Abstract
Immune checkpoint inhibitors (ICIs) deeply changed the treatment landscape of breast cancer (BC). In particular, anti-programmed-death (ligand) 1 antibodies were approved for the treatment of triple-negative breast cancer (TNBC), both in first line for metastatic disease and in neoadjuvant setting, on the basis of a demonstrated improvement of the survival outcomes. In light of these results, current clinical trials aim at improving this benefit investigating novel combinations and strategies, at exploring the role of ICIs beyond TNBC, and at better selecting the patients in order to spare non-responders from avoidable toxicities. This narrative review aims at summarizing and discussing the evolving landscape of immunotherapeutic treatments for BC, highlighting the current challenges and the future perspectives.
Keywords
Introduction
Breast cancer (BC) is the most common cancer in women and represents the leading cause of death from all cancers.1,2 BC is traditionally classified according to the expression of hormone receptors (HRs) and of human epidermal growth factor receptor 2 (HER2) in three subtypes: HR-positive (HR+)/HER2-negative (HER2−) BC, HER2+ BC, and HR−/HER2− or triple-negative breast cancer (TNBC), which account for approximately 70%, 15–20% and 10–15% of all BC diagnoses, respectively. 3
immuno-oncology (IO) drugs and in particular immune checkpoint inhibitors (ICIs) have revolutionized the treatment landscape of several cancer types, including TNBC, despite BC has been for long time considered immunogenically quiescent, and less likely to derive benefit from IO approaches. 4 For example, the tumor mutational burden (TMB) in BC is on average lower than in other tumor types (e.g. melanoma and lung carcinoma) and varies by subtype, with HER2+, and triple-negative BC having higher burden than HR+ tumors: TMB has been correlated with a higher chance of immunogenicity, for a potential likelihood to mount an immunogenic, tumor-specific neoantigen restricted, immune response. Similarly, enrichment in tumor-infiltrating lymphocyte (TIL) appears higher in HER2+ and TNBC, when compared with HR+ BC and their expression is positively associated with improved prognosis.
TNBC is considered the most aggressive but immunogenic subtype: this cancer type is characterized by higher levels of programmed death-ligand 1 (PD-L1) expression on both tumor and immune cells, and non-synonymous mutations, which give rise to tumor-specific neoantigens.5–11 ICIs, in particular anti-programmed-death (ligand) 1 (PD-[L]1) antibodies, are currently only approved for patients with TNBC, both in first line for metastatic disease and in neoadjuvant setting, on the basis of a demonstrated clinical benefit in terms of overall survival (OS) and event-free survival (EFS), respectively.12–16
In light of the accumulating evidence of a role of IO in BC treatments, current clinical trials aim at improving IO benefit investigating novel compounds and strategies, for exploring the role of IO in BC in other populations and disease subtypes, and at better selecting the patients to spare non-responders from toxicities.
IO agents in metastatic TNBC
A lesson from previous clinical trials: first-line chemotherapy and immunotherapy is effective in biomarker-selected TNBC
Prior clinical trials enrolling patients with locally advanced and inoperable or metastatic TNBC highlighted that ICIs work in first-line setting, in combination with chemotherapy and in PD-L1+ patients (Table 1). Attempt to develop IO in biomarker-unselected populations resulted in no added benefit, and the use of IO agents in the second and later lines showed poor general activity of IO.
Trials with results enrolling patients with TNBC and evaluating ICI activity or efficacy as primary endpoint.

Tested hierarchically first in the PD-L1-positive [immune cell expression 1%, VENTANA PD-L1 (SP142) assay] population, and then in the ITT population.
AC, doxorubicin + cyclophosphamide; ACT, paclitaxel QW x C12 + (anthracycline + cyclophosphamide) Q3W x C4; ADC, antibody–drug conjugate; AE, adverse events; AUC, area under curve; BID, twice a day; C, cycle; CNS, central nervous system; CPS, combined positive score; CR, complete response; CT, chemotherapy; ctDNA, circulating tumor DNA; D, day; DCR, disease control rate; DFI, disease-free interval; DFS, disease-free survival; DLT, dose limiting toxicity; EC, epirubicin + cyclophosphamide; EFS, event-free survival; ET, endocrine therapy; FEC, 5-fluorouracil + epirubicin + cyclophosphamide; G, grade; gBRCA1/2m(ut), germline BRCA1/2 mutated; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; ICI, immune checkpoint inhibitor; IO, immuno-oncology; ITT, intention to treat; iv, intravenously; L, line; mo, months; (m)BC, (metastatic) breast cancer; (m)DOR, (median) duration of response; (m)OS, median overall survival; (m)PFS, (median) progression-free survival; n, number; NA, not available; NACT, neoadjuvant chemotherapy; ORR, objective response rate; P, pembrolizumab; pCR, pathological complete response; PD, progression of disease; PD-(L)1, programmed death-(ligand) 1; Ph, phase; PLD, peghilated lyposomial doxorubicin; PO, per os; PR, partial response; pre-op, preoperative; QnW/M, every n weeks/months; RCB, residual cancer burden; RT, radiation therapy; SABR, stereotactic body radiation therapy; T, taxane; TAM, tumor-associated macrophage; TCb, paclitaxel + carboplatin; T-DXd, trastuzumab deructecan; TIL, tumor-infiltrating lymphocyte; TKI, tyrosine kinase inhibitor; (e/m)TNBC, (early/metastatic) triple-negative breast cancer; TRAE, treatment-related adverse event; TT, targeted therapy; W, week.
