Abstract
Background:
The effect of gender on the prognosis of bladder cancer (BCa) in different metastatic sites is insufficiently understood. We aimed to assess the impact and potential mechanisms of a combination of gender dimorphism and BCa metastasis sites on the risk of death.
Methods:
Independent predictors of overall survival and cancer-specific survival were analyzed after stratification by gender and metastasis sites from the Surveillance, Epidemiology, and End Results database. Furthermore, gender-differentially expressed genes (DEGs) and function-enriched annotations for patients with lymph node metastasis (LNM) were identified from The Cancer Genome Atlas (TCGA) database. A gender-associated signature was constructed in TCGA and validated in the IMvigor210 trial, and the magnetic resonance imaging-based radiomics signature was developed in our center to predict the gender-associated signature.
Results:
In patients with metastatic BCa, the most common site of metastasis is bone in men and lung in women. Moreover, stratified by sex, LNM had a better prognosis in men than visceral metastasis, which was not observed in female. Similarly, stratified by the metastasis site, the prognosis of men in patients with LNM is better than that of women, which was not observed in visceral metastasis patients. Enrichment of DEGs between sexes in patients with LNM may be related to metastasis and tumor immunity, especially the role of neutrophils. Moreover, the gender-associated signature is related to the clinicopathological characteristics of patients, and patients in the high-risk group had worse survival outcomes, and higher susceptibility to cisplatin, docetaxel, camptothecin, and paclitaxel. A nomogram combined with the signature and clinical staging showed significant predictive power in survival prediction. Furthermore, patients with high radiomics scores had a strong tendency for high-risk group.
Conclusion:
These results may improve the understanding of the differences in tumor biology between sexes and thus provide additional evidence for individualized treatment in BCa.
Introduction
It is estimated that bladder cancer (BCa) was the fourth common cancer and the eighth fatal tumor in men in 202. 1 Muscle-invasive or metastatic BCa occurs in 25% of patients at the time of initial diagnosis, and the 5-year relative survival for metastatic BCa is only 12%.2,3 Chemotherapy is currently generally the preferred treatment for metastatic BCa. 2 However, the median survival of untreated metastatic BCa rarely exceeds 3–6 months.4,5 Therefore, early diagnosis and treatment of metastatic BCa are of paramount importance. It has been reported that some factors, such as gender, age, circulating tumor cell, and treatment, may be related to the survival of metastatic BCa.6–9 However, the effects of these factors on the site of metastasis are unknown.
In recent years, some studies on epidemiology and translational research have fully revealed the gender differences in the incidence and outcome of BCa.10–12 Although the incidence of BCa is higher in men, women are more likely to be diagnosed with advanced disease and are more likely to disease recurrence, progression, and poorer outcomes than men.13,14 In addition, the 5-year survival difference between men and women in patients with stage IV BCa has been reported to be estimated at 12%.15,16 There are multiple theoretical explanations for this gender difference, including genetic risk factors, different carcinogen exposures, differences in sex hormone regulation, and changes in treatment.13,17 To the best of our knowledge, the difference in prognosis between genders with distant metastasis patterns in BCa patients is currently unknown.
In this study, we used clinical and demographic factors from the Surveillance, Epidemiology, and End Results (SEER) Program. Our study found that in patients with metastasis BCa, the prognosis of women is worse than that of men, especially in patients with lymph node metastasis (LNM), but not in visceral metastasis. In addition, to further explore the reasons for the worse prognosis of LNM of female, we analyzed the gender-differentially expressed genes (gDEGs) in the LNM population and constructed a gender-associated signature using The Cancer Genome Atlas (TCGA) database. The gender-associated signature can independently predict the drug susceptibility and prognosis of BCa patients. Moreover, the radiomics signature from our center was constructed to predict gender-associated signature to more easily identify drug sensitivity and prognosis in BCa patients. Therefore, this study is focused on raising the awareness on gender differences in the prognosis of BCa patients, and could provide a new perspective for research and for proposing gender-based options for treatment of BCa.
Materials and methods
Database and patient selection
The SEER database is a basic cancer registry that covers approximately 28% of the population of the United States, recording basic demographic and clinicopathological features of cancer patients. In all, 1829 patients were screened through the flow chart (Figure S1).
The original counts and corresponding clinical information from RNA sequencing data for 408 BCa were obtained from TCGA dataset, which included 129 pathologically proven BCa tissues with LNM (97 males and 32 females). Transcriptome data from the IMvigor210 trial, including patients with metastatic urothelial carcinoma treated with atezolizumab (programmed cell death-Ligand 1 [PD-L1] inhibitor), were downloaded from was downloaded from the R package ‘IMvigor210CoreBiologies’ (version 1.0.0).
The studies involving human participants were reviewed and approved by the Ethics Committee of Shanghai Tenth People’s Hospital (approval number 2021KN108). Informed consent of patients was waived due to its retrospective nature. BCa patients who visited the Shanghai Tenth People’s Hospital between November 2019 and July 2021were collected through the flow chart (Figure S2). The protocols of magnetic resonance imaging (MRI) acquisition, total RNA extraction, paired-end libraries generation, and RNA-sequence can be obtained from our previous studies.18,19
Specific methods of bioinformatics analysis, radioomics feature extraction and construction, and immunohistochemistry are described in the supplemental methods.
