Abstract
Background:
Nivolumab has been approved in China as second-line treatment for advanced non-small-cell lung cancer (NSCLC) via weight-based infusion, based on the CheckMate 078 study. We investigated the safety and efficacy of 240 mg flat-dose nivolumab in patients with advanced NSCLC, including those with hepatitis B virus (HBV) and epidermal growth factor receptor (EGFR) mutation/ALK receptor tyrosine kinase (ALK) translocation due to high prevalence in China.
Methods:
CheckMate 870 was a single-arm, open-label, phase IIIb trial in Asian (primarily Chinese) patients with previously treated advanced NSCLC. Patients received flat-dose nivolumab 240 mg every 2 weeks (Q2W) for up to 2 years. The primary endpoint was the incidence and severity of treatment-related select adverse events (TRsAEs) in non-HBV patients; secondary and exploratory endpoints included severity of high-grade TRsAEs in HBV-infected patients, and safety, efficacy and patient-reported outcomes (PROs) in the whole population.
Results:
Out of 404 patients enrolled, 400 received treatment. Median (standard deviation) age was 60.5 (8.68) years and the majority were male (78.5%). At a median follow-up of 37.6 months, no Grade 5 TRsAEs were reported, and the frequency of Grade 3–4 TRsAEs was low (0.0–5.9%) in non-HBV and HBV NSCLC patients. Median overall survival (OS) and progression-free survival (PFS) in all treated patients were 14.7 (12.3–18.1) and 3.6 (2.3–3.8) months, respectively. Median OS was 14.2 (12.3–18.1) and 22.3 (10.0–NA) months for non-HBV and HBV-infected patients, 19.3 (11.2–31.7) and 13.7 (11.5–18.1) months for EGFR-positive and wild-type subgroups, and 19.3 (12.9–23.5) and 13.3 (10.9–17.7) months for those with programmed death-ligand 1 (PD-L1) expression ⩾1% and <1%, respectively. No notable changes from baseline were observed in PROs throughout the study.
Conclusion:
Nivolumab 240 mg infusion Q2W was well tolerated, efficacious, and maintained health status and quality of life in Asian patients with previously treated advanced NSCLC regardless of HBV, EGFR, or PD-L1 status.
Introduction
Lung cancer is one of the most common causes of cancer-related deaths globally. 1 Non-small-cell lung cancer (NSCLC) is the dominant form of lung cancer (accounting for 85%), and consists mainly of non-squamous (NSQ, ~70%) and squamous (SQ, ~30%) histological subtypes.2,3 Most patients are diagnosed at the advanced stages of the disease and ineligible for surgical resection. 4
Nivolumab is a first-in-human programmed death receptor-1 (PD-1) immune checkpoint inhibitor antibody that disrupts the binding of PD-1 and its ligands, programmed death-ligand 1 (PD-L1) and PD-L2, to promote antitumor immunity 5 and is currently approved for the treatment of several cancer types, including advanced NSCLC. 6 In June 2018, the Chinese National Medical Products Administration approved nivolumab monotherapy for previously treated advanced NSCLC (SQ and NSQ) based on results of the CheckMate 078 study, a randomized, open-label phase III trial in a predominantly Chinese population.7,8 This regimen is approved in most Asian countries, including Hong Kong, Taiwan, Singapore, and Thailand. Pooled analysis of 5-year outcomes from the pivotal global CheckMate 017 and CheckMate 057 studies (primarily non-Asian patients) demonstrated a fivefold higher overall survival (OS) rate among previously treated NSCLC patients receiving nivolumab [3 mg/kg, once every 2 weeks (Q2W)] compared with those who received docetaxel (13.4% versus 2.6%). 9 Consistent with the CheckMate 017 and CheckMate 057 studies, nivolumab significantly improved OS versus docetaxel [median OS: 12.0 (10.4–14.0) versus 9.6 (7.6–11.2) months] in the CheckMate 078 study. 8
Patients with epidermal growth factor receptor (EGFR) mutation, ALK receptor tyrosine kinase (ALK) translocation, and positive for hepatitis B surface antigen (HBsAg) were excluded from the CheckMate 078 study, although ALK and EGFR are the two most common oncogenic driver mutations in NSCLC, with EGFR mutation more common in Asian compared with Western patients (40–50% versus 10–16%).10,11 Moreover, China has the largest number of individuals infected with hepatitis B virus (HBV) worldwide. 12 Previous studies have shown that PD-1 upregulation is associated with HBV-specific T cell dysfunction, while in patients with NSCLC, PD-L1 expression has been reported to be significantly higher in those with chronic HBV infection. 13 Hepatitis B reactivation can occur in NSCLC patients undergoing treatment with immunosuppressive agents, which can lead to increased HBV-related morbidity and mortality. 14 For patients receiving chemotherapy, there are additional safety concerns relating to hepatotoxicity secondary to treatment. 15
In this study, we aimed to investigate the safety and tolerability of nivolumab in a population more representative of that found in clinical practice in Asia, to provide additional insight into the frequency of high-grade select adverse events (AEs) and their outcomes, thus supplementing the growing pool of safety data for nivolumab-treated patients in Asia. In addition, we aimed to investigate the safety of flat-dose nivolumab as a 30-min infusion, in light of the fact that flat-dose nivolumab is likely to be favored in China in the future due to fewer dose calculation errors and reduced preparation time, as well as the shorter and more convenient infusion time for patients.16–20
Methods
Study design
CheckMate 870 (ClinicalTrials. gov number, NCT03195491, date of registration: 20 June 2017) was a single-arm, open-label, phase IIIb trial in patients with advanced NSCLC previously treated with one or two systemic therapies. Patients received nivolumab 240 mg via intravenous infusion over 30 min Q2W for 24 months or until disease progression, unacceptable toxicity, or withdrawal of consent, whichever occurred first. See Supplemental Table S1 for criteria for dose interruption, delay, and discontinuation. Patients who discontinued nivolumab were followed for collection of outcomes and/or survival data as required, until death, or conclusion of the study.