Monotherapy with anti-PD-1/PD-L1 antibodies showed a limited antitumor activity [objective response rate (ORR) of 5–20%] and provided a small clinical benefit [median progression-free survival (mPFS) of 1–2 months], which was strongly influenced by the number of previous lines of therapy for metastatic disease and by the PD-L1 expression.17–23 In other words, a monotherapy approach is limited by the high rate of primary immune resistance (i.e. 60–85% of patients experience an early progression within the first 2–3 months) undermined by a demonstrated immunosuppressing milieu that occur during metastatic progression of BC. 11
The development of IO strategies for BC has then moved toward a combination approach with ICIs, to overcome primary and acquired immune resistance and to provide a major clinical benefit. Chemotherapy represents an ideal partner because of its immunomodulatory properties yielding immunogenic cell death and promoting antigen presentation11,24; in fact, both pembrolizumab plus physician’s choice chemotherapy (KEYNOTE-355) and atezolizumab plus nab-paclitaxel (Impassion130) showed to improve survival in patients with PD-L1-positive TNBC in first-line setting, soon becoming a new standard of care.13,14,16 PFS and OS were the co-primary endpoints of both trials, but, as far as OS, the Impassion130 failed to demonstrate a statistically significant benefit in the intention-to-treat (ITT) population and, according to the hierarchic design, the OS was not tested in the PD-L1+ population; anyway, an exploratory analysis in this subgroup showed a clinically significant benefit [mOS: 25.4 versus 17.9 months; hazard ratio (HR): 0.67, 95% CI: 0.53–0.86, median follow-up: 19 months]. 25 Instead, on the basis of a different statistical plan, in the KEYNOTE-355 the OS was tested first in PD-L1+ patients [with combined positive score (CPS) ⩾ 10 and ⩾1] and second in the ITT only if OS in the CPS ⩾ 1 subgroup was met; after a median follow-up of 44 months, a statistically significant OS benefit was assessed only in the CPS⩾10 subgroup (mOS: 23 versus 16 months; HR: 0.73, 95% CI: 0.55–0.95, p = 0.0185).
In contrast with these results, the phase III IMpassion131 trial investigating the combination of atezolizumab and paclitaxel failed to demonstrate a PFS benefit in untreated PD-L1-positive population. 26 This difference may be due to many factors, for example: the previous administration of taxanes in early setting (50% of patients enrolled), the lesser immunomodulatory properties of paclitaxel (perhaps dampened by steroid premedication), and patient heterogeneity.
Novel strategies for improving the clinical benefit of IO agents in BC
The next step to the initial demonstration of a benefit in the first-line setting with IO combined to chemotherapy aimed at potentiating the amplitude and the duration of the clinical benefit and to identify IO therapies for PD-L1-negative BCs. The deeper characterization of the tumor microenvironment (TME) of BC, the comprehension of resistance mechanisms to ICIs, and the investigation of novel ICI combination strategies, also with other IO agents have driven the formulation of innovative approaches.
In detail, resistance to ICIs in BC is strongly related to the intrinsic immune-phenotype of the tumor tissue and is characterized by poorer outcomes 27 ; the emerging resistance mechanisms comprise tumor-specific alterations, loss of tumor-specific antigens, and TME reshaping driven by extrinsic immune factors. 28 These mechanisms may be bypassed by facilitating the trafficking of the expanded cytotoxic cells into the tumor mass, improving the antigen presentation, or decreasing the inhibitory functions of the components of TME, such as tumor-associated macrophages (TAMs), regulatory T and B cells, or myeloid-derived suppressor cells, and also decreasing the activity of inhibitory cytokines. 29 Therefore, many combination strategies are under investigation to overcome these resistances and ultimately maximize ICI benefit. In particular, the ‘druggability’ of novel immune checkpoints, such as lymphocyte-activation gene 3 (LAG3) and T-cell immunoglobulin and mucin domain 3 (TIM3), is currently being explored at preclinical and early-phase clinical studies for patients with BC, with early signs of promising antitumor activity 30 (Table 2). Pending these data, we do not have robust evidences about biomarkers of ICI resistance.
Ongoing clinical trials investigating ICI combinations in mTNBC.

AC, doxorubicin + cyclophosphamide; ACT, paclitaxel QW x C12 + (anthracycline + cyclophosphamide) Q3W × C4; ADC, antibody–drug conjugate; AE, adverse events; AUC, area under curve; BID, twice a day; C, cycle; CNS, central nervous system; CPS, combined positive score; CR, complete response; CT, chemotherapy; ctDNA, circulating tumor DNA; D, day; DCR, disease control rate; DFI, disease-free interval; DFS, disease-free survival; DLT, dose limiting toxicity; EC, epirubicin + cyclophosphamide; EFS, event-free survival; ET, endocrine therapy; FEC, 5-fluorouracil + epirubicin + cyclophosphamide; G, grade; gBRCA1/2m(ut), germline BRCA1/2 mutated; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; ICI, immune checkpoint inhibitor; IO, immuno-oncology; ITT, intention to treat; iv, intravenously; L, line; (m)BC, (metastatic) breast cancer; (m)DOR, (median) duration of response; mo, months; n, number; NA, not available; NACT, neoadjuvant chemotherapy; ORR, objective response rate; (m)OS, median overall survival; P, pembrolizumab; pCR, pathological complete response; PD, progression of disease; PD-(L)1, programmed death-(ligand) 1; (m)PFS, (median) progression-free survival; Ph, phase; PLD, peghilated lyposomial doxorubicin; PO, per os; PR, partial response; pre-op, preoperative; QnW/M, every n weeks/months; RCB, residual cancer burden; RT, radiation therapy; SABR, stereotactic body radiation therapy; T, taxane; TAM, tumor-associated macrophage; TCb, paclitaxel + carboplatin; T-DXd, trastuzumab deructecan; TIL, tumor-infiltrating lymphocyte; TKI, tyrosine kinase inhibitor; (e/m)TNBC, (early/metastatic) triple-negative breast cancer; TRAE, treatment-related adverse event; TT, targeted therapy; W, week.