Statistical analysis
The chi-square test was used to compare categorical variables between men and women and the Student’s t-test was used for continuous variables. We used the Kaplan–Meier method to generate survival curves and performed log-rank test to compare the cancer-specific survival (CSS) and overall survival (OS) of each group. Multivariate Cox proportional risk regression models were used to assess the factors associated with CSS and OS, respectively, and to compare the association between gender and metastases sites and survival by adjusting for potential confounders. The hazard ratio (HR) and 95% confidence interval (CI) were estimated. Log rank was used to test Kaplan–Meier survival analysis to compare the survival differences between the two groups, and time-dependent receiver-operating characteristic (ROC) analysis was performed to compare the risk scores. All statistical tests were bilateral and p values <0.05 were considered statistically significant. The GraphPad Prism version 7.0, SPSS version 25.0, and R software (version v4.0.3) were used for statistical analysis and graphical work. The workflow schematic is shown in Figure 1.

Workflow schematic.
Results
Patient characteristics
A total of 79,312 eligible BCa patients were obtained from the SEER database, and 22% of these patients were women (Table S1). Furthermore, we selected 1829 BCa patients with a single metastasis, including 1314 male patients (72%) and 515 female patients (28%) (Table 1). Women were more likely than men to be divorced/widowed (44.3% versus 24.5%; p < 0.01), Black (14% versus 8.8%; p < 0.01), and more likely Grade I to Grade II (6.4% versus 3.8%; p = 0.042) and non-transitional urothelial carcinoma (TUC; 26.2% versus 17.7%; p < 0.01). Females were less likely than males to receive chemotherapy (47.2% versus 57.1%; p < 0.01). There was also no difference in distant metastasis between male and female patients. However, among the sites of metastasis, we found that bone remained the most common site of metastasis for male BCa patients (34.9%), followed by lung (29.4%), lymph nodes (21.2%), liver (12.9%), and brain (1.6%), while in female patients, the most common site of metastasis was lung (33%), followed by bone (28.3%), lymph nodes (22.5%), liver (15.1%), and brain (1.0%). Table 1 summarizes the differences in demographic characteristics between the sexes.
Sex comparisons of demographic and clinical features in patients with metastatic BCa.

BCa, bladder cancer; TUC, transitional urothelial carcinoma; TURBT, transurethral resection of bladder tumor.
Gender differences in the association of single metastatic sites with risk of mortality
In analyzing the effect of gender on the prognosis of BCa, we found that female patients had worse CSS and OS (p < 0.05; Figure S3A, B), and a similar phenomenon was observed in patients with metastatic BCa (p < 0.05; Figure S3C, D). Next, we explored the impact of different sites of metastasis on the prognosis. Kaplan–Meier analysis showed that patients with LNM had a better prognosis than those with visceral metastasis (p < 0.05; Figure 2(a) and (b)). Furthermore, through multivariate cox regression analysis, we found that metastatic sites, together with age, tumor T stage, surgery, and chemoradiotherapy, were all independent prognostic predictors of OS and CSS (p < 0.05; Table 2). Moreover, compared with LNM, patients with visceral metastasis (bone, lung, liver, and brain) had worse prognosis (p < 0.05). Furthermore, we explored whether there were gender differences in the prognostic implications of different metastasis sites. Then the data stratified by gender, and Kaplan–Meier survival curve showed that among male patients, the prognosis of patients with LNM was better than that of patients with visceral metastasis (p < 0.05; Figure 2(c) and (d)). However, a similar phenomenon was not observed in female patients (p > 0.05; Figure 2(e) and (f)). Subsequently, multivariate cox analysis revealed that in male patients, metastatic sites, together with age, tumor T-stage, and chemotherapy, were independent prognostic predictors of CSS and OS (p < 0.05; Table 3). Moreover, the prognosis of visceral metastasis is worse than that of LNM, and the prognosis of patients with brain metastasis is the worst (p < 0.05). In contrast, in female patients, only tumor T-stage, surgery, and chemotherapy were independent prognostic predictors of CSS and OS (p < 0.05; Table 3).
Multivariate cox regression analysis based on demographic factors.

CI, confidence interval; CSS, cancer-specific survival; HR, hazard ratio; OS, overall survival; TUC, transitional urothelial carcinoma; TURBT, transurethral resection of bladder tumor.
Multivariate survival analysis of patients with metastatic BCa by sex stratification.

BCa, bladder cancer; CI, confidence interval; CSS, cancer-specific survival; HR, hazard ratio; OS, overall survival; TURBT, transurethral resection of bladder tumor.

Kaplan–Meier curves for OS and CSS, by site of metastasis in the overall population (a, b), and for males (c, d) and females (e, f).
