Abstract
Background:
Cabozantinib is approved, in various settings, for the treatment of renal cell carcinoma, medullary thyroid cancer, and hepatocellular carcinoma, and it has been investigated for the treatment of other cancers. With the available evidence and the real-world performance of cabozantinib compared with clinical trial data, we performed a systematic review of cabozantinib monotherapy as treatment for solid tumors in adults.
Methods:
This study was designed in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses and registered with PROSPERO (CRD42020144680). We searched for clinical and observational studies of cabozantinib monotherapy for solid tumors using Embase, MEDLINE, and Cochrane databases (October 2020), and screened relevant congress abstracts. Eligible studies reported clinical or safety outcomes, or biomarker data. Small studies (n < 25) and studies of cabozantinib combination therapies were excluded. Quality was assessed using National Institute for Health and Care Excellence methodology, and study characteristics were described qualitatively.
Results:
Of 2888 citations, 114 were included (52 randomized studies, 29 observational studies, 32 nonrandomized phase I or II studies or pilot trials, and 1 analysis of data from a randomized study and a nonrandomized study). Beyond approved indications, other tumors studied were castration-resistant prostate cancer, urothelial carcinoma, Ewing sarcoma, osteosarcoma, uveal melanoma, non-small-cell lung cancer, Merkel cell carcinoma, glioblastoma, pheochromocytomas and paragangliomas, cholangiocarcinoma, gastrointestinal stromal tumor, colorectal cancer, salivary gland cancer, carcinoid and pancreatic neuroendocrine tumors, and breast, endometrial and ovarian cancers. The most common adverse events were hypertension, diarrhea, and fatigue.
Conclusion:
The identified evidence demonstrates the positive efficacy/effectiveness of cabozantinib monotherapy in various solid tumor types, with safety findings being consistent with those observed with other VEGFR-targeting tyrosine kinase inhibitors. When available, real-world findings were consistent with the data reported from clinical trials. A limitation of this review is the high proportion of abstracts; however, this allowed us to capture the most up-to-date findings.
Keywords
Introduction
Vascular endothelial growth factor (VEGF) receptor and hepatocyte growth factor receptor (MET) are involved in signaling pathways that regulate angiogenesis, epithelial to mesenchymal transition, cell proliferation, and cell migration. 1 Overexpression of VEGF receptor and MET promotes tumor growth and metastasis. 1 The kinase AXL is also linked to tumor pathogenesis, signaling to promote metastasis. 2 As well as promoting tumor growth, dysregulation of VEGF, MET, and AXL signaling pathways is also associated with immune suppression, leading to inhibition of antitumor immunity.3,4 Cabozantinib is the only approved tyrosine kinase inhibitor (TKI) that targets VEGF receptors (VEGFRs), MET, and AXL. 5
Cabozantinib is approved for several indications both in the Europe and in the United States. In Europe, cabozantinib monotherapy is approved for the treatment of the following patients: adult patients with advanced renal cell carcinoma (RCC) who are naive to treatment and have intermediate or poor risk in terms of prognosis [tablets, 60 mg once daily (QD)] 6 ; adults with advanced RCC who have received prior VEGF-targeted therapy (tablets, 60 mg QD) 6 ; adults with progressive, unresectable locally advanced or metastatic medullary thyroid cancer (MTC; capsules, 140 mg QD) 7 ; and adults with hepatocellular carcinoma (HCC) who have previously been treated with sorafenib (tablets, 60 mg QD). 6 In Europe, the Committee for Medicinal Products for Human Use endorsed the use of cabozantinib in differentiated thyroid cancer (DTC), based on the results of the COSMIC-311 study; a decision from the European Commission is awaited, at the time of writing.8,9 In the United States, cabozantinib monotherapy is approved for the treatment of patients with advanced RCC (tablets, 60 mg QD) 10 ; patients with progressive, metastatic MTC (capsules, 140 mg QD) 11 ; and patients with HCC who have previously been treated with sorafenib (tablets, 60 mg QD) 10 and adult and pediatric patients (aged 12 years and older) with locally advanced or metastatic DTC that has progressed following prior VEGFR-targeted therapy and who are radioiodine refractory, or ineligible (tablets, 60 mg QD). 10 These approvals were based on the following: a randomized phase III study demonstrated improved objective response rate (ORR), progression-free survival (PFS), and overall survival (OS) in patients with advanced RCC compared with everolimus; a phase III trial comparing cabozantinib with placebo showed improved ORR, PFS, and OS in patients with advanced progressive HCC; and a phase III trial comparing cabozantinib with placebo showed improved PFS in progressive MTC.6,7,10–14 It should be noted that cabozantinib capsules and tablets are not bioequivalent and cannot be used interchangeably.6,7 Cabozantinib is also approved in combination with nivolumab for the first-line treatment of adults with advanced RCC in the European Union and in the United States.6,10,15
Cabozantinib has also been investigated for the treatment of other types of solid tumors. For example, the use of cabozantinib in metastatic castration-resistant prostate cancer (mCRPC) has been investigated in two phase III randomized controlled trials (RCTs; COMET-1 and COMET-2),16,17 its use in non-small-cell lung cancer (NSCLC) has been investigated in a phase II RCT,18,19 and has recently been assessed in DTC in a phase III RCT (COSMIC-311). 9 With evidence growing in multiple disease areas, there is a need to establish an up-to-date understanding of the use of cabozantinib in the treatment of solid tumors, focusing on the clinical efficacy, comparative effectiveness, and safety profile of cabozantinib as a monotherapy for solid tumors.
