Abstract
Immunotherapy has been used in the clinical treatment of colorectal cancer (CRC); however, most patients fail to achieve satisfactory survival benefits. Biomarkers with high specificity and sensitivity are being increasingly developed to predict the efficacy of CRC immunotherapy. In addition to DNA alteration markers, such as microsatellite instability/mismatch repair and tumor mutational burden, immune cell infiltration and immune checkpoints (ICs), epigenetic changes and no-coding RNA, and gut microbiomes all show potential predictive ability. Recently, the hypoxic tumor microenvironment (TME) has been identified as a key factor mediating CRC immune evasion and resistance to treatment. Hypoxia-inducible factor-1α is the central transcription factor in the hypoxia response that drives the expression of a vast number of survival genes by binding to the hypoxia response element in cancer and immune cells in the TME. Hypoxia regulates angiogenesis, immune cell infiltration and activation, expression of ICs, and secretion of various immune molecules in the TME and is closely associated with the immunotherapeutic efficacy of CRC. Currently, various agents targeting hypoxia have been found to improve the TME and enhance the efficacy of immunotherapy. We reviewed current markers commonly used in CRC to predict therapeutic efficacy and the mechanisms underlying hypoxia-induced angiogenesis and tumor immune evasion. Exploring the mechanisms by which hypoxia affects the TME will assist the discovery of new immunotherapeutic predictive biomarkers and development of more effective combinations of agents targeting hypoxia and immunotherapy.
Introduction
Colorectal cancer (CRC) is the third most common cancer and has a high mortality due to poor treatment options. 1 Over the past decade, immunotherapy has achieved satisfactory efficacy in a variety of highly mutated solid tumors. Immune checkpoint inhibitors (ICIs) primarily combine with immunosuppressive molecules, such as programmed cell death 1 (PD-1), programmed death ligand 1 (PD-L1), cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), indoleamine 2, 3-dioxygenase, and lymphocyte-activation gene 3 (LAG-3), on the surface of immune cells and tumor cells to block cancer immune escape. 2 Three ICIs (pembrolizumab, nivolumab, and ipilimumab) have been approved by the US Food and Drug Administration (FDA) for CRC containing mismatch repair defects (dMMR) or with high levels of microsatellite instability (MSI-H). 3 Pembrolizumab and nivolumab combined with PD-1 to block the binding of PD-1 and PD-L1/PD-L2-mediated immune evasion, while ipilimumab mainly blocked the binding of CTLA-4 to cluster of differentiation 80/86 (CD80/CD86) on the surface of antigen-presenting cells (APCs) (Figure 1). However, the use of ICIs has been greatly limited by the lack of sensitive predictive biomarkers and emergence of immunotherapy resistance. The complex immunotherapy resistance mechanisms suggest that we need to develop more effective combination therapy regimens and predictive biomarkers to provide improved survival benefits to patients with CRC.

ICIs for CRC. Pembrolizumab and nivolumab combined with PD-1 to block the binding of PD-1 and PD-L1/PD-L2-mediated immune escape, while ipilimumab mainly blocked the binding of CTLA-4 and increased binding of CD28 to CD80/CD86 on the surface of APCs and T cells.
Hypoxia is a common feature of the tumor microenvironment (TME) and an independent factor for poor prognosis in tumors. 4 Tumor hypoxia is mainly regulated by hypoxia-inducible factor (HIF), a heterodimer composed of an O2-sensitive α-subunit (HIF-1α, HIF-2α, or HIF-3α) and a constitutively expressed β-subunit (HIF-1β), in which HIF-1α expression and stability play key roles in tumors adapting to hypoxia. 5 Hypoxia-induced HIF-1α expression promotes the formation of disordered blood vessels, which does not improve hypoxia in the TME, and increases the risk of metastasis of CRC.6,7 Hypoxia also regulates the immune response of CRC patients by impairing antitumor innate and adaptive immunity through multiple mechanisms.8,9 A hypoxic environment induces the expression of immune checkpoints (ICs) such as PD-L1 through HIF-1α, promotes immunosuppressive cells such as myeloid-derived suppressor cells (MDSCs), regulatory T cells (Treg cells), and tumor-associated macrophages (TAMs), and inhibits tumor-infiltrating lymphocyte (TILs) infiltration and activation.10,11 Recently, therapies targeting HIF-1α and its downstream molecules have demonstrated promising antitumor effects.12,13
In this review, we focus on the predictors of immunotherapy in CRC and the impact of hypoxia on the TME and explore the complex relationship between HIF-1α and antitumor immunotherapy, which will provide new ideas for predicting and improving the efficacy of immunotherapy.
Predictors of immunotherapy for CRC
Although several ICIs have been used to treat CRC, their efficacy remains poor. The difference in therapeutic efficacy among different CRC patients is attributed to tumor heterogeneity, including DNA alterations, immune cell infiltration, ICs expression, epigenetic changes, noncoding RNA, and the gut microbiome. Biomarkers predicting the efficacy of immunotherapy are important for screening potential beneficiaries. Here, we summarize some predictive markers for CRC immunotherapy (Table 1), and explain their relationship to hypoxia.
Predictive markers of immunotherapy for CRC.

