Abstract
Brain metastasis of nasopharyngeal carcinoma (NPC) is a rare event with limited research. In this report, we discuss the details of a case of brain metastasis from NPC that presented with a solitary, cystic lesion in the frontal lobe. We also reviewed the related literature published in the last 20 years. We analyzed the patient’s clinical characteristics, which indirectly suggested hematogenous spread rather than a cerebrospinal fluid route. Although there are no standard treatments for brain metastasis of NPC, previous studies reported that combined surgery and radiotherapy was a good treatment option, with long survival. Our patient achieved intracranial complete response after the combination of conventional chemoradiotherapy and novel immunotherapy. This treatment option could be useful in future similar cases.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a rare malignancy in Europe and the United States; however, this cancer is endemic in Southern China. 1 The world age-standardized incidence of NPC was 3.0 per 100,000 and 0.4 per 100,000 people in China and Western countries, respectively, in 2018. 2 Radiotherapy is the most powerful treatment modality for NPC. 3 Unfortunately, approximately 15% to 30% of patients with NPC develop metastasis after initial treatment. 4 The common metastatic sites of NPC are bone, lung, and liver. Brain metastasis is extremely rare, although intracranial invasion by direct extension from the nasopharynx through the skull base in patients with locally advanced disease is not uncommon. 5 In this report, we present a case of NPC with solitary, cystic brain metastasis confirmed by pathology. The diagnosis was confirmed after synchronous chemoradiotherapy in our hospital. Intracranial complete response (CR) was achieved after the patient received conventional chemoradiotherapy and novel immunotherapy. We also reviewed previous reports of central nervous system (CNS) metastases from NPC to gain insight into this uncommon phenomenon.
Case report
A 50-year-old man from Guangdong Province, China, was admitted to our hospital in February 2018, with the complaint of persistent left nasal cavity bleeding and hearing impairment. Nasopharyngeal fiber optic endoscopy and histopathological biopsy revealed a poorly differentiated nonkeratinizing NPC. Magnetic resonance imaging (MRI) was performed and showed an irregularly-shaped tumor located mainly in the left lateral nasopharynx, extending to the left parapharyngeal space, with multiple lymph node metastases in the left neck. Therefore, the patient was diagnosed with stage II (T2N1M0) NPC based on the criteria of the American Joint Committee on Cancer/Union Internationale Contre le Cancer (AJCC/UICC) 7th edition. 6 He then received chemoradiotherapy using intensity-modulated radiation therapy (IMRT), concomitant with one cycle of chemotherapy with paclitaxel and cisplatin (TP), and three cycles chemotherapy with cisplatin weekly.
The patient remained well until 12 months after the conclusion of radiotherapy, when he suddenly developed right chest pain and bloody sputum. Multiple lung metastases were identified with chest computed tomography (CT). Therefore, six cycles of palliative chemotherapy with a cisplatin-based regimen were administered to control the lung metastases. This was followed by anlotinib (12 mg on days 1–14) for maintenance treatment. Additionally, lung lesion stereotactic body radiotherapy (SBRT) was commenced in May 2021 (40 Gy/5 fractions).
Three months after lung SBRT, routine MRI of the nasopharynx revealed a solitary, cystic brain lesion in the left frontal lobe; contrast-enhanced MRI confirmed the lesion (Figure 1a and b). The patient refused intracranial neoplasm resection; therefore, stereotactic biopsy with CT guidance was immediately performed to clarify the histological diagnosis. The pathological findings were consistent with the results of the nasopharyngeal biopsy (Figure 2a and b). Epstein–Barr virus-encoded RNA (EBER) staining using in situ hybridization was positive in the brain lesion (Figure 2c), which further confirmed brain metastasis from NPC. The patient then received immunotherapy and whole-brain radiotherapy (WBRT) (total radiation dose: 30 Gy/10 fractions). The brain lesion and the multiple enhanced sites along the puncture needle tract demonstrated by MRI (Figure 3a) received 45 Gy radiotherapy in 15 fractions. Nearly 2 months after WBRT, brain lesion CR was achieved (Figure 3b). The patient was still alive with continued intracranial CR at the last follow-up (June 2022).

A solitary, cystic lesion with rim enhancement in the left frontal lobe in T1-weighted axial (a) and sagittal (b) MRI.
