Abstract
Background:
The efficacy and safety of human epidermal growth factor receptor 2 (HER2)-targeted therapy and androgen deprivation therapy (ADT) for locally advanced or recurrent or metastatic (LA/RM) salivary duct carcinoma (SDC) have been reported in prospective studies. However, the survival benefit of these therapies to conventional therapy remains controversial, and whether HER2-targeted therapy or ADT should be chosen in HER2- and androgen receptor (AR)-positive SDC patients remains unknown.
Methods:
Overall, 323 LA/RM SDC patients treated at seven institutions between August 1992 and June 2020 were retrospectively enrolled. The primary aim was to analyze the effect of HER2-targeted therapy and ADT on overall survival from the diagnosis of LA/RM disease to death from any cause (OS1). The secondary indicators included the overall response rate (ORR), clinical benefit rate (CBR), overall survival from therapy initiation for LA/RM disease (OS2), progression-free survival (PFS), time to second progression (PFS2), duration of response (DoR), and duration of clinical benefit (DoCB) of HER2-targeted therapy or ADT as first-line therapy for HER2-positive/AR-positive SDC.
Results:
Patients treated with HER2-targeted therapy or ADT had longer OS1 than those treated without these therapies (Median OS1: historical control, 21.6 months; HER2-targeted therapy, 50.6 months; ADT, 32.8 months; HER2-targeted therapy followed by ADT, 42.4 months; and ADT followed by HER2-targeted therapy, 45.2 months, p < 0.001). Among HER2-positive/AR-positive SDC patients, although HER2-targeted therapy had better ORR, CBR, and PFS than those of ADT as first-line therapy, we found no significant differences between HER2-targeted therapy and ADT regarding OS2, PFS2, DoR, and DoCB.
Conclusion:
Patients treated with HER2-targeted therapy and ADT showed longer survival in LA/RM SDC. HER2-targeted therapy can be recommended prior to ADT for HER2-positive/AR-positive SDC. It is warranted to establish a biomarker that could predict the efficacy of clinical benefit or better response in ADT.
Keywords
Introduction
Salivary gland cancer (SGC) is a rare condition that accounted for 0.3% of the total cancer cases and 0.2% of the total cancer deaths worldwide in 2020. 1 According to the World Health Organization classification, there are more than 20 histopathological subtypes of SGC, and their biological behaviors are different. 2 Among them, salivary duct carcinoma (SDC) is defined as an adenocarcinoma, which histologically resembles invasive breast ductal carcinoma. SDC, which accounts for 10–20% of all salivary gland malignancies and is one of their most aggressive forms, is frequently associated with the development of distant metastases.3,4 The standard treatment for resectable SDC is radical surgery, including neck dissection followed by postoperative radiotherapy.4,5 The median overall survival (OS) of patients with SDC with distant metastases who received the best supportive care (BSC) was approximately 5 months. 6 Furthermore, the United States National Cancer Database indicated that patients with Stage IVC SDC had a median OS of 21 months, and no patient survived for 5 years. 7 Even in the recent Surveillance Epidemiology and End Results database, the 5-year survival rate for M1 cases at initial presentation was 0%. 8 Furthermore, there are no reports of improved prognosis of SDC in recent years compared to those in the sieve era.8,9
Although systemic therapy has been developed for metastatic SGC, its efficacy remains unclear.7,10–12 The overall response rate (ORR) of combination therapy with platinum and taxane for metastatic SDC was 40–50%, and the median OS in Japanese studies was 10–20 months.13–15 Recently, the efficacy of immune checkpoint inhibitors (ICIs) has been shown in SGC cases; however, its efficacy in patients with SDC has not been fully elucidated.16,17 We reported that the ORR of nivolumab was 5%, and the median OS was 11.5 months among patients with SDC in a retrospective setting. 18
Most SDCs express androgen receptor (AR), and approximately 40% are positive for human epidermal growth factor receptor 2 (HER2).4,19,20 The efficacy and safety of HER2-targeted therapy and androgen deprivation therapy (ADT) in patients with locally advanced (LA) and/or recurrent or metastatic (RM) SDC, in accordance with the treatment strategies for HER2-positive breast cancer and AR-positive prostate cancer, have been evaluated in prospective and retrospective studies (Table 1).6,21–33 These therapies are recommended by the latest National Comprehensive Cancer Network (NCCN) 34 and American Society for Clinical Oncology (ASCO) 35 guidelines. However, as results from randomized phase III studies comparing HER2-targeted therapy or ADT to conventional therapy are not available, because of the rarity of SDC, the superiority of HER2-targeted therapy or ADT over conventional therapy remains controversial.10,36,37 The latest NCCN guidelines mentioned that there are no preferred regimens. 34 The ASCO guidelines also described that no single-agent or combination therapy has been shown to have a survival advantage, and there are no randomized trials comparing treatment with supportive care alone. 35
Summary of literature reports on HER2-targeted therapy and ADT for salivary gland carcinoma.

