Abstract
Ovarian clear cell carcinoma (OCCC) is a distinct epithelial ovarian cancer subtype with unique molecular features, and a notable resistance to conventional platinum-based chemotherapy in advanced disease. Key molecular hallmarks include frequent ARID1A loss and PIK3CA activation, which often co-occur and contribute to early tumorigenesis. Emerging targeted therapies—including anti-angiogenic agents, immune checkpoint inhibitors, ATR and PI3K pathway inhibitors, and antibody-drug conjugates—demonstrate promising activity, particularly in molecularly defined subgroups, though most evidence to date remains largely limited to early-phase trials. Given its rarity, chemoresistance, and underrepresentation in large trials, OCCC requires histology-specific therapeutic strategies informed by molecular profiling. Ongoing research into biomarker-driven therapies and combination regimens holds the potential to transform outcomes for this challenging ovarian cancer subtype.
Plain language summary
Ovarian clear cell carcinoma (OCCC) is a rare type of ovarian cancer that behaves differently from other forms. It often does not respond well to standard chemotherapy, especially in advanced stages. Scientists have discovered that many OCCC tumors share certain genetic changes, such as loss of the ARID1A gene and activation of the PIK3CA pathway, which are thought to drive cancer growth early on. Because of these unique features, researchers are testing new treatments that target these changes. These include drugs that block blood vessel growth, stimulate the immune system, or directly target faulty DNA repair or cell growth pathways. Some early studies show encouraging results, especially when treatments are matched to specific tumor characteristics, but most of the evidence so far comes from small, early-stage trials. Since OCCC is uncommon and usually excluded from large clinical studies, doctors and researchers emphasize the need for treatment strategies designed specifically for this cancer type. Future advances are likely to come from tailoring therapies to each tumor’s molecular profile and exploring drug combinations to improve outcomes.
Introduction
Ovarian cancer (OC) is the eighth leading cause of cancer-related mortality among women worldwide, with a disproportionate burden in low- and middle-income countries, where 70% of cases are diagnosed. 1 Ovarian cancer continues to be a growing oncological concern, with projections indicating that by 2050 the number of women diagnosed worldwide will increase by more than 55%, exceeding half a million cases. 1 Epithelial tumors account for more than 90% of cases, with ovarian clear cell carcinoma (OCCC) being the second most prevalent subtype after high-grade serous ovarian cancer (HGSOC), representing approximately 10% of all epithelial ovarian cancer (EOC) cases. 2 While OCCC represents around 10% of OC cases in Western Europe and North America, it is more common in East Asia where it accounts for up to 25% of cases in Japan, Singapore, and the Republic of Korea.3,4 The reason for such demographic differences remains unclear and may explain the underrepresentation of clear cell carcinomas in clinical trials so far. OCCC is predominantly diagnosed at an early stage of disease (45%–81% of patients diagnosed with stage I disease vs 4%–35% at stage III5,6) OCCC has a more favorable prognosis in early stage disease, however prognosis remains poor in the advanced stage due to chemoresistance. 4 The mainstay of treatment for early OCCC remains oncological surgery with aim of complete resection and adequate staging including total hysterectomy with bilateral salpingo-oophorectomy, peritoneal biopsies, omentectomy, bilateral pelvic and para-aortic lymphadenectomy, and peritoneal washings. 7 In advanced disease, surgical debulking with the aim to achieve complete cytoreduction (defined as total macroscopic tumor clearance with no residual visible disease) has been shown to improve overall survival (OS). 8 If upfront cytoreduction is not feasible, then neoadjuvant chemotherapy followed by interval cytoreductive surgery is non-inferior to primary cytoreductive surgery followed by adjuvant chemotherapy in advanced FIGO IIIC-IV disease. 9 Historically, treatment for OCCC (such as olaparib or bevacizumab maintenance) has been extrapolated from its other EOC counterparts based on pivotal trials comprised of the most frequent histologic types. However, in recent years, several trials have considered OCCC independently and are progressively reshaping their therapeutic landscape. This review examines the distinct molecular features of OCCC that differentiate it from other EOC subtypes and explores how these features may confer susceptibility to established and emerging targeted therapies.
