Abstract
Background:
Excessive waiting time intervals for the diagnosis and treatment of patients with pancreatic cancer can influence their prognosis but they remain unclear. The objective was to describe time intervals from the medical visit to diagnostic imaging and to treatment and their prognostic impact in pancreatic cancer in one French region.
Methods:
This retrospective observational multicentre study included all patients with pancreatic cancer seen for the first time in 2017 in multidisciplinary team meetings (MTMs), where clinical data were collected. A probabilistic matching with the medico-administrative data from the French national healthcare database (Système National des Données de Santé) was performed to define the care pathway from clinical presentation to the beginning of treatment. Median key time intervals were estimated for both resected and unresected tumours. Factors associated with 1-year survival were studied using Cox model.
Results:
A total of 324 patients (88% of total patients with MTM presentation) were matched and included: male 54%, mean age 72 years ±9.2, Eastern Cooperative Oncology Group (ECOG) PS > 1 19.5%, metastatic disease at diagnosis 47.4%, tumour resection 16%. At 1 year, 57% had died (65% in the unresected group and 17% in the resected group). The median time interval from the medical visit to diagnostic imaging was 15 days [Q1–Q3: 8–44]. After imaging, median time intervals to definite diagnosis and to first treatment were 11 and 20 days, respectively. Significant prognostic factors associated with the risk of death at 1 year were ECOG PS > 1 (hazard ratio (HR) 2.1 [1.4–3.0]), metastasis (HR 2.7 [1.9–3.9]), no tumour resection (HR 2.7 [1.3–5.6]) and time interval between the medical visit and diagnostic imaging ⩾25 days (HR 1.7 [1.2–2.3]).
Conclusion:
Delay in access to diagnostic imaging impacted survival in patients with pancreatic cancer, regardless of whether tumour resection had been performed.
Introduction
Pancreatic cancer is the fourth cause of cancer-related death in France1–3 and the third one in the United States. 4 Its incidence increased over the last 30 years, ranking it at the second cause of cancer-related death in developed countries by 2030.5,6 Pancreatic ductal adenocarcinoma, a fast-growing aggressive tumour, represents 85% of all pancreatic cancer.7,8 The 5-year survival rate can reach 20–30% in case of complete surgical resection followed by adjuvant chemotherapy9,10 but drops to 5–7% in case of unresectable disease.
Diagnosis is established at an advanced stage in 80% of cases, due to the non-specific presenting symptoms of the disease and long investigation procedures.11,12 A large number of patients seems to be oriented to specialized tertiary centres after an extended time interval,11,13 with a negative impact on the therapeutic approach and poor prognosis.
Key time points of the care pathway represent essential indicators to study the quality of the care continuum, in order to highlight disparities and to access inequality to healthcare.14,15 Time to diagnosis or treatment varies according to the cancer type, geographical area, and socioeconomic status of patients.15–17 Care pathways also depend on the resectability status of pancreatic cancer at the time of diagnosis. Regarding pancreatic cancer, multiple independent studies support contradictory results, difficult to compare as healthcare systems differ tremendously in European and non-European developed countries. In France, very few studies have been carried out over the last decade, suggesting that time intervals from the first symptoms to the first specialized visit and from the first visit to the first treatment often exceed 1 month.18–20 However, there are no recommendations in the Guidelines concerning time intervals in diagnostic and treatment.
The primary objective of our study was to provide an accurate description of the different care pathways and key time intervals for patients with a new diagnosis of pancreatic cancer in 2017 in one vast French region (2.5 million of inhabitants). The secondary objectives were to identify factors associated with delay from diagnostic imaging to definitive diagnosis and from diagnostic imaging to first treatment, and eventually to identify factors associated with survival at 1 year.
Materials and methods
Study design and data sources
The Dossier Communicant en Cancérologie (DCC; Shared Files in Cancerology) is a French regional register gathering data about cancer and treatment decision of all new cancer patients diagnosed, discussed in a multidisciplinary team meeting (MTM; Réunion de Concertation Multidisciplinaire). Each adult patient (⩾18 years) with a clinical presentation of pancreatic or related cancer discussed in MTM in 2017 was selected and checked by reviewing the electronic medical report before inclusion (exclusion of other pancreatic or similar pathological diagnosis of peri ampullary malignant tumour, other digestive malignant tumours with pancreatic location and cases of histological definitive diagnosis made before 2017).
As the DCC does not include all follow-up data, particularly death, the French Health Insurance Database [Système National des Données de Santé (SNDS)] was also used to describe the entire care pathway of patients. The SNDS represents the French medico-administrative database including all care reimbursement data as well as the date of death. A probabilistic matching was performed between the two anonymous databases based on the following variables: sex, month and year of birth and dates of all major acts of the medical care registered (imaging, definitive diagnosis, first treatment). The research data set then allowed to describe the included patients and their care pathway.