Unconfirmed partial response.
Castration-resistant, androgen receptor-expressing salivary gland carcinoma.
Interquartile range.
14 (70%); Bicalutamide + leuprorelin, 1 (5%); leuprorelin, 2 (10%); bicalutamide, 3 (15%); enzalutamide.
Results of total 27 cases treated with ADT as first-line therapy or after the second-line therapy.
ADT, androgen deprivation therapy; CI, confidence interval; HER2, human epidermal growth factor receptor 2; LHRH, luteinizing hormone-releasing hormone analogs; NA, not available; NR, not reached; ORR, overall response rate; OS, overall survival; PFS, progression-free survival.
Furthermore, selecting appropriate treatment for HER2-positive/AR-positive patients with SDC, who account for 21–62% of patients with SDC,19,38,39 is another unresolved issue.10,12,32,33,36,37 Compared to the previously recommended systemic therapy, a high response rate for HER2-targeted therapy and more tolerable adverse events for ADT can be expected, although this is an indirect comparison.
Therefore, this retrospective study aimed to elucidate the survival benefit of HER2-targeted therapy or ADT compared to that of historical control, and the impact of HER2-targeted therapy or ADT on HER2-positive/AR-positive patients with SDC.
Materials and methods
Patients and tumor assessment
This study was conducted in accordance with the Declaration of Helsinki and was approved by each Institutional Review Board listed in Supplemental Table S1. Data were obtained from each institutional database following Institutional Review Board approval. Written informed consent for the participation and publication of this study was obtained from all patients included in this study. Patients who were deceased or did not revisit for examination were also included. Therefore, we provided information regarding the research plan via a web-based public release, and patients and their families were provided an opportunity to opt-out of the study in accordance with the policy of the Japanese government.
We retrospectively evaluated LA/RM patients with SDC who were treated at seven Japanese institutions between August 1992 and June 2020. Moreover, we performed restaging according to the UICC TNM classification, eighth edition. LA SDC was defined as an unresectable tumor fulfilling at least one of the following criteria: (a) T4b primary lesion and (b) cervical lymph node metastasis invading the carotid artery.22,29,35 Several patients in this study have been included in previous studies.14,18,19,22,29,48,51
All included patients had SDC that was histopathologically confirmed by expert pathologists according to rigorous histomorphological criteria for SDC. 3 Tumors were also assessed to determine the HER2 and AR status.22,29 HER2 positivity was defined as immunohistochemical 3+ or the presence of HER2 gene amplification detected by fluorescence in situ hybridization, according to the ASCO/College of American Pathologists guidelines for breast cancer. 40 If a minimum of 1% of tumor cell nuclei were immunoreactive, the tumor was considered positive for AR. 29
HER2-targeted therapy and ADT and tumor assessment
HER2-targeted therapy and ADT were performed at the International University of Health and Welfare, Mita Hospital. We previously described details of the HER2-targeted therapy [trastuzumab and docetaxel (Tmab/DTX)] and ADT [combined androgen blockade (CAB) therapy with leuprorelin acetate and bicalutamide] regimens.22,29 Briefly, for Tmab/DTX therapy, a loading dose of 8 mg/kg of trastuzumab was administered intravenously, followed by a dose of 6 mg/kg once every 3 weeks. Docetaxel (70 mg/m2) was administered intravenously once every 3 weeks. For CAB therapy, leuprorelin acetate (11.25 mg) was administered subcutaneously every 12 weeks. A dose of 3.75 mg every 4 weeks or 22.5 mg every 24 weeks was permitted if the patient desired. Bicalutamide was administered orally at a daily dose of 80 mg.
Tumor assessments were performed within 4 weeks before the initiation of Tmab/DTX and CAB therapy (using computed tomography or magnetic resonance imaging) and were repeated every 6–8 weeks until disease progression (PD), death, or up to 2 years after treatment initiation. Thereafter, the assessment was continued every 3 months in surviving patients. Patient response was determined based on the Response Evaluation Criteria in Solid Tumors (version 1.1). 41
Data collected and outcomes
Data on patient demographic characteristics, Charlson Comorbidity Index (CCI), 42 and treatment modalities (systemic therapy, surgery, and radiation therapy for LA/RM disease) were collected. According to the CCI, patients with RM disease were assigned a score of 6 points.
The primary objective was the effect of HER2-targeted therapy and ADT on overall survival (OS1), defined as the time from the diagnosis of LA/RM disease to death from any cause or the last follow-up. In this study, we included patients treated with Tmab/DTX, CAB, or both. In addition, among patients receiving both therapies, those treated with Tmab/DTX before CAB and those treated with CAB before Tmab/DTX were also included. Furthermore, it remains unclear whether there is a significant difference in prognosis in patients treated with both Tmab/DTX and CAB, depending on the prior treatment. Therefore, the patients were divided into the following five groups according to the presence or absence of Tmab/DTX and CAB during the entire period of treatment for LA/RM SDC, to elucidate the survival benefit from these therapies compared to that of conventional therapy: historical control (surgery ± radiotherapy, radiotherapy, cytotoxic chemotherapy, ICIs, and BSC), Tmab/DTX followed by CAB (with/without conventional chemotherapy), CAB followed by Tmab/DTX (with/without conventional chemotherapy), Tmab/DTX without CAB (with/without conventional chemotherapy), and CAB without Tmab/DTX (with/without conventional chemotherapy) groups.