Molecular profile
Alterations in the PI3K signaling cascade and the switch/sucrose non-fermentable (SWI/SNF) chromatin-remodeling complex represent key molecular hallmarks of OCCC and frequently co-exist.10–12 ARID1A (also known as BAF250a), encoded by the ARID1A gene on chromosome 1p36, is an essential subunit of the SWI/SNF chromatin-remodeling complex, which regulates gene expression, cellular differentiation, and tumor suppression. 13 ARID1A inactivation is thought to promote cell-cycle progression, potentially through c-myc–mediated pathways, thereby driving uncontrolled cellular proliferation. 14 Mutations in ARID1A and PIK3CA are each detected in approximately 50% of OCCC cases.10,11 Of note, nearly all ARID1A mutations are truncation mutations resulting in the loss of ARID1A expression. 15
OCCC is the most frequent EOC that arises from or is associated with endometriosis, with approximately 50% of OCCC patients also having a diagnosis of endometriosis. 16 Loss of ARID1A has also been identified in contiguous atypical endometriosis, with mutations more frequently observed in endometriosis-associated OCCC. 12 Comparative analyses of benign endometriosis, atypical endometriosis, and OCCC reveal a stepwise accumulation of molecular alterations. The concurrent loss of ARID1A expression, upregulation of HNF-1β, and loss of ER expression were absent in benign endometriosis, present in 23% of atypical endometriosis, and further increased to 42% of OCCC tumors. 15 Furthermore, ARID1A deficiency has been associated with poor prognosis in several studies, particularly in advanced-stage disease. 17 It must be noted that ARID1A is also altered in many other malignancies, such as prostate cancer where its expression is significantly decreased, 18 as well as in non-malignant tissues such as placental tissue, in which it serves as a marker of poor trophoblast differentiation. 19
Similar to ARID1A alterations, activation of the PI3K/Akt pathway has been demonstrated in endometriosis, suggesting its role as an early event in clear cell tumorigenesis. 20 OCCC also exhibits a higher frequency of telomerase reverse transcriptase (TERT) promoter mutations (up to 16%) compared with other EOC histotypes. TERT codes for the enzyme telomerase and TERT promoter (TERTp) mutations have conflicting prognostic significance in OCCC. One study demonstrated that TERTp mutations are associated with platinum resistance and worse disease-free survival (DFS) in OCCC, 21 while another found the -124C>T TERTp mutation may serve as a favorable prognostic marker in OCCC. 22 Notably, TERT mutations do not appear to represent an early event in OCCC pathogenesis, as they are absent in endometriosis lesions progressing to carcinoma. 23 Additional recurrent mutations include PTEN deletions (37% of cases), 24 MET proto-oncogene amplification (24%–37%), 25 KRAS mutations (21%), and, less frequently, alterations in ARID1B and PPP2R1A. 26