Differences in metastatic sites between sex and risk of mortality
The above results show that the prognosis of LNM in male patients is better than that of visceral metastasis, but there is no significant difference in female patients. On this basis, we further explore whether LNM and visceral metastasis can affect the prognosis of gender. Then we stratified according to the metastasis site, and Kaplan–Meier survival curve showed that the prognosis of male patients with LNM was better than that of female patients (p < 0.05; Figure 3(a) and (b)), whereas in patients with visceral metastasis a similar phenomenon was not observed (p > 0.05; Figure 3(c) and (d)). Subsequently, multivariate cox regression analysis revealed that in patients with LNM, gender, together with age, tumor T-stage, surgery, and chemotherapy, were independent prognostic predictors of OS and CSS (p < 0.05; Table 4). The prognosis of female patients was worse than that of male patients (p < 0.05). In addition, the survival of female patients receiving radical cystectomy (RC) and chemotherapy can be significantly prolonged, while that of male patients only receiving chemotherapy (Tables S2 and S3). In contrast, in patients with visceral metastasis, only age, tumor T-stage, grade, surgery, and chemotherapy were independent prognostic predictors of OS (p < 0.05; Table 4); Besides, chemotherapy and grade were independent prognostic factors of CSS (p < 0.05; Table 4). There is also no difference in prognosis between male and female patients. Moreover, OS could be significantly prolonged after transurethral resection of bladder tumor (TURBT) and chemotherapy in male patients, while that of female patients only receiving chemotherapy (Tables S4 and S5).
Multivariate survival analysis of patients with metastatic BCa by metastatic site stratification.

BCa, bladder cancer; CI, confidence interval; CSS, cancer-specific survival; HR, hazard ratio; LNM, lymph node metastasis; OS, overall survival; TURBT, transurethral resection of bladder tumor.
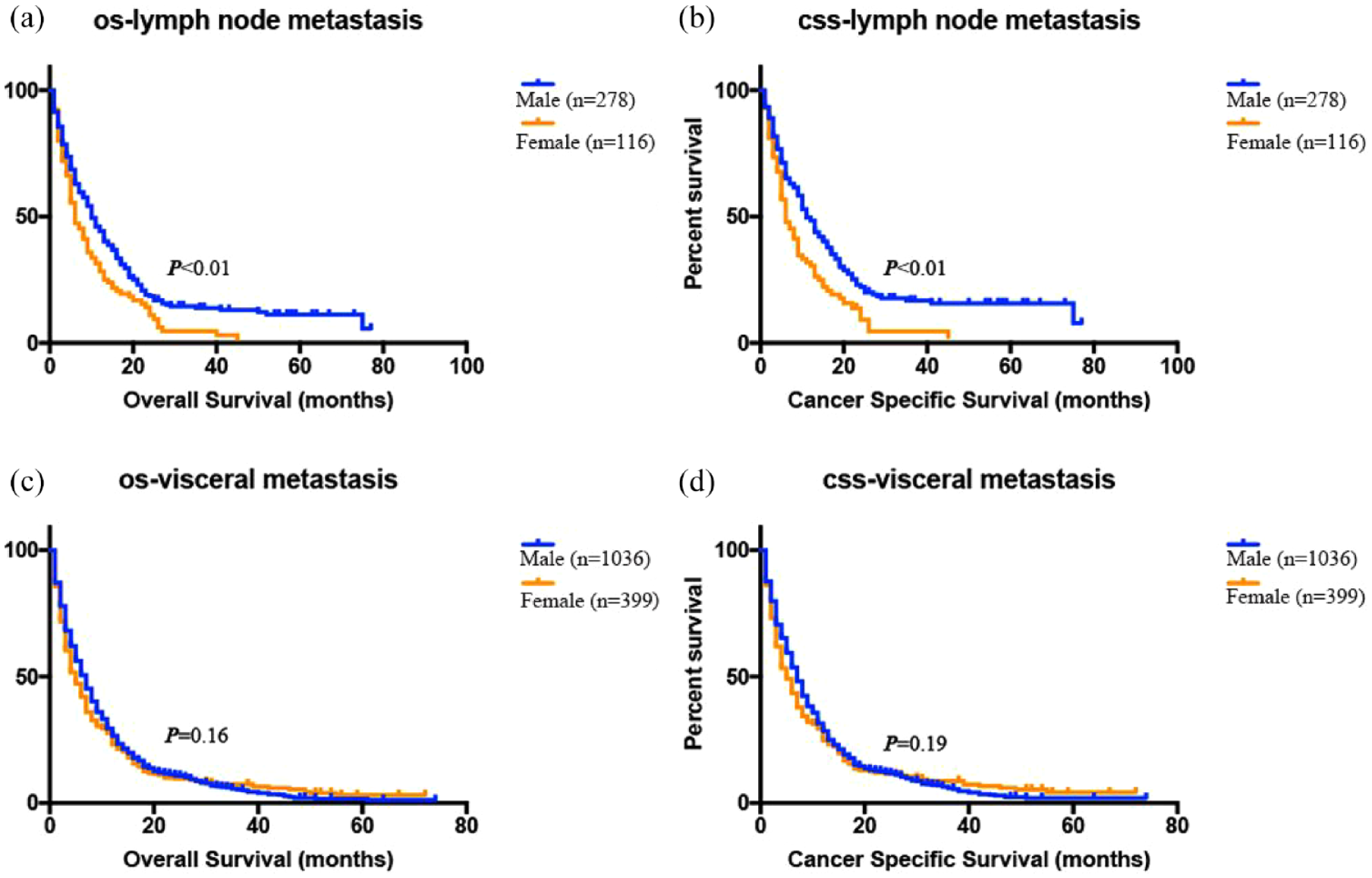
Kaplan–Meier curves for OS and CSS, stratified by sex in patients with LNM (a, b), and patients with visceral metastasis (c, d).