This study was conducted in accordance with the Declaration of Helsinki, followed Good Clinical Practice guidelines as defined by the International Conference on Harmonization, and followed the ethical principles underlying European Union Directive 2001/20/EC and the United States Code of Federal regulations, Title21, Part 50 (21CFR50). Study approval was granted by the Ethical Review Committee at each site (refer to Supplemental Methods for more details). All participants provided written informed consent prior to enrollment.
Patients
Eligible patients were aged ⩾18 years and with histologically or cytologically diagnosed stage IIIB/IV NSCLC (SQ or NSQ), 21 or recurrent or progressive disease following multimodal therapy (radiation therapy, surgical resection, or definitive chemoradiotherapy for locally advanced disease), with an Eastern Cooperative Oncology Group performance status (ECOG PS) ⩽1. Patients with active central nervous system (CNS) metastasis were excluded. Patients with CNS metastases that were adequately treated and neurologically returned to baseline (except for residual signs or symptoms related to the CNS treatment) for at least 2 weeks prior to enrollment were eligible. All patients were required to have had at least one prior systemic therapy for advanced disease, including patients who received platinum-containing adjuvant, neoadjuvant, or definitive chemoradiation therapy for locally advanced disease, and who developed recurrent disease after completing therapy.
Patients with NSQ histology were tested for EGFR mutation and ALK translocation. Patients who were negative on EGFR mutation or ALK translocation testing [wild-type (WT)], and patients with indeterminate results or for whom no tissue was available for testing, were eligible if they had received one prior systemic therapy as described above. Those positive on EGFR-sensitizing mutation [exon 19 deletion, exon 21 (L858R) substitution] or ALK translocation testing were eligible to enroll after progression from first-line tyrosine kinase inhibitor (TKI) therapy and one systemic therapy. Patients positive on non-sensitizing EGFR mutation testing were eligible to enroll following two lines of systemic therapy (which could be TKI and chemotherapy).
Patients previously treated with immune checkpoint inhibitors were excluded, as were HBsAg-positive patients with HBV DNA >500 IU/ml. Hereafter, the term HBV infected is used to describe patients who were HBsAg positive with HBV DNA ⩽500 IU/ml. All patients who were HBsAg positive were required to be on antiviral therapy.
Data collection
Collection of baseline and clinical characteristics [including mutational status of EGFR, ALK, ROS proto-oncogene (ROS), MET proto-oncogene (MET), KRAS proto-oncogene (KRAS), and B-Raf proto-oncogene (B-RAF)], safety assessments, laboratory tests (including HBV testing), PD-L1 and molecular testing, and radiographic tumor assessments were performed at the time of screening. Safety, laboratory, and efficacy assessments were performed at Cycle 1 Day 1, each subsequent cycle (Q2W), and at follow-up visits [Day 35 (±7 days) and Day 100 (±7 days) after last dose] (Supplemental Table S2). Patients were followed for at least 100 days after the last dose of study treatment; survival follow-up visits occurred every 12 weeks (±14 days) for 2 years (Supplemental Table S2). Patient-reported outcomes (PROs) were assessed on Day 1 (±5 days) and every 4 weeks for the first 6 months, then every 6 weeks for the remainder of the study. PROs were assessed using the three-level version of the EuroQol five-dimensional questionnaire (EQ-5D-3L), the EuroQol visual analog scale (EQ-VAS), and the Lung Cancer Symptom Scale (LCSS). Tumor response was evaluated by investigators using the Response Evaluation Criteria in Solid Tumors (RECIST) v1.1.