The secondary objectives included the comparison of the following parameters between Tmab/DTX and CAB therapy as first-line systemic therapy for HER2-positive/AR-positive patients with SDC: ORR [the proportion of patients who experienced a best overall response of complete response (CR) or partial response (PR)], clinical benefit rate (CBR) [the proportion of patients who experienced a best overall response of CR, PR, or stable disease (SD) for ⩾24 weeks], OS2 (the time from initiation of Tmab/DTX or CAB therapy to death from any cause or last follow-up examination), progression-free survival (PFS) (the time from the start of Tmab/DTX or CAB therapy to PD), PFS2 (the time from the start of Tmab/DTX or CAB therapy to PD after Tmab/DTX, CAB, or combination therapy with Tmab/DTX and CAB), duration of response (DoR) [the time from initiation of Tmab/DTX or CAB therapy to progression (or death from any cause) in patients who had a CR or PR], and duration of clinical benefit (DoCB) [the time from initiation of Tmab/DTX or CAB therapy to progression (or death from any cause) in patients who had a CR, PR, or SD for ⩾24 weeks].
Statistical analysis
The association between systemic therapy and survival parameters was evaluated using the Kaplan–Meier product limit method, log-rank test, and univariate and multivariate Cox proportional hazards models. The measure of association in this study was the hazard ratio (HR) with 95% confidence intervals (CIs). In addition, we estimated the odds ratio (OR) and 95% CI to evaluate the association between treatment modality and response rate using unconditional univariate and multivariate logistic regression models. Potential confounders considered in the multivariate analysis were age, sex, CCI, Eastern Cooperative Oncology Group performance status (ECOG-PS), primary site, disease status (LA/RM), HER2 status, AR status, and surgery and radiotherapy for LA/RM disease.
All statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS) for Windows (versions 23 and 25) (IBM Corp., Armonk, NY, USA). All tests were two-sided, and p < 0.05 was considered statistically significant.
Results
Demographic characteristics
Between August 1992 and June 2020, 424 patients with SDC were treated at the seven institutions. The data cutoff date was on 30 October 2020. Of the 424 patients, the following were excluded from this study: 92 with no evidence of disease after definitive treatment, four with an unknown outcome, three with data for <3 months after definitive treatment, and two who did not complete the first-line treatment for LA/RM disease. Finally, 323 patients (275 men and 48 women) with LA/RM SDC (31 and 292 with LA and RM disease, respectively) were enrolled in this study (Figure 1, Table 2). The median age was 64 years (26–90 years). The median follow-up period of the 323 patients was 26.4 (range: 0.7–131.0) months, while the median OS1 was 35.6 (95% CI: 29.4–41.8) months.