BRAF, B-type Raf kinase; CCL, CC chemokine ligand; CMS, consensus molecular subtype; CRC, colorectal cancer; CT, core tumor; CTLA-4, cytotoxic T-lymphocyte-associated protein 4; CXCL, C-X-C motif chemokine; CXCR, C-X-C receptor; DCs, dendritic cells; FoxP3, forkhead box P3; HLA, human leukocyte antigen; ICs, immune checkpoints; IL, interleukin; IM, invasive margin; KRAS, Kirsten rat sarcoma; LAG-3, lymphocyte-activation gene 3; m1A, N1-methyladenosine; m6A, N6-methyladenosine; MDSCs, myeloid-derived suppressor cells; MMR, mismatch repair; MSI, microsatellite instability; NK cells, natural killer cells; PD-1, programmed cell death 1; PD-L1, programmed death ligand 1; POLD, polymerase δ; POLE, polymerase ε; TAA, tumor-associated antigens; TAMs, tumor-associated macrophages; TGF-β, transforming growth factor β; Th17 cells, T-helper 17 cells; TIGIT, T-cell immunoreceptor with Ig and ITIM domains; TILs, tumor-infiltrating lymphocytes; TIM-3, T-cell immunoglobulin-3; TMB, tumor mutational burden; Treg cells, regulatory T cells.
DNA alterations
MSI and MMR systems are considered important markers for predicting the therapeutic effects of ICIs for CRC.15,49 MSI is divided into MSI-H, MSI-low (MSI-L), and microsatellite stability (MSS). 50 The MMR system can detect DNA replication errors in the S phase, while MSI may be derived from defects in the MMR system. CRC with dMMR/MSI-H has a higher density of TILs than MSS and sustained ICIs response. 14 Generally, MSI status testing involves polymerase chain reaction, next-generation sequencing to identify short repeating DNA segments and alterations in MMR genes, and immunohistochemistry analysis for the MMR proteins.51,52 A new method [single-molecule molecular inversion probes (smMIPs)] targets high-throughput sequencing of numerous microsatellite loci for MSI using smMIPs with 100% diagnostic sensitivity and specificity in CRC. 53 In addition, artificial intelligence has been used to predict MSI/dMMR directly. 54
Tumor mutational burden (TMB) is caused by DNA deletion, insertion, and frameshift mutations in the replication process of cancer cells and considered a useful biomarker for identifying patients for whom immunotherapy might be advantageous.16,55 CRC with a high TMB generates more neoantigens than that with lower TMB, which increases the immunogenicity of tumors. 56 The FDA-approved pembrolizumab for the treatment of unresectable or metastatic solid tumors with TMB ⩾10 mutations per megabase (mut/Mb). 57 Furthermore, TMB ⩾12 mut/Mb has also been used as a threshold for predicting immunotherapy efficacy in CRC populations 17 . TMB in MSI-H mCRC is generally elevated but still quite variable. 17 High TMB can still be detected in CRC patients with MSS and benefits from immunotherapy. 58 In addition, polymerase ε and polymerase δ mutations occur in a subset (less than 3%) of patients with MSS/MMR proficiency (pMMR), which is associated with a higher number of TILs and leads to the upregulation of genes encoding PD-1, PDL-1, and CTLA-4, suggesting a favorable prognosis with immunotherapy.18–20 In addition, according to the CRC molecular gene expression profile, a consensus molecular subtype (CMS) classification is developed based on both tumor and infiltrating stroma gene expression. 22 CMS includes four ‘consensus’ molecular subtypes: CMS1–CMS4. 21 CMS1 and CMS4 show greater infiltration of immune cells, whereas CMS2 and CMS3 lack immunoactivating components. In CRC with MSI-H/dMMR, CMS1 is mainly accompanied by infiltration of CD8+ TILs, and CD68+ macrophages and upregulated expression of ICs, which also predicts the efficacy of ICIs.23,24 CMS4 was infiltrated by Treg cells, MDSCs, monocyte-derived cells, and T-helper 17 cells (Th17 cells). These immunosuppressive cells should be monitored when targeting ICs.29,59 Other DNA alterations, such as BRAF mutations, need to be investigated further. Hypoxia is also one of the main features of solid tumors. HIF-1α expression may represent a novel marker to separate the MSI-L group from the MSS and MSI-H groups. A study shows downregulation of HIF-1α in the MSI-L group compared with MSS and MSI-H groups. 60 Moreover, suppression of the MMR pathway by hypoxia has been previously documented with specific downregulation of the MMR proteins MLH1, MSH2, and MSH6, leading to genomic instability.61,62 Therefore, additional studies are necessary to shed lighter on potential biomarkers and to explore the molecular mechanism of hypoxia regulating DNA alterations.