Identification of DEGs between sexes in patients with LNM
Based on the above findings, the prognosis of women with LNM is worse than that of men. To further explore the potential mechanism, the ‘Assistant of Clinical Bioinformatics’ was used to conduct DEGs analysis on mRNA expression profile data of LNM BCa tissues of men and women in the TCGA database. A total of 30 DEGs were identified, of which 21 genes were highly expressed in male patients and 9 genes were highly expressed in female patients (Figure S4A-B). And then, GO enrichment analysis was performed on DEGs. Cell junction assembly, zinc homeostasis, and neutrophil migration were highly abundant terms for GO in biological processes (Figure 4(a)). Furthermore, in LNM BCa tissues, the expression of neutrophil infiltration in women was relatively high (Figure 4(c)), although there was no statistical difference, possibly due to the small sample size. The overall population was then analyzed and found to have higher neutrophil infiltration in women than in men (Figure 4(d)). Similarly, samples stained by IHC from our center also showed consistent findings (Figure 4(e) and (f)). In addition, both the TIMER database and the IMvigor210 database show that BCa patients with high neutrophil expression have poor prognosis (Figure 4(g) and (h)). The results of the KEGG enrichment analysis indicated that PI3K-Akt signaling pathway and extracellular matrix (ECM)-receptor interaction were highly enriched KEGG terms (Figure 4(b)).

The function of DEGs between sexes in LNM patients. (a) Functional enrichment: the enrichment result of differential gene KEGG pathway. (b) Enrichment results of DEGs GO term; among them, different colors represented the significance of differential enrichment results, and a larger value represented the smaller FDR value. p < 0.05 or FDR < 0.05 for enrichment results. The sex difference in neutrophils from TCGA for the LNM population (c) and the overall population (d). Immunohistochemistry showed that the sex difference in neutrophils from our central population (e, f). The effect of neutrophils on the prognosis of BCa from TIMER (g) and in the validation set IMvigor210 trial (h).
Construction and evaluation of BCa risk model based on gender difference in patients with LNM
LASSO-penalized Cox analysis was used to reduce the number of 30 DEGs. Finally, seven genes (PRKY, AKR1B15, ALDH3B2, KRT23, PLA2G2F, SAA1, and LAMB3) were used to construct a gender-associated signature (Figure 5(a) and (b)). The risk score for each sample was calculated using the following method: Riskscore = (–0.0955) * PRKY + (0.1546) * AKR1B15 + (−4 × 10−4) * ALDH3B2 + (0.0496) * KRT23 + (−0.078) * PLA2G2F + (−0.0048) * SAA1 + (−0.0478) * LAMB3.

Prognosis value of gender-associated signature in BCa. (a, b) The coefficient of the selected feature is shown by the lambda parameter, the horizontal axis represents the value of the independent variable lambda, and the vertical axis represents the coefficient of the independent variable. The relationship between the partial likelihood deviation and log(λ) was plotted using LASSO Cox regression model. (c, d) Riskscore and survival time and survival state in the TCGA dataset. The Riskscore was shown as a scatter chart from low to high, and different colors represented different expression groups (c); scatter diagram of survival time and survival state corresponding to Riskscore of different samples, and heat diagram of gene expression in this signature (d). (e) The Kaplan–Meier survival curve distribution of this risk model in the training set TCGA database; median time represented the time (median survival time) in unit year corresponding to the survival rate of 50% in the high-expression group and the low-expression group. (f) The Kaplan–Meier overall survival curves for the two risk groups in the validation set IMvigor210 trial. (g) ROC curve and AUC of the risk model at different times, wherein the higher the AUC value, the stronger the prediction ability of the model.
BCa samples were divided into low-risk or high-risk group according to the median risk score (Figure 5(c)). The survival state and survival time of patients in the low-risk or high-risk group, and the relative expression criteria of the seven gender-related genes in each patient (Figure 5(d)). Results from the Kaplan–Meier survival curve showed that the OS was shorter in the high-risk group than in the low-risk group (p < 0.05; Figure 5(e)). Next, the IMvigor210 database was used to validate the accuracy of gender-associated signature and found that patients treated with PD-L1 inhibitors were divided into low-risk and high-risk groups according to the optimal cutoff value (−0.076). Kaplan–Meier survival analysis showed significantly lower OS in high-risk patients than in low-risk patients (p < 0.05; Figure 5(f)). To evaluate the efficiency of risk scoring in predicting 1-, 3-, and 5-year survival, we performed a ROC curve using data from the TCGA dataset. The AUC was 0.658 at 1 year, 0.687 at 3 years, and 0.69 at 5 years, indicating a high prediction (Figure 5(g)).