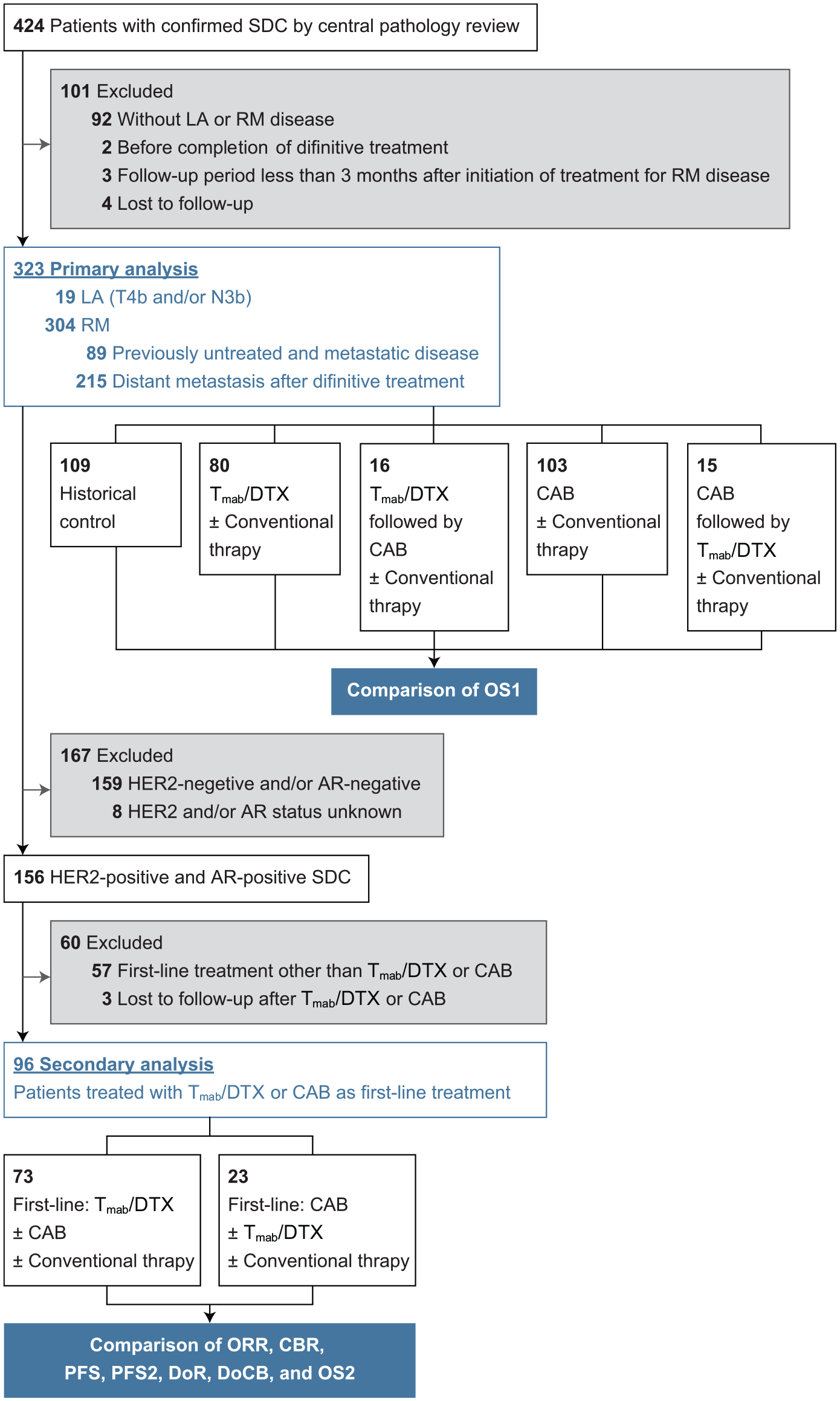
Flow diagram of study cohort selection steps.
Patient characteristics.

Excludes docetaxel combined with trastuzumab.
AR, androgen receptor; BSC, best supportive care; CAB, combined androgen blockade; CCI, Charlson Comorbidity Index; ECOG-PS, Eastern Cooperative Oncology Group performance status; HER2, human epidermal growth factor receptor 2; ICIs, immune checkpoint inhibitors; LA, locally advanced; RM, recurrent or metastatic; Tmab/DTX, trastuzumab and docetaxel.
Altogether, 109 patients were classified into the historical control group, including surgery (n = 33), radiotherapy (n = 39), and BSC only (n = 24) (Table 2). Patients who received Tmab/DTX or CAB initiated treatment in March 2012 or later, whereas the historical control group included patients whose treatment started 20 years earlier. First, we examined whether there was a difference in prognosis according to the timing of treatment in the 109 patients of the historical control group. For this reason, OS1 was analyzed by dividing the patients into two groups: the Pre-Tmab/CAB group (55 patients, RM diagnosis date: 9/14/1992–12/26/2011) and the Post-Tmab/CAB group (54 patients, RM diagnosis date: 5/9/2012–3/25/2020). The results showed that the median OS1 was 20.2 months in the Pre-Tmab/CAB group (95% CI: 13.8–23.0) and 22.7 months in the Post-Tmab/CAB group (95% CI: 17.5–34.6) (p = 0.212). As there was no significant difference in prognosis in the historical control group in this cohort, we judged that this group could be combined into one group and compared its findings with those of other groups.
As systemic treatment, Tmab/DTX and CAB was administered to 111 and 134 patients, respectively. Especially, there were 16 patients who received Tmab/DTX followed by CAB and 15 patients who received CAB followed by Tmab/DTX (Table 2, Figure 1). The ORR and CBR of all 111 patients treated with Tmab/DTX were 72.1% (95% CI: 62.9–79.7%) and 83.8% (95% CI: 75.6–89.6%), respectively, and those of all 134 patients treated with CAB were 28.4% (95% CI: 21.3–36.6%) and 47.8% (95% CI: 39.4–56.3%), respectively (online-only Supplemental Table S2). The median durations of Tmab/DTX and CAB treatments were 16.5 (range: 1.4–97.9) and 8.5 (range: 0.9–79.8) months, respectively.