On the basis of a strong preclinical rational and the clinical benefit demonstrated as monotherapy, combinations of antibody–drug conjugates (ADCs) with ICIs represent a promising strategy.31,32 In particular, in two phase Ib/II trials enrolling patients with TNBC and HER2− low TNBC, respectively, the combinations of both ladiratuzumab vedotin [(LV), an antizinc transporter LIV-1 ADC] plus pembrolizumab and trastuzumab deruxtecan [(T-Dxd), an anti-HER2 ADC] plus durvalumab showed an acceptable safety profile to continue the drug development and promising ORRs (54% and 67%, respectively).33,34 Many phase II clinical trials evaluating this strategy are ongoing; in particular, in first-line setting, the SACI-IO TNBC trial (NCT04468061) is assessing the activity of pembrolizumab plus sacituzumab govitecan, an antitrophoblast-antigen-2 (Trop-2) ADC, which has demonstrated to provide an OS benefit as monotherapy in pretreated patients with metastatic TNBC. 31
The associations of ICIs and targeted therapy (TT) may potentiate the efficacy of ICIs too, but preliminary results were scarce. Of note, both cobimetinib, a MEK inhibitor, and entinostat, a class I-selective histone deacetylase (HDAC) inhibitor, failed to demonstrate a benefit in unselected patients with TNBC.35,36 Other targeted agents under investigation comprise: antiangiogenic tyrosine kinase inhibitors (TKI), like apatinib or famitinib; binimetinib, a MEK inhibitor; eganelisib, a PI3K-γ inhibitor. Moreover, the combination of ICIs and Poly-(ADP-ribose)-polymerase (PARP) inhibitors (PARPi) was evaluated in two single-arm phase II trials (TOPACIO and MEDIOLA),37,38 which showed results similar to those reported in clinical trials assessing PARPi monotherapy (OlympiAD and EMBRACA),39,40 suggesting that biomarkers are key to identify patients who will benefit most.
The use of tumor-directed treatments has been for long viewed as promising to enhance cancer immunogenicity by exposing tumor-derived antigens to the immune system. Locoregional treatments include radiation therapy, cryoablation and microwave ablation, among others; through different mechanisms of action, they determine local tumor destruction, neoantigen release, and a subsequent enhanced and tumor-specific immune response, which can also extend outside the primary tumor (i.e. abscopal effect). Therefore, locoregional treatments represent an ideal partner of ICIs, whose action requires a pre-existing immune ‘recognition’ of tumor-associated antigens.41–43 However, a clinical trial assessing the non-central nervous system disease control rate (DCR) after the administration of tremelimumab and brain radiotherapy to 20 patients with metastatic HER2− BC and brain metastasis did not meet its primary endpoint 44 ; anyway, enrolled patients were heavily pretreated and non-selected according to PD-L1 status. Data from ongoing clinical trials assessing this abscopal effect in a more favorable setting of disease are awaited.
Lastly, as far as IO therapies other than ICIs are concerned, chimeric antigen receptor (CAR)-T cells have emerged as a promising immunotherapeutic strategy in TNBC: this approach combines the antigen specificity of an antibody with the effector function of T cells and is under investigation in several phase I clinical trials. 45 Despite numerous antigens have been identified as potential targets (e.g. Trop2, GD2, ROR1, MUC1, EpCAM), the target selection represents the most relevant obstacle, to minimize on-target/off-tumor toxicities, as well as to reduce tumor escape via antigen loss and intrinsic heterogeneity.
Current challenges for immunotherapy in BC
The evolving landscape of immunotherapy for BC presents a multitude of issues to be further clarified by the ongoing and future clinical trials (Figure 1). First, the advent of promising but toxic partners such as ADCs requires the development of novel strategies to manage the safety, such as the maintenance with only ICIs following the response to few cycles of induction treatment with only ADCs; in this regard, for example, the phase II SAFIR02-BREAST IMMUNO trial randomized 199 patients with metastatic HER2− BC whose disease did not progress after six to eight cycles of first or second line chemotherapy to receive either durvalumab or maintenance chemotherapy. 22 Despite the trial failed to demonstrate a PFS (primary endpoint) and OS benefit in the ITT population, in an exploratory subgroup analysis, durvalumab improved OS in the 82 patients with TNBC.

Immunotherapy in BC: current issues.
Another important issue is represented by the lack of evidence of ICI efficacy in patients with early distant recurrence after neoadjuvant chemotherapy (NACT), excluded from KEYNOTE-355 and Impassion130, because a disease-free interval (DFI) of ⩾6 and ⩾12 months was respectively required. In this regard, the placebo-controlled randomized phase III IMpassion132 trial (NCT03371017) is assessing atezolizumab with first-line chemotherapy (capecitabine, mandatory in platinum-pretreated patients, or gemcitabine/carboplatin) for metastatic TNBC and a DFI of ⩽12 months. 46
With the advent of ICIs in early setting, an emerging question is represented by the efficacy of the retreatment with ICIs and chemotherapy as first-line therapy; clinical trials with a strong translational design to identify biomarkers of ICI resistance and guide the retreatment are needed.
Lastly, a deeper comprehension of immune TME to identify response predictive biomarkers represents the most urgent need to better select the patients beyond the PD-L1 expression. In fact, according to the ICI, the PD-L1 assessment is performed with different companion diagnostic assays and is therefore burdened by a lack of homogeneity. 47
Challenges of immunotherapy for early-stage TNBC
Trials investigating neoadjuvant immunotherapy
TNBC is diagnosed at stage II or III in more of 60% of cases48–50; nevertheless, because of its aggressive behavior, it is characterized by early recurrences (3-year distant recurrence rate of 30%–35%) and the poorest prognosis among BC subtypes (5-year estimate OS of 64% for stages I–III combined).51,52 In this regard, on the basis of positive results in advanced setting, ICIs moved to early TNBC setting, which represents the ideal context for ICI treatment, before the acquisition of multiple mechanisms of immune escape and when the host immune system is less compromised. 11
Emerging evidences confirmed these hypotheses: a trial-based meta-analysis of five randomized trials enrolling 1496 TNBC patients showed a statistically significant association between ICI addition and pathological complete response [(pCR), odds ratio (OR): 1.72, 95% CI: 1.22–2.42]. 53 Furthermore, the phase III randomized KEYNOTE-522 clinical trial randomizing 1174 patients with stage II–III TNBC to NACT with paclitaxel–carboplatin followed by doxorubicin–cyclophosphamide, with or without the addition of pembrolizumab, demonstrated a benefit in terms of EFS (primary endpoint; 3-year EFS: 84.5% versus 76.8%), regardless of PD-L1 expression (CPS ⩾ 1 or <1) (Table 1). As far as the safety profile of this five-drug combination, 77% and 73% of patients, respectively, from the experimental and the control arm, experienced a treatment-related adverse event of grade 3 or more; furthermore, the rate of immune-mediated adverse events of grade 3 or higher was of 13% and 1%. These results led to full approval for neoadjuvant pembrolizumab in combination with chemotherapy by the Food and Drug Administration for patients with high-risk TNBC. 54
In contrast with positive clinical trials (I-SPY2, Impassion031, KEYNOTE-522),12,15,55,56 the phase II GeparNuevo, randomizing patients to receive nab-paclitaxel followed by dose dense anthracyclines and cyclophosphamide with either durvalumab or placebo, failed to demonstrate a statistically significant improvement in the primary endpoint of pCR (53.4% versus 44.2%, OR: 1.45) but showed potential clinical benefit after 42 months of follow-up in a descriptive analysis, in terms of 3-year invasive disease-free survival (iDFS, 84.9% versus 76.9%; HR 0.54, 95% CI 0.27–1.09), 3-year distant disease-free survival (DDFS, 91.4% versus 79.5%; HR 0.37, 95% CI 0.15–0.87) and 3-year OS (95.1% versus 83.1%, HR 0.26, 95% CI 0.09–0.79).57,58 Unlike the other three similar positive trials, which enrolled only patients with stage II–III TNBC, 35% of patients from GeparNuevo had a TNBC in stage I and seemed to derive a lower benefit in terms of pCR from the addition of durvalumab. Although the study was formally negative for its primary endpoint, the significant difference found in iDFS represents an intriguing finding, suggesting that pCR may be not the sole driver of IO benefit in early TNBC, and that clinical outcomes may also be independent of pCR, especially when investigating IO agents.