Immune cells infiltration and ICs
Although MSS/pMMR and MSI/dMMR are important predictors of CRC, abundant TILs more directly indicate antigenicity of tumor cells. 63 Immune cells in TME include CD4+ T cells, CD8+ T cells, natural killer (NK) cells, dendritic cells (DCs), MDSCs, Treg cells, and TAMs. As the main immune cells that kill tumors, the number of CD8+ T cells and NK cells infiltrating the TME reflects the antitumor immune response. DCs mainly recognize tumor antigens through the combination of major histocompatibility complex (MHC) molecules and T-cell receptor, present them to CD8+ T cells and other lymphatic T cells, and participate in the elimination of tumor cells through DC-NK crosstalk. Immune status is prognostic for survival in CRC and is associated with hypoxia and angiogenesis.23,27 Fewer CD8- and CD3-positive immune cells were found in hypoxic tumor centers than in tumor margins, suggesting an insensitivity to immunotherapy drugs. Apart from the CD8 and CD3 phenotypes, the number of immune cells with a CD4 phenotype has also been identified as a biomarker of the immune status and prognosis of CRC. 27 Treg cells are a subset of CD4+ T lymphocytes that predict a worse outcome in CRC with a higher Treg proportion.28,29,64 Immunoscore is based on the quantification of cytotoxic and memory T cells in the central and invasive margins (IMs) of the tumor.65,66 Immunoscore is based on the numeration of two lymphocyte populations (CD3/CD45RO, CD3/CD8, or CD8/CD45RO) in the core of the tumor (CT) and in the IM of tumors. 34
The expression of various ICs, including PD-1, PD-L1, CTLA4, T-cell immunoreceptor with Ig and ITIM domains (TIGIT), LAG-3, human leukocyte antigen (HLA), and T-cell immunoglobulin-3 (TIM-3), suggests the potential efficacy of ICIs.30,32 The high expression of ICs provides targets for immunotherapy and predicts its efficacy. 67 PD-1 is mainly expressed on the surface of CD8+ T cells and inhibits the immune-killing effect by binding to PD-L1 or PD-L2 on the surface of tumor cells. CTLA-4 on CD4+ and CD8+ T cells participate in immune tolerance and immune suppression by competing with CD28 to bind to CD80/CD86 on the surface of APCs. 68 MSS CRC constitutes the majority of cases and responds poorly to immunotherapy targeting PD-1, PD-L1, and CTLA-4. Nevertheless, targeting TIM-3 or TIGIT in conjunction with blocking PD-1/PD-L1 or other immunotherapies can restore the function of CD8+ T cells and enhance immunotherapy efficacy. 69 Hypoxia is a key factor affecting immune cell infiltration and ICs expression in TME, which predict the effect of immunotherapy. The specific molecular mechanisms by which hypoxia regulates immune cell infiltration and ICs expression will be detailed in the following sections.
Epigenetic change and noncoding RNA
Gene epigenetic modifications and noncoding RNA can also predict the efficacy of immunotherapy in CRC. CD8+ T-cell distribution in the TME of CRC was associated with the methylation of CD8+ T cells. A DNA methylation signature for the CD8+ TILs characteristic score system was constructed based on CD8+ T-cell differential methylation sites, with lower scores, indicating increasing CD8+ T-cell infiltration. 36 N6-methyladenosine (m6A), a critical regulator of transcript expression, is the most frequent internal modification of mRNA in the human body. The m6A modification decreases the mutation burden and immune activation and is related to reduced neoantigen load and poor response to immunotherapy. 37 The m6A methylation-mediated intercellular communication between TME and tumor cells contributes to CRC progression. 38 The epigenetic inheritance of CRC is also regulated by hypoxia, and it has been reported that fat mass and obesity-associated protein (FTO), an m6A demethylase, inhibited CRC metastasis and progression. 70 Hypoxia restrained FTO protein expression, mainly due to an increase in ubiquitin-mediated protein degradation. Except for m6A, epigenetic modification of m1A, alternative polyadenylation, and adenosine-to-inosine RNA editing are also predictors of CRC immunotherapy. 39 Long noncoding RNAs (lncRNAs) and microRNAs (miRNAs) have been screened as prognostic biomarkers for cancers and used to construct predictive models to predict immunotherapy efficacy.71,72 lncRNAs can be epigenetically regulated via m6A and are involved in predicting the tumor-immune-stromal microenvironment and immunotherapy efficacy. 73 Liu et al. 40 proposed an integrated algorithm, immuMiRNA, to identify miRNA modulators of immune-associated pathways and established an immune-associated miRNA prognostic signature consisting of three miRNAs (miR-194-3P, miR-216a-5p, and miR-3677-3p) to predict immunotherapy efficacy. In addition, small nucleolar RNA host gene 11 (SNHG11) is upregulated by promotor hypomethylation in CRC and can promote CRC cell migration and metastasis under hypoxia. 74 Mechanistically, SNHG11 binds to the von Hippel-Lindau tumor suppressor product (pVHL) recognition sites on HIF-1α, thus blocking the interaction of pVHL with HIF-1α and preventing its ubiquitination and degradation. Interestingly, HIF-1α was also able to regulate CRC proliferation by inducing noncoding RNA such as LINC00511. 75 More studies are still needed to elucidate the mechanism of action of hypoxia and noncoding RNA.
Gut microbiome
Hypoxia is an important environmental factor affecting the abundance of intestinal flora. 76 Studies have shown that changes in oxygen content caused by external and internal factors have regulatory effects on a variety of intestinal bacteria, including Enterococcaceae, Prevotella, Enterococci, Actococcus, Fusobacteria, Pseudomonas, and Escherichia–Shigella.77,78 Dysregulation of gut microbiota is not only an important inducement to CRC, but also modulate the immune system and affect immunotherapy efficacy. Bacteroidales improved the efficacy of CTLA-4 blockade, whereas Bifidobacterium was associated with the clinical benefits of PD-L1 blockade.43,79 Escherichia coli, Bacteroides fragilis and Firmicutes stimulate T-cell trafficking into CRC tumor tissues by increasing the expression of T cell-recruiting chemokines. 41 However, colibactin-producing Escherichia coli impairs CD3+ and CD8+ T-cell infiltration in CRC and leads to tumor resistance to immunotherapy. 80 Interestingly, Fusobacteria correlates with tumor evasion by inhibiting NK cells and TILs and increasing the expression of inflammatory mediators.44,45 Fusobacterium nucleatum induces PD-L1 expression by activating stimulator of interferon genes signaling and increasing the accumulation of interferon-gamma (IFN-γ)+ CD8+ TILs, and enhances the antitumor response to PD-1/PD-L1 blockade during treatment. 81 Fusobacterium and Bacteroidetes in CRC were shown to be associated with lower-level T-cell infiltrates and MSI, whereas enrichment of Bacteroides fragilis and Fusobacterium nucleatum was shown in dMMR CRC.42,46
Hypoxia regulated the TME of CRC
Hypoxia affects tumor immune cell infiltration, immune cell activity, ICs, and cytokine secretion and plays a predictive role in immunotherapy. 82 HIF-1α is the primary molecule responsible for hypoxia. The stability and activity of HIF-1α are regulated by post-translational modifications, hydroxylation, acetylation, and phosphorylation. Under normoxia, HIF-1α is rapidly degraded via pVHL-mediated ubiquitin-proteasome pathway, which is triggered by the hydroxylation of prolines and acetylation of lysine within a polypeptide segment known as the oxygen-dependent degradation domain of HIF-1α. Under hypoxic conditions, the HIF-1α subunit becomes stable and interacts with coactivators (such as the response element-binding protein), regulating the expression of target genes, including angiogenesis, epithelial mesenchymal transition, maintenance of cancer stem cells, metabolic reprogramming, tumor cell survival and proliferation, drug resistance, and tumor immune regulation. 83 In the following sections, we review the effect of hypoxia and its marker HIF-1 on the antitumor immunity of CRC.