Relationship between BCa risk score and clinical features and drug sensitivity
Furthermore, we analyzed the distribution of the corresponding samples for risk scores by age, gender, smoking history, subtype, tumor TNM stage, clinical stage, and gene mutation landscape.Although the association of risk scores with age, smoking history, tumor NM stage, and gene mutation landscape was not significantly different (Figure 5(a), S5A, B), a higher risk score is more likely for females and basal squamous cell subtypes and is associated with higher T-staging and advanced pathological staging (p < 0.05; Figure 6(a)). In addition, the inhibitory concentration (IC)50 values for cisplatin, docetaxel, camptothecin, and paclitaxel were significantly lower in high-risk patients (Figure 6(b)–(e)), while there was no significant difference in IC50 values for gemcitabine and FGFR3 inhibitors (Figure 6(f) and (g)). To assess whether the risk score was an independent prognostic feature of BCa, univariate Cox regression analysis found that the gender-associated score was significantly associated with poorer OS, and other variables associated with poorer survival included age, clinical stage, and (TNM)-staging (p < 0.05; Figure 6(h)). In multivariate Cox regression analysis, only the risk score [p < 0.001; hazard ratio (HR): 4.62; 95% confidence interval (CI), 2.76–7.76) and clinical stage (p = 0.005, HR: 1.56, 95% CI, 1.14–2.13) were independent predictors of OS (Figure 6(i)).

Relationship between BCa risk score and clinical features and drug sensitivity in BCa. (a) The relationship between risk score and clinical pathological characteristics. (b) Cisplatin IC50 of the two risk groups. (c) Docetaxel IC50 of the two risk groups. (d) Paclitaxel IC50 of the two risk groups. (e) Camptothecin IC50 of the two risk groups. (f) Gemcitabine IC50 of the two risk groups. (g) FGFR3 inhibitors IC50 of the two risk groups. (h, i) Forest plots for univariate and multivariate Cox regression analysis in the TCGA cohort. **p < 0.05, ***p < 0.001.
Construction and evaluation of prognostic nomograms
A nomogram containing clinical staging and risk scores was constructed for OS prediction in BCa samples. The gender-associated signature, combined with clinical staging, shows significant predictive power in nomograms (Figure 7(a)). The graphs show that the observed and predicted OS at 1, 3, and 5 years show ideal agreement (Figure 7(b)–(d)). In addition, the AUC favored OS for 1 year (AUC = 0.656), 3 years (AUC = 0.689), and 5 years (AUC = 0.703) (Figure 7(e)).
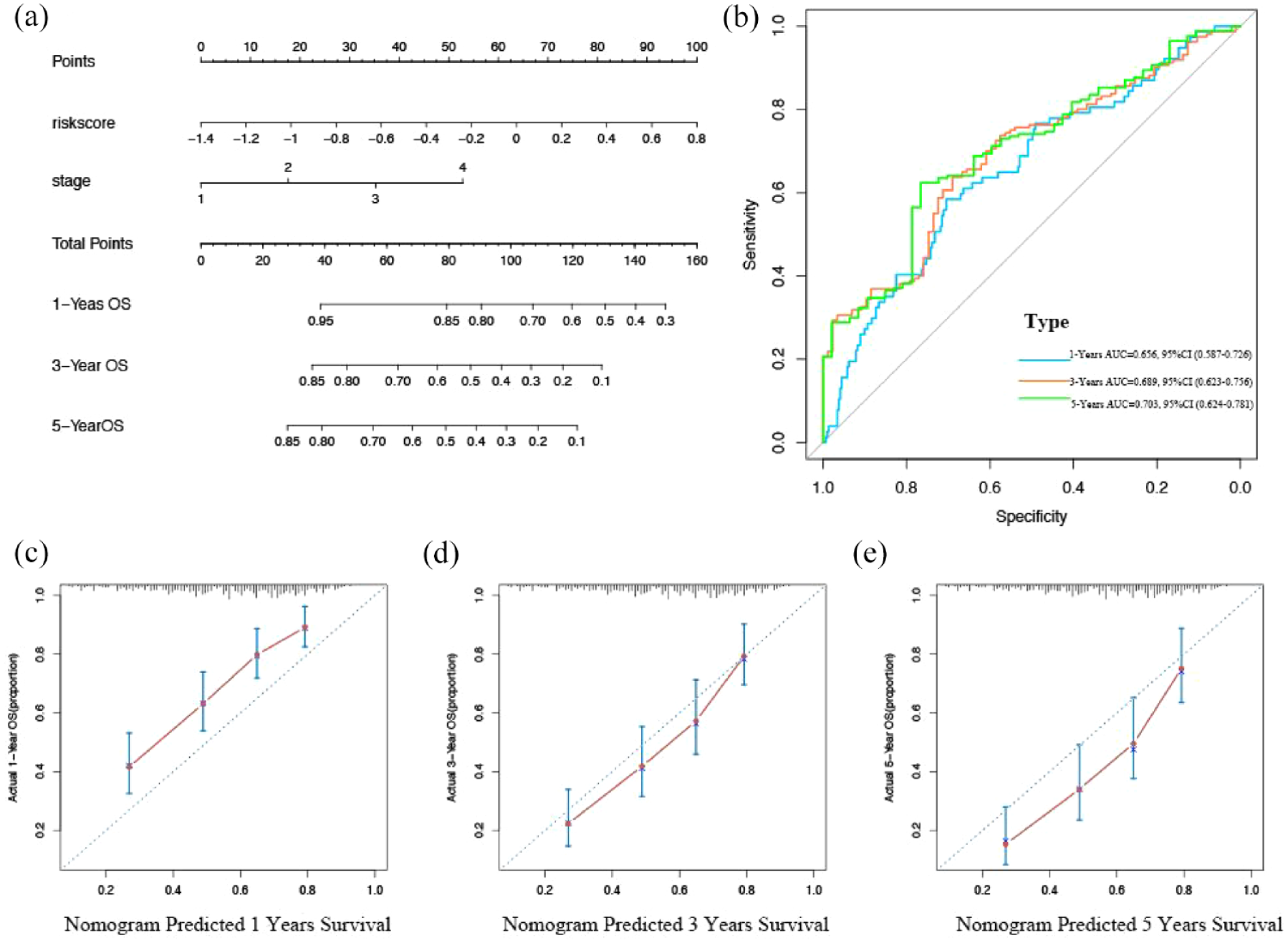
Predictive value of gender-related scoring model combined with clinical pathological characteristics for OS of patients with BCa. (a) Nomograms predict OS from TCGA cohort of BCa patients. (b) ROC curve and AUC of the nomogram at different times, wherein the higher the AUC value, the stronger the prediction ability of the model. (c–e) Calibration diagram for nomogram; the x-axis is the survival predicted by the nomogram, and the y-axis is the actual survival.