To address these needs, we performed a systematic literature review (SLR) to identify the clinical and observational data on cabozantinib monotherapy for the treatment of different types of solid tumor in adults and to understand how the real-world performance of cabozantinib monotherapy compares with the data reported in pivotal clinical trials. As a secondary aim of this SLR, we also assessed which biomarkers are being explored to guide treatment decisions relating to the use of cabozantinib monotherapy. Although associations between biomarkers and patient response to cancer therapy have huge potential in guiding treatment decisions, a thorough evaluation of this broad topic was beyond the scope of this review.
Materials and methods
Search strategy
The protocol for this SLR was registered with the International Prospective Register of Systematic Reviews (PROSPERO; registration name: ‘A systematic literature review of cabozantinib for the treatment of solid tumors’; registration number: CRD42020144680). Published studies of cabozantinib as a monotherapy for solid tumors were identified through a systematic search. MEDLINE In-Process & Other Non-Indexed Citations and Ovid MEDLINE (1946–present), Embase (1974–present), and the Cochrane Library were searched on 9 October 2020 (see Supplemental Resource 1 for search terms used in Embase).
Supplementary searches
Congress abstracts were searched from 1 January 2016 to 9 October 2020. The congresses included were as follows: American Society of Clinical Oncology (ASCO), ASCO Gastrointestinal Cancers Symposium, ASCO Genitourinary Cancers Symposium, European Society for Medical Oncology, and American Association for Cancer Research. The bibliographies of studies identified in the electronic searches were reviewed to identify the additional relevant references.
Study selection and data collection