Effects of hypoxia on angiogenesis in CRC
Tumor angiogenesis is critical to the TME. However, the distribution of the tumor vasculature network is irregular and does not improve hypoxia in the tumor center. Hypoxia results in angiogenesis via binding to the hypoxia response element (HRE) and inducing more vascular endothelial growth factor (VEGF), specifically VEGF-A. 84 VEGF-A promotes the differentiation of CD8+ T cells to exhausted subsets with high expression of PD-1 and TIM-3.85,86 Interestingly, VEGF-A is partly regulated by HIF-1α, and VEGF-A expression in CD8+ T cells contributes to T-cell infiltration in breast cancer. 87 The binding of VEGF to the VEGF receptor (VEGFR) 2 on the DCs membrane upregulates the expression of cofilin 1, which impairs the motility and differentiation of DCs. 88 HIF-1α is induced by leucine-rich-alpha-2-glycoprotein 1 in a concentration- and time-dependent manner, leading to VEGF-A expression. 89 HIF-1α binds to the HRE element in the promoter region of cyclooxygenase-2 (COX-2). The expression of COX-2 leads to elevated levels of prostaglandin E2 (PGE2), which induces PGE2-mediated angiogenesis. 90 PGE2 can also promote escape from immune surveillance by activating PD-L1 expression and suppressing DCs maturation.91,92 Furthermore, noncoding RNA also regulates HIF-α-mediated angiogenesis. Circ-Erbin, a circular RNA highly expressed in CRC cells, induces HIF-1α expression by activating the miR-125a-5p-5p/miR-138-5p/4E binding protein 1 axis to promote angiogenesis. 93 HIF-1α can transactivate myocyte enhancer factor 2D (MEF2D) expression by binding to the MEF2D promoter, inducing the expression of proangiogenic cytokines. 94 Dyskeratosis congenita 1 promotes HIF-1α expression by modulating its promoter activity, which promotes CRC angiogenesis and metastasis by increasing VEGF expression. 6 Hypoxic CRC promotes tumor growth and angiogenesis by delivering exosomes. Hypoxia is dependent on HIF-1α to promote exosomal Wnt4 upregulation, followed by β-catenin activation in endothelial cells to promote endothelial cell proliferation and migration. 95 A possible interaction between HIF-1α and tumor necrosis factor receptor-associated factor 6 (TRAF6) has been identified. 96 TRAF6, an upstream effector of the nuclear factor-kappa B (NF-κB) pathway, increases perfused vasculature and vessel permeability in hypoxic CRC. In conclusion, in CRC, multiple mechanisms, including hypoxia, induce HIF-1α to promote disordered angiogenesis in the TME and mediate immune evasion (Figure 2).
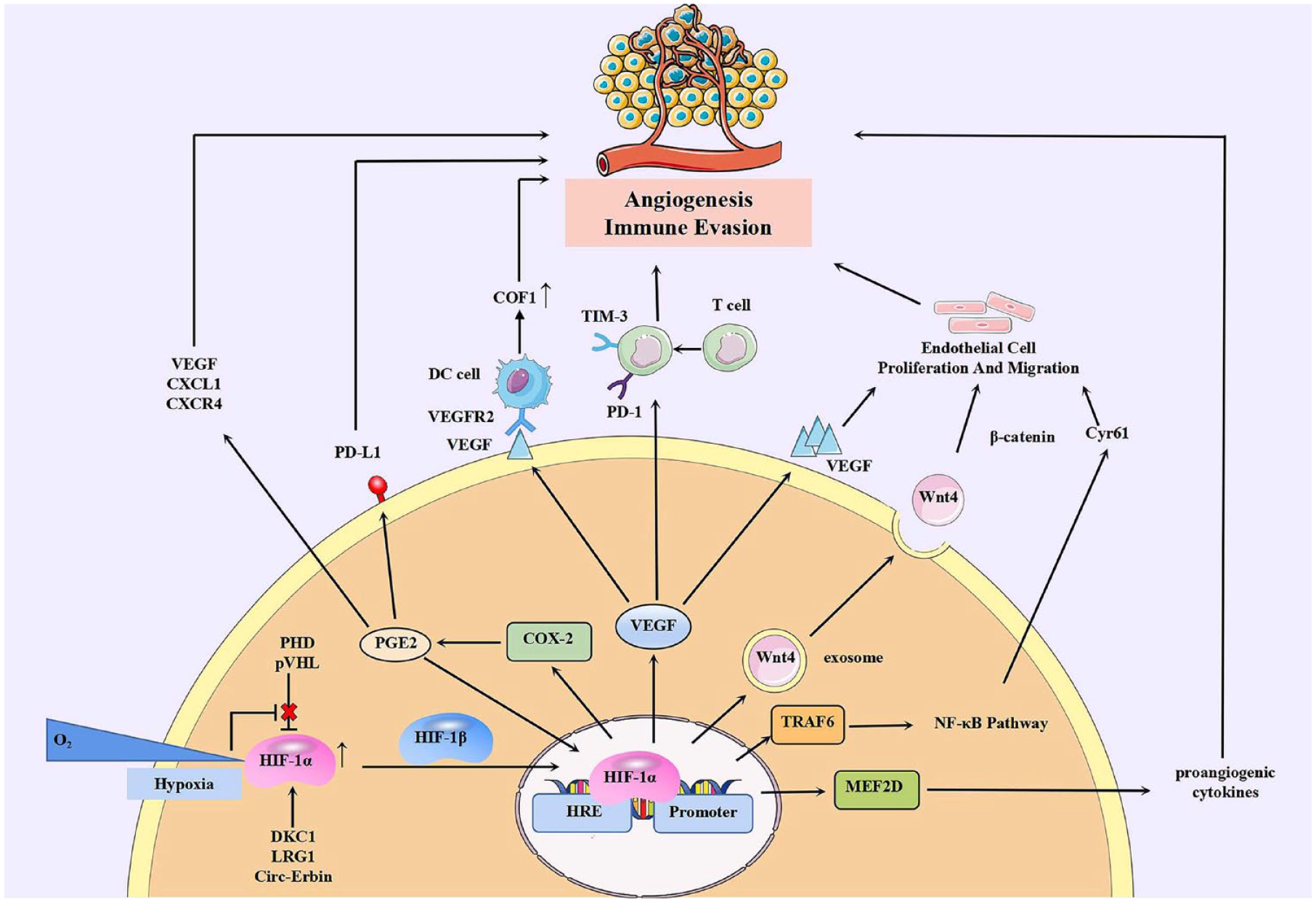