Novel agent combinations in neoadjuvant setting
Many innovative IO combination strategies are under investigation with the aim of further improve outcomes of patients with early TNBC; they include: TT/ADC-ICI, other IO agents-ICI and locoregional treatment–ICI combinations, and cancer vaccine (CV)–chemotherapy combinations (Table 3). 59
Ongoing clinical trials investigating ICI combinations in early TNBC (neoadjuvant and post-neoadjuvant setting).
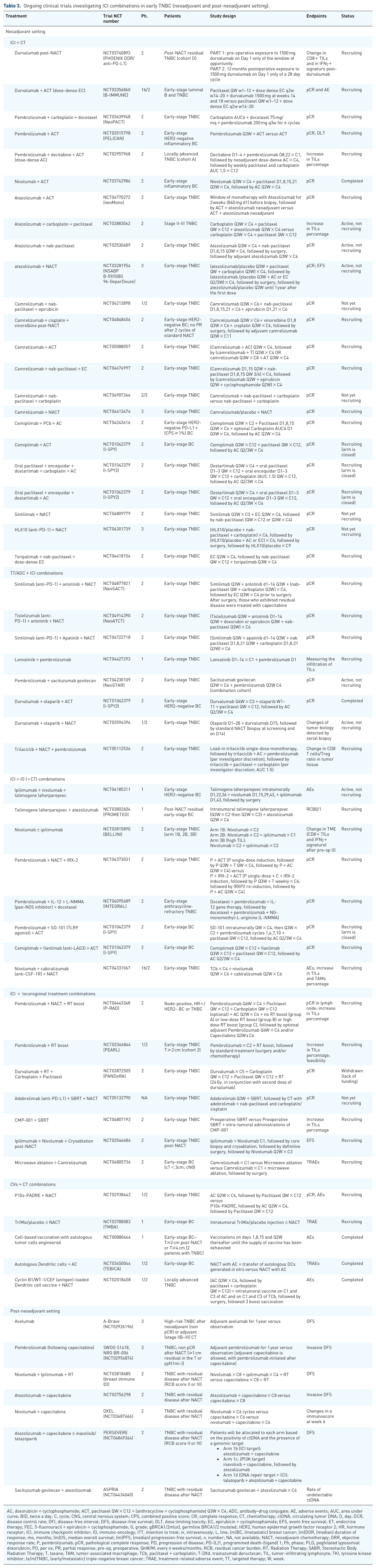
AC, doxorubicin + cyclophosphamide; ACT, paclitaxel QW × C12 + (anthracycline + cyclophosphamide) Q3W × C4; ADC, antibody–drug conjugate; AE, adverse events; AUC, area under curve; BID, twice a day; C, cycle; CNS, central nervous system; CPS, combined positive score; CR, complete response; CT, chemotherapy; ctDNA, circulating tumor DNA; D, day; DCR, disease control rate; DFI, disease-free interval; DFS, disease-free survival; DLT, dose limiting toxicity; EC, epirubicin + cyclophosphamide; EFS, event-free survival; ET, endocrine therapy; FEC, 5-fluorouracil + epirubicin + cyclophosphamide; G, grade; gBRCA1/2m(ut), germline BRCA1/2 mutated; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; ICI, immune checkpoint inhibitor; IO, immuno-oncology; ITT, intention to treat; iv, intravenously; L, line; (m)BC, (metastatic) breast cancer; (m)DOR, (median) duration of response; mo, months; (m)OS, median overall survival; (m)PFS, (median) progression-free survival; n, number; NA, not available; NACT, neoadjuvant chemotherapy; ORR, objective response rate; P, pembrolizumab; pCR, pathological complete response; PD, progression of disease; PD-(L)1, programmed death-(ligand) 1; Ph, phase; PLD, peghilated lyposomial doxorubicin; PO, per os; PR, partial response; pre-op, preoperative; QnW/M, every n weeks/months; RCB, residual cancer burden; RT, Radiation Therapy; SABR, Stereotactic Body Radiation Therapy; T, taxane; TAM, tumor-associated macrophage; TCb, paclitaxel + carboplatin; T-DXd, trastuzumab deructecan; TIL, tumor-infiltrating lymphocyte; TKI, tyrosine kinase inhibitor; (e/m)TNBC, (early/metastatic) triple-negative breast cancer; TRAE, treatment-related adverse event; TT, targeted therapy; W, week.