Comparison of OS in patients with SDC who received HER2-targeted therapy, ADT, or conventional therapy
In comparison to patients treated with conventional therapy, those treated with Tmab/DTX or CAB had a significantly longer median OS1 [historical control: 21.6 (95% CI: 18.3–25.0) months; Tmab/DTX: 50.6 (95% CI: 38.3–62.9) months; CAB: 32.8 (95% CI: 25.5–40.0) months; Tmab/DTX followed by CAB: 42.4 (95% CI: 27.0–57.7) months; and CAB followed by Tmab/DTX: 45.2 (95% CI: 26.5–63.9) months; log-rank test, p < 0.001] (Figure 2). Table 3 shows the results of the univariate and multivariate analyses of OS1 for systemic therapies using Cox proportional hazards models. After adjustment for potential confounders, Tmab/DTX or CAB therapy was consistently associated with longer OS1 than that with the historical control group in LA/RM patients with SDC [Tmab/DTX: HR, 0.32 (95% CI: 0.20–0.50); CAB: HR, 0.53 (95% CI: 0.36–0.77); Tmab/DTX followed by CAB: HR, 0.23 (95% CI: 0.10–0.52); and CAB followed by Tmab/DTX: HR, 0.19 (95% CI: 0.08–0.50)] (Table 3). In addition, patients with an ECOG-PS of ⩾2 and HER2-positive SDC were at a significantly greater risk of developing worse OS1 outcomes in multivariate analysis.

OS1 according to systemic therapy.
Impact of HER2-targeted therapy and ADT on OS1 in patients with LA and/or RM SDC.
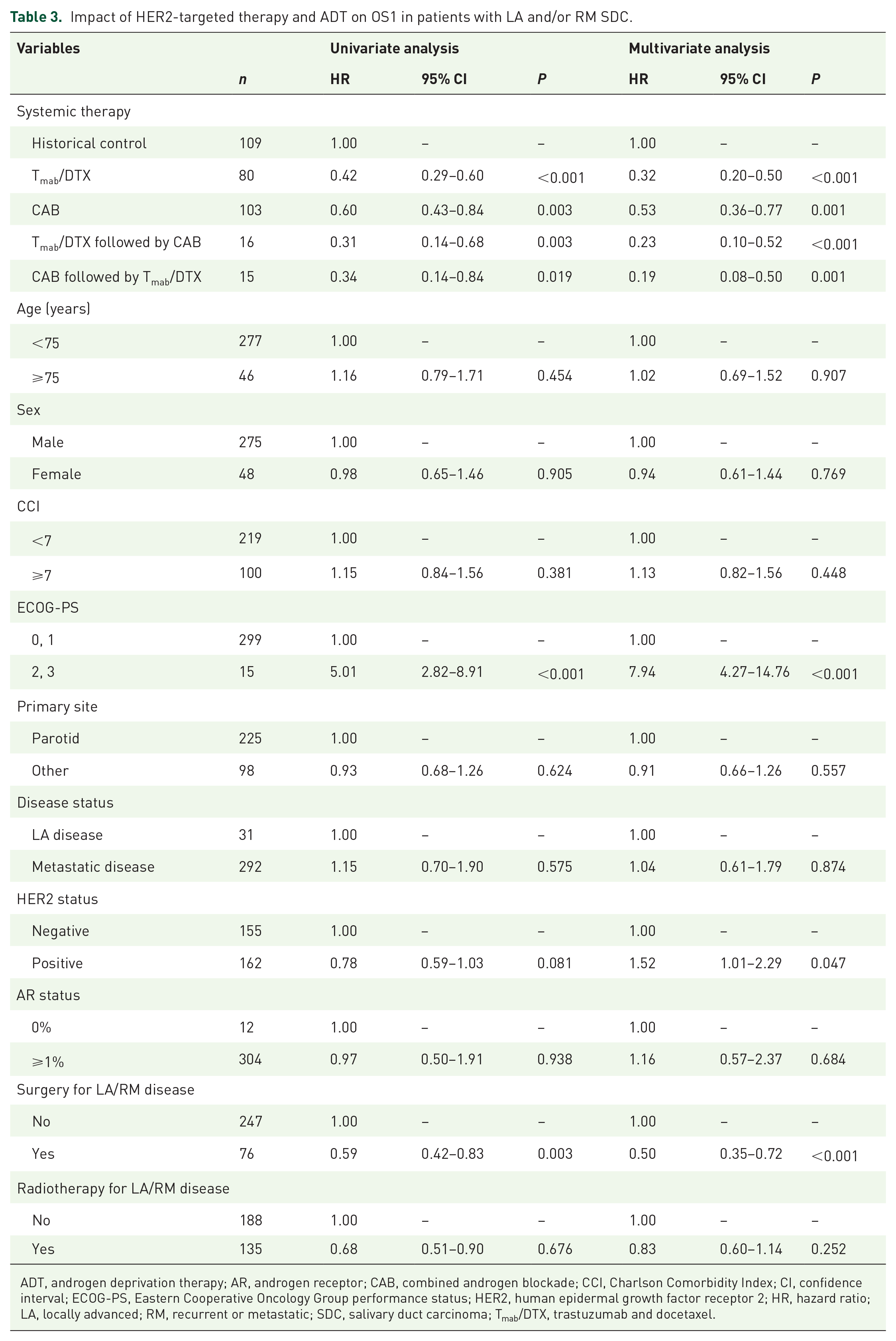
ADT, androgen deprivation therapy; AR, androgen receptor; CAB, combined androgen blockade; CCI, Charlson Comorbidity Index; CI, confidence interval; ECOG-PS, Eastern Cooperative Oncology Group performance status; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; LA, locally advanced; RM, recurrent or metastatic; SDC, salivary duct carcinoma; Tmab/DTX, trastuzumab and docetaxel.