Hypoxia-induced angiogenesis and immune evasion via HIF-1α in CRC. HIF-1α is elevated by hypoxia and other molecules. After heterodimerization with HIF-1β, HIF-1α binds to HRE and activates transcription of VEGF, COX-2, TRAF6, MEF2D, and the production of exosomes containing Wnt4. These molecules directly or indirectly mediate disordered angiogenesis in TME and mediated immune evasion induced by hypoxia.
Effects of hypoxia on cancer stem cells
Cancer stem cells are tightly linked with PD-1/PD-L1 regulation and hypoxia. Hypoxic TME through promoting core-to-edge transition of tumor cells increases their stemness and resistance, and predisposes cancer into metastasis. 97 By regulating pH in tumor cells and by regulating the function of cell surface molecules such as extracellular carbonic anhydrase, cancer stem cell function is increased and cell invasion is enhanced in hypoxic TME, while also contributing to immune evasion. 98 Enriched PD-L1 expression in cancer stem-like cells (CSCs) contributes to CSCs immune evasion. 99 PD-L1 is closely associated with cancer stem cell immune escape and has been reported to regulate self-renewal of cancer stem cells, which is dependent on hypoxic HIF-1α and HIF-2α activation. 100 The interaction of CSCs with the immune system is an important mechanism of tumor immune escape. 101 Some studies have shown that CSC can interact with TAM to promote M2 polarization by secreting immunosuppressive molecules, such as transforming growth factor β (TGF-β) and interleukin (IL)-4.102,103 Meanwhile, hypoxia-induced CSCs can secrete CC chemokine ligand 20 to recruit TAMs to inhibit T-cell responses and promote immune tolerance. 104 In turn, TAM-derived IL-6 can promote the expansion and phenotypic expression of CSCs through signal transducer and activator of transcription (STAT) 3 signaling pathway. 105
Effects of hypoxia on immunosuppressive cells in CRC
Hypoxia contributes to the infiltration and activation of immunosuppressive cells, mediates immune evasion by secreting immunosuppressive molecules, or directly inhibits the function of immune cells. Here, we examined the effects of hypoxia and HIF-1 on the infiltration, differentiation, and function of immunosuppressive cells, such as Treg cells, MDSCs, and TAMs (Figure 3).

Effects of hypoxia on immunosuppressive cells in CRC. Hypoxia can influence multiple aspects of immunosuppressive cells. Generally, hypoxia can induce immune tolerance and immune escape by supporting survival, migration, and functions of immunosuppressive cells (Tregs, MDSCs, and TAMs), and causing an increase in the production of immunosuppressive cytokines.