Construction and performance of the radiomics signature
The above results have shown that gender-associated signature can significantly predict the drug sensitivity and prognosis of BCa. However, not every patient can conduct genome sequencing to construct a gender-associated signature. At present, most patients with BCa will undergo MRI examination. Next, we will predict the gender-associated scores through radiomics signature to more conveniently identify the drug sensitivity and prognosis of BCa patients.
There were 111 BCa patients with RNA-sequence data and preoperative MRI in our center. The number of BCa patients in the training and validation sets were 77 and 34, respectively. Table S6 demonstrated the clinical characteristics of BCa patients. No statistical differences were found between training and validation sets. The intraclass correlations between two radiologists ranged from 0.762 to 0.908, suggesting a high inter- and intra-observer reproducibility. The top 10 features were selected by Max-Relevance and Min-Redundancy (mRMR) before radiomics signature construction. A LASSO model based on 10 radiomics features was constructed (Figure 8(a) and (b)). The coefficients of these 10 features are shown in Figure S6A. These 10 features were not highly correlated with each other (Figure S6B; mean absolute Spearman, ρ = 0.138). The AUCs of the LASSO model were 0.854 and 0.838 in the training and validation sets, respectively (Figure 8(c)). The accuracies of the LASSO model were 79.2% and 70.6% in the training and validation sets, respectively (Figure 8(d)). Patients in high-risk group had significantly higher radiomics scores than patients in low-risk group in both training and validation sets (Figure 8(e) and (f), both p < 0.001). The waterfall plot showed that patients with high radiomics scores had a strong tendency for high-risk group in the combined training and validation sets (Figure 8(g)).
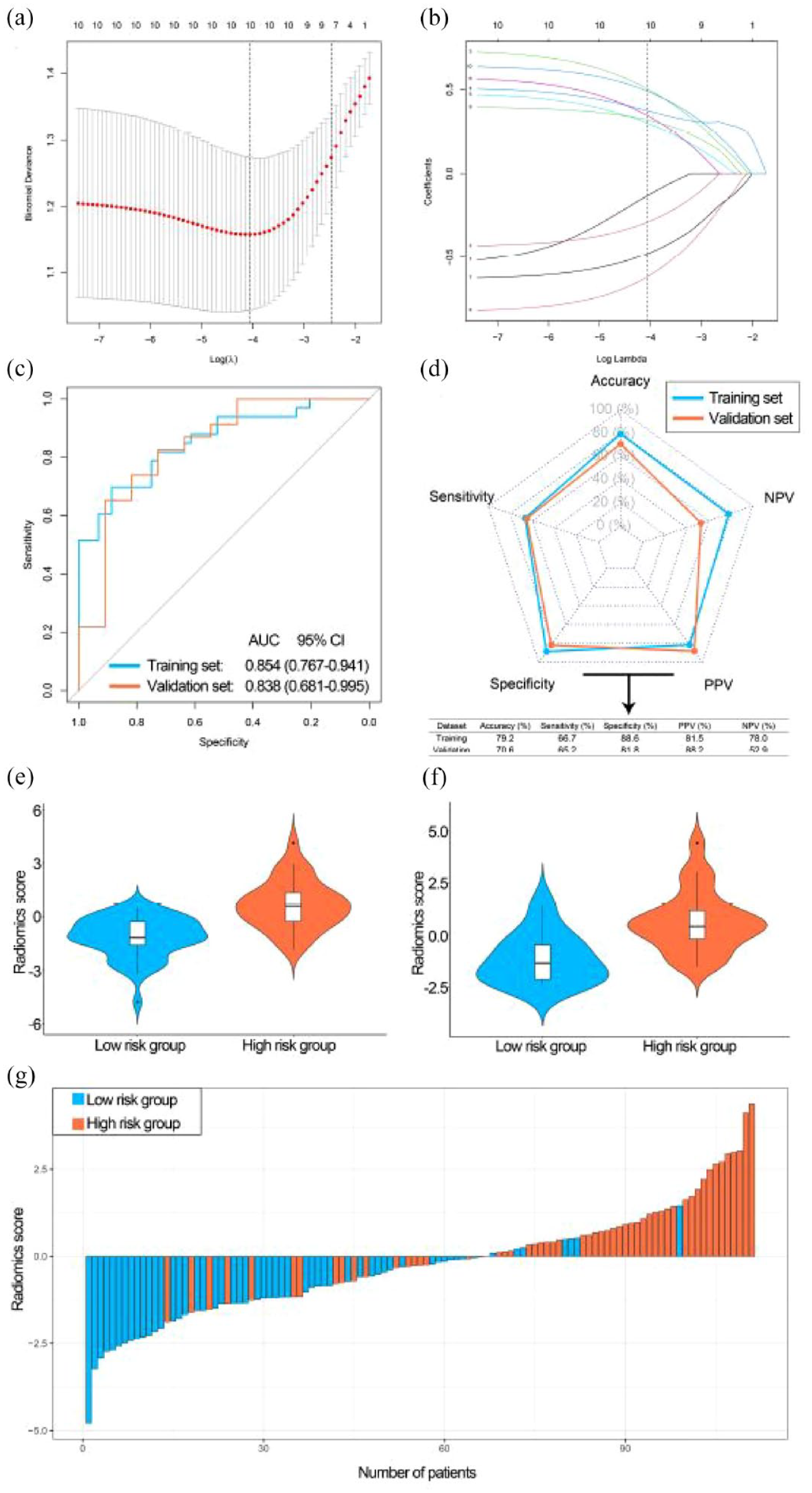
Development and performance of the radiomics signature (LASSO model). (a) Selecting the optimal number of features in the LASSO model based on minimum criteria in the training set. (b) Based on the optimal λ value, 10 radiomics features were selected. (c) The ROC curves of the radiomics signature in training and validation sets. (d) The performance of the radiomics signature in training and validation sets. (e) The violin plot of the radiomics score in the training set. (f) The violin plot of the radiomics score in the validation set. (g) Waterfall plot of the distribution of radiomics scores and risk groups of individual patients in the combined training and validation sets.
Discussion
Gender dimorphism shows significant differences in the incidence and mortality of BCa. In general, the incidence is higher in males, but the prognosis is more severe in females.13,20 Genetic and environmental factors as well as clinical management issues have been widely associated with gender-related differences in BCa outcomes.13,17,21,22 Metastatic BCa is the end stage of this malignancy and the survival after diagnosis is quite low. Despite a good initial response to chemotherapy, few patients achieved long-term OS.2,23 Previous studies have shown that some factors such as