Study endpoints
The primary endpoint was incidence and severity of high-grade [combined National Cancer Institute Common Terminology Criteria for Adverse Events Version 4 (NCI CTCAE v4), Grade 3–4 and Grade 5] treatment-related select adverse events (TRsAEs) in non-HBV patients. Secondary endpoints included incidence and severity of high-grade TRsAEs in HBV-infected patients, as well as OS, progression-free survival (PFS), objective response rate (ORR), and duration of response (DOR) by PD-L1 expression (PD-L1 <1% and ⩾1%, and <50% and ⩾50%), HBV status (HBV infection +/-), and molecular genotype (EGFR/ALK status). Primary and secondary endpoint definitions are found in Supplemental Table S3. Exploratory endpoints included EQ-5D-3L, EQ-VAS, and LCSS scores, and patient molecular profiles (ROS1, MET, KRAS, and B-RAF). All on-study and drug-related AEs and serious AEs were tabulated using worst grade per NCI CTCAE v4 criteria by system organ class and Medical Dictionary for Regulatory Activities preferred term.
Statistical analysis
Sample size was determined based on the frequency and outcomes of infrequent high-grade treatment-related AEs. Target sample size was 400 patients (assuming 340 non-HBV-infected patients and 60 HBV-infected patients), and 95% confidence intervals (CIs) were estimated to be 0.06–1.79%, 0.27–2.54%, and 1.56–5.18% for AE incidence rates of 0.5%, 1%, and 3%, respectively. Based on a 15% screen failure rate, we aimed to screen 470 patients. In addition, the EGFR-positive population was capped at 40 participants due to a large percentage of patients with NSCLC harboring EGFR mutation in China (approximately 30%). 22
Statistical analyses were performed for all treated patients by HBV status; three HBV status groups were analyzed (non-HBV, HBV, and total). General descriptive statistics, including mean and standard deviation (SD) for continuous variables, and count and percentage for categorical variables were used. OS, PFS, and DOR were estimated using the Kaplan–Meier method (Supplemental Table S3). Rates at fixed timepoints (12 and 24 months) were derived from the Kaplan–Meier estimate (Supplemental Table S3). Secondary efficacy endpoints were additionally analyzed by ROS1, MET, KRAS, and B-RAF status post-hoc.
Results
Baseline patient characteristics
Out of a total 470 patients screened, 404 were enrolled between December 2017 and July 2018, and 400 (99.0%) patients received study treatment; four (1.0%) were excluded (two died and two withdrew with no reported reason). There were 383 HBsAg-negative and 17 HBsAg-positive patients. All patients had discontinued study treatment by data cut-off; the most common reasons were disease progression (n = 274, 68.5%), completion of treatment (n = 38, 9.5%), study drug toxicity (n = 28, 7.0%), and AEs unrelated to the study drug (n = 21, 5.3%) (Supplemental Figure S1).
Baseline characteristics and patient demographics are presented in Table 1. Mean (SD) age was 60.5 (8.68) years, the majority of patients were male (n = 314, 78.5%), and all patients were from Asia, including 394 (98.5%) Chinese patients. The majority of patients had a baseline ECOG PS score of 1 (n = 340, 85.0%). Over half the patients (67.0%) were former (n = 244, 61.0%) or current (n = 24, 6.0%) smokers. Most patients had stage IVA (n = 149, 37.3%) or IVB (n = 160, 40.0%) disease at study entry, and the majority of patients had metastatic disease (n = 328, 82.0%). Only 1.8% (n = 7) of patients had CNS metastasis. Overall, 99.8% (399/400) of the patients had received prior systemic cancer therapy; of those, 87.8% and 10.5% had received one and two prior therapies, respectively. Median (95% CI) duration of nivolumab therapy for non-HBV patients, HBV-infected patients, and total patients was 3.25 (2.60, 3.45), 1.54 (0.95, 12.68), and 3.24 (2.60, 3.45) months, respectively. In addition, EGFR, KRAS, ALK, B-RAF, ROS, and MET mutations were detected in 34 (8.5%), 27 (6.75%), 10 (2.5%), seven (1.75%), five (1.25%), and four (1.0%) patients, respectively (Supplemental Table S4). Patients who were positive, negative, or had indeterminate results on mutation testing are hereafter described using the terms ‘positive’ (e.g. EGFR positive), WT (e.g. EGFR WT), and ‘undetermined’ (e.g. EGFR undetermined), respectively. The HBV reactivation rate at baseline was 17.6% (3/17).
Baseline characteristics and patient demographics: all treated patients.

Staging of lung cancer according to version 8 of the International Association for the Study of Lung Cancer Staging Manual in Thoracic Oncology. All data shown as n (%) unless otherwise stated.
CNS, central nervous system; ECOG PS, eastern cooperative oncology group performance status; HBV, hepatitis B virus; SD, standard deviation.