Citations identified by the searches were screened against prespecified criteria in accordance with the 2009 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. During screening, abstracts and titles were reviewed by a single reviewer against the eligibility criteria and uncertainties were resolved by a second reviewer. Eligibility criteria are presented in Table 1. During data extraction, study population data and key results relating to efficacy, safety and biomarkers were extracted manually for each included study.
Eligibility criteria for studies identified by SLR.
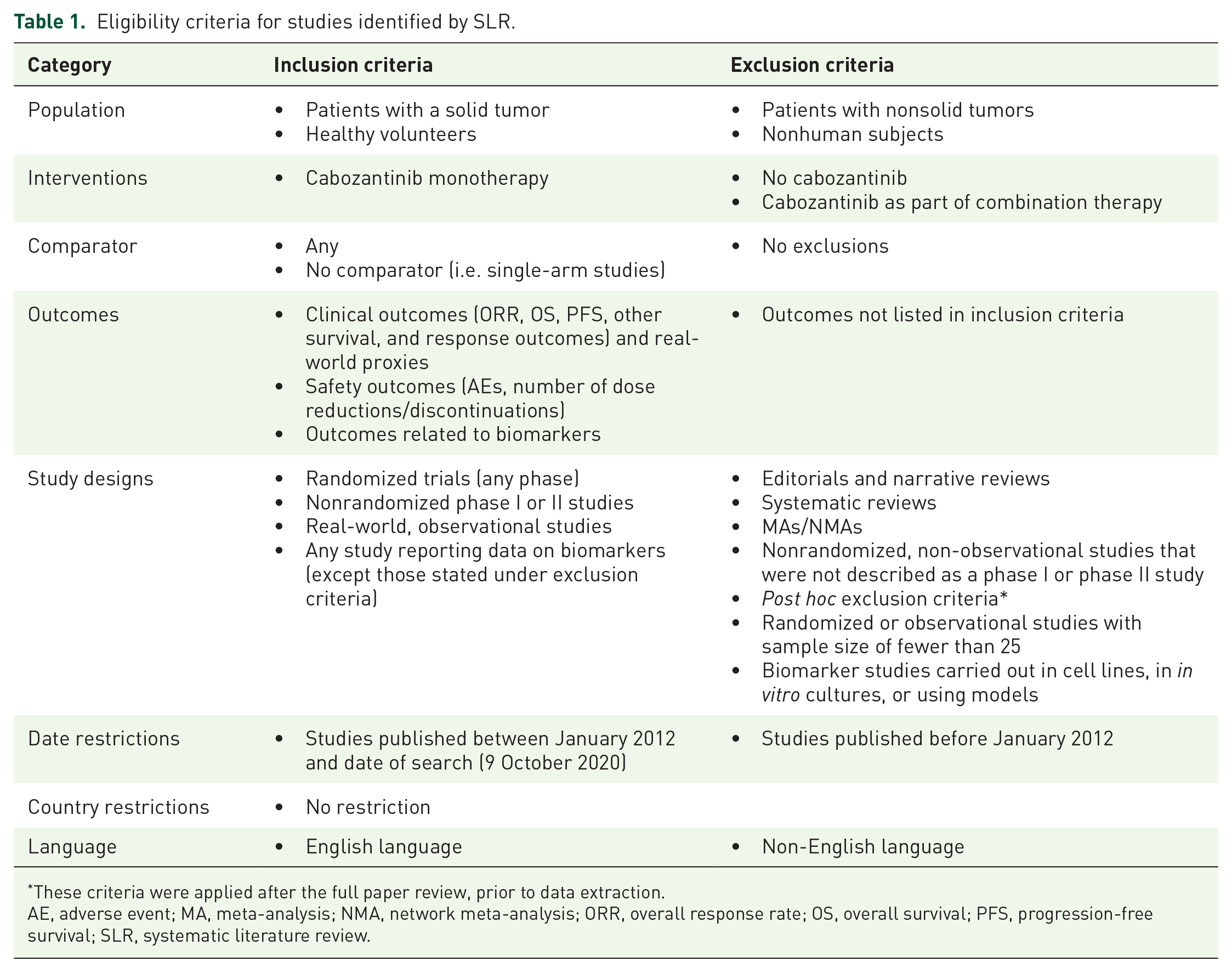
These criteria were applied after the full paper review, prior to data extraction.
AE, adverse event; MA, meta-analysis; NMA, network meta-analysis; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; SLR, systematic literature review.
Quality assessment
Quality was assessed using National Institute for Health and Care Excellence methodology checklists. Studies were given a rating according to their level of potential bias in terms of internal and external validity. 20 All studies are included in the Results section, regardless of their quality rating.
Results
Overview of evidence
Results of SLR
In total, 2888 citations were identified by the electronic searches after de-duplication; of these, 386 proceeded to full paper review. A further 20 articles were considered relevant for full-text review, including 17 from congress searches and 3 from bibliographies of studies identified in the electronic searches. Following full paper review, 292 articles were excluded, of which 77 were excluded according to post hoc exclusion criteria, including RCTs and real-world studies with a sample size of fewer than 25; biomarker studies carried out in cell lines; in vitro cultures or in vivo models; encores of abstracts already included; or abstracts that had been superseded by articles that were included. The final number of studies included in this review was 114 (Figure 1).