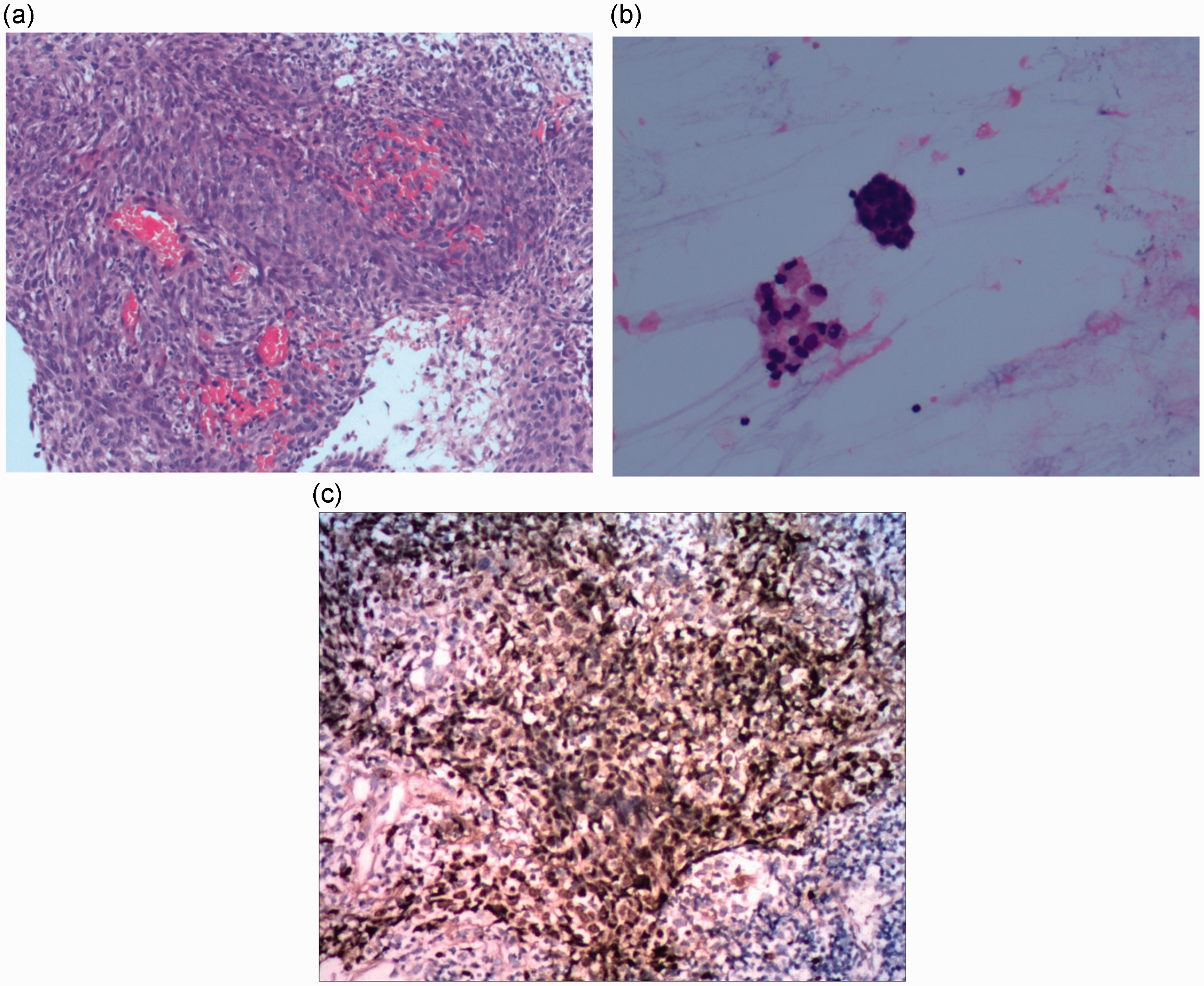
Pathological examination of the cystic wall (a) of the lesion. The cystic fluid smear examination (b) shows the presence of tumor cells (hematoxylin and eosin, original magnification ×200). (c) Photomicrograph showing positive Epstein–Barr virus-encoded RNA staining in tumor cells (in situ hybridization, original magnification ×200).

T1-weighted sagittal MRI images with contrast. (a) Multiple well-enhanced sites along the needle tract; (b) Intracranial complete response after combined therapy.
Discussion
Distant metastasis is the main treatment failure in NPC; however, metastasis to the brain is extremely rare, ranging from an incidence of 0.3% to 0.7% in previous reports.7,8 A PubMed search of cases of brain metastasis from NPC in the last 20 years is summarized in Table 1.4,5,7,9–12 It is generally acknowledged that lymphatic and hematogenous spread are the main routes of NPC metastasis. 11 Tumor cell spread frequently occurs hematogenously via the circulatory system, primarily to the liver, lung, and bone. 9 In our institution, most patients present with lung, bone, or other organ metastases preceding brain metastasis after the completion of the primary treatment. Based on our experience, we consider that brain metastases from NPC may be more likely to disseminate hematogenously. In addition to the hematogenous route, spread through the CSF is also considered a possible pathway for CNS metastasis of NPC because tumor cells may enter the CSF by invading the skull base and disrupting the dura. 11 Shen et al. 10 reported a woman who developed multiple CNS metastases without distant metastases to the common metastatic sites of NPC during a 20-month follow-up, suggesting that CSF spread was most likely. The findings in Shen et al.’s 10 case were supported by the observations of Ngan et al. 5 who found an absence of other organ involvement more than 3 years after a diagnosis of intradural spinal metastasis in an NPC patient. However, NPC cell spread to the brain through the CSF does not occur frequently. Even though cases of spinal cord involvement have been reported, these patients did not have synchronous brain metastases.13,14 In our patient, the initial primary nasopharyngeal tumor extended only to the left parapharyngeal space and not to the skull base and cavernous sinus. Before being confirmed as having brain metastasis, the patient received palliative therapy for lung metastases over 2 years. The clinical characteristics in this case indirectly suggest hematogenous spread rather than spread via CSF.