Tregs identified by the expression of forkhead box P3 (FoxP3) are a subset of CD4+ T lymphocytes that dampen the immune response against cancer cells. High FoxP3 expression may be an independent prognostic factor for patients with MMR-proficient CRC. 106 High Treg infiltration in CRC is associated with increased expression of ICs, implying susceptibility to immunotherapy. 29 The role of HIF-1α in Treg differentiation is complex. HIF-1α can identify the HRE site in the FoxP3 promoter to promote its expression and directly bind to FoxP3 protein to promote its ubiquitination and degradation, thereby inhibiting Treg cell differentiation. 107 Th17 and Treg cells are derived from a common precursor and can be induced by different cytokines to perform opposing functions. HIF-1α can directly induce the transcription of receptor-related orphan receptor γt to promote Th17 cell differentiation and drive the transcription of IL-17 genes, with the assistance of IL-6. 108 Under hypoxic conditions, HIF-1α also promotes peripheral Treg cell activity, inducing carcinogenesis and the progression of CRC cells. 109 Hypoxia also promotes CD68+ macrophages in CRC to express C-X-C motif chemokine (CXCL) 11 and attract FoxP3+ Treg cell infiltration. 110 In response to hypoxia, FoxP3+ Treg cells express IL-17 and become FoxP3+ IL-17+ Treg cells, which can suppress tumor-specific CD8+ T cells. 111
TAMs are critical components of TME. TAMs mainly have two phenotypes: an M1-like phenotype (antitumor) and an M2-like phenotype (protumor). 112 The number of M2 macrophages increases in the hypoxic TME of CRC. 112 HIF-1α induced metabolic reprogramming of macrophages in the TME, including upregulation of amino acid metabolism and oxidative stress pathways, induces the transformation of macrophages into TAM with a tumor-promoting effect. 113 HIF-1α-induced lncRNA pituitary tumor-transforming 3, pseudogene (PTTG3P) contributes to CRC glycolysis and the M2 phenotype of macrophages under hypoxic conditions. 114 M2 macrophages were induced by hypoxia-triggered forkhead box O1 (FoxO1) deficiency, which could negatively regulate MHC-II genes. 115 Hypoxia augments macrophage-mediated T-cell suppression in a manner dependent on the expression of HIF-1α and targeting HIF-1α in macrophages can promote the proliferation and activation of CD8+ T cells. 116 TAMs produce TGF-β to support HIF-1α expression in CRC cells, thereby upregulating tribbles pseudokinase 3 (TRIB3), which results in activation of the β-catenin/Wnt signaling pathway. 117 TRIB3 can also reduce tumor-infiltrating T cells by inhibiting STAT 1-mediated CXCL10 transcription by enhancing the epidermal growth factor receptor signaling pathway in CRC. 118 HIF1-α is significantly associated with the expression of VEGFR2 in M2-TAM, and TGF-β1 is produced through the VEGF/VEGFR2 signaling pathway. 119 Hypoxia also activates RAS signaling in CRC, which drives the production of colony-stimulating factor 2 (CSF2) and lactate. 112 CSF2 synergizes with lactate to elicit functional reprogramming of TAMs and exerts a tumor-supportive capacity. 120 Succinate exerts its effects via succinate receptor 1, which mediates signaling through the phosphatidylinositol 3-kinase (PI3K)/HIF-1α pathway. Multiple cancer cells, including CRC cells, secrete succinate into the TME, facilitating macrophage migration and mediating TAMs polarization. 121 However, some studies have reported different results, that upregulation of HIF-1α in macrophages contributes to the overexpression of M1 markers, whereas HIF-2α promotes M2 polarization.122,123
MDSCs, with the ability to suppress immune cell responses, are a heterogeneous group of cells derived from the bone marrow and are considered precursors of DCs and macrophages. MDSCs, considered a predictive marker of immunotherapy, limit the therapeutic efficacy of ICIs by leading to T-cell dysfunction and promoting angiogenesis. 124 MDSCs comprise two main populations: monocytic MDSCs (M-MDSCs) and granulocytic MDSCs (G-MDSCs). HIF-1α redirects differentiation toward the classically activated (M1) phenotype and alters the function of MDSCs. 125 MDSCs from tumors primarily use arginase (Arg) and nitric oxide (NO) to suppress T-cell function, and hypoxia causes the upregulation of Arg1 and inducible nitric oxide synthase (iNOS) in MDSC. 126 HIF-1α directly binds to a transcriptionally active HRE in the miR-210 proximal promoter. 127 MiR-210 modulated MDSCs function by increasing Arg1, NO, CXCL12, and IL-16 production. HIF-1α also upregulates lncRNA PVT1 expression in G-MDSCs under hypoxia, which can increase the levels of Arg1 and reactive oxygen species in G-MDSCs and regulate their immunosuppressive ability. 128 S100A9, a G-MDSC exosome, is mediated by hypoxia-induced Rab27a (a GTPase required for exosome secretion) and enhances the stemness of CRC. 129 Overexpression of the V-domain Ig suppressor of T-cell activation (VISTA) is associated with MDSC function. In CRC, hypoxia upregulates VISTA expression by HIF-1α binding to the VISTA promoter. 130 VISTA, in turn, contributes to MDSC-mediated T-cell suppression under hypoxia. Increased HIF-1α upregulates CXCL1, CXCL3, and CXCL8 expression in MDSCs, contributing to enhanced recruitment of MDSCs to tumors. 131
Effects of hypoxia on immunoeffector cells in CRC
Hypoxia promotes the infiltration and activation of immunosuppressive cells and inhibits cells that mediate antitumor immunity. In this section, we discuss the mechanism by which hypoxia and its marker, HIF-1, inhibit DCs, NK cells, and CD8+ T cells (Figure 4).

Effects of hypoxia on immunoeffector cells in CRC. Hypoxia leads to immune evasion of CRC by inhibiting infiltration, activation, maturation, and function of immune effector cells and antigen-presenting cells through multiple pathways.