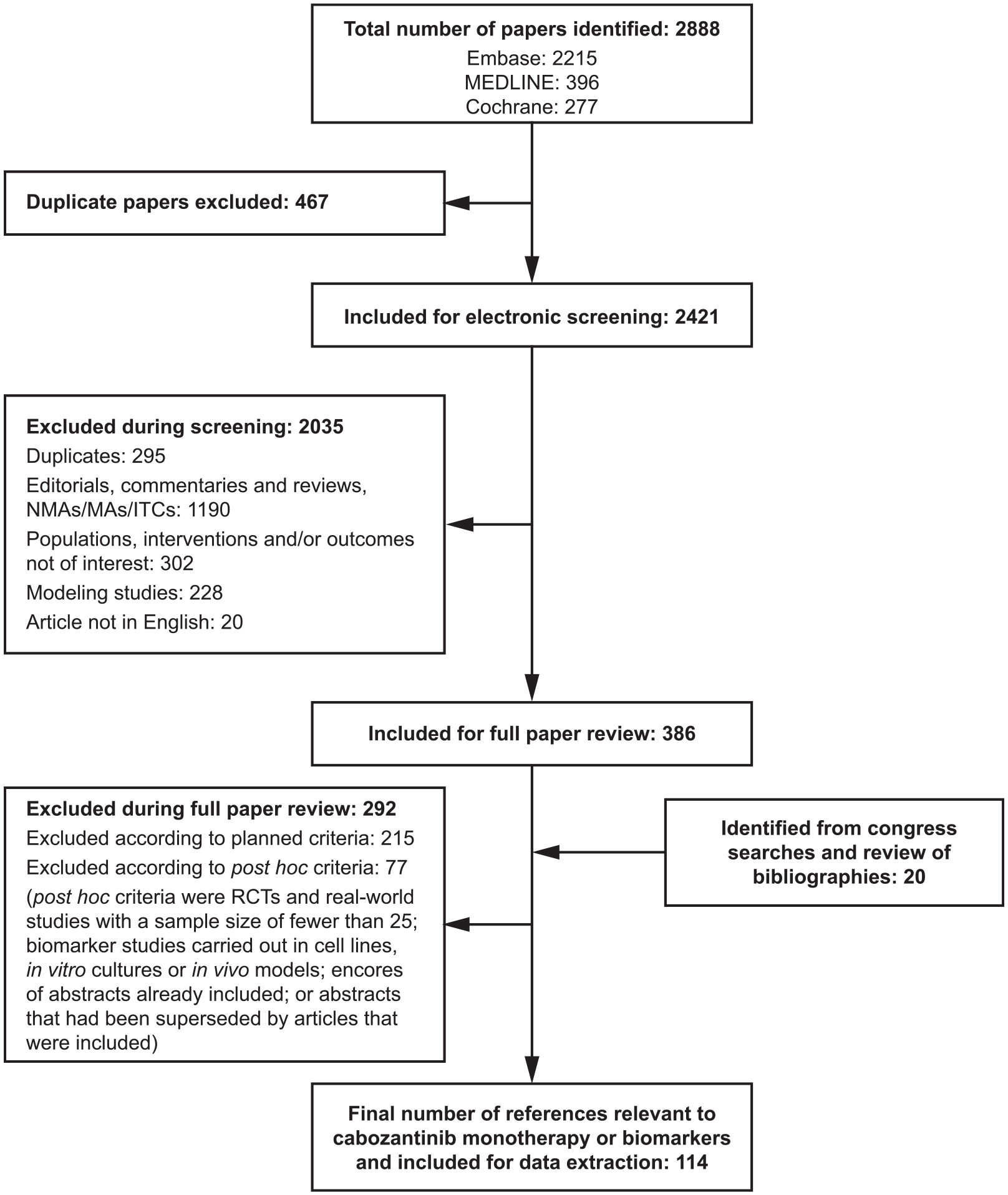
PRISMA diagram of included and excluded studies in the SLR.
Characteristics of included studies
Of the 114 included articles, 106 reported efficacy, effectiveness, and/or safety outcomes; of these, 47 reported findings from RCTs or randomized discontinuation trials (RDTs), 28 reported findings from observational or retrospective studies, and 31 reported findings from nonrandomized phase I or II studies (Supplemental Resource 2). Among the identified RCTs and RDTs, cabozantinib was evaluated versus placebo or versus the active comparators everolimus, temozolomide, dacarbazine, mitoxantrone, prednisone, or sunitinib. The most commonly studied disease type in terms of efficacy was RCC (Figure 2). Among the included studies, 54 articles contained data on biomarkers.

Summary of clinical efficacy studies of cabozantinib monotherapy identified by the SLR, by study, and tumor type.
In terms of quality assessment, 21/114 studies had the highest rating for internal validity. Poorer ratings were predominantly due to a lack of study details reported in congress abstracts (58 were abstracts, and 35 were articles).