As it pertains TT and ADCs, on the basis of immunosuppressive properties showed by vascular endothelial growth factor (VEGF), many combinations of ICIs with antiangiogenic agents are under evaluation in clinical trials, including apatinib, a selective VEGFR2 inhibitor, and anlotinib and lenvatinib, two multi-target tyrosine kinase inhibitors (TKI) inhibiting VEGFR, FGFR, PDGFR, c-Kit, and Ret.60–62 Morover, the phase II NeoSTAR clinical trial (NCT04230109) has investigated the activity and safety of neoadjuvant sacituzumab govitecan plus pembrolizumab on the basis of a strong preclinical rational,63,64 reporting 30% ORR. In regard with PARPis, following the results of MEDIOLA trial, 38 the I-SPY2 study evaluated the combination of durvalumab and olaparib concurrent with weekly neoadjuvant paclitaxel, followed by AC regimen, in early-stage HER2-negative setting. 65 73 patients, whose 21 with TNBC, were enrolled in the experimental arm and 299 in the control arm (chemotherapy only); at the final efficacy analysis, the combination improved estimated pCR rates over control from 20% to 37% in HER2-negative cancers, and from 27% to 47% in TNBC. Lastly, TNBC has been considered a poor candidate for CDK4/6 inhibitors (CDK4/6i), because it holds the loss of retinoblastoma (Rb) protein in 50% of cases, critical for CDK4/6i-induced cell cycle arrest. 66 Nevertheless, the neutrophil-preserving CDK4/6i trilaciclib has shown an antitumor activity in TNBC preclinical models and has enhanced antitumor immune responses through T-cell effector stimulation and regulatory T-cell suppression,67–69 demonstrating also an intriguing and unexpected OS benefit in association with chemotherapy in an unpowered phase II trial.70,71
Combinations of IO agents are also under investigation in early TNBC; they comprise ipilimumab, 72 an antibody anticytotoxic t-lymphocyte antigen (CTLA)-4; fianlimab, an antibody anti-LAG-3 which enhances cytotoxic T-lymphocyte (CTL)-mediated tumor cell lysis 73 ; cabiralizumab, an antibody anticolony-stimulating factor 1 receptor (CSF-1R) which reduces immunosuppressive TAMs and promotes a proinflammatory TME, stimulating T-cell responses 74 ; IRX-2, a mixture of cytokines which increases the immune activation in TME 75 ; IL-12 and monomethylated L-arginine (L-NMMA), a pan-NOS-inhibitor which showed the ability to remodel the immune TME 76 ; SD-101, a toll-like receptor 9 (TLR9) agonist which promotes a T helper 1-like chemokine milieu in TME and induces an antitumor CD8+ T-cell response. 77
As stated, the combination of ICI with locoregional treatments is based on a strong preclinical rational and may represent an opportunity to de-escalate NACT and optimize its toxicity; in fact, in the phase Ib/II PEARL clinical trial, enrolling patients with early-stage TNBC, the administration of pembrolizumab and RT prior to physician’s choice NACT provided a 56% of pCR rate, overlapping results from control arm of KEYNOTE-522, despite only 1/3 of study participants received the same chemotherapy regimen. 78
Lastly, after almost two decades of limited benefit from clinical trial investigating CVs, the coronavirus disease 2019 pandemic boosted a significant technological advancement and many neoantigens are emerging as the preferred targets to develop therapeutic CVs.79,80 Moreover, in light of a deeper comprehension of immune escape mechanisms, clinical trials moved from metastatic disease, where the TME is more likely compromised by inhibitory mechanisms, to premalignant or adjuvant setting and investigated combination treatments (i.e. CV plus ICI). Examples of CVs under investigation in patients with TNBC comprise: P10s-PADRE, a peptide-based CVs contains a carbohydrate mimetic peptide P10s fused to the pan-HLA-DRe binding epitope (PADRE) peptide, with a CTL-stimulating activity, 81 TriMix, a cell-based CVs consisting of a mRNA encoding a mixture of three immune modulating molecules (TLR-4, CD40L, and CD70) with a T-cell stimulatory capacity.82,83
Post-neoadjuvant setting
For patients with early TNBC, NACT is preferred to adjuvant therapy in order to in vivo test the sensitivity to chemotherapy and to personalize treatments in post-neoadjuvant setting on the basis of pCR, which represents a valid patient-level prognosticator of better outcomes. 84 For patients with high-risk TNBC, for example those who do not experience pCR after NACT, the use of treatment intensification in the adjuvant setting has showed to portend survival benefits: specifically, capecitabine and Olaparib in patients without and with germline BRCA (gBRCA) pathogenetic mutations.85,86
Of interest, the KEYNOTE-522 scheduled included nine cycles of post-neoadjuvant pembrolizumab, regardless of the pCR, and neither concomitant olaparib nor capecitabine were admitted, because the trial was designed before the approval of these indications. 87
Therefore, there is an urgent need to incorporate these treatment options and to adapt them to the risk of recurrence. In particular, the role of ICI in post-neoadjuvant setting has to be clarified, both in ICI-naïve patients and in those who received neoadjuvant immunotherapy. 88
In this regard, many clinical trials are investigating the role of ICIs as alternative or in association (sequential or concomitant) to capecitabine in ICI-naïve patients with high-risk TNBC and residual disease after NACT (Table 3). Furthermore, ASPRIA trial is investigating another escalation option with sacituzumab govitecan plus atezolizumab or alone in patients with residual disease after NACT.
However, on the basis of safety profile assessed in other settings, the combination of post-neoadjuvant pembrolizumab plus capecitabine or olaparib, according to gBRCA status, may be considered in clinical trials for patients not reaching the pCR and at higher risk of recurrence. 88
Contrarily, the 3-year EFS rate of patients enrolled in KEYNOTE-522 who reached the pCR (94.4% versus 92.5%, HR: 0.73, 95% CI: 0.39–0.36) suggests the exploration of de-intensification strategies to spare post-neoadjuvant pembrolizumab and, consequently, clinical, psychological, and financial toxicities; this hypothesis is corroborated by GeparNuevo trial, which demonstrated an underpowered but significant OS benefit and did not include post-neoadjuvant durvalumab.15,58
Current issues
The introduction of ICIs for the treatment of patients with early TNBC raises many interesting issues warranting new translational studies and clinical trials59,89,90 (Figure 1).
First, the choice of the best backbone chemotherapy is not fully understood, in particular platinum salts demonstrated to provide an EFS benefit when added to standard NACT but GeparNuevo trial showed a survival benefit also without carboplatin58,91; therefore, the need for platinum in the presence of pembrolizumab has to be assessed. Furthermore, KEYNOTE-522 did not include dose-dense anthracyclines and cyclophosphamide (AC) regimen, which has demonstrated to be superior to standard AC 92 ; hence, also the role of dose dense AC in association to ICIs needs to be clarified.