Definition of the care pathway key points and time intervals
Patients were classified in two major groups according to the final treatment performed: resection with curative intent and or no resection, using the medico-administrative data (International Classification of Diseases, Tenth Revision) and the national Common Classification of Medical Procedures (Classification Communes des Actes Médicaux) or clinical data (DCC). Key points and time intervals of these two different major care pathways were defined by the multidisciplinary research team including epidemiologists and gastroenterologists, based on the major determinant investigations and treatments performed according to French Recommendations, including medical visit with a general practitioner (GP) and/or a specialist prior to diagnostic imaging, diagnostic imaging, cancer definitive pathological diagnosis, first treatment and death. The time intervals were estimated for the most common order of management (investigation then treatment) of pancreatic cancer, including the following steps: (i) from the last medical visit performed to diagnostic imaging, (ii) from diagnosis imaging to cancer definitive diagnosis, (iii) from diagnosis imaging to first treatment and (iv) from diagnosis imaging to death. In some cases, the sequence between two steps in the care pathway was reversed and results were excluded. Eventually, only 3% of the time intervals (negative or not available due to missing data) were excluded from the analyses.
Statistical analyses
First, analyses were performed to describe the characteristics of patients and the different steps and median time intervals of each care pathway, according to the performance of resection surgery or not.
Second, factors associated with 1-year survival were identified by a Cox regression model giving hazard ratio (HR) along with their 95% confidence intervals (95% CI). A sensitivity analysis for 6-month survival was performed on the subgroup of patients with unresectable disease. Factors associated with time to diagnosis and to first treatment were identified by multiple linear regression models, giving estimates and their p-value. For patients with resected disease, the first treatment coincides with diagnostic confirmation in most cases (surgical resection) with an expected bias in time intervals; therefore, those patients were not included in the analysis of factors associated with time to diagnosis.
All variables with p < 0.2 in bivariate analysis were included in the initial multivariate model for each care pathway. Then, a descending stepwise process was used to select the final model, including all the statistically significant variables at the threshold p < 0.05. The analyses were performed with SAS Enterprise Guide 71 64-bit (SAS Institute Inc., Cary, NC, USA), version available on the SNDS website at the moment of the analyses.
This health assessment study fell within the framework of research not involving the human being and was conducted in accordance with the French and European laws and regulations in force, as well as any other applicable guidelines. This project has obtained an authorization from the French data protection board (Commission Nationale de l’Informatique et des Libertés) allowing the access to SNDS data, decision DR-2019-113. Moreover, the study was granted a waiver of the individual information requirement by the French data protection board, as the majority of patients were unfortunately deceased at the time of the retrospective study and their personal details were not known. As an alternative measure, collective information about the study was made available and required by the protection board on the websites of the regional epidemiology unit and of the regional oncology centre, mentioning the rights of access, rectification and opposition, in accordance with the provisions of article R. 1461-9 of the CSP, as well as the following rights and information, in accordance with the European Data Protection Regulation.
Results
Baseline characteristics of patients
Among the 367 patients eligible for the study, 324 (88%) were included after matching (Figure 1). Mean age was 72 ± 9.2 years old. Most patients were males (53.7%) and presented metastasis at diagnosis (47.4%). Two hundred and sixty-five patients (85%) presented at least one symptom before diagnosis. The most common symptoms were abdominal or back pain (n = 141, 45.5%) and jaundice (n = 137, 44.2%) (Supplemental Table 1). The last medical visit before diagnostic imaging was performed by a GP in 65.2% of cases. A total of 84 patients (25.9%) were treated in the Regional University Hospital, 116 patients (35.8%) in peripheral hospital centres and 124 patients (38.3%) in private healthcare facilities. Two hundred and three patients (62.7%) received chemotherapy (43% treated by FOLFIRINOX regimen and 43% by gemcitabine), of which 89 as a first line treatment. Curative resection concerned 53 patients (16%): 30 as a first treatment, 3 after chemotherapy and 20 after biliary endoscopic drainage where cancer was mostly located at the head region of the pancreas. The characteristics of patients with unresectable disease (84% of cases) were comparable to the whole cohort (Supplemental Table 2).

Flow chart of the study population selection. MTM, multidisciniplary team meeting; DCC, Dossier Communicant en Cancérologie (Shared Files in Cancerology); SNDS, Système National des Données de Santé (French Health Insurance Database).