Efficacy and effectiveness of cabozantinib monotherapy
Renal cell carcinoma
Evidence from randomized studies: Two RCTs evaluated cabozantinib in patients with RCC: the phase III METEOR trial13,21–27 and the phase II CABOSUN trial.28,29
The METEOR trial compared cabozantinib 60 mg (n = 330) with everolimus 10 mg (n = 328) QD in patients who received prior treatment with at least one VEGFR-targeting TKI; approximately 70% of patients received study treatment as a second-line therapy. Results demonstrated a superior efficacy for cabozantinib in all three endpoints: PFS (primary endpoint, with 90% statistical power), and OS and ORR (secondary endpoints). The disease control rate [DCR; defined as complete response (CR) + partial response (PR) + stable disease (SD)] was 83% in the cabozantinib group and 66% in the everolimus group. Subgroup analyses of patients in this study with or without prior nephrectomy showed cabozantinib improved PFS, ORR, and OS compared with everolimus in patients with advanced RCC, irrespective of nephrectomy status. 26 Similarly, a retrospective analysis of patients in the METEOR trial stratified by age group (<65, 65–74, and ⩾75 years) reported improved PFS, ORR, and OS compared with everolimus, irrespective of age group. 27 A post hoc pooled analysis of patients from METEOR and a Japanese, open-label, phase II study showed similar efficacy results, irrespective of prior treatment with immuno-oncology agents. 30
The CABOSUN trial compared cabozantinib 60 mg (n = 79), as a first-line therapy in patients at intermediate and poor risk only, with sunitinib 50 mg (n = 78). Patients receiving cabozantinib had a significantly lower risk of progression (primary endpoint, with 85% statistical power) than those receiving sunitinib (p = 0.0008). Risk of death was not significantly different between treatment groups; however, the study was not powered for survival differences. 29 The DCRs for cabozantinib and sunitinib were 75% and 47%, respectively. Characteristics and key findings from these studies are presented in Table 2.
Summary of clinical efficacy and safety findings from key randomized controlled trials of cabozantinib.

AE, adverse event; aHCC, advanced hepatocellular carcinoma; ALT, alanine aminotransferase; aRCC, advanced renal cell carcinoma; AST, aspartate aminotransferase; BID, twice daily; CI, confidence interval; HCC, hepatocellular carcinoma; HR, hazard ratio; IMDC, International Metastatic Renal Cell Carcinoma Database Consortium; mCRPC, metastatic castration-resistant prostate cancer; MKI, multikinase inhibitor; mMTC, metastatic medullary thyroid cancer; MP, mitoxantrone plus prednisone; mRCC, metastatic renal cell carcinoma; MTC, medullary thyroid cancer; ORR, objective response rate; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PPES, palmar–plantar erythrodysesthesia syndrome; PR, partial response; Q3W, once every 3 weeks; QD, once daily; SD, stable disease; TKI, tyrosine kinase inhibitor; TTP, time to progression; VEGFR, vascular endothelial growth factor receptor.
Evidence from real-world studies: In all, 21 publications relating to 16 retrospective or observational analyses investigated OS, PFS, or response (primary endpoints were not specified) of cabozantinib (60 mg or not reported) in RCC (Figure 3).35–55 Of the 16 studies, 13 were in patients with advanced or metastatic RCC in which the histological subtypes were either mixed or not reported.35–52 Two studies evaluated patients with advanced or metastatic non-clear-cell RCC,53,54 and one study was conducted in patients with metastatic clear-cell RCC. 55 Effectiveness data reported in real-world studies were broadly similar to the efficacy data reported in the RCTs (Figure 3). OS was reported in 12 publications relating to 11 studies and ranged from 9.1 to 25.4 months.35–43,53–55 PFS was reported in 10 studies and ranged from 5.6 to 12.5 months.35–38,42,44,45,47,53,54 The ORR (or the proportion of patients with CR + PR) was reported in 13 publications relating to 12 studies and ranged from 14% to 52%.35,37–39,41–44,46,47,53–55 The DCR or clinical benefit rate (or proportion of CR + PR + SD) was reported by 12 publications relating to 11 studies and ranged from 50% to 96%.35,37–39,41–44,46,47,53–55 The time to treatment failure was reported in four studies and ranged from 5.7 to 7.4 months.39,43,53,55

Comparison of outcomes for key trials of cabozantinib in patients with RCC: (a) OS, (b) PFS, (c) ORR, and (d) DCR.