High-grade TRsAEs
Grade 3–4 TRsAEs occurred with low frequency (0.0–2.1%) in NSCLC patients without HBV, and the most common were hepatic (2.1%), pulmonary (1.3%), and skin (1.8%) AEs (Table 2). There were no Grade 5 TRsAEs in non-HBV patients (Table 2). The median time to onset for high-grade TRsAEs ranged from 10.64 (8.1–13.1) weeks for skin AEs to 30.07 (21.1–39) weeks for gastrointestinal (GI) AEs. Time to resolution differed depending on the type of AE, with the shortest time being 1.36 (0.4–2.3) weeks (GI), and longest being 28.57 (3.1–30.0) weeks (pulmonary) (Supplemental Table S5).
TRsAEs in non-HBV patients.

Included events reported between first dose and 30 days after last dose of study treatment. All data shown as n (%) where applicable.
AE, adverse event; ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamma-glutamyl transferase; GI, gastrointestinal; HBV, hepatitis B virus; TRsAEs, treatment-related select adverse events.
With the exception of Grade 3–4 endocrine (diabetic ketoacidosis) and skin (rash) AEs, each experienced by one (5.9%) patient, there were no other Grade 3–4 and Grade 5 select AEs among HBV-infected NSCLC patients (Table 3). Time to onset of the high-grade endocrine and skin AEs was 69.0 weeks and 64.57 weeks, respectively. The skin AE resolved in 9.14 weeks, while the endocrine AE remained unresolved (Supplemental Table S6).
TRsAEs in HBV-infected patients.

Included events reported between first dose and 30 days after last dose of study treatment. All data shown as n (%) where applicable.
AE, adverse event; ALT, alanine aminotransferase; AST, aspartate aminotransferase; GI, gastrointestinal; HBV, hepatitis B virus; TRsAEs, treatment-related select adverse events.
Overall safety summary in all treated patients
Any-grade TRAEs occurred in 301 (75.3%) patients, including 288 (75.2%) non-HBV patients and 13 (76.5%) HBV-infected patients. Grade 3–4 TRAEs occurred in 60 (15%) patients, including 55 (14.4%) non-HBV patients and five (29.4%) HBV-infected patients. The most common TRAEs among all treated patients included increased alanine aminotransferase (ALT; 12.0%), hypothyroidism (12.0%), anemia (11.5%), increased aspartate aminotransferase (AST; 10.5%), rash (10.5%), decreased appetite (10.5%), malaise (10.3%), and fatigue (10.0%) (Supplemental Table S7).
Any-grade interstitial lung disease occurred in 11 (2.9%) and 1 (5.9%) of non-HBV and HBV-infected patients, respectively (0.8% and 0.0% were grade 3–4), while pneumonitis occurred in 3 (0.8%) of non-HBV patients (0.3% grade 3–4). There were no cases of pneumonitis among HBV-infected patients (Tables 2 and 3).
Grade 3–4 TRAEs resulted in discontinuation in nine (2.3%) patients, with the most common reason being interstitial lung disease, reported in two non-HBV patients (Table 4). Treatment-related death occurred in nine (2.3%) patients, including six (1.6%) non-HBV patients and three (17.6%) HBV-infected patients. The reported causes of death were malignant neoplasm progression and pneumonia in two patients each, and immune-mediated myocarditis, myocarditis, ventricular fibrillation, sudden death, and unknown death in one patient each (Supplemental Table S7). Treatment-related serious AEs of any grade occurred in 68 (17.0%) patients, including 61 (15.9%) non-HBV patients and seven (41.2%) HBV-infected patients, while treatment-related Grade 3–4 serious AEs occurred in 30 (7.5%) patients, including 27 (7.0%) non-HBV patients and three (17.6%) HBV-infected patients (Table 4). The most common treatment-related Grade 3–4 serious AEs were pneumonia (1.3%), interstitial lung disease (0.8%), diarrhea (0.5%), diabetes mellitus (0.5%), and diabetic ketoacidosis (0.5%) (Supplemental Table S8). Grade 3–4 serious interstitial lung disease or pneumonitis occurred in 3 (0.8%) and 1 (0.3%) non-HBV patient(s), respectively, while there were no cases of these Grade 3–4 serious AEs among HBV-infected patients.
Overall safety summary.

All data shown as n (%).
AE, adverse event; DC, discontinuation; HBV, hepatitis B virus; TRAE, treatment-related adverse event.
Overall efficacy
The overall median OS was 14.65 months (95% CI: 12.32–18.10) (Figure 1), and overall OS rates were 56% (95% CI: 51–61), 34% (95% CI: 29–39), and 29% (95% CI: 24–33) at 12, 24, and 30 months, respectively (Table 5). Median PFS was 3.61 months (95% CI: 2.33–3.75) (Figure 2), and the PFS rate at 12 months, 24 months, and 30 months was 19% (95% CI: 15–23), 10% (95% CI: 7–13), and 7% (95% CI: 5–11), respectively (Table 5). ORR was 15.00% (95% CI: 11.65–18.88), and median DOR was 19.38 (95% CI: 11.04–24.97) months (Table 5).