The median time intervals from prior medical visit to diagnostic imaging, from diagnostic imaging to cancer diagnosis (pathological diagnosis when possible/available; 38% of missing data) and from diagnostic imaging to the first treatment performed were 24.5 days, 11 days and 19.5 days, respectively [Figure 2(a)].

Care Pathway Key Points of Pancreatic Cancer, regarding: (a) All patients (n = 324). (b) Patients with unresected tumour (n = 271). (c) Patients with resected tumour (n = 53).
Patients with unresectable disease had median interval times that seemed longer compared to those of patients with resectable disease: 27 days versus 13 days for the median interval time from prior medical visit to diagnostic imaging and 22 days versus 14 days for the median interval time from diagnostic imaging to the first treatment [Figure 2(b) and (c)].
The median time interval from prior medical visit to diagnostic imaging varied according to the specialty of the practitioner, longer when patients consulted a GP before diagnostic imaging as compared with a specialist (24 versus 15 days).
Factors associated with the time interval from diagnostic imaging to definitive diagnosis
For patients with unresectable disease (n = 256 with exhaustive data, 95%), the presence of a venous thromboembolism was associated with an increased time interval from imaging to cancer diagnosis (+25.8 days; p < 0.01) (Table 1). Moreover, without reaching significance, jaundice as first symptom (p = 0.06) and a WHO performance status (WHO PS) ⩾ 2 (p = 0.06) tended to be associated with a decreased time interval (−8.7 days and −9.7 days, respectively).
Factors associated with the time interval from diagnostic imaging to cancer diagnosis.
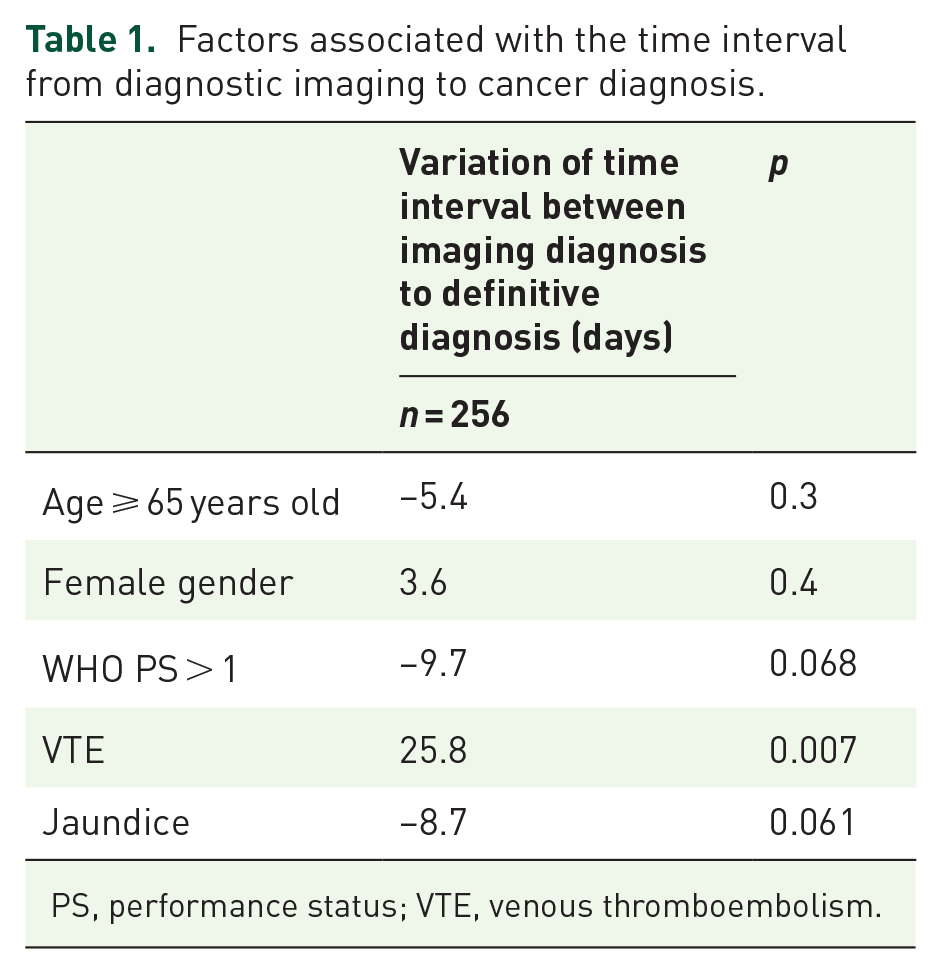
PS, performance status; VTE, venous thromboembolism.