Gene expression profiling further indicates that the transcriptional signature of OCCC is distinct from other EOC subtypes, with enrichment in pathways related to carbohydrate metabolism and hypoxic adaptation. Hepatocyte nuclear factor 1β is overexpressed in more than 80% of cases of OCCC and plays a pivotal role in promoting the Warburg effect through drastically altering the glycolytic process and carbohydrate metabolism in OCCC cells. 27 While OCCC is predominantly glycolytic, HGSOC presents more variation; specifically, its high-oxidative phosphorylation variant is defined by an enriched expression of electron transport chain components and higher oxygen consumption rates, making it more responsive to chemotherapy than its glycolytic counterparts. 28 Transcriptomic analyses stratify OCCC into subgroups with distinct biology and prognosis. The aggregation of 222 gene expression profiles identified two distinct molecular subtypes: epithelial and mesenchymal (Figure 1). 29 The epithelial subtype exhibits a higher expression of cell-cell adhesion genes (CDH1, CLDN3, and CLDN4) and is characterized by frequent SWI/SNF alterations, while the mesenchymal subtype showed enriched expression of genes involved in the extracellular matrix (ECM) and collagen V binding. 29 Furthermore, the epithelial subtype was associated with endometriosis, lower disease stage and lower risk of progression while the mesenchymal subtype was associated with poorer prognosis, advanced disease and greater chemoresistance. 29 A separate series of whole-transcriptome sequencing of OCCC tumors incorporating both Asian and non-Asian patients reported similar findings and also identified two similar subgroups: firstly, the more common “classic-OCCC” subtype, which was characterized by AT-rich interactive domain-containing protein 1A (ARID1A) mutations and frequent co-alterations (e.g., PIK3CA, TERT), demonstrated metabolic pathway enrichment involving carbohydrate metabolism, and typically presented at early stage. 30 In contrast, the second TP53-mutant subtype showed upregulation of ECM, mesenchymal, and immune-related pathways and was more often diagnosed at advanced stage with poorer survival.30 Unlike high-grade serous ovarian carcinoma, in which TP53 mutations are nearly universal, OCCC typically retains wild-type TP53, with alterations identified in only ~20% of cases. 31 Immune gene expression profiling also showed that tumors with high expression of immune checkpoint molecules PD1 and CTLA4 were associated with poorer 5-year DFS and OS outcomes. 32
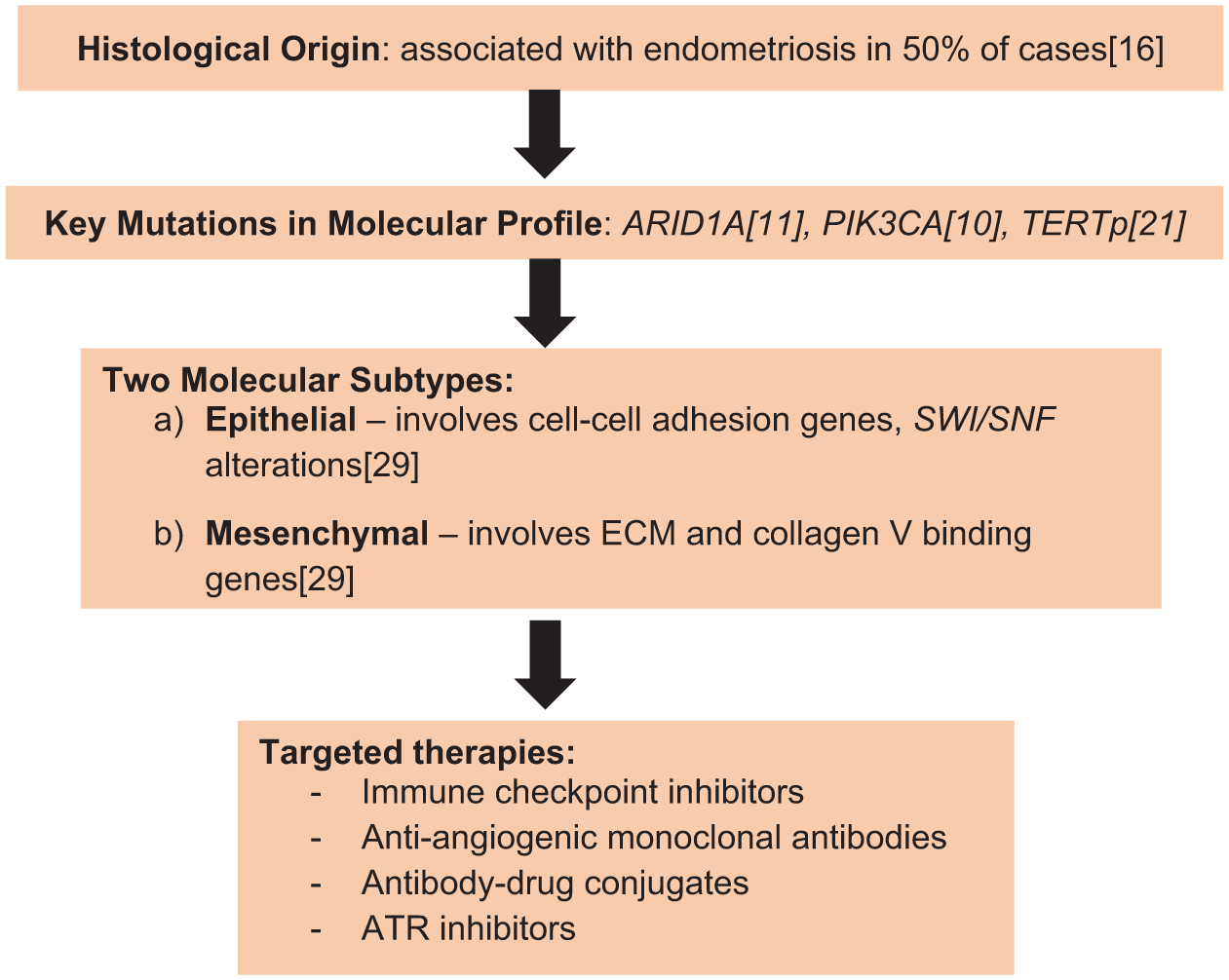
Schematic representing molecular profile and targeted therapies for OCCC.