HER2-targeted therapy and ADT as first-line systemic therapy for HER2-Positive/AR-positive patients with SDC
We evaluated the impact of Tmab/DTX and CAB as first-line systemic therapy on 96 HER2-positive/AR-positive LA/RM patients with SDC (Figure 1). The characteristics of these patients are presented in online-only Supplemental Table S3. The median follow-up time was 28.3 (5.5–95.2) months. Tmab/DTX had a significantly higher response rate than that with CAB in HER2-positive/AR-positive patients with SDC [Tmab/DTX: ORR, 71.2% (95% CI: 59.7–80.6%) and CBR, 83.6% (95% CI: 73.0–90.5%); CAB: ORR, 13.0% (95% CI: 4.0–35.1%); and CBR, 21.7% (95% CI: 8.9–44.2%)] (Table 4). These associations were consistent with those identified in the multivariate logistic regression models adjusted for potential confounders [CAB: ORR, 0.05 (95% CI: 0.01–0.20); CBR, 0.05 (95% CI: 0.01–0.17)] (Table 4).
Impact of HER2-targeted therapy and ADT as first-line systemic therapy on the clinical outcomes of patients with HER2-positive/AR-positive LA and/or RM SDC.

Confirmed CR or PR according to an independent review and the Response Evaluation Criteria for Solid Tumors (version 1.1).
Adjustment for age, sex, CCI, and primary site for ORR and CBR in the logistic regression model.
CR, PR, or SD for ⩾24 weeks.
Adjustment for age, sex, CCI, primary site, ECOG-PS, and surgery, radiotherapy, or conventional therapy after Tmab/DTX or CAB for overall survival 2 in the Cox proportional hazards model.
Adjustment for age, sex, CCI, and primary site for PFS, PFS2, DoR, and DoCB in the Cox proportional hazards model.
Time to second progression.
Time from treatment initiation to progression in patients who had a best overall response of CR or PR.
Time from treatment initiation to progression in patients who had a best overall response of CR, PR, or SD for ⩾24 weeks.
ADT, androgen deprivation therapy; CAB, combined androgen blockade; CBR, clinical benefit rate; CCI, Charlson Comorbidity Index; CI, confidence interval; CR, complete response; DoCB, duration of clinical benefit; DoR, duration of response; ECOG-PS, Eastern Cooperative Oncology Group performance status; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; N/A, not available; OR, odds ratio; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; PR, partial response; SD, stable disease; Tmab/DTX, trastuzumab and docetaxel.
Similarly, patients treated with Tmab/DTX as first-line systemic therapy had longer PFS than those treated with CAB as a first-line regimen, even after adjustment for potential confounders in the Cox proportional hazards models (HR, 3.51; 95% CI: 2.02–6.10; p < 0.001) (Table 4, Figure 3). However, we did not find significant differences between Tmab/DTX and CAB therapies in terms of OS2, PFS2, DoR, and DoCB in HER2-positive/AR-positive patients with SDC (Table 4, Figure 3). The results of the other subgroups are presented in online-only Supplemental Table S4.

Kaplan–Meier survival curves according to systemic therapy in patients with HER2-positive/AR-positive SDC. (a) The 3-year overall survival 2 rate was 62.4% (95% CI: 49.3–73.0%) for Tmab/DTX and 65.1% (95% CI: 36.1–83.5%) for CAB. (b) The 1-year PFS rate was 31.3% (95% CI: 19.6–43.8%) for Tmab/DTX and 13.0% (95% CI: 3.3–29.7%) for CAB. (c) The 1-year PFS2 rate was 47.3% (95% CI: 34.3–59.2%) for Tmab/DTX and 51.0% (95% CI: 29.1–69.3%) for CAB. (d) The 1-year DoR rate was 39.3% (95% CI: 24.3–53.9%) for Tmab/DTX and N/A for CAB. (e) The 1-year duration of the CBR was 35.2% (95% CI: 22.0–48.7%) for Tmab/DTX and 60.0% (95% CI: 12.6–88.2%) for CAB.
Discussion
In our cohort of LA/RM patients with SDC, which included the largest number of such patients examined to date, those on HER2-targeted therapy and ADT had significantly longer OS1 than those on historical control.