Subgroup analyses of a UK study (CERES) of 100 patients with advanced RCC reported found that patients who were enrolled early in the study had similar outcomes to those that who enrolled later, 36 but lower (⩽6 versus >6) Charlson Comorbidity Index scores were associated with longer OS and PFS. 48 In a US-based retrospective study (n = 65), increased body mass index 6 weeks after initiation of cabozantinib therapy was significantly associated with prolonged OS [hazard ratio (HR) 0.90; 95% confidence interval (CI): 0.83–0.98; p = 0.016]. 52 The CABOREAL study investigated the real-world use of cabozantinib in the French Early Access Program.40,49,50 A subgroup analysis of patients enrolled in CABOREAL by age showed similar clinical outcomes regardless of age; median (95% CI) OS was 13.6 (10.2–15.4) months for patients younger than 65 years, 16.2 (14.1–19.5) months for those aged 65–75 years and 13.3 (6.5–18.3) months for aged 75 years and older. 49 A post hoc analysis in 47 patients with non-clear-cell RCC enrolled in CABOREAL by subtype showed that cabozantinib is particularly effective in papillary RCC type 1, with median OS of 16 months. 50 Two abstracts relating to the same French retrospective single-center study based on the Institut Gustave Roussy RCC database were included,41,42 the second with an additional eight patients. 42 This later abstract reported that ORR was higher in patients who had received prior immune checkpoint blockade (ICB; 49%; 54% clear-cell RCC versus 17% non-clear-cell RCC) than in those who had received prior TKI (21%; 27% clear-cell RCC versus 14% non-clear-cell RCC). 42 Finally, another retrospective analysis, using a multinational database, investigated the effectiveness of cabozantinib in patients with metastatic RCC. Median (95% CI) OS was 30.7 (15.8–36.8), 17.8 (11.9–23.3), 12.6 (9.3–21.7) and 14.9 (10.2–21.7) months in the first-, second-, third- and fourth-line settings, respectively. ORR was maintained over the different lines of treatment, at 32% in the first line, 26% in the second line, 51% in the third line, and 29% in the fourth line. 51
Evidence from nonrandomized phase I and II clinical studies: In phase I study, patients (n = 25) who received prior therapies (median: two) received cabozantinib (capsule, 140 mg QD). Median OS and PFS were 15.0 months and 12.9 months, respectively. Numbers of patients with PR, SD, and PD were 7 (28%), 13 (52%), and 1 (4%), respectively. 56 A phase II, open-label, single-arm study of cabozantinib (60 mg QD) in Japanese patients (n = 35) with advanced RCC who had received at least one prior TKI (one prior TKI: 70%; two prior TKIs: 23%; three or more prior TKIs: 9%) reported an ORR of 20%, a clinical benefit rate (CR + PR + SD) of 86% and a 6-month PFS estimate of 72.3% [median PFS and OS were not reached (NR)]. 57
Hepatocellular carcinoma
Evidence from randomized studies: One phase III RCT (CELESTIAL trial) compared cabozantinib 60 mg (n = 470) with placebo (n = 237) in patients with HCC who had been treated with sorafenib and had received up to two previous systemic treatments. The primary endpoint was median (95% CI) OS (with 90% power to detect an HR of 0.76), which was significantly greater with cabozantinib than with placebo [10.2 (9.1–12.0) months versus 8.0 (6.8–9.4) months; HR 0.76; 95% CI: 0.63–0.92; p = 0.005]. Median (95% CI) PFS as 5.2 (4.0–5.5) for cabozantinib versus 1.9 (1.9–1.9) for placebo (HR 0.44; 95% CI: 0.32–0.52; p < 0.001). 14 A further analysis of tumor response reported that 47% of patients who received cabozantinib, versus 11% of those who received placebo, had a post-baseline reduction in tumor target lesion size; median time to progression was 5.4 months for cabozantinib versus 1.9 months for placebo (HR 0.41; 95% CI: 0.34–0.49). 32 Further analyses of a subset of patients who received sorafenib as their only prior therapy demonstrated greater median (95% CI) OS with cabozantinib, at 11.3 (9.5–13.9) months, than with placebo, at 7.2 (5.8–9.3) months (HR 0.70; 95% CI: 0.55–0.88). 58 Another retrospective analysis showed clinical efficacy in patients in CELESTIAL who had deterioration of liver function to Child–Pugh B by study week 8. Median OS was 8.5 months with cabozantinib versus 3.8 months with placebo (HR 0.32; 95% CI: 0.18–0.58), and median PFS was 3.7 months versus 1.9 months (HR 0.44; 95% CI: 0.25–0.76). 59 An analysis of patients in CELESTIAL who had received prior transarterial chemoembolization (TACE) showed that cabozantinib improved outcomes versus placebo, irrespective of prior TACE. Median OS was 11.4 months with cabozantinib versus 8.6 months with placebo for patients with prior TACE and 9.5 months versus 7.2 months for patients with no prior TACE. 60 Characteristics and key findings from the CELESTIAL study are presented in Table 2.