While the overall mutational landscape of OCCC is broadly shared between ethnicities, subtle differences in frequency and clinical presentation have been observed. According to Bolton et al., there was a trend toward a higher frequency of ARID1A-mutated tumors in women of East Asian descent compared to other groups, though this difference did not reach statistical significance. 30 Their molecular clustering further revealed that the TP53-mutant, ARID1A wild-type cluster was predominantly enriched for advanced-stage disease and associated with worse survival outcomes. 30 In contrast, tumors belonging to the ARID1A-mutant clusters were more likely to occur in individuals of Asian ancestry and those with a documented history of endometriosis. 30
Supplementing these findings, Heong et al. affirmed that OCCC tumor samples from Asian and Caucasian populations are molecularly and clinically similar. 32 By analyzing cohorts from Singapore, Japan, and the United Kingdom, they identified four novel immune subtypes—PD1-high, CTLA4-high, antigen-presentation, and pro-angiogenic—characterized by distinct differentially expressed pathways. 32 Notably, the PD1-high and CTLA4-high subtypes were consistently associated with poorer clinical outcomes across all three countries. 32
Tumor microenvironment
The molecular landscape of OCCC demonstrates a highly immunosuppressive tumor microenvironment (TME). Unlike most malignancies, where increased CD8+ T-cell infiltration correlates with improved survival, in OCCC, it has been associated with adverse outcomes. 33 Notably, both PD-L1 and CD8+ T-cell expression increase significantly following tumor recurrence. 34 Immune checkpoints, such as PD-1/PD-L1, are expressed on tumor cells to deliver inhibitory signals that dampen antitumor immunity. While no standardized threshold for PD-L1 positivity exists, approximately 34%–64% of OCCCs demonstrate >5% stromal cell PD-L1 expression.35,36 The role of PD-1 and PD-L1 inhibitors in OCCC will be discussed in detail further on in this review.
Current standards and emerging targeted strategies in OCCC
The clinical management of OCCC is currently predicated on maximal cytoreductive surgery followed by platinum-based adjuvant chemotherapy. 7 While early-stage disease often presents a favorable prognosis, the chemo-resistant nature of relapsed and metastatic OCCC has prompted the investigation of targeted agents, such as ataxia telangiectasia and Rad3-related (ATR) inhibitors and PI3K pathway inhibitors that exploit the tumor’s distinct genomic landscape
Targeted therapies in the maintenance setting: Anti-angiogenics and Poly (ADP-ribose) Polymerase inhibitors
The role of maintenance strategies for OCCC warrants further exploration. Around 50% of OC exhibit some degree of homologous recombination deficiency (HRD) which can be attributed to germline or somatic mutations in homologous recombination repair (HRR) genes such as BRCA1 or BRCA2, or other mechanisms. In contrast, HRR mutations are not commonly observed in OCCC, accounting for only around 5% of cases, and only 13% of OCCC are considered HRD based on genomic instability assays. 37
BRCA1/2 mutations occur in approximately 13%–15% of ovarian cancers overall, 38 with the highest prevalence in high-grade serous carcinoma, but are detected in only ~6% of OCCC. 39 In stage III or IV high-grade serous or endometrioid ovarian cancer, primary peritoneal cancer, or fallopian-tube cancer harboring BRCA1/2 alterations or exhibiting high genomic instability, maintenance therapy with Poly (ADP-ribose) Polymerase (PARP) inhibitors has been shown to markedly improve both progression-free survival (PFS) and OS, establishing these agents as a major therapeutic advance in ovarian cancer. 40 No dedicated studies have addressed their efficacy in OCCC, however given the paucity of effective alternatives, OCCC patients should still be tested for BRCA1/2 mutations and broader HRD to determine potential eligibility for PARP inhibition, which remains a reasonable treatment strategy.