As far as predictive biomarkers, pembrolizumab demonstrated to provide a clinical benefit regardless of PD-L1 status (CPS ⩾ 1 or <1); there is an urgent need to optimize the patient selection in order to develop de-escalation and escalation strategies for the upfront responders to ICI and non-responders, respectively. Also in this setting, PD-L1 assessment is burdened by a lack of homogeneity: in fact, three different companion diagnostic assays with different thresholds were used. Furthermore, the role of TILs is not fully understood because their high expression is predictive of response also to chemotherapy alone. 57 Other biomarkers predictive of ICI response in other tumor histology, but not yet evaluated and validated in early TNBC, are circulating tumor cells and circulating tumoral DNA (ctDNA), mismatch repair deficiency/microsatellite instability, CD274 amplification. 59 In particular, a recent meta-analysis has demonstrated that the detection of ctDNA, both at baseline and after completion of NACT, is significantly associated to worse recurrence-free survival [(RFS), HR 4.22, 95%CI: 1.29–13.82 and HR: 5.67, 95%CI: 2.73–11.75, respectively] and worse OS (HR 19.1, 95% CI: 6.9–53.04 and HR 4.00, 95% CI: 1.90–8.42, respectively). 93 This dynamic and circulating biomarker represents an interesting tool for monitoring tumor evolution, predicting treatment response and determining prognosis. Probably, the ideal prognostic and predictive immune biomarker will derive from the integration of different features into a unique immunogram and will require a dynamic assessment before, during and after the neoadjuvant treatment.
Another issue regards the role of neoadjuvant ICIs in patients with stage I TNBC; in fact these patients were not included in KEYNOTE-522 and currently cannot receive pembrolizumab. Anyway, they represent the 35% of patients enrolled in GeparNuevo trial, which demonstrate to provide a benefit in terms of iDFS regardless of the stage (stage 0 or I: HR for iDFS: 0.55, 95% CI: 0.09–3.31; stage IIA or higher: HR for iDFS: 0.51; 95% CI: 0.24–1.12). 58
Lastly, there is an urgent need for surrogate endpoints to optimize and accelerate the drug development. In fact, in early TNBC setting, the pCR has shown to be an adequate endpoint of response to NACT, being associated with EFS (HR 0.24, 95% CI 0.18–0.33) and OS (HR 0.16; 95%CI 0.11–0.25) only at patient level, so regardless of treatment group. 84 Instead, at a trial level (i.e. considering trial arms), an increase in pCR rate between treatment groups does not predict improvements in EFS and OS; therefore, pCR may not represent a perfect surrogate endpoint for clinical outcomes, maybe because it does not capture the whole effect of treatment upon the true endpoint: for example, pCR does not consider pathological partial responses [measured by residual cancer burden (RCB)] and the clearance of micrometastatic systemic disease, which could be measured with liquid biopsy.84,94,95 This complex scenario is further complicated by the advent of ICIs, whose kinetic of action is even less framed by a response endpoint such as pCR. 11 In fact, in both KEYNOTE-522 and GeparNuevo, survival outcomes improved to an extent greater than expected on the basis of pCR rate increases (by 7.5% and 9.2%, respectively) 11 ; furthermore, GerparNuevo showed an improvement in DFS, EFS, and OS (secondary endpoints), despite the primary endpoint (pCR) was not met. On the basis of these evidences, a solution may be the validation of new composite response endpoints (RCB, the clearance of ctDNA and/or the dynamic modification of tissue immune biomarkers), in order to test and eventually validate a response surrogate endpoint for long-term outcomes.
Immunotherapy in other subtypes beyond TNBC
After the demonstration of a clinical benefit in patients with TNBC, the next step was moving IO agents to other BC subtypes, namely HR+/HER2− BC and HER2+ BC (Tables 4 and 5). Furthermore, the finding of some cases of non-TNBCs expressing high level of TMB, TILs, and PD-L1 supported this expansion. 96
Results from clinical trials investigating ICIs in HR+/HER2− and HER2+ BC.

AC, doxorubicin + cyclophosphamide; ACT, paclitaxel QW × C12 + (anthracycline + cyclophosphamide) Q3W × C4; ADC, antibody–drug conjugate; AE, adverse events; AUC, area under curve; BID, twice a day; C, cycle; CNS, central nervous system; CPS, combined positive score; CR, complete response; CT, chemotherapy; ctDNA, circulating tumor DNA; D, day; DCR, disease control rate; DFI, disease-free interval; DFS, disease-free survival; DLT, dose limiting toxicity; EC, epirubicin + cyclophosphamide; EFS, event-free survival; ER, estrogen receptor; ET, endocrine therapy; FEC, 5-fluorouracil + epirubicin + cyclophosphamide; G, grade; gBRCA1/2m(ut), germline BRCA1/2 mutated; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; ICI, immune checkpoint inhibitor; IO, immuno-oncology; ITT, intention to treat; iv, intravenously; L, line; mo, months; (m)BC, (metastatic) breast cancer; (m)DOR, (median) duration of response; (m)OS, median overall survival; (m)PFS, (median) progression-free survival; n, number; NA, not available; NACT, neoadjuvant chemotherapy; NR, not reached; ORR, objective response rate; P, pembrolizumab; pCR, pathological complete response; PD, progression of disease; PD-(L)1, programmed death-(ligand) 1; Ph, phase; PLD, peghilated lyposomial doxorubicin; PO, per os; PR, partial response; pre-op, preoperative; QnW/M, every n weeks/months; RCB, residual cancer burden; RT, Radiation Therapy; SABR, Stereotactic Body Radiation Therapy; T, taxane; TAM, tumor-associated macrophage; TCb, paclitaxel + carboplatin; T-DXd, trastuzumab deructecan; TIL, tumor-infiltrating lymphocyte; TKI, tyrosine kinase inhibitor; (e/m)TNBC, (early/metastatic) triple-negative breast cancer; TRAE, treatment-related adverse event; TT, targeted therapy; W, week.
Ongoing clinical trials investigating IO agents in HR+/HER2− or HER2+ BC.