Kaplan–Meier plot of OS: all treated patients.
Key efficacy summary: all treated patients (secondary endpoints).

Indicates a censored value. Median (in months) was computed using the Kaplan–Meier method.
CI, confidence interval; CR, complete response; HBV, hepatitis B virus; NA, not assessable; OS, overall survival; PD, progressive disease; PFS, progression-free survival, PR, partial response; SD, stable disease.
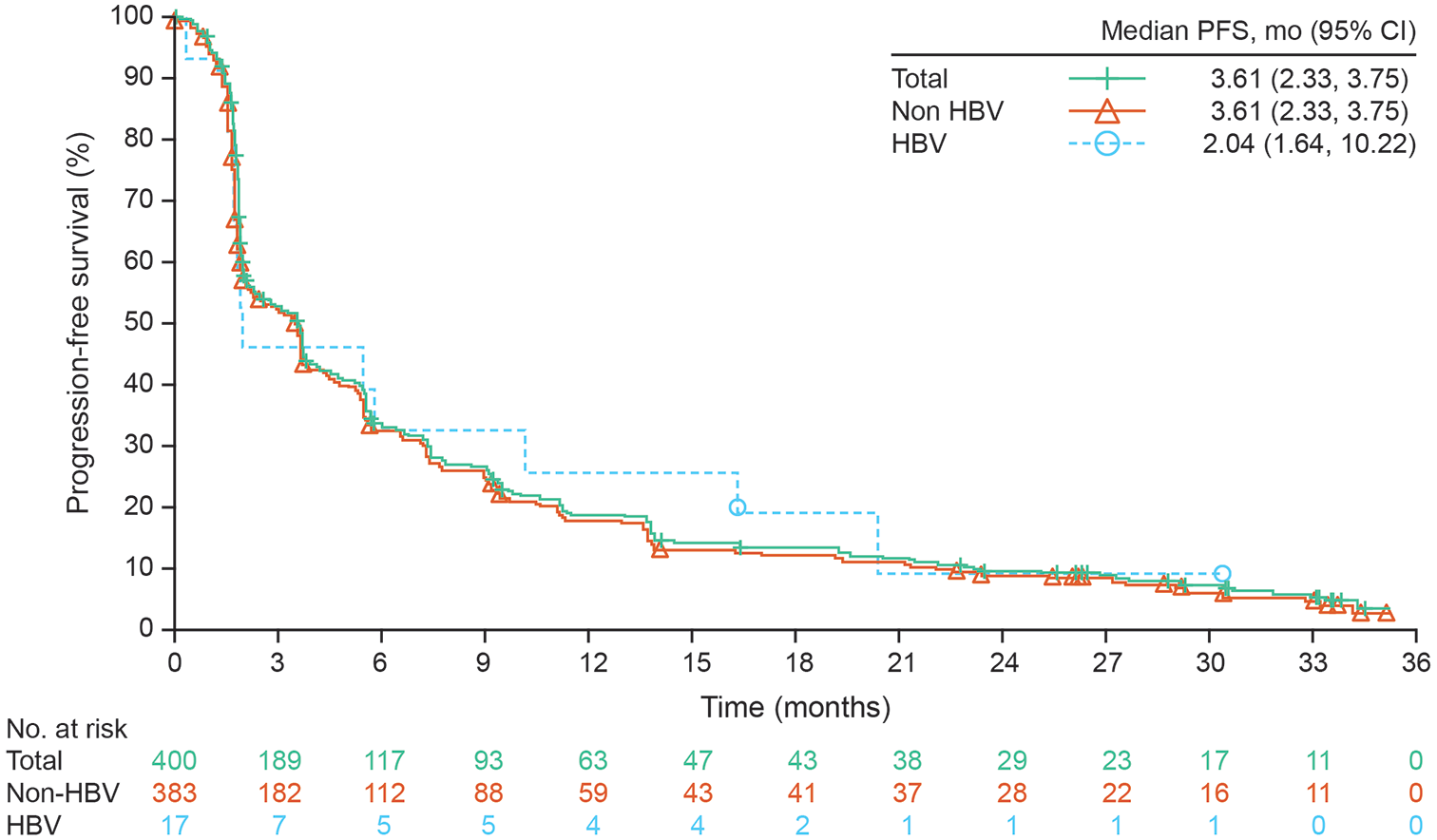
Kaplan–Meier plot of PFS per investigator assessment: all treated patients.
Efficacy in patient subgroups
The median OS was 14.6 months (95% CI: 12.25–18.07) and 22.31 months (95% CI: 10.02–NA) in non-HBV and HBV-infected patients, respectively (Figure 1). Median PFS was 3.61 months (95% CI: 2.33–3.75) and 2.04 months (95% CI: 1.64–10.22) in non-HBV and HBV-infected patients, respectively (Figure 2).