In contrast, the evidence for bevacizumab maintenance in OCCC is somewhat more favorable, with potential benefit observed both as a first-line therapy and in recurrent disease. Bevacizumab, a humanized monoclonal antibody directed against vascular endothelial growth factor (VEGF), was the first targeted therapy to show a PFS benefit in ovarian cancer. Its role in OCCC, however, is extrapolated from large phase III studies such as GOG-0218 41 and ICON-7, 42 which included patients across histological subtypes but only a limited number with OCCC. In GOG-0218, patients with stage III (incompletely resected) or stage IV ovarian cancer treated with carboplatin and paclitaxel plus concurrent and maintenance bevacizumab achieved a median PFS of 14.1 months compared with 10.3 months for chemotherapy alone, although no OS advantage was demonstrated. 41 Retrospective analyses specific to OCCC suggest that bevacizumab may provide substantial benefit in advanced-stage disease, extending median PFS from 12.5 to 29.7 months and increasing median OS from 34.7 to 51.4 months. 43 Retrospective data are promising, and the use of bevacizumab is supported by National Comprehensive Cancer Network (NCCN) guidelines for OCCC. 44 The highest level of evidence (prospective, randomized, subtype-specific) is still lacking but clinicians use bevacizumab in real-world clinical practice to treat stage III–IV OCCC based on extrapolated benefit, clinical guidelines, and limited viable alternatives.
Immune checkpoint inhibition in the relapsed or metastatic setting
Clear cell gynecologic malignancies remain highly resistant to chemotherapy, with limited survival benefits in the recurrent or metastatic setting. In relapsed OCCC, chemotherapy achieves a median PFS of only 2–3 months,45,46 underscoring the urgent need for new therapeutic approaches. Immune checkpoint inhibiton (ICI) has tumor agnostic approval in the mismatch repair deficient (dMMR) setting for all patients, including OCCC, based on KEYNOTE-158. This phase II study enrolled 233 dMMR/MSI-H (microsatellite instability-high) patients and demonstrated 33% objective response rate (ORR) in 25 dMMR OC patients enrolled. 47 Approximately 5%–8% of OCCC patients are dMMR 48 and Lynch syndrome accounts for ~ 12% of OCCC cases. 32 Of note, MMR status and ARID1A loss are closely related: dMMR OCCC cases show significantly higher frequency of loss of ARID1A expression compared to proficient MMR (pMMR) cases, suggesting that ARID1A deficiency, via its role in DNA damage repair, is the core mechanism of microsatellite instability rather than its consequence.49,50
Phase II trials evaluating ICI monotherapy in clear cell gynecologic malignancies have yielded mixed results and limited activity. In contrast, more encouraging efficacy signals have been observed with dual ICI regimens and with combinations of ICI and anti-angiogenic therapy. The single arm PEACOCC trial (conducted in 5 UK centers) reported encouraging activity of pembrolizumab monotherapy in 41 relapsed OCCC patients, with an ORR of 25% and a median response duration approaching 12 months. 51 In contrast, the MOCCA trial, a phase II trial conducted in Singapore, Korea, and Australia, which randomized pMMR recurrent OCCC patients to durvalumab versus physician’s choice chemotherapy, demonstrated no ORR or PFS benefit. 45
To improve efficacy, dual ICI strategies have been evaluated. Three phase II studies—BrUOG (NCT03355976), DART/SWOG S1609, and MoST-CIRCUIT—tested nivolumab (PD-1 inhibitor) plus ipilimumab (CTLA-4 inhibitor) in clear cell gynecologic cancers, reporting ORRs of 26.6%, 52 37%, 53 and 50%, 54 respectively. Median PFS was 5.1 months in BrUOG 52 and 3.7 months in OCCC within DART/SWOG S1609. 53 In the MoST-CIRCUIT trial, the 6-month PFS was 58%. 54 It is important to note that the doses and scheduling of ipilimumab varied across these three trials. The three trials were also conducted in different populations: BrUOG 52 was conducted in the United States with most sites in Rhode Island, DART/SWOG S1609 53 was a large U.S.-wide basket trial and MoST-CIRCUIT was a multicenter study conducted in Australia and New Zealand. 54 However, the clinical benefit of dual ICI must be weighed against toxicity, with the MoST-CIRCUIT trial reporting grade ⩾3 adverse events in 25% of patients, including 1 G5 myocarditis event, despite an impressive 50% ORR. 54 Another phase II trial comparing nivolumab with nivolumab plus ipilimumab in recurrent or persistent ovarian cancer showed OCCC patients were five times more likely to respond to nivolumab/ipilimumab than those with other histologic subtypes. 55 This improved response may be underpinned by the distinct immunosuppressive TME characteristic of clear cell tumors, marked by increased expression of inhibitory receptors such as LAG3 and PD-1. 