most common cancer in women and approximately 60% of patients have localized disease at the time of diagnosis, while 6%–10% are diagnosed at the metastatic stage. While 5-year survival is 100% in localized disease, this rate decreases to 30% in metastatic disease. 1 Although clinical trials are ongoing for many drugs for metastatic disease, CDK 4/6 inhibitors have been shown to significantly improve survival in metastatic HR+ (hormone receptor positive) HER2- (human epidermal growth factor receptor 2) patients. CDKs play a critical role in the cell cycle. CDKs ensure the transition of the cell from G1 to S phase by hyperphosphorylation of the retinoblastoma gene product. Overactivation of CDKs and the resulting dysregulation of the cell cycle and uncontrolled cell proliferation play an important role in the pathogenesis of breast cancer. CDK 4/6 inhibitors also have an effect by preventing the transition of the tumor cell from G1 to S phase. Clinical studies have shown that these drugs significantly improve progression-free survival (PFS) and overall survival (OS) in HR+ HER2-patients.2,3
Since they are recommended as a first-line treatment in metastatic HR+ HER2-patients in current guidelines, they are the most commonly used drugs in current practice. Although generally well tolerated, CDK 4/6 inhibitors can cause a wide spectrum of side effects. In the PALOMA-2 and PALOMA-3 studies, neutropenia, leukopenia and increased risk of infection were observed in patients receiving palbociclib, while in the MONALEESA-2 study, in addition to these, increased liver enzymes, hypertension, QTC prolongation and diarrhea were observed in patients receiving ribociclib. In addition to hematologic side effects, gastrointestinal side effects such as diarrhea, nausea, and vomiting were observed more frequently than other CDK 4/6 inhibitors in clinical trials of abemaciclib. In the literature, there are studies showing that the use of CDK 4/6 inhibitors is associated with an increase in serum creatinine and a decrease in glomerular filtration rate. Although the frequency of this condition is not clearly known in phase III trials of palbociclib and ribociclib, it was observed in 25% of patients receiving abemaciclib.4,5 Primarily, it has been suggested that these drugs affect creatinine secretion in the proximal tubule and accordingly cause a 20-30% decrease in eGFR (estimated glomerular filtration rate) without causing actual acute kidney injury.6–8 However, some recent studies have shown that they may also cause intrinsic acute kidney injury. 9
Since breast cancer patients are usually diagnosed at middle-aged and advanced age and their glomerular filtration capacity may be low, it is important to evaluate the risk of CDK 4/6 inhibitor-induced nephrotoxicity in these patients and the factors that may lead to it in order to follow-up and treat the patient effectively.
The aim of our study was to determine the risk of developing nephrotoxicity secondary to CDK 4/6 inhibitors and which factors may play a role in this process and to evaluate the effect of this situation on the treatment response and survival of the patients.
Materıals and methods
Study design and patients
This retrospective study included women with HR+ HER2- advanced breast cancer who were admitted to the Medical Oncology Outpatient Clinic of Manisa Celal Bayar University Hafsa Sultan Hospital and received at least four cycles of palbociclib or ribociclib treatment between October 2018 and July 2024. Patient demographic data, clinical, and treatment-related characteristics were collected from hospital records.
Nephrotoxicity was defined as a ⩾20% reduction in estimated glomerular filtration rate (eGFR) from baseline, calculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) 2021 equation, at any point during the treatment period. The CKD-EPI 2021 formula was selected for its superior accuracy in estimating GFR compared to older formulas, particularly in patients with near-normal renal function, and its widespread recommendation by current nephrology guidelines. To mitigate confounding factors, patients were screened for the initiation of concomitant nephrotoxic medications during the study timeline. Those who commenced such drugs and subsequently exhibited a significant decline in eGFR were excluded from the final analysis to enhance the specificity of attributing renal function changes to the CDK4/6 inhibitors.
Treatment efficacy was assessed at 12-week intervals with 18F-fluorodeoxyglucose positron emission tomography–computed tomography (18F-FDG PET-CT) or computed tomography (CT). Tumor response was categorized according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. Definitions were applied as follows: complete response (CR) indicated the disappearance of all target lesions; partial response (PR) was defined as a reduction of at least 30% in the sum of target lesion diameters; and progressive disease (PD) was characterized by a minimum 20% increase in the sum of diameters or the emergence of new metastatic lesions. Cases that did not qualify as CR, PR, or PD were classified as stable disease (SD).