The median OS was 19.25 months (95% CI: 12.94–23.46) and 13.31 months (95% CI: 10.87–17.71) in patients with PD-L1 expression ⩾1% and < 1%, respectively (Supplemental Figure S2). The 12- and 24-month OS rates were 60% (95% CI: 52–67) and 52% (95% CI: 44–59) in patients with PD-L1 ⩾1%, and 54% (95% CI: 46–62) and 30% (95% CI: 23–38) in patients with PD-L1 <1%; 12- and 24-month OS rates were 58% (95% CI: 51–64) and 34% (95% CI: 28–40) in patients with PD-L1 ⩾50%, and 49% (95% CI: 34–62) and 25% (95% CI: 14–38) in patients with PD-L1 <50%, respectively (Supplemental Table S9). Median PFS was 4.73 months (95% CI: 3.68–5.65) and 2.33 months (95% CI: 1.94–3.68) in patients with PD-L1 ⩾1% and PD-L1 <1%, respectively (Supplemental Figure S3). At 12 and 24 months, patients with PD-L1 ⩾1% had PFS rates of 27% (95% CI: 20–34) and 16% (95% CI: 11–22), while patients with PD-L1 <1% had PFS rates of 14% (95% CI: 9–20) and 5% (95% CI: 2–9), respectively; the equivalent PFS rates in patients with PD-L1 ⩾50% and <50% were 28% (95% CI: 17–39) and 21% (95% CI: 12–32), and 18% (95% CI: 13–23) and 7% (95% CI: 4–11), respectively (Supplemental Table S10). The ORR (95% CI) was 25.60% (19.18–32.89) and 6.32% (3.20–11.03) in patients with PD-L1 ⩾1% and <1%, respectively (Supplemental Table S11).
The median OS in EGFR positive, EGFR WT, and EGFR undetermined subgroups was 19.32 (95% CI: 11.24–31.74), 13.70 (95% CI: 11.47–18.07), and 15.11 (95% CI: 11.56–19.81) months, respectively (Supplemental Figure S4). OS rates at 1 and 2 years, respectively, were 63% (95% CI: 44–77) and 49% (95% CI: 31–65) for patients who were EGFR positive, and 60% (95% CI: 25–83) and 40% (95% CI: 12–67) for patients who were ALK positive (Supplemental Table S9). Median PFS in EGFR positive, EGFR WT, and EGFR undetermined subgroups was 1.87 (95% CI: 1.71–4.73), 3.25 (95% CI: 2.07–3.68), and 5.36 (95% CI: 3.42–6.01) months, respectively (Supplemental Figure S5). The corresponding 12-month PFS rates were 19% (95% CI: 7–35), 8% (95% CI: 1–21), and 17% (95% CI: 12–22), and the corresponding 24-month PFS rates were 9% (95% CI: 6–14), 25% (95% CI: 16–34), and 11% (95% CI: 6–11), respectively (Supplemental Table S10). The ORR (95% CI) was 17.65% (95% CI: 6.76–34.53) in patients who were EGFR positive, and 12.93% (95% CI: 9.12–17.59) in patients who were EGFR WT (Supplemental Table S11).
PROs
Completion rates for EQ-5D-3L, EQ-VAS, and LCSS questionnaires at baseline were all 99.5% (381/383) for non-HBV patients, and 100% (17/17) for HBV-infected patients. Completion rates were also consistent between PRO instruments at 6, 12, and 24 months post-treatment. In non-HBV patients, completion rates were 94.1% (127/135), 100% (73/73), and 100% (37/37) at 6, 12, and 24 months, and in HBV-infected patients, completion rates were 100% (7/7), 100% (4/4), and 100% (2/2) at 6, 12, and 24 months, respectively. Completion rates were also high across EGFR and PD-L1 subgroups for all PRO instruments.
There was no notable change in LCSS, EQ-5D-3L, and EQ-VAS scores throughout the study among all treated patients and across patient subgroups (Supplemental Table S12).
Discussion
CheckMate 870 evaluated the efficacy and tolerability of flat-dose nivolumab in a predominantly Chinese population with advanced NSCLC who had progressed during or after one prior systemic therapy, including patients with HBV (HBsAg positive with HBV DNA ⩽500 IU/ml) and those who tested positive for EGFR mutation or ALK translocation. The occurrence of AEs in this study appeared consistent with the known safety profile of nivolumab, including the CheckMate 078 study, which utilized weight-based dosing. 8 High-grade TRsAEs were uncommon, and the most frequently reported were hepatic, pulmonary, and skin AEs in non-HBV patients, and endocrine and skin AEs in HBV-infected patients.