Evidence from real-world studies: Two European real-world studies of cabozantinib in HCC were identified, the findings of which were consistent with data reported from the pivotal CELESTIAL trial (Figure 4).61,62 Both real-world studies evaluated cabozantinib as a second or later line of therapy, with median OS ranging from 7.761 to 12.9 months. 62 In the first study, conducted in Austria and Germany, four patients (5%) achieved PR. 61 The second study, conducted in Italy, reported a median (95% CI) PFS of 5.1 (2.7–7.5) months 62 and DCR of 59%. 62
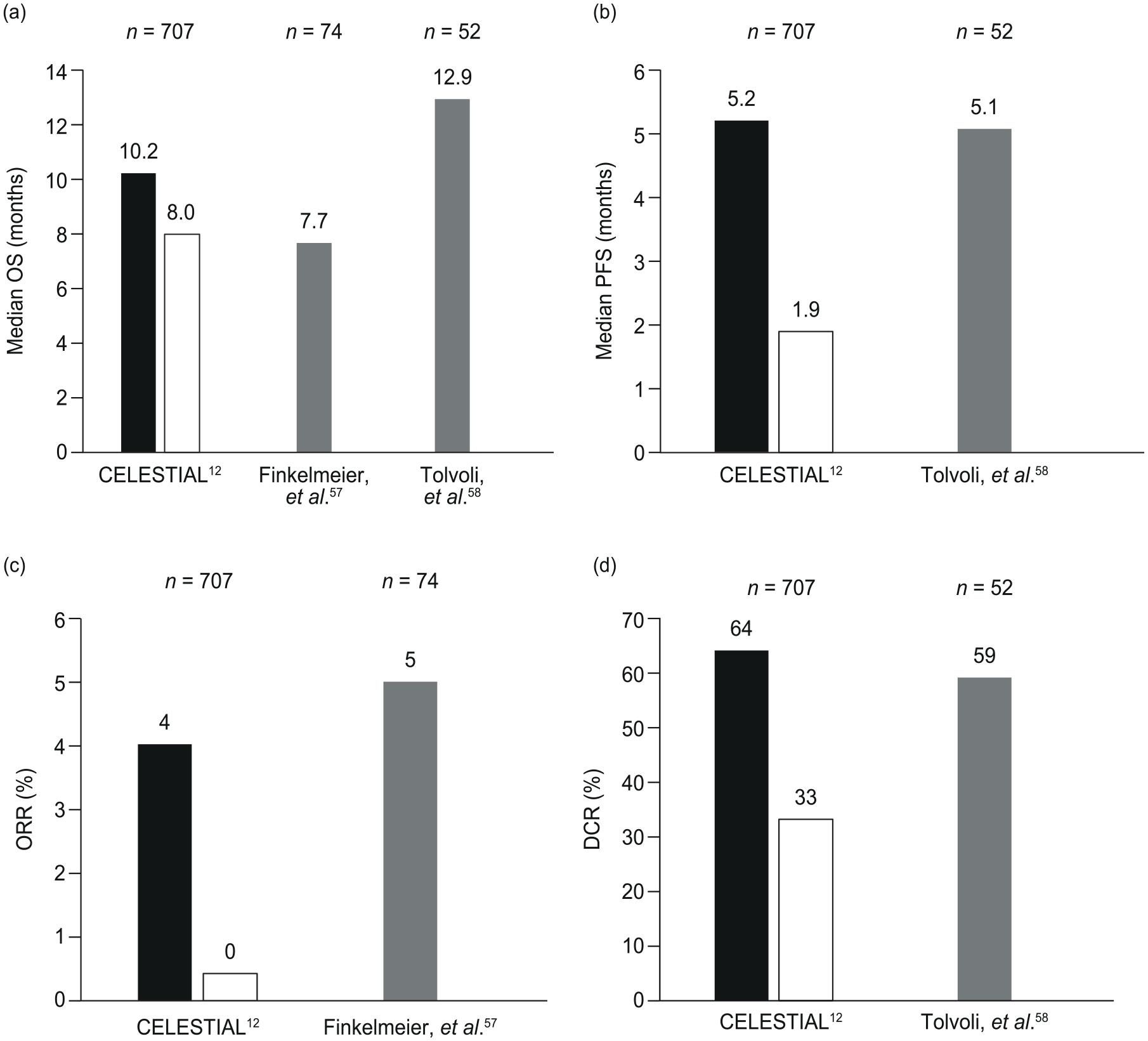
Comparison of outcomes for key trials of cabozantinib in patients with HCC: (a) OS, (b) PFS, (c) ORR, and (d) DCR.