AC, doxorubicin + cyclophosphamide; ACT, paclitaxel QW x C12 + (anthracycline + cyclophosphamide) Q3W x C4; ADC, antibody–drug conjugate; AE, adverse events; AUC, area under curve; BID, twice a day; C, cycle; CNS, central nervous system; CPS, combined positive score; CR, complete response; CT, chemotherapy; ctDNA, circulating tumor DNA; D, day; DCR, disease control rate; DFI, disease-free interval; DFS, disease-free survival; DLT, dose limiting toxicity; EC, epirubicin + cyclophosphamide; EFS, event-free survival; ET, endocrine therapy; FEC, 5-fluorouracil + epirubicin + cyclophosphamide; G, grade; gBRCA1/2m(ut), germline BRCA1/2 mutated; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; ICI, immune checkpoint inhibitor; IO, immuno-oncology; ITT, intention to treat; iv, intravenously; L, line; mo, months; (m)BC, (metastatic) breast cancer; (m)DOR, (median) duration of response; (m)OS, median overall survival; (m)PFS, (median) progression-free survival; n, number; NA, not available; NACT, neoadjuvant chemotherapy; ORR, objective response rate; P, pembrolizumab; pCR, pathological complete response; PD, progression of disease; PD-(L)1, programmed death-(ligand) 1; Ph, phase; PLD, peghilated lyposomial doxorubicin; PO, per os; PR, partial response; pre-op, preoperative; QnW/M, every n weeks/months; RCB, residual cancer burden; RT, Radiation Therapy; SABR, Stereotactic Body Radiation Therapy; T, taxane; TAM, tumor-associated macrophage; TCb, paclitaxel + carboplatin; T-DXd, trastuzumab deructecan; TIL, tumor-infiltrating lymphocyte; TKI, tyrosine kinase inhibitor; (e/m)TNBC, (early/metastatic) triple-negative breast cancer; TRAE, treatment-related adverse event; TT, targeted therapy; W, week.
HR+/HER2− BC
HR+/HER2− BC is characterized by an immune suppressive TME, with low TIL infiltration, low HLA class I expression and abundant TAMs, which limit the antitumor immune activity. 97 In fact, only 6% of HR+ BC is characterized by high (⩾50%) TILs-infiltration and the prognostic role of TILs in this subtype is controversial, probably mirroring a disease that is less differentiated and more aggressive. 98 Anyway, on the basis of these limited signs of immune response, the activity and efficacy of IO agents was investigated also in this subtype, which is the most prevalent.
The first proof of concept phase I/II clinical trial demonstrated that in metastatic endocrine-resistant non-selected patients ICIs as monotherapy or in combination with endocrine therapy are associated with very limited ORRs of 0–3% and mPFS lower than 3 months 18,99,100; furthermore, the KEYNOTE-028 showed that neither the patient selection on the basis of PD-L1 (CPS ⩾ 1) provides clinically significant results (ORR 12%) 101 (Table 4).
As in TNBC, ICI–chemotherapy combination were then explored, but without any evidence of clinical benefit in terms of PFS in unselected patients22,102,103 (Table 4). For example, on the basis of preclinical data demonstrating an upregulation of immune-related genes in invasive lobular BC (ILBC), 104 the phase II GELATO trial enrolled patients with metastatic ILBC of any type, of which 18 had endocrine-resistant HR+ BC and investigated the activity of carboplatin plus atezolizumab, but only 2 (11%) patients had a clinical benefit in terms of DCR. 105
Moreover, the randomized phase II SAFIR02-IMMUNO clinical trial investigated the maintenance with durvalumab or chemotherapy also in patients with HER2− mBC who did not progress on 6–8 cycles of chemotherapy; in fact, 108 of 199 enrolled patients had an endocrine-resistant HR+ disease. Despite the trial failed to demonstrate a benefit (mPFS: 2.7 versus 4.6 months), an exploratory analysis showed that seven of 67 (10%) patients with HR+ disease presented CD274 gene gains/amplifications, six received durvalumab and five were alive after 15, 16, 19, 24, and 26 months, thus providing a strong rational for further exploring this predictive biomarker also beyond TNBC subtype. 22
As far as early setting, the phase II GIADA trial investigated the activity in terms of pCR of neoadjuvant epirubicin, cyclophosphamide, nivolumab, exemestane, and triptoreline in 43 premenopausal patients with stage II–IIIA luminal B-like BC. 106 This trial did not meet its primary endpoint (pCR: 16%) but showed that pCR was associated with higher TILs at baseline (15% in pCR subgroup versus 2% in non-pCR) and basal subtype at PAM50, and that chemotherapy determined an increase of TILs and CD8+ cells and a decrease of intratumoral stromal CD4+ cells.
In contrast, the randomized phase II I-SPY2 clinical trial evaluating the activity of the combination of pembrolizumab with standard NACT in HER2− early BC demonstrated a pCR benefit (30% versus 13%) in patients with HR+ disease. 56 As in TNBC, these data highlight the importance of investigating ICIs in early setting, in association with chemotherapy and possibly in selected patients; in this sense, the ongoing randomized phase III CheckMate 7FL clinical trial is assessing the benefit in terms of pCR and EFS from the addition of Nivolumab to standard NACT in 1200 patients with stage II–III high-grade HR+/HER2− BC. 107
Chemotherapy-free options were also explored. On the basis of preclinical evidences demonstrating an increase of immunogenic cell death driven by CDK4/6 inhibitors (CDK4/6i) and, in general, a synergistic activity of ICIs and CDK4/6i through modulation of the TME, this combination was investigated both in early and in metastatic setting.69,108 Anyway, because of safety concerns, in particular interstitial lung disease and severe liver enzyme abnormalities, the enrolment was discontinued in both settings.109–111 HDAC inhibitors represented another potential partner for ICIs, but the triplet of vorinostat, pembrolizumab and tamoxifen showed initial signs of a clinical benefit only in patients with increased CTLA4+/PD-1+/CD8+ T cells in either blood or tumor (mPFS: 8.6 versus 2.8 months). 112
As far as the combination with PARPi, on the basis of preclinical evidences showing a synergistic activity, the phase I/II MEDIOLA clinical trial investigated the combination of olaparib and durvalumab in patients with germline BRCA-mutated HER2-negative mBC and showed results similar to those reported in OlympiAD and EMBRACA trials, with PARPi monotherapy.38–40,113,114 In particular, considering the 13 evaluable for ORR patients with HR+/HER2− disease, nine of them (70%) had partial response as best response.