Overall survival (OS) was defined as the time from the start of treatment to the date of death from any cause. Progression-free survival (PFS) was measured from the date of treatment initiation to the date of the first documented disease progression or the date of the visit at which progression was detected.
The reporting of this study conforms to the STROBE statement. 10 The completed checklist is provided as a Supplemental File.
Statistical analysis
SPSS 15.0 for Windows program was used for statistical analysis. Descriptive statistics were given as number and percentage for categorical variables and median, minimum and maximum for numerical variables. The proportions in the groups were compared with the chi-square test. Comparisons of numerical variables in the groups were made with Student t test when the normal distribution condition was met and Mann–Whitney U test when the condition was not met. Survival rates were analyzed by Kaplan–Meier Analysis and risk factors were analyzed by Cox Regression Analysis. Alpha significance level was accepted as p < 0.05.
Results
The study included 120 patients. The median age at diagnosis was 53.9 years (26–83), while the median age at the start of treatment was 57.3 years (31–83). Forty-five patients received ribociclib (37.5%) and 75 patients received palbociclib (62.5%). Nephrotoxicity developed in 28 patients (23.3%). The median age of patients who developed nephrotoxicity was higher than those who did not. The proportion of patients over 65 years of age was 46% in those who developed nephrotoxicity compared to 21% in those who did not, and this rate was statistically significant (p = 0.01). Other demographic and clinical characteristics of the patients are given in Table 1 and were similar to each other.
Demographic and clinical characteristics of the patients.

Chi-square test, *Student’s t-test, **Mann–Whitney U test.
Statistically significant p-values are shown in bold.
BMI, body mass index; CAD, coronary artery disease; CR, complete response; DM, diabetes mellitus; ECOG PS, Eastern Cooperative Oncology Group Performance score; HT, hypertension; Med, median; PD, progressive disease; PR, partial response; SD, stable disease; SDe, standard deviation.
When the renal functions of the patients at the beginning of treatment were analyzed, the median serum urea and creatinine levels of the patients who developed nephrotoxicity were higher, while their eGFR was statistically significantly lower than the patients who did not develop nephrotoxicity (Table 2).
Renal function parameters of the patients before treatment.

eGFR, estimated glomerular filtration rate; med, median.