As the most important APC, DCs can specifically recognize, process, and present diverse cancer antigens and mediate tumor immunity via the activation of CD8+ and CD4+ T cells. The effects of hypoxia on the differentiation and maturation of DCs have been well studied. Hypoxia keeps DCs in the TME in an immature state and contributes to the inhibition of T-cell response, which results in decreased motility, phagocytosis, and CD83 expression. 132 The hypoxic TME also promotes apoptosis of DCs by inhibiting the PI3K/Akt pathway. 133 Plasmacytoid DCs (pDCs) play immunosuppressive roles in the TME. Hypoxia-induced extracellular adenosine (ADO) significantly enhances pDCs recruitment into tumors. 134 ADO-stimulated pDCs promote Treg cell induction and suppress proliferation and cytotoxicity of CD8+ T cells. In addition, hypoxia-induced VEGF expression and downregulation of IFN hamper DCs maturation. 135 Hypoxic DCs have been shown to alter chemokine expression and promote the upregulation of proinflammatory cytokines such as IL-1β and tumor necrosis factor (TNF)-α 4 . HIF-1α upregulates CC chemokine receptor (CCR) 5 and impairs the upregulation of CCR7, a necessary molecule for the homing of mature DCs. 136 Changes in CCR5 and CCR7 may originate from hypoxia-induced expression of IL-10, which also inhibits the differentiation and maturation of DCs. 137 Interestingly, under exogenous inflammatory stimulation, hypoxia was able to increase the activation of mouse DCs through HIF-1α, which was manifested by the expression of costimulatory molecules CD80 and CD86 and may be caused by increasing glycolysis.138,139
NK cells are a subtype of innately toxic lymphocytes that can directly kill tumor cells after activation by cytokines and participate in adaptive immunity through intercellular crosstalk. In CRC patients, hypoxia-induced HIF-1α inhibits IL-18-mediated antitumor activity of NK cells. 140 Deletion of HIF-1α in NK cells results in an enriched NF-κB pathway and enhanced IFN-γ, which markedly increases human NK cell responses. NK cell receptors (NKp46, NKp30, NKp44, and NKG2D), the major phenotypic features of activating NK cells, are downregulated by HIF-1α in hypoxic environments. 141 Furthermore, HIF-1α promotes the expression of metalloproteinase, a disintegrin and metalloproteinase 10 (ADAM10) on the tumor cell surface. ADAM10 leads to shedding of the NK cell-activating ligand MHC class I chain-related protein A (MICA) from the surface of tumor cells. 142 Shedded MICA can also downregulate the expression NKG2D in NK cells, resulting in tumor escape. 143 In addition, TGF-β secreted by Treg cells and tumor cells also mediates the downregulation of NKG2D expression in the hypoxic TME. 144
CD8+ T cells, major cytotoxic T lymphocytes (CTLs), are the key antitumor immune cells in CRC, mainly engaged in adaptive immunity, and their activity and quantity are vital for determining the efficacy of immunotherapy. 145 The infiltration of CD8+ T cells in the hypoxic regions of CRC tumors is significantly reduced. 146 PTTG3P not only affects the phenotype of TAMs, but also inhibits the infiltration of CD8+ T cells.114,147 Genes related to cellular response hypoxia are upregulated in CD8+ T cells at an advanced stage, which can potentially lead to T-cell apoptosis and increased secretion of the suppressive cytokine IL-10. The effects of hypoxia and HIF-1α on CD8+ T cells remain controversial. In a CRC cell model, hypoxia was shown to enhance CD8+ T-cell activity and IFN-γ expression by promoting MHC I folding and cell surface expression. 148 In CD8+ T cells, hypoxia can enhance effector responses to persistent antigens by increasing cytolytic activity and production of IFNγ. 149 Anti-VEGF treatment enhances CD8+ T-cell antitumor activity by amplifying hypoxia. Furthermore, hypoxia can drive the activity of CD8+ T cells by enhancing glycolysis in CD8+ T cells. 150 However, hypoxia simultaneously induces the accumulation of lactate and adenosine in the TME, which leads to the inhibition of CD8+ T-cell proliferation and cytotoxicity. 151
Effects of hypoxia on the alteration of ICs in CRC
As inhibitory regulatory molecules, ICs may be regulated by hypoxia. We discussed several ICs and how they caused immune evasion in the hypoxic TME (Figure 5). Elevated PD-L1 expression causes escape from adaptive immunity via the promotion of apoptosis of T cells. On the surface of MDSCs, TAMs, DCs, and tumor cells in hypoxic TME, PD-L1 expression was rapidly, significantly and selectively upregulated, which was dependent on HIF-1α but not HIF-2α. 11 HIF-1α can upregulate PD-L1 expression by binding to transcriptionally active HRE in the PD-L1 proximal promoter.11,152 Pyruvate kinase isoform M2 (PKM2) assists HIF-1α in binding to HRE. 153 PKM2 regulates PD-L1 expression not only in CRC tumor cells but also in TAMs, DCs, and T cells. 153 Hypoxia can induce the secretion of circEIF3K exosomes from cancer-associated fibroblasts (CAFs), which increases the stabilization of PD-L1 by reducing the production of miR-214 in CRC. 154 In addition to PD-L1, hypoxia also causes upregulation of CTLA-4 on MDSCs, TAMs, DCs, and tumor cells through HIF-1α. 11 CTLA-4 expression appears to be associated with hypoxic-induced VEGF and angiogenesis, and targeting both the hypoxic pathway and CTLA-4 has shown promising efficacy.155,156 Hypoxic tumor regions in the TME are associated with increased VISTA expression in tumor infiltrating MDSCs. Targeting VISTA under hypoxia relieved MDSC-mediated T-cell suppression. 130 Further studies showed that HIF-1α binding to a conserved HRE in the VISTA promoter upregulated VISTA in myeloid cells. HLA-G is a non-classical MHC-I molecule that was considered to be involved in immune evasion and prognosis of CRC. 31 HLA-G suppresses IFN-γ and TNF-α, leading to a reduction in the antitumor effects of NK and T cells. 157 HLA-G has been shown to be induced in melanoma and glioma by HIF-1α binding to HRE.158,159 However, hypoxia may decrease HLA-G expression in constitutively expressed HLA-G cell lines. Hypoxia maintenance may result in channeling cell energy into productive gene expression at the cost of HLA-G. 158 Similarly, the overexpression of CD47, CD137, and other ICs may be associated with hypoxia. 160 Further investigations are needed to elucidate the relationship between hypoxia and ICs in CRC.