56 Recent research, specifically a landmark study published in Nature by Dai et al., 57 analyzed post-ICI treatment TME changes in patients with recurrent, platinum-resistant or refractory OCCC via serial biopsies. The study found that patients with PPP2R1A mutations—a key biomarker for exceptional response—demonstrated significantly enhanced immune infiltration and a marked expansion of CD45RO+ CD8+ memory T cells in the TME following immunotherapy. 57 Additionally, alterations in the PI3K pathway and loss of ARID1A function have been linked to elevated PD-L1 expression, 58 enhanced IL-6 and IL-8 signaling, and further immune evasion. 56 Another strategy, pembrolizumab combined with the IDO-1 inhibitor epacadostat, achieved an ORR of 21% and median PFS of 4.8 months, but the trial was terminated early due to insufficient drug supply. 59
The addition of anti-angiogenic therapy represents another evolving strategy to enhance ICI efficacy in OCCC. As monotherapy, anti-angiogenics show limited activity: in the NICCC/ENGOT-OV36 trial, nintedanib (a Vascular Endothelial Growth Factor Receptor, Platelet-Derived Growth Factor Receptor, and Fibroblast Growth Factor Receptor inhibitor) produced a 2% ORR and no PFS advantage over chemotherapy, which itself demonstrated 0% ORR. 45 These findings highlighted both the ineffectiveness of chemotherapy in this setting and the need for rational combination strategies. VEGF signaling is a key immunosuppressive driver within the TME, suppressing T-cell function and promoting regulatory T-cell recruitment.60–62 Bevacizumab, a VEGF inhibitor, may reverse these effects by normalizing tumor vasculature and augmenting ICI response. Several early-phase studies support this approach. The single-arm INOVA trial (NCT04735861) conducted in China, evaluating bevacizumab plus sintilimab in recurrent OCCC, reported an ORR of 40.5% and median PFS of 6.9 months. 63 The LARA study (NCT04699071) was conducted across three tertiary hospitals in Singapore and South Korea and investigated the use of pembrolizumab (a PD-1 inhibitor) plus lenvatinib (a multi-kinase inhibitor that primarily inhibits VEGFR1-3, FGFR1-4, PDGFRα, RET, and KIT) in recurrent OCCC and endometrial clear cell carcinoma. 64 It demonstrated a 24-week ORR of 44.0% and median PFS of 23.4 weeks. 65 Another single cohort phase II trial conducted in the United States evaluating the use of pembrolizumab and lenvatinib in OCCC (NCT05296512) demonstrated a median PFS of 10.9 months. 65 In the global BOUQUET umbrella trial (NCT04931342), the atezolizumab–bevacizumab arm yielded an interim ORR of 14% (including one OCCC patient) and a disease control rate of 71%. 66 The ongoing global DOVE trial (APGOT-OV7/ENGOT-ov80) is further evaluating dostarlimab with or without bevacizumab versus chemotherapy in recurrent gynecologic clear cell cancers. 67 Taken together, phase II evidence suggests that the most promising regimens include pembrolizumab plus lenvatinib, dual ICI with nivolumab plus ipilimumab, and bevacizumab plus PD-1 inhibition.
ARID1A as a novel therapeutic target
ARID1A is one of the most frequently mutated genes in clear cell carcinomas and represents a promising therapeutic target, although no clinically approved therapies currently exist. ARID1A functions as a chromatin-remodeling tumor suppressor and is recruited to double-strand DNA breaks via interaction with ataxia telangiectasia and Rad3-related (ATR) kinase. 68 Loss of ARID1A leads to aberrant cell-cycle regulation 69 and replicative stress, which has been linked to increased sensitivity to gemcitabine in ARID1A-mutated OCCC, though additional mechanistic data are needed. 70 ARID1A-deficient cancers also display impaired DNA damage response and heightened vulnerability to ATR inhibition. 71 Preclinical studies further suggest that ARID1A modulates HRR through PI3K/Akt1 signaling, thereby conferring enhanced sensitivity to PARP inhibitors. 72 The combination of ATR and PARP inhibition has shown synergistic activity in preclinical models, and the ongoing ATARI trial is investigating this strategy in rare gynecologic tumors including OCCC. Preliminary results reported an ORR of 14% in both ARID1A-deficient patients treated with ceralasertib (ATR kinase inhibitor) alone and ARID1A-proficient patients treated with ceralasertib plus olaparib (PARP inhibitor), suggesting modest activity with further data awaited. 73
Another agent, ENMD-2076, an oral multitarget kinase inhibitor selective against Aurora A and VEGFR, failed to meet its 6-month PFS threshold in an unselected OCCC cohort. 74 Interestingly, ARID1A loss correlated with improved PFS, warranting further exploration of ARID1A as a predictive biomarker for response. Additionally, epigenetic therapies such as EZH2 inhibitors and BET inhibitors have demonstrated selective antitumor activity in ARID1A-mutant ovarian cancer models.75–77 ARID1A deficiency may represent a target for novel therapeutic strategies designed to exploit synthetic lethality.