surgery, histological subtype, and sociodemographic disparities, etc. have been considered to be associated with the prognosis of metastatic BCa.24–26 However, the effect of a combination of gender dimorphism and BCa metastasis sites on the risk of death is currently unknown. Therefore, exploring the effect of gender dimorphism on metastatic BCa will help provide insight into the progression of BCa and thus have important guiding significance for a novel, gender-based approach to BCa management and clinical decision-making.
Our study found that in patients with metastatic BCa, the diagnosis was older in women than in men. This may be due to the fact that women have a delayed diagnosis due to hematuria and urinary tract infections, and are more likely to have an advanced disease at the time of diagnosis.27,28 Moreover, women are more often Black and in a non-TUC and divorced/widowed state. Some studies have shown that women and Black are more likely to suffer from the basal subtype of BCa, and the prognosis of BCa with the basal subtype is worse.21,29 In addition, women are less likely to receive chemotherapy than men. This is consistent with the study by Rose et al. 6 The above results may partly explain the low survival of female patients. Interestingly, in patients with metastatic BCa, the most common site of metastasis is bone in men and lung in women. Some studies have found that some biological processes such as hemodynamics and the ‘seed and soil’ hypothesis may affect the occurrence of bone metastasis. 30 However, there are a few studies on the mechanism that is responsible for lung metastasis to be more frequent in females, so elucidating the mechanism of lung metastasis in female patients with BCa remains a challenge.
To date, this is the first study to investigate the effect of a combination of gender dimorphism and BCa metastasis on the risk of death. First, our study has proved the gender difference associated with the risk of death in the single metastatic site. Compared with LNM, the prognosis of visceral metastasis is worse, especially that of brain metastasis. However, a similar phenomenon was not observed in female patients. Dong et al. 31 also found a better prognosis in LNM than in visceral metastasis in the general population, but did not perform sex stratification. Furthermore, chemotherapy can significantly improve the survival of BCa patients regardless of gender and metastatic site. In addition, RC can significantly improve the prognosis of women with LNM, while TURBT cannot. And TURBT can significantly improve the prognosis of men with visceral metastasis, while RC cannot. Therefore, it is essential for patients with BCa to choose the appropriate treatment. Second, to explore the effect of gender on the prognosis of BCa in different metastatic sites, we found that gender was an independent prognostic factor in patients with LNM through multivariate cox regression analysis. Furthermore, female patients have a poorer prognosis than male patients. A similar phenomenon was not observed in patients with visceral metastasis. Based on the above results, the prognosis of female patients with LNM is worse than that of male patients, and there is no gender dimorphism in patients with visceral metastasis.