Effects of hypoxia on the alteration of ICs in CRC. Hypoxia induces the expression of multiple ICs (PD-L1, HLA-G, and VISTA) in the TME of CRC through the binding of HIF-1α to HRE. Hypoxia can also induce CAFs to secrete exosome circEIF3K to promote PD-L1 stability. These ICs promote the immune evasion of CRC.
Targeting hypoxia to enhance the efficacy of immunotherapy for CRC
Currently, improvements in the hypoxic TME are being explored to enhance the efficacy of immunotherapy in CRC. Some treatments have been found to improve immune status and immunotherapy efficacy by improving hypoxia in CRC. Approaches to improve hypoxia mainly include targeting HIF-1α, antiangiogenesis, promoting vascular normalization, and increasing tumor oxygenation (Table 2).
Summary of improving hypoxia agents with potential to improve immunotherapy in CRC.

COX-2, cyclooxygenase-2; HIF-1α, hypoxia-inducible factor 1α; IL, interleukin; iNOS, inducible nitric oxide synthase; LAG-3, lymphocyte-activation gene 3; PD-1, programmed cell death 1; PD-L1, programmed death ligand 1; MDSCs, myeloid-derived suppressor cells; mTOR, mechanistic target of rapamycin; ROS, reactive oxygen species; STAT3, signal transducer and activator of transcription 3; TAMs, tumor-associated macrophages; TIGIT, T-cell immunoreceptor with Ig and ITIM domains; TILs, tumor-infiltrating lymphocytes; TIM-3, T-cell immunoglobulin-3; TLR4, Toll-like receptor 4; Treg cells, regulatory T cells; VEGF, vascular endothelial growth factor; YYFZBJS, Yi-Yi-Fu-Zi-Bai-Jiang-San.
Decursin is a novel HIF-1α inhibitor that promotes proteasomal degradation and improves T-cell activation in the TME. Decursin reduces the hypoxic area; PD-L1 expression; and accumulated CD3+ T, CD4+ T, and CD8+ T-cell infiltration, but FoxP3+ Treg cells and MDSC-mediated Arg1 are attenuated. 161 Yi-Yi-Fu-Zi-Bai-Jiang-San, a traditional Chinese medicine, improves the TME of CRC by inhibiting HIF-1α-mediated Treg cell activation. 109 Panaxadiol enhances the activity of CTLs and restores their capacity to kill tumor cells. 162 The HIF-1α inhibitor echinomycin potentiates the cancer immunotherapeutic effects of anti-CTLA-4 therapy and prevent the high incidence of immune-related adverse events induced by the combination of anti-CTLA-4 and anti-PD-1/PD-L1. 163 The combination of targeting TIGIT and HIF-1α is a novel strategy against CRC. 172
Antiangiogenesis therapy improves the efficiency of ICIs in VEGFA-overexpressing CRC. Apatinib, a VEGFR2 tyrosine kinase inhibitor, has the potential to reverse immunosuppression in hypoxic CRC. Apatinib plus anti-PD-1 increases CD8+ TILs, restores CD8+ T-cell exhaustion, and reduces the accumulation of Treg cells, M2-like TAMs and MDSCs. 164 CU06-1004, an endothelial dysfunctional blocker, causes increased levels of TILs and tumor apoptosis within the TME. 165 Catalpol inhibits the expression of IL-1β, IL-6, IL-8, COX-2, and iNOS by inhibiting the expression of angiogenic markers. 166 The combination of fruquintinib and anti-PD-1 decreases angiogenesis, enhances vascular normalization, and reprograms the immune microenvironment. 167 A Cu2+ ion-based intracellular bio-nanoreactor stimulates robust tumoricidal immunity by delivering small interfering RNA targeting VEGF. 168 A phase IB clinical study (NCT02298959) showed that ziv-aflibercept, an antiangiogenic agent, plus pembrolizumab in patients with advanced solid tumors, including CRC, demonstrates an acceptable safety profile with antitumor activity. 169 Oxygen supplementation is the simplest way to improve hypoxia and improve immunotherapy. Hyperbaric oxygen therapy and melatonin can alter the hypoxic microenvironment, resulting in decreased HIF-1α expression and diminished immune evasion in CRC. 170 Employing metformin as a method to improve hypoxic TME could increase TILs and enhance T-cell cytotoxicity activity, thus improving sensitivity to anti-PD-1 immunotherapy.171,173
Conclusion
The efficacy of immunotherapy is not as expected in most CRC patients. More sensitive and extensive biomarkers are needed to predict the efficacy of immunotherapy for CRC. We summarized some common markers and systems for efficacy prediction. CRC can create a hypoxic TME that impairs the efficacy of immunotherapy and antitumor immunity. Markers of hypoxia, especially HIF-1, showed strong correlations and predictors of immunotherapy outcomes, and some strategies for improving hypoxia have shown synergy with immunotherapy. In the future, more effective strategies and immunotherapy combinations are needed to improve hypoxia, and further searches should be made for the most appropriate combination approaches to provide a broader space for the treatment of CRC patients.