PI3K pathway inhibitors
Another potential target based on the molecular profile of OCCC includes the PI3K pathway. Since more than 80% of OCCC demonstrate AKT-mTOR pathway activation, 77 the GOG268 trial combined temsirolimus, an mTOR inhibitor, with postoperative carboplatin and paclitaxel followed by 1-year maintenance in OCCC. The phase II study failed to demonstrate PFS or OS benefit. 78 However, a Japanese phase II study of combination chemotherapy with temsirolimus and trabectedin (an alkylating agent which binds to the minor groove of DNA) for recurrent OCCC demonstrated a clinical benefit rate of 47%. 79 OCCC tumor cells may derive sensitivity to trabectedin via their increased nucleotide excision repair activity. 80 Similarly, the ongoing phase II CYH33-G201 trial is investigating CYH-33, an alpha-selective PIK3CA inhibitor, in patients with recurrent or persistent OCCC with PIK3CA hotspot mutations. 81
Antibody drug conjugates
Antibody-drug conjugates (ADCs) represent an emerging therapeutic avenue in OCCC, leveraging specific surface antigens to deliver cytotoxic payloads. Reported rates of HER2 overexpression in OCCC vary considerably across studies, reflecting methodological differences. 82 HER2 overexpression (immunohistochemistry 3+ score) was observed in 9% of OCCC tumors in a Korean study, with notable concordance between HER2 3+ scores and ERBB amplification. 83 Trastuzumab deruxtecan (T-DXd) functions as an ADC that targets HER2-expressing cells to deliver a potent topoisomerase I inhibitor payload, which is released intracellularly upon lysosomal cleavage of its tetrapeptide linker. 84 A distinguishing characteristic of this agent is the membrane-permeability of its payload, which enables a “bystander effect” that eliminates neighboring tumor cells regardless of their specific HER2 expression levels. 84 The efficacy of T-DXd is likely modulated by the local TME. One study in gastric cancer reported a trend toward increased levels of PD1+CD8+ T cells during T-DXd treatment in responders compared with nonresponders, suggesting that the density of CD8+ T cells may impact ADC response by inducing immunogenic cell death. 85 In ovarian cancer, T-DXd has shown encouraging efficacy: the DESTINY-PanTumor02 phase II trial reported an ORR of 63.6% in ovarian cancer patients with HER2 IHC 3+ tumors treated in the second-line or later setting, 86 while the HERALD trial in HER2-amplified solid tumors demonstrated a 100% ORR in ovarian cancer, though inclusion of the clear cell subtype was not specified in either study. 87 T-DXd may represent a promising ADC strategy for HER2-positive OCCC, though further investigation of its efficacy in this subtype is needed (Tables 1 and 2).
Molecular alterations, key trials, and targeted therapies for OCCC.

ARID1A, AT-rich interactive domain-containing protein 1A; ATR, ataxia telangiectasia and Rad3-related; OCCC, ovarian clear cell carcinoma; SWI/SNF, switch/sucrose non-fermentable.
Summary of key clinical trials and evidence in OCCC.

Bev, bevacizumab; CA, cancer; CCGC, clear-cell gynecological carcinoma; dMMR, deficient mismatch repair; EOC, epithelial ovarian cancer; IHC, immunohistochemistry; MSI-H, microsatellite instability-high; OCCC, ovarian clear cell carcinoma; ORR, objective response rate; OS, overall survival; PFS, progression-free survival.