Furthermore, the I-SPY2 study demonstrated an improvement in terms of pCR (28% versus 14%) by the addition of olaparib and durvalumab to the standard NACT in 52 patients with stage II-III HR+/HER2− BC. 65 Interestingly, these patients were classified as MammaPrint High1 (MP1) or MammaPrint (ultra) High2 (MP2), with MP2 defined as MammaPrint score ⩽0.154, and MP2 cases benefited selectively from durvalumab and olaparib (pCR: 64% versus 22%), while no benefit was seen in MP1 cancers (pCR: 9% versus 10%).
Lastly, with the advent of ADCs in HR+ endocrine-resistant mBC and in light of ADCs immunomodulatory activity by interacting with cancer and immune cells, there is a strong rationale to combine ICIs with ADCs in this setting32,115,116; in this regard, two ongoing clinical trials (NCT04448886 and NCT04556773) are evaluating the safety and the active of anti-PD-(L)1 in association with sacituzumab govitecan and T-DXd, respectively (Table 5).
In conclusion, there is an urgent need to better characterize the immunologic aspects of the TME in HR+ BC and to identify novel IO agents impacting also on immune cells other than T cells, such as TAMs.
HER2+ BC
In comparison with HR+ subtype, HER2+ BC is characterized by a less immunosuppressive TME. In fact, in this subtype, PD-L1 is expressed in more than 50% of patients and high (⩾50%) TILs infiltration is found in 16% of cases. In general, TILs have a median level of 15–20% and are associated with improved prognosis and response to chemotherapy and trastuzumab4,98,117–119; in fact, trastuzumab is also characterized by immune-mediated mechanisms of action, and preclinical studies suggest immune-mediated mechanisms of trastuzumab resistance that can be overcome with checkpoint inhibition combinations. 120 These evidences provide the rational to combine anti-HER2 therapies with ICIs in this BC subtype.
The single arm phase Ib/II PANACEA clinical trial investigated the activity of trastuzumab and pembrolizumab in patients with metastatic HER2+ BC progressing on trastuzumab or TDM-1; 58 patients were enrolled, 46 of them had a PD-L1-positive disease. 121 The activity of this combination was limited (ORR: 15%) and observed only in PD-L1-positive cases; anyway, the biomarker analysis showed a statistical significant correlation between TILs and PD-L1 status and an association between stromal TILs at baseline and better response and DC. Interestingly, a significant difference in 12-month OS rate emerged between PD-L1+ and PD-L1 tumors (65% versus 12%), but, because of the very limited population and the absence of a control arm, it is difficult to define the prognostic or predictive value of PD-L1 expression.
The combination of ICIs and chemotherapy was also investigated in early-stage setting, in the Impassion-050 trial. 122 It was a phase III clinical trial assessing the clinical benefit in terms of pCR provided by the addition of atezolizumab to standard neoadjuvant therapy. The trial was stopped prematurely due to an unfavorable risk–benefit ratio for patients receiving atezolizumab: despite the absence of a pCR benefit, also in PD-L1+ patients, as stated, a clinical benefit in terms of EFS cannot be excluded; the similar ongoing phase III APTneo trial is powered for EFS and its result may clarify this issue (Table 5).
As far as the combination of ICIs and ADCs, the randomized phase II KATE2 trial investigated the addition of atezolizumab to trastuzumab emtansine (T-DM1) in patients with HER2+ mBC progressing to trastuzumab and taxanes. 123 It did not meet its primary endpoint (PFS in ITT), but in PD-L1+ subgroup (84/202, 42% of patients) a significant difference emerged (mPFS: 8.5 versus 4.1 months; HR: 0.60, 95% CI: 032–1.11); no differences in OS were observed (secondary endpoint). The ongoing phase III KATE3 clinical trial is investigating this combination in the same setting but only in PD-L1-positive patients, with PFS and OS as coprimary endpoints; moreover, the benefit of the addition of atezolizumab to T-DM1 is under investigation also in the post-neoadjuvant setting, in patients with residual disease following neoadjuvant therapy (ASTEFANIA trial).
Another promising ADC is T-DXd, which demonstrated an extraordinary antitumor activity both in HER2+ and in HER2-low BC115,124; the safety profile of the combination of T-DXd and Nivolumab was evaluated in a phase Ib trial enrolling 48 patients (32 HER2+ and 16 HER2 low) of whom 50% experienced a TRAE of grade 3 or more, and 37% discontinue the treatment because of TEAEs (25% related to T-DXd, 21% to nivolumab). 125 The ongoing phase Ib/II DESTINY-Breast 07 trial is assessing the combination of T-Dxd with durvalumab and paclitaxel.
Novel promising IO strategies other than ICIs comprise the CAR T cell therapy, which seems to be promising in HER2+ BC on the basis of preclinical data but is burdened by serious adverse events, and vaccines, which – on the basis of initial negative trials – have been moved from heavily pretreated patients to early setting, where an immune engagement is eventually more predictable. 118
Conclusions
The pharmacopoeia of TNBC deeply changed after the advent of ICIs, which demonstrated to improve outcomes in terms of OS and EFS in metastatic and early setting, respectively. Clinical trials confirmed that IO agents should be administrated as upfront therapy in both setting, when the immune microenvironment is more permissive and the degree of tumor evasion is lower.
In order to increase this benefit among non-responders and to expand this treatment approach beyond triple-negative subtype, which accounts only for 15% of all BCs, a deeper characterization of BC immune landscape and a better definition of BC immunogram are paramount. In fact, escalation and de-escalation strategies require the identification and prospective validations of biomarkers of response and resistance.
Moreover, many clinical trials investigating IO agents and aiming at improving clinical outcomes are ongoing; in this regard, the ADC-ICI combination seems to be the most promising escalation strategy, despite burdened by important toxicities. Instead, as far as IO beyond ICIs, CAR-T-cell therapy and CVs represent the most intriguing and innovative strategies in advanced and in early setting, respectively.
Lastly, because efficacy endpoints (e.g. EFS, PFS, OS) better measure the immune-mediated antitumor effects over time compared to response endpoints (e.g. pCR, ORR), the acceleration of drug development requires the identification and validation of novel composite surrogate endpoints considering the mechanism of action of IO agents.
Footnotes
Acknowledgements
None.