First, the data of participants in the historical control group were analyzed to examine whether there was any difference in prognosis depending on the treatment timing. However, we did not find any interaction between the treatment period and survival in LA/RM patients with SDC in the historical control group. Conventional therapy, mainly with cytotoxic chemotherapeutic drugs, did not improve prognosis between the past 20 years and the most recent 10 years. This result was similar to that reported in the database-based analysis and re-affirmed that there was a need for conducting a new treatment.8,9
The use of ADT has been reported to be associated with better survival than that with BSC or cytotoxic chemotherapy in retrospective studies.6,31 Although Sousa et al. reported that patients on HER2-targeted therapy had better ORR and PFS than those on conventional therapy,36 to the best of our knowledge, our study is the first to report that patients on HER2-targeted therapy have longer OS than those on conventional therapy. Thus, further development of HER2-targeted therapy and ADT as systemic therapy in these patients, as well as in patients with breast, stomach, and prostate cancer, is warranted.10,12 To date, the efficacy of pertuzumab,24,25,43 ado-trastuzumab emtansine,25–27,43 and trastuzumab deruxtecan 28 as a HER2-targeted therapy, and the efficacy of receiving abiraterone 32 and enzalutamide 33 as an ADT have been evaluated. Furthermore, HER2-targeted therapy and ADT are also expected to have a role as adjuvant therapy.36,44–46 The results of a clinical trial (ClinicalTrials.gov; NCT04620187) investigating the efficacy and safety of ado-trastuzumab emtansine as postoperative therapy for HER2-positive SDC are awaited.
Based on the secondary analysis of the comparison between HER2-targeted therapy and ADT as first-line systemic therapy, we found that patients with HER2-positive/AR-positive SDC on HER2-targeted therapy had significantly better ORR, CBR, and PFS than those on ADT. Furthermore, for ADT use, the ORR and CBR were lower, while the PFS was shorter, in HER2-positive patients with SDC than in all patients or those with HER2-negative SDC (Table 3, online-only Supplemental Tables S1, S3). This result was similar to that recently reported in the phase II study of enzalutamide.33 Therefore, HER2-targeted therapy can be recommended prior to ADT for HER2-positive/AR-positive SDC. These findings support the treatment algorithm for systemic therapy in patients with SDC advocated by Sousa et al. 36 Conversely, the OS2 and PFS2 of patients who received ADT were not inferior to those of patients who received HER2-targeted therapy. Taken together, as ADT is tolerable in older patients or those with multiple comorbidities or a poor ECOG-PS, ADT may be considered for HER2-positive/AR-positive patients with SDC who are unable to tolerate HER2-targeted therapy. Similarly, the DoR and DoCB for ADT were not inferior to those for HER2-targeted therapy in HER2-positive/AR-positive patients with SDC. The retrospective study of Boon et al. mentioned that 17 patients with SDC who achieved better response (PR or SD) had better OS than the 17 patients who had worse response (PD) among the 34 patients with SDC treated with ADT (29 versus 8 months). 6 Recently, a phase II study of enzalutamide reported similar results.33 Based on these results, if biomarkers that can predict clinical benefit or better response are established, ADT may be recommended even in patients who can tolerate HER2-targeted therapy because ADT is better tolerated than HER2-targeted therapy. The AR pathway activity score47,48 and the expression of SRD5A1 48 have been reported as factors predictive of the clinical benefit of ADT. In addition, AR-V7,49,50 FOXA1 mutations, 50 and EZH2 expression 51 are also potential predictive factors for response to ADT. Thus, the application of these biomarkers in clinical practice should be investigated in the future.10,12,36,37,47,48,51
To our knowledge, this was the largest study to investigate the clinical outcomes of LA/RM patients with SDC with a confirmed diagnosis of SDC based on a central pathology review. However, this study had some limitations. First, this was a retrospective study. Therefore, the results of a randomized phase II study (ClinicalTrials.gov; NCT01969578) comparing ADT and conventional therapy are awaited. Second, as conventional therapy included different treatments (e.g. cytotoxic chemotherapy, ICIs, and BSC), the resultant heterogeneity may have affected the comparisons between conventional therapy, HER2-targeted therapy, and ADT. Third, there might have been a selection bias in the choice between HER2-targeted therapy and ADT for HER2-positive/AR-positive patients with SDC. Fourth, we did not perform a detailed examination of the treatment of HER2-negative/AR-negative patients with SDC owing to the small sample size. The development of further treatment strategies for HER2-negative/AR-negative SDC is warranted.