Factors associated with the time interval from diagnostic imaging to first treatment
For all patients (n = 283 with exhaustive data and having a first treatment registered, 87%), an increased time interval was found when the diagnosis was made in a private healthcare facility (+7.5 days; p = 0.04) (Table 2). On the contrary, jaundice as first symptom (p < 0.0001) and a WHO PS ⩾ 2 (p = 0.01) were significantly associated with an important decrease of this time interval (−30 days and −12 days, respectively).
Factors associated with the risk of death at 1 year – all patients.

PS, performance status; VTE, venous thromboembolism; CVL, Centre-Val de Loire.
Missing data = 16.
Factors associated with death at 1 year
One year after their inclusion, 57% of patients were dead (65% for patients with unresectable disease versus 17% of patients with resectable disease) (Table 3). At 2 years, 81.7% of the cohort population was dead (n = 265);
Factors associated with the time between imaging diagnosis and first treatment – all patients.

PS, performance status.
A WHO PS ⩾ 2 (HR 2.4 [1.4–3.0]), metastatic disease at diagnosis (HR 2.7 [1.9–3.9]), unresectable disease (HR 2.7 [1.3–5.6]), care pathway undertaken in the administrative area of Eure-et-Loir (HR 2.7 [1.3–5.6]), and a median interval time from consultation to imaging ⩾25 days (HR 1.7 [1.2–2.3]) were independently associated with a higher 1-year mortality rate. Moreover, the management in the administrative area of Indre tended to be associated with a higher mortality rate (HR 2.9 [0.9–9.5]).
Similar results were found after the sensitivity analysis in the subgroup of patients with unresectable disease, 6 months after inclusion (Supplemental Table 3).
Discussion
This is the first study to date allowing a comprehensive approach of healthcare pathways for patients with pancreatic cancer at all stages in France. Thanks to the matching of an exhaustive medico-administrative database with medical records of MTM, completeness and analysis strength were greatly enhanced. Hence, an accurate description of the care pathway in real-life conditions of these patients was provided in a vast French region of more than 2.5 million inhabitants. We observed that delay in access to diagnostic imaging impacted survival, regardless of the tumour resection.
Median diagnostic and treatment time intervals appeared to be shorter in our study compared to others European population-based studies, for both resected and unresected patient groups.21–23 These estimated time intervals could not be easily compared to the literature, where a majority had a starting point at the first symptom occurrence or from the first consultation; these data related to the very first steps of the care pathway were not available in our study. However, our median time intervals from prior medical visit to diagnostic imaging and from diagnostic imaging to definitive diagnosis seem longer as compared to a median diagnosis time interval of 14 days reported in the French REPERE study of metastatic patients and 0.51 month (about 15 days) in another recent French population-based study.19,20 However, the limitation of these two studies is that they are oligocentric and not population-based and do not accurately reflect the entire healthcare system. Similar trends were shown concerning factors associated with a shortened time interval between diagnostic imaging and first treatment for both patients with resected or unresected disease in several international studies where, however, the role of jaundice remains paradoxical, sometimes associated with delays, sometimes with shortened time intervals.19,23–25 A British study specifically dedicated to the analysis of symptoms occurring prior to pancreatic cancer diagnosis highlighted diabetes, back pain and self-reported anxiety or depression as factors associated with longer diagnostic delays. 24 In our study, the delay in obtaining a definitive pathological diagnosis in the presence of a deep venous thromboembolism might be explained by the postponement of invasive explorations, in particular biopsy, due to the high risk of bleeding under anticoagulation treatment.
It is the first study to provide evidence about the relationship between the waiting time from the medical consultation to diagnostic imaging and prognosis in real-life setting with a time interval over 25 days between medical visit and diagnostic imaging significantly associated with 1-year survival. Prognosis of these patients was also largely influenced by patient and tumour features, as well as the first geographical area of the medical facility taking the patient in charge. Those factors previously emerged as independent and significant prognosis factors in other studies.26–28 For both patients with resectable and unresectable tumours, Gobbi et al. showed significant differences in survival when considering diagnosis was made before 4 weeks, versus between 4 and 16 weeks and after 16 weeks. 27 In the study of Apollos et al., survival was better in the group where less additional investigations were performed resulting in a shorter diagnostic time 29 whereas Raptis et al. 25 showed that a delay from symptoms to referral had minimal clinical relevance to survival.