Next, to further explore the underlying mechanism of a worse prognosis in women than in men with LNM, we performed DEGs on the expression profiles of LNM BCa in men and women in the TCGA database. And then, GO enrichment analysis was performed on DEGs, and it was found that the highly enriched terms were mainly cell junction assembly, zinc ion homeostasis, neutrophil chemotaxis, and neutrophil migration. Studies have shown that cell junction assembly is related to tumor invasion and metastasis, 32 and zinc homeostasis may play an important role in maintaining T-cell function and cell-mediated immune function. 33 In addition, studies have found that tumor-associated neutrophils can play a pro-tumor role by promoting metastasis, angiogenesis, and tumor cell survival. 34 This study showed that neutrophil infiltration was relatively higher in women than in men in BCa patients with LNM, although there was no statistical difference, probably due to the small sample size in this population. Furthermore, in the general population, we found that neutrophil infiltration was significantly higher in women than in men, and BCa patients with high neutrophil infiltration had a worse prognosis. These results further prove that high neutrophil infiltration may be an important reason for the poor prognosis in women. Markman et al. 35 also found tumor metastasis burden in female and castrated male mice is higher than in sham male mice, which partially explains the possibility of loss of testosterone impairing neutrophil maturation and function to promote tumor progression. Furthermore, the results of the KEGG enrichment analysis indicated that the highly enriched terms mainly include PI3K-Akt signaling pathway and ECM-receptor interaction. These two pathways also play an important role in tumor progression.36,37 The above results further explain the gender difference in prognosis of patients with LNM.
In this study, LASSO Cox regression analysis was used to reduce the number of DEGs, and we further established a gender-associated signature with seven DEGs. The signature was used to predict OS in patients with BCa to better understand the role of these genes. Among them, studies have shown that PRKY only plays an important role in the male reproductive system, 38 and AKR1B15 has a unique effect on the higher activity of steroids and ketones. 39 Subsequently, we found that compared with the low-risk group, patients in the high-risk group had a worse prognosis and higher sensitivity to cisplatin, docetaxel, camptothecin, and paclitaxel. Analysis of the ROC curve and nomogram showed that the model had greater prognostic accuracy at 1, 3, and 5 years. In addition, the gender-associated scores predicted by radiomics signature were quite accurate. Due to the clinical limitation that not every patient can perform genome sequencing, the radiomics signature can more conveniently help clinicians to identify the drug sensitivity and prognosis of BCa patients by predicting the gender-associated scores.
This study has a number of limitations. First, we evaluated a single distant metastasis, but lacked detailed information on metastases such as the size of a specific organ and the exact number of metastatic lesions. Second, the database includes information about the use of surgery and chemotherapy, but does not record detailed protocols for these treatments. Third, we focus only on gene expression levels and ignore other events such as gene mutations, methylation, and copy number amplification, which are also important in tumor progression. Finally, the gender-associated scores were not validated with our center data. Therefore, we will collect clinical samples from our center and conduct external verification for the accuracy of the model.
Conclusions
In summary, our study found that there is a combination of gender-related factors affecting the prognosis of BCa metastatic pattern. Specifically, (1) male patients with LNM had a better prognosis than visceral metastasis, but similar phenomena were not observed in female patients and (2) in patients with LNM, the prognosis is better in men than in women, while no similar phenomenon was observed in visceral metastasis. (3) Enrichment of DEGs between sexes in patients with LNM may be related to metastasis and tumor immunity, especially the role of neutrophils. (4) Gender-associated signature is related to the clinicopathological characteristics of patients, and the prognosis of the high-risk group is worse, and higher susceptibility to cisplatin, docetaxel, camptothecin, and paclitaxel. (5) Patients with high radiomics scores had a strong tendency for high-risk group. Therefore, the gender-associated signature can be used as an accurate and efficient prediction model to guide clinical practice. These results may provide additional evidence for differences in tumor biology between sexes and for individualized treatment.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221108690 – Supplemental material for Gender dimorphism in survival of patients with lymph node metastasis of bladder cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359221108690 for Gender dimorphism in survival of patients with lymph node metastasis of bladder cancer by Yadong Guo, Zongtai Zheng, Wentao Zhang, Shiyu Mao, Fuhan Yang, Wei Li, Yang Yan and Xudong Yao in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We thank Feijia Xu and Shuai Wang (pathologists at the Department of Radiology, Shanghai Tenth People’s Hospital, Tongji University, China) for their radiology expertise and technical assistance. And we thanked Bullet Edits for editing this manuscript.
Compliance with Ethics Requirements
The studies involving human participants were reviewed and approved by the Ethics Committee of Shanghai Tenth People’s Hospital (approval number 2021KN108). Informed consent of patients was waived due to its retrospective nature.
Consent for publication
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by grants from the Shanghai Science Committee Foundation (#19411967700).
Conflict of interest statement
Yadong Guo, Zongtai Zheng, Wentao Zhang, Shiyu Mao, Fuhan Yang, Wei Li, Yang Yan, and Xudong Yao declare that they have no conflict of interest.
Availability of data and materials
The Surveillance, Epidemiology, and End Results (SEER) Program data are available upon reasonable request and with permission of the registry. All data generated or analyzed during this study are included in this published article and its additional information files.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