Compared to conventional therapy, HER2-targeted therapy and ADT have remarkable survival benefits in LA/RM patients with SDC. Among HER2-positive/AR-positive patients with SDC, although the administration of HER2-targeted therapy should be considered first, ADT is suitable when HER2-targeted therapy is contraindicated owing to poor ECOG-PS, older age, or multiple comorbidities. Our findings should be further validated in a prospective multi-institutional study because of the rarity of SDC cases. Moreover, studies to identify factors predicting the response to HER2-targeted therapy and ADT are warranted to facilitate the selection of optimal treatment strategies for patients with SDC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221119538 – Supplemental material for Survival benefit of HER2-targeted or androgen deprivation therapy in salivary duct carcinoma
Supplemental material, sj-docx-1-tam-10.1177_17588359221119538 for Survival benefit of HER2-targeted or androgen deprivation therapy in salivary duct carcinoma by Daisuke Kawakita, Toshitaka Nagao, Hideaki Takahashi, Satoshi Kano, Yoshitaka Honma, Hideaki Hirai, Natsuki Saigusa, Kohei Akazawa, Kaori Tani, Hiroya Ojiri, Kiyoaki Tsukahara, Hiroyuki Ozawa, Kenji Okami, Takahito Kondo, Takafumi Togashi, Chihiro Fushimi, Tomotaka Shimura, Akira Shimizu, Isaku Okamoto, Takuro Okada, Yorihisa Imanishi, Yoshihiro Watanabe, Kuninori Otsuka, Akihiro Sakai, Koji Ebisumoto, Yuichiro Sato, Keisuke Yamazaki, Yushi Ueki, Toyoyuki Hanazawa, Yuki Saito, Mizuo Ando, Takashi Matsuki, Masato Nakaguro, Yukiko Sato, Makoto Urano, Yoshitaka Utsumi, Shinji Kohsaka, Takashi Saotome and Yuichiro Tada in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359221119538 – Supplemental material for Survival benefit of HER2-targeted or androgen deprivation therapy in salivary duct carcinoma
Supplemental material, sj-docx-2-tam-10.1177_17588359221119538 for Survival benefit of HER2-targeted or androgen deprivation therapy in salivary duct carcinoma by Daisuke Kawakita, Toshitaka Nagao, Hideaki Takahashi, Satoshi Kano, Yoshitaka Honma, Hideaki Hirai, Natsuki Saigusa, Kohei Akazawa, Kaori Tani, Hiroya Ojiri, Kiyoaki Tsukahara, Hiroyuki Ozawa, Kenji Okami, Takahito Kondo, Takafumi Togashi, Chihiro Fushimi, Tomotaka Shimura, Akira Shimizu, Isaku Okamoto, Takuro Okada, Yorihisa Imanishi, Yoshihiro Watanabe, Kuninori Otsuka, Akihiro Sakai, Koji Ebisumoto, Yuichiro Sato, Keisuke Yamazaki, Yushi Ueki, Toyoyuki Hanazawa, Yuki Saito, Mizuo Ando, Takashi Matsuki, Masato Nakaguro, Yukiko Sato, Makoto Urano, Yoshitaka Utsumi, Shinji Kohsaka, Takashi Saotome and Yuichiro Tada in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359221119538 – Supplemental material for Survival benefit of HER2-targeted or androgen deprivation therapy in salivary duct carcinoma
Supplemental material, sj-docx-3-tam-10.1177_17588359221119538 for Survival benefit of HER2-targeted or androgen deprivation therapy in salivary duct carcinoma by Daisuke Kawakita, Toshitaka Nagao, Hideaki Takahashi, Satoshi Kano, Yoshitaka Honma, Hideaki Hirai, Natsuki Saigusa, Kohei Akazawa, Kaori Tani, Hiroya Ojiri, Kiyoaki Tsukahara, Hiroyuki Ozawa, Kenji Okami, Takahito Kondo, Takafumi Togashi, Chihiro Fushimi, Tomotaka Shimura, Akira Shimizu, Isaku Okamoto, Takuro Okada, Yorihisa Imanishi, Yoshihiro Watanabe, Kuninori Otsuka, Akihiro Sakai, Koji Ebisumoto, Yuichiro Sato, Keisuke Yamazaki, Yushi Ueki, Toyoyuki Hanazawa, Yuki Saito, Mizuo Ando, Takashi Matsuki, Masato Nakaguro, Yukiko Sato, Makoto Urano, Yoshitaka Utsumi, Shinji Kohsaka, Takashi Saotome and Yuichiro Tada in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-4-tam-10.1177_17588359221119538 – Supplemental material for Survival benefit of HER2-targeted or androgen deprivation therapy in salivary duct carcinoma
Supplemental material, sj-docx-4-tam-10.1177_17588359221119538 for Survival benefit of HER2-targeted or androgen deprivation therapy in salivary duct carcinoma by Daisuke Kawakita, Toshitaka Nagao, Hideaki Takahashi, Satoshi Kano, Yoshitaka Honma, Hideaki Hirai, Natsuki Saigusa, Kohei Akazawa, Kaori Tani, Hiroya Ojiri, Kiyoaki Tsukahara, Hiroyuki Ozawa, Kenji Okami, Takahito Kondo, Takafumi Togashi, Chihiro Fushimi, Tomotaka Shimura, Akira Shimizu, Isaku Okamoto, Takuro Okada, Yorihisa Imanishi, Yoshihiro Watanabe, Kuninori Otsuka, Akihiro Sakai, Koji Ebisumoto, Yuichiro Sato, Keisuke Yamazaki, Yushi Ueki, Toyoyuki Hanazawa, Yuki Saito, Mizuo Ando, Takashi Matsuki, Masato Nakaguro, Yukiko Sato, Makoto Urano, Yoshitaka Utsumi, Shinji Kohsaka, Takashi Saotome and Yuichiro Tada in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