Statistically significant p-values are shown in bold.
PFS was a median of 24 months in the general population. Although not statistically significant, PFS was 10 months longer in those who developed nephrotoxicity (median 30 months vs 20 months, respectively, p = 0.188). 3-year PFS was also higher in those who developed nephrotoxicity (46.9% vs 32.5%, respectively) (Figure 1).

Progression-free survival.
When patients were compared in terms of overall survival, the median OS was not reached, and the 3-year overall survival rate was higher in those who developed nephrotoxicity, but this was not statistically significant. (77.9% vs 63.8%, p = 0.80, respectively; Figure 2).
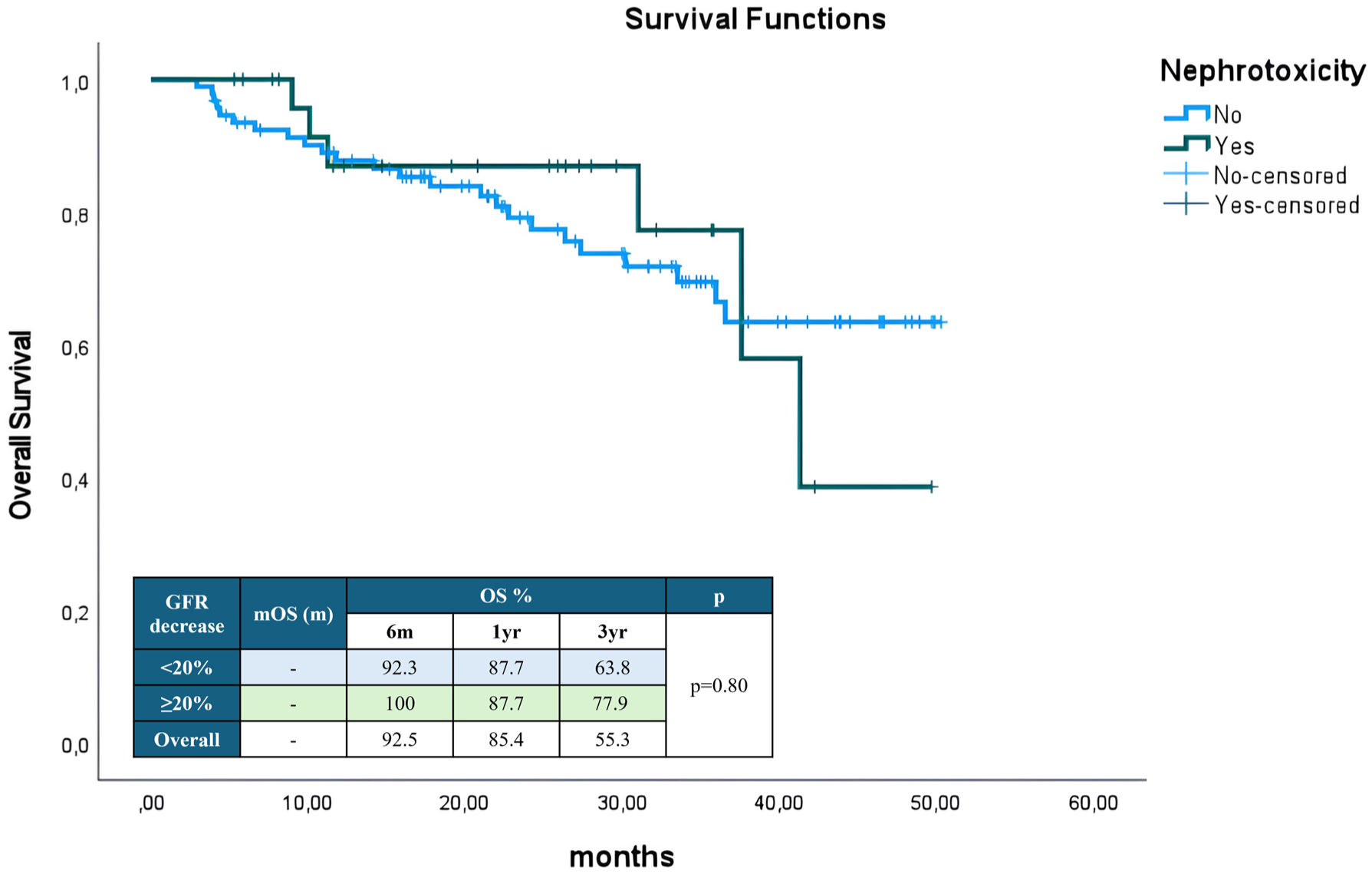
Overall survival.
When the factors affecting survival were analyzed by Cox regression analysis, the risk of death was lower in recurrent patients than in de novo metastatic patients in univariate analysis, this situation lost its statistical significance in multivariate analysis (Table 3). The presence of coronary artery disease increased the mortality risk 9.6-fold, and DM was associated with a reduced risk of death, contrary to expectations, but this should be interpreted with caution. In addition, the risk of mortality was 4.4 times lower in patients receiving ribociclib than in those receiving palbociclib, and a 3.2-fold risk reduction was observed in patients receiving fulvestrant therapy compared to letrozole. In patients who developed nephrotoxicity, there was an approximately 3-fold risk reduction in the risk of death, which did not reach statistical significance but showed a positive trend.
Risk factors affecting overall survival (Cox regression analysis).

BMI, body mass index; BUN, blood urea nitrogen; CAD, coronary artery disease; DM, diabetes mellitus; HT, hypertension; OR, Odds ratio.
Statistically significant p-values are shown in bold.
Discussion
CDK 4/6 inhibitors offer advantages in both PFS and OS (ribociclib, abemaciclib) in HR+/HER2- advanced breast cancer; therefore, their use in the first-line setting is recommended with a high level of recommendation by international guidelines.11,12 Recent clinical studies have also demonstrated that the use of CDK 4/6 inhibitors for a specific duration during the adjuvant period in patients with breast cancer who have undergone surgery and are at high risk of recurrence provides an event-free survival advantage, and their use has been approved in these patients.13,14 Therefore, CDK 4/6 inhibitors are frequently used treatments in breast cancer patients, and the side effects that develop secondary to these treatments must also be evaluated with real-life data.