Summary of previous studies reporting brain metastases of NPC.

The sites of brain metastasis of NPC can include the frontal and temporal lobes, and even the cerebellum; 12 however, the most commonly reported site in the literature is the occipital lobe. Ngan et al. 5 speculated that there may be a particular microenvironment that promotes circulating NPC cells to settle in the occipital lobe. Nevertheless, there is no acknowledged hematological or lymphatic pathway directly linking the nasopharynx and occipital lobes, and more research is needed to elucidate the exact mechanism of action. Although solid lesions were the main morphology of NPC brain metastases in previous reports, cystic lesions are not rare.4,5,7,10–12 It is difficult to distinguish radiation encephalopathy (RE) and metastasis by conventional MRI because cystic necrosis is a form of RE, 4 similar to the characteristics of cystic brain metastasis. Therefore, when an NPC patient presents with cystic RE in the radiation field, brain metastasis should be considered a differential diagnosis.
The prognosis of cancer patients who develop brain metastasis is poor. A meta-analysis 15 of studies of brain metastasis showed that the median survival of the best prognosis group was 7.1 months while that of the worst prognosis group was only 2.3 months. Currently, there are no standard treatments for brain metastasis of NPC. Considering that the majority of cases present with a solitary brain lesion, local treatment comprising surgery or radiotherapy may be the most effective and direct treatments for brain metastasis of NPC. Zeng et al. 11 reported a 44-year-old man who was diagnosed with temporal lobe metastasis of NPC following combined treatment with surgery and IMRT. CR of the brain lesion was eventually achieved, and the patient survived for over 17 months after the confirmation of brain metastasis. Similarly, Shen et al. 10 described a patient who achieved 31-months’ overall survival after undergoing intracranial neoplasm resection and irradiation. However, surgical resection alone might be insufficient to treat brain metastases from NPC because of poor disease control. There are two reported cases4–5 of brain metastasis from NPC treated with surgery alone and both developed progression within 6 months of treatment. WBRT has been widely used for the management of brain metastasis. However, some scholars prefer stereotactic radiosurgery (SRS) to WBRT because the former results in less decline in cognitive function. 16 As NPC is sensitive to ionizing radiation, SRS can be recommended as a salvage option to address brain metastasis of NPC. Park et al. 12 reported three cases of brain metastasis from NPC treated with SRS and two achieved a significant decrease in tumor size within 3 months; however, the long-term effect was unsatisfactory. Based on these cases, it appears that combined therapy with surgery and radiotherapy is a more appropriate approach compared with either therapy alone. Our patient had fears regarding the adverse effects of craniotomy, and he refused intracranial neoplasm resection but accepted stereotactic biopsy. Importantly, we observed multiple well-enhanced signals along the needle tract from the initial CT-guided biopsy in brain MR images. These imaging changes should not be excluded simply as postoperative changes, and implantation metastasis should be considered a differential diagnosis. To our knowledge, this is the first report of suspected needle tract implantation after stereotactic biopsy in brain metastasis from NPC. Because of concerns regarding the needle tract lesions, our patient underwent WBRT instead of SRS after stereotactic biopsy.
The main cause of death in cancer patients with brain metastasis is not the brain lesion itself, but progression of systemic disease. 16 Therefore, systemic treatment should be considered seriously in NPC patients who develop brain metastases, especially in patients with other organ involvement concurrently. Unlike patients in previous studies, in addition to radiotherapy, our patient also received chemotherapy, WBRT, and immunotherapy with camrelizumab, an immune checkpoint inhibitor that is widely used in metastatic NPC. 17 As of June 2022, CR of the brain lesion had been achieved for 6 months without obvious neurological impairment, and the lung metastases remained stable under immunotherapy alone.
In conclusion, NPC is not a tumor with brain tropism, and the underlying mechanisms of metastasis to this uncommon site remain unclear. Hematogenous NPC cell spread to the CNS may be the most probable route, as opposed to the CSF, especially for patients with earlier initial tumor T stage and other organ involvement. Although there is no standard treatment for brain metastasis of NPC, the combination of surgery and radiotherapy appears best. Given the satisfactory short-term effects achieved with combined therapy in our patient, we suggest this alternative regimen to treat this rare brain metastases in similar cases, until a better treatment mode is identified.
Footnotes
Acknowledgements
We thank Dongguan People's Hospital for support with data collection.
Author contributions
Liji Jiang and Chun Zhang made substantial contributions to the study conception and design, acquisition of data, or the analysis and interpretation of the data. All authors reviewed and approved the final version of the article.
Availability of data and material
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
CARE checklist (2013) statement
The authors have read the CARE checklist (2013), and the authors confirm that the reporting of this study conforms to the CARE guidelines. 18
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
Ethical approval is not required for case reports by the Medical Ethics Committee of Dongguan People's Hospital, Guangdong, China. Written informed consent was obtained from the patient for publication of this case report and any accompanying images. Consent for treatment was obtained from the patient.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