Time interval from diagnosis to first treatment was not reported as a prognostic factor as it could have been expected, results shown by other international studies including all stages of pancreatic cancer20,21,27,30; however, it has recently been shown by a Swedish study and one very large-scale American study that this delay has a negative prognostic impact when it is prolonged in patients with a resectable tumour.31,32
Diagnostic imaging is determining for pancreatic cancer and before discussing any surgical treatment with curative intent, French recommendations require a recent imaging performed within the last 4 weeks.33, 34 In France, abdominal CT scan is the second most performed imaging exam in the Emergency departments, but waiting time intervals still remain variable. 35 In CVL region, CT scan equipment is probably not related to the diagnostic imaging time intervals, as the equipment density seems lighter only in the Loir-et-Cher and the Eure-et-Loir 36 compared to the national density. Organizational and professional factors should be preferentially targeted; as a matter of fact, the population density of regional specialists eligible to take care of pancreatic cancer patients is lower as compared to the national level. 37 Education of primary care professionals about the necessity of an increased awareness of the risk of pancreatic cancer in patients with identified risk factors (obesity, diabetes, smoking, family history of pancreatic or related cancer, hereditary chronic pancreatitis) might help improving pancreatic cancer diagnosis.38,39 No evidence of specific symptomatic presentations preceding a pancreatic cancer diagnosis was shown in the ‘SYMPTOM pancreatic study’. 24 Systematic and fast recourse to diagnostic imaging such as C -scan in the presence of any symptom potentially compatible with pancreatic neoplasia seems hardly feasible in practice in terms of organization and cost.
Our study has several limitations. The study included only patient presented at the MTM. It is possible that a small number of patients could not have been discussed on time at the MTM due to very early death. As previously reported, the percentage of these patients is certainly small.40,41 Moreover, due to a lack of completeness of the DCC medical forms, it was not possible to distinguish non-operable patients with borderline tumours (due to possible co-morbidities) from patients with locally advanced unresectable tumours. In the DCC, the classification of tumours according to the standardized TNM Classification of Malignant Tumours (Tumor Node Metastatis classification) is requested but not mandatory. In the SNDS, only the date of the pathology examination is known, without the detailed result. It was possible to obtain an approximative number of metastatic cases by coding (CIMX C77-C79 diagnostic codes during hospitalizations). With a matching process, 12% of patients were excluded due to missing matching data between the medical register (lack of completeness on dates) and the SNDS (missing diagnosis codes of pancreatic cancer). As previously highlighted, data related to the very first steps of the care pathway were not available in our study. Finally, these results are only the reflection of a territorial organization at the scale of our region. However, an exhaustive collection of data from medical registers (including pathological diagnosis, when available) offsets a potential inclusion bias, as medical files were checked one by one by a resident of the gastroenterology ward, giving a better assessment on high-certainty cases. SNDS provided a major contribution to this study, as clinical data could be completed after matching and their quality checked, tracing precisely all the steps of the different care pathways. Using data from clinical data warehouses could be a good substitute through cost-saving automated extractions of useful structured and unstructured data. 42
In conclusion, our study showed that a delay in access to diagnostic imaging impacted survival in patients with pancreatic cancer, regardless of tumour resection. However, a fast, generalized access to abdominal CT scan in case of occurrence of suspicious symptoms seems unrealistic for both economic and organizational reasons. Additional struggle to target high-risk populations, to educate primary care professionals and to develop effective treatment for pancreatic cancer remains a challenge for the future. This appears all the more justified since pancreatic cancer incidence increases dramatically. Thanks to the matching between clinical and medico-administrative electronic databases, one comprehensive approach of healthcare pathways for patients with cancer; this approach should be enhanced in future national studies, as big data in health research is in progress with a huge French legacy in the historic French medico-administrative databases and the development of French clinical data warehouses.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221113264 – Supplemental material for Time interval from last visit to imaging diagnosis influences outcome in pancreatic adenocarcinoma: A regional population-based study on linked medico-administrative and clinical data
Supplemental material, sj-docx-1-tam-10.1177_17588359221113264 for Time interval from last visit to imaging diagnosis influences outcome in pancreatic adenocarcinoma: A regional population-based study on linked medico-administrative and clinical data by Vittoria Balzano, Emeline Laurent, Aline-Marie Florence, Anne-Isabelle Lecuyer, Carole Lefebvre, Patrick Heitzmann, Pascal Hammel, Thierry Lecomte and Leslie Grammatico-Guillon in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Authors thank Florence Cocqueel-Tiran and Céline Raveneau (Réseau Régional de Cancérologie – Oncocentre), Hortense Richardson and Béatrice Cottu (Département d’Information Médicale, Centre Hospitalier de Blois), Esther Palanghi (Centre de Coordination en Cancérologie de Blois-Vendôme) and all the 3C teams members of Centre-Val de Loire region who contributed to the feasibility of this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