Our study retrospectively evaluated the incidence of nephrotoxicity, risk factors, and survival effects in 120 advanced breast cancer patients treated with CDK4/6 inhibitors. Although this incidence varies depending on the definition of kidney damage and the specific CDK4/6 inhibitor used, the findings show that the risk of nephrotoxic side effects is increased in HR+/HER2- breast cancer patients treated with CDK 4/6 inhibitors, both in clinical trials and in real-world data. In a meta-analysis of 17 clinical trials involving 19,638 patients, the rate of nephrotoxicity of all grades secondary to CDK 4/6 inhibitors (abemaciclib, ribociclib, palbociclib, and dalpiciclib) was 9.73%. 15 This rate was 3.14% in the control group, 8.85% in those receiving ribociclib, and 7.58% in those receiving palbociclib. In a case series of 32 patients receiving ribociclib, grade 1 nephrotoxicity was observed in 28% of patients. 16 Similarly, in another study involving 474 patients, the rate of early decline in eGFR by more than 20% was 61% in the abemaciclib arm, 22% in the palbociclib arm, and 20% in the ribociclib arm. 17 In our study, nephrotoxicity was defined as a ⩾20% decrease in creatinine clearance and occurred in 23.3% of patients, similar to the literature.
Our results show that advanced age and higher baseline urea and creatinine levels are important risk factors for nephrotoxicity, which is consistent with the findings of Keskinkılıç et al., who reported higher risk in patients with elevated renal parameters prior to treatment. 18 The strong association with age highlights the importance of careful renal monitoring in elderly adults receiving CDK4/6 inhibitors, as age-related decline in renal function may exacerbate drug-induced nephrotoxicity.
The most intriguing finding of our study is the paradoxical relationship between nephrotoxicity and the trend toward improved survival outcomes. The median PFS of patients who developed nephrotoxicity was 10 months longer than those who did not (30 vs 20 months), and their 3-year OS was higher (77.9% vs 63.8%), although these differences were not statistically significant despite the positive trend. In multivariate Cox regression analysis, although there was a 70.7% reduction trend in mortality risk in patients who developed nephrotoxicity, this was not statistically significant (HR = 0.293, p = 0.078). These findings challenge the traditional view that drug-induced kidney injury is an unfavorable prognostic factor. A possible explanation for this finding is that nephrotoxicity may serve as a surrogate marker for effective drug exposure. It is known that CDK4/6 inhibitors, particularly abemaciclib, inhibit renal tubular creatinine secretion without causing actual glomerular damage, leading to a reversible increase in serum creatinine and a decrease in eGFR.6–8 This “pseudo-nephrotoxicity” may reflect higher drug levels or longer-term target inhibition, which may be associated with improved antitumor efficacy. Targeted side effects have also been shown to be associated with improved treatment response in tyrosine kinase inhibitors. 19
However, it is important to note that recent studies have also reported cases of intrinsic acute kidney injury associated with CDK4/6 inhibitors, suggesting that not all declines in kidney function are benign. 9 Therefore, it is important to carefully distinguish between functional eGFR changes and structural kidney damage in clinical practice.
Our study has some limitations, including its retrospective nature, relatively small sample size, and single-center experience. However, our study goes beyond merely reporting the frequency of a side effect and presents an innovative and thought-provoking paradox that may influence clinical decision-making processes.
Conclusion
CDK4/6 inhibitors are widely used as first-line therapy in patients with HR+/HER2- advanced breast cancer. Our study found that patients over 65 years of age had a higher risk of developing nephrotoxicity. Those with elevated pretreatment urea and creatinine levels also had an increased risk of nephrotoxicity. While 3-year survival analysis showed higher PFS and OS in patients who developed nephrotoxicity, this difference was not statistically significant. However, our study highlights the need to examine the relationship between nephrotoxicity and survival in larger patient populations.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251411133 – Supplemental material for Nephrotoxicity secondary to CDK 4/6 inhibitors in advanced breast cancer patients and its impact on survival
Supplemental material, sj-docx-1-tam-10.1177_17588359251411133 for Nephrotoxicity secondary to CDK 4/6 inhibitors in advanced breast cancer patients and its impact on survival by Tugay Avci, Ahmet Anil Altunbas, Mustafa Sahbazlar, Ferhat Ekinci and Atike Pinar Erdogan in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