Additional ADCs under investigation include those targeting folate receptor alpha (FOLR1), 88 Claudin-6 (CLDN6), 89 and CDH6, 90 among others. The CATALINA-2 (ENGOT-ov83) study evaluates TORL-1–23, a novel ADC targeting CLDN6 with a monomethyl auristatin E payload, in patients with platinum-resistant EOC. 91 CLDN6 is a member of the claudin family of tight junction proteins whose aberrant expression is limited to selected epithelial malignancies, including ovarian cancers, while remaining nearly absent in healthy adult tissues. 91 Early results from the phase I trial enrolling patients with multiple tumor types demonstrated a promising ORR of 55% in CLDN6+ platinum-resistant ovarian cancer patients. 92 The therapeutic utility and clinical efficacy of these agents specifically within the OCCC cohort remain undetermined.
OCCC also has a distinct surface marker profile that differs from HGSOC. A Canadian cohort of over 1500 ovarian cancer patients reported high FOLR1 expression was detected in 44% of HGSOC, but only 8% of OCCC, potentially limiting the utility of folate-targeted ADCs in this subtype compared to HGSOC. 93 Conversely, OCCC shows a unique enrichment for other targets; for instance, HER2 overexpression is more frequently observed in OCCC with a Korean cohort of almost 400 patients reporting IHC 2+ or 3+ HER2 expression rates in 17.6% of HGSOC samples, compared to 45.2% of OCCC samples, 94 indicating a potentially greater utility for T-DXd in OCCC.
While cancer vaccines have primarily been deployed in HGSOC, 95 the unique genomic and immunologic profile of OCCC suggests potential for subtype-specific applications. Unlike HGSOC, which is characterized by near-universal TP53 mutations, OCCC frequently harbors ARID1A and PIK3CA mutations, which may generate a distinct set of targetable neoantigens. Two shared antigen vaccines used in HGSOC target Wilms Tumor 1 (WT1) 96 or MUC1 (Mucin 1). 97 While WT1 is a hallmark of serous differentiation 98 and is generally not expressed in OCCC, MUC1 is overexpressed in over 90% of all EOCs, 99 including the clear cell subtype, 100 making MUC1-based vaccines (e.g., CVac) a potential cross-over strategy. 101 The definitive role of these vaccine strategies specifically in the OCCC population remains to be determined, as prospective data are currently lacking, yet the unique mutational landscape of OCCC provides a compelling rationale for future subtype-specific trials.
Discussion
OCCC represents a distinct and clinically challenging subset of EOC, characterized by unique epidemiological and molecular features. The disproportionately high incidence of OCCC in East Asian populations and its relative rarity may contribute to its historical underrepresentation in pivotal phase III trials, emphasizing the need for targeted investigation in this histologic subtype.
At the molecular level, OCCC exhibits a unique landscape defined by frequent co-alterations in the SWI/SNF chromatin-remodeling complex (ARID1A) and the PI3K/Akt signaling pathway. The high prevalence of truncating ARID1A mutations, coupled with PIK3CA activation, highlights early events in tumorigenesis and potential vulnerabilities exploitable by targeted therapy. Transcriptomic stratification into epithelial and mesenchymal subtypes further delineates prognostic and therapeutic heterogeneity. These findings underscore the value of molecular profiling in guiding personalized treatment strategies.
The TME in OCCC presents additional therapeutic challenges. Contrary to most solid tumors, increased CD8+ T-cell infiltration does not correlate with improved survival, reflecting a uniquely immunosuppressive milieu. Although single-agent ICI has shown limited and variable activity, dual ICI strategies and combinations with anti-angiogenic therapy appear more promising, suggesting that modulation of the TME may enhance therapeutic efficacy.
From a clinical perspective, although HRD in OCCC is less common, PARP inhibition remains a viable maintenance strategy in those patients with HRD OCCC. The integration of molecularly targeted approaches—such as ATR, EZH2, or PI3K pathway inhibitors—and ADCs offers additional therapeutic promise, particularly for ARID1A-deficient or HER2-overexpressing tumors. Preclinical and early-phase clinical studies suggest that exploiting synthetic lethality in ARID1A-mutant tumors, either through PARP and/or ATR inhibition, may represent a viable strategy, although further validation in larger, dedicated trials is required.
Overall, these findings highlight the necessity of histology-specific approaches to OCCC. While many therapies have been extrapolated from high-grade serous ovarian cancer studies, the distinct molecular and immunologic features of OCCC necessitate tailored strategies, incorporating both molecular profiling and innovative combination therapies. Future research should focus on refining biomarker-driven selection, understanding mechanisms of chemoresistance, and expanding access to clinical trials for patients with this aggressive ovarian cancer subtype.