Based on the accumulated evidence for nivolumab safety, efficacy, and pharmacokinetics across a range of patient body weights, population pharmacokinetic modeling demonstrated that the benefit–risk profile of the 240 mg flat dose was comparable to a 3 mg/kg dosing regimen in patients with advanced tumors (including NSCLC), 16 including in Asian patients. 23 The CheckMate 870 study provides timely evidence for flat-dose nivolumab in a clinically relevant patient population. Considering these data and the improved convenience of flat dosing for both patients and physicians,16–20 flat-dose nivolumab appears to be a good choice for Chinese patients.
CheckMate 078 provided crucial evidence for the efficacy and safety of second-line nivolumab in a predominantly Chinese population, 8 but there is still a paucity of data on the effect of nivolumab, and immune checkpoint inhibitors more generally, in patients with NSCLC in China. As patients with EGFR mutation, ALK translocation, and HBV infection were excluded from the CheckMate 078 study, a substantial knowledge gap on the effect of nivolumab in these patient populations remained. This data deficit is particularly relevant to clinical practice in China given the higher prevalence of HBV infection and EGFR mutation in Chinese patients with NSCLC.10–12 Hence, CheckMate 870 included a more representative patient population to supplement the safety database of nivolumab-treated patients and better inform clinical practice in China.
Overall, the safety profile of flat-dose nivolumab was broadly consistent between HBV-infected and non-HBV populations. There was a higher frequency of Grade 3–4 and Grade 5 treatment-related AEs in HBV-infected patients compared with non-HBV patients. However, it is unknown to what degree the small sample size of HBV-infected patients contributed to these results.
Current evidence for the safety of immune checkpoint inhibitors in patients with NSCLC and hepatitis B infection report mixed results with respect to hepatic AEs.24–26 In this study, any-grade hepatic AEs were observed in a similar proportion of non-HBV and HBV-infected patients following treatment with nivolumab. Increased ALT and AST were reported in 12.0% and 10.5% of patients overall, respectively, and none were Grade 3–5. Furthermore, the frequency of ALT and AST increases in patients with HBV infection was lower or comparable to that in non-HBV patients. The majority of hepatic AEs were managed successfully with supportive care, including all hepatic TRsAEs reported in HBV-infected patients, which resolved in a median 4.4 weeks. There are limited available data on HBV reactivation in patients with NSCLC, although a study from Sun Yat-sen University Cancer Center, China reported no cases of HBV reactivation among patients with NSCLC with HBV infection treated with anti-PD-1/PD-L1 therapies (including nivolumab) who did not discontinue antiviral therapy. 24 HBV reactivation in patients with HBV infection was not assessed throughout the CheckMate 870 study, and so further studies are needed to confirm the safety of nivolumab in this population.
Due to prior pulmonary dysfunction, patients with NSCLC may be more susceptible to pulmonary AEs. A meta-analysis of incidence and risk of PD-1-inhibitor-related pneumonitis in cancer patients, comprising 6360 patients from 16 phase II/III studies, found that anti-PD-1 immunotherapy increased the risk of pneumonitis compared with routine chemotherapy, with NSCLC patients among those at highest risk. 27 The frequency of pulmonary AEs may also be higher in Asian compared with non-Asian populations. 28 Moreover, EGFR mutation, which is more prevalent in Asian patients with NSCLC, has been shown to increase the risk of immune-related AEs including pneumonitis. 29 Treatment-related pulmonary AEs were reported in 4%, 5%, and 7% of patients treated with nivolumab in CheckMate 017, 057, and 078 studies, respectively. In the present study, treatment-related select pulmonary AEs occurred in 4.4% and 5.9% of non-HBV and HBV-infected patients, respectively, and were implicated in two cases of treatment discontinuation and one death. While pulmonary AEs were not overrepresented in this study, it remains unclear whether Asian patients are more susceptible to pulmonary AEs than non-Asian patients.
The efficacy profile of nivolumab in this study appears consistent with pivotal trials in international and Asian populations: overall median OS was 12.0, 9.2, and 12.2 months in the CheckMate 078, 8 017, 30 and 057 trials, 31 respectively. A systematic review and meta-analysis on the value of PD-L1 expression in predicting efficacy of anti-PD-1/PD-L1 therapy in cancer patients reported that anti-PD-1/PD-L1 therapy correlated with prolonged survival in patients with NSCLC and PD-L1 ⩾1%.32 In this study, we observed a numerically higher ORR, longer median OS, and longer median PFS in nivolumab-treated patients with higher PD-L1 expression (PD-L1 ⩾1% versus PD-L1 <1%). A similar observation was made in CheckMate 057, 31 where the ORR was numerically higher in nivolumab-treated patients with PD-L1 ⩾1% versus <1% [31% (95% CI: 23–40) versus 10% (95% CI: 5–16)], and the median OS and median PFS were longer in patients with PD-L1 ⩾1% versus <1% (OS: 17.7 versus 10.5 months; PFS: 4.2 versus 2.1 months).
Efficacious second-line treatment options are limited for patients who progress on or after first-line therapy. For example, in the 2-year follow-up of the CheckMate 078 study, Asian patients treated with second-line docetaxel achieved a median (95% CI) OS of just 9.6 (7.6–11.2) months. The efficacy of second-line TKI therapy in patients with mutation-positive NSCLC may also be attenuated compared with first-line therapy. 33 Recent evidence suggests that immune checkpoint inhibitors may provide clinical benefit regardless of EGFR/ALK mutation status in certain patients.34–36 In this study, 63% and 49% of EGFR-positive patients, and 60% and 40% of ALK-positive patients, were alive at 1 and 2 years, respectively. Moreover, 1- and 2-year survival rates were 71% and 43%, respectively, in B-RAF-positive patients. No clear association was observed between OS rates in patients with detectable tumor, MET, KRAS, or ROS activating gene mutation. Based on evidence available to date, Chen and colleagues proposed that in EGFR positive patients with TKI-resistant disease who have a low number of previous treatments, immune checkpoint inhibitor therapy may be a preferred treatment option in the future. 35 Additional data from future clinical trials is required to confirm the potential role of immune checkpoint inhibitors in the treatment of patients with EGFR-positive NSCLC.
Overall, in CheckMate 870, second-line nivolumab monotherapy offered a durable efficacy benefit independent of HBV status, PD-L1 expression, EGFR mutation, and ALK translocation. We additionally collected data on PROs during nivolumab treatment and during the 2-year follow-up period as part of an exploratory analysis. NSCLC has a substantial impact on patients’ quality of life and health-related utility, particularly for those with advanced disease, disease progression, and those requiring subsequent lines of therapy. 37 Across all PRO measures, we observed no notable changes from baseline through treatment and follow-up among HBV, EGFR, and PD-L1 patient subgroups, providing further support for nivolumab as a second-line treatment option in patients with advanced NSCLC. It should be noted that PRO data were summarized by nominal visits and were not analyzed by longitudinal modeling, and no adjusted mean change was reported.
Considering the limited number of HBV-infected patients in this study (n = 17), more evidence is needed to further discuss the efficacy and safety of nivolumab in patients with HBV infection. In addition, real-world data will be critical in verifying the efficacy and safety of second-line nivolumab in these patients, including other special populations such as EGFR/ALK positive. This study was initiated prior to immuno-oncology regimens becoming standard of care in first-line treatment of advanced/metastatic NSCLC, and it excluded patients who were previously treated with immune checkpoint inhibitors. Consequently, the optimal treatment strategy for these patients remains a substantial unmet need. Immune checkpoint inhibitor and chemotherapy combinations are more frequently being used in first-line treatment, and further studies are needed to establish the role of second-line nivolumab, and immune checkpoint inhibitors more broadly, in these patients.
In conclusion, the long-term results from CheckMate 870 demonstrated that flat-dose nivolumab 240 mg administered as a 30-min infusion Q2W was well tolerated and efficacious, and maintained health status and quality of life in Asian patients with advanced NSCLC, regardless of HBV, EGFR/ALK, and PD-L1 status. Nivolumab 240 mg flat dose also appeared to show similar efficacy and safety to weight-based nivolumab dosing (3 mg/kg Q2W) in the CheckMate 078 trial. Moreover, no new safety signals were observed in the long-term follow up.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221138380 – Supplemental material for An open label, safety study of Asian patients with advanced non-small-cell lung cancer receiving second-line nivolumab monotherapy (CheckMate 870)
Supplemental material, sj-docx-1-tam-10.1177_17588359221138380 for An open label, safety study of Asian patients with advanced non-small-cell lung cancer receiving second-line nivolumab monotherapy (CheckMate 870) by Shun Lu, Ying Cheng, Jianying Zhou, Mengzhao Wang, Jun Zhao, Baocheng Wang, Gongyan Chen, Jifeng Feng, Zhiyong Ma, Lin Wu, Changli Wang, Kewei Ma, Shucai Zhang, Jun Liang, Yong Song, Jie Wang, Yi-Long Wu, Ang Li, Yizhi Huang and Jianhua Chang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors thank the patients and families who made this trial possible; the investigators and the clinical study teams who participated in the trial; the protocol manager for this study, Qian Feng; Dako for collaborative development of the PD-L1 IHC 28-8 pharmDx assay; Bristol-Myers Squibb (Princeton, New Jersey); and ONO Pharmaceutical Company Ltd (Osaka, Japan). Professional medical writing and editorial assistance was provided by Alice Carruthers, BSc (Hons), PhD, and Juanyu Piao, PhD, of Nucleus Global, and funded by Bristol-Myers Squibb, China.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