Evidence from nonrandomized phase I and II clinical studies: One phase II study evaluated cabozantinib in 41 patients with HCC, most (78%) of whom had 1–2 lines of systemic therapy previously. At the end of a 12-week lead-in phase of an RDT, patients receiving cabozantinib 100 mg QD had an ORR (measured by Response Evaluation Criteria in Solid Tumors version 1.0) of 5%, and the DCR was 66% overall. 63
Medullary thyroid cancer
Evidence from randomized studies: One phase III RCT (EXAM study) compared cabozantinib (140 mg as capsules; n = 219) with placebo (n = 111) in patients with MTC, with no limit on the number of prior therapies. The study met the primary endpoint (PFS) with an HR (95% CI) of 0.28 (0.19–0.40); there was 80% power to detect an HR of 0.667. There was also a longer median OS with cabozantinib than with placebo (26.6 months versus 21.1 months). Further characteristics and key findings of the EXAM study12,31,64–67 are presented in Table 2.
Evidence from real-world studies/nonrandomized phase I and II clinical studies: No real-world studies or phase I or II studies of cabozantinib monotherapy in MTC were identified.
Differentiated thyroid cancer
Evidence from randomized studies/real-world studies: No randomized or real-world studies of cabozantinib monotherapy in DTC were identified. The results of the phase III COSMIC-311 trial of cabozantinib in patients with radioiodine-refractory DTC previously treated with BEGFR-targeted therapy were published after the searches for this review had been conducted. For completeness, the key efficacy results of COSMIC-311 are included in the Discussion. 9
Evidence from nonrandomized phase I and II clinical studies: Three phase I or II studies were identified for the evaluation of patients with DTC. In one phase II study, in the first-line setting, patients receiving cabozantinib 60 mg QD had a PR rate of 54% (19/35 patients). 68 Cabanillas et al.69,70 conducted a phase I study (n = 15) with cabozantinib 140 mg and a phase II study (n = 25) with cabozantinib 60 mg. The phase I study measured response in patients who were refractory to standard therapy with radioactive iodine (RAI). The numbers of patients with PR and SD were 8 (53%) and 6 (40%), respectively, out of a total of 15 patients. 69 In the phase II study of patients who received up to two lines of prior VEGFR-targeted therapy, ORR (primary endpoint) was 40% and median (95% CI) OS was 34.7 (18.3–upper limit NR) months. 70
Castration-resistant prostate cancer
Evidence from randomized studies: The COMET-1 and COMET-2 trials were phase III RCTs that compared cabozantinib 60 mg with prednisone, and cabozantinib 60 mg with mitoxantrone plus prednisone (MP), respectively. The study samples included men with mCRPC, more than 90% of whom had received three or more prior cancer therapies.17,34,71 COMET-1 did not meet its primary endpoint (OS); there was no significant difference between cabozantinib (n = 682) and prednisone (n = 346; the study had 90% power to detect an HR of 0.75). However, there were favorable results for cabozantinib in terms of the secondary endpoint, PFS (Table 2). 17 COMET-2 had a primary endpoint of pain response, which was not significantly different for cabozantinib versus MP (15% versus 17%; p = 0.8) and is outside the scope of this SLR. In terms of additional endpoints, patients receiving cabozantinib (n = 61) demonstrated longer OS than those receiving MP (n = 58; 9.0 months versus 7.9 months; p = 0.1). 16 Characteristics and key findings of these studies are presented in Table 2.
In a phase II RDT, the primary endpoints were ORR during the 12-week lead-in stage and PFS after randomization. 72 Patients had received no more than one prior standard chemotherapy regimen, completed at least 4 weeks prior to study entry, and received cabozantinib 100 mg QD or placebo during the trial. At the end of the 12-week lead-in phase, during which all 171 patients received cabozantinib, numbers of patients with PR, SD, and PD were 9 (5%), 127 (75%), and 18 (11%), respectively. After randomization of 31 patients, median (95% CI) PFS was 23.9 (10.7–62.4) weeks in the cabozantinib arm (n = 14) and 5.9 (5.4–6.6) weeks in the placebo arm (n = 17). HR for cabozantinib versus placebo was 0.12 (p < 0.001).
Evidence from real-world studies: No real-world studies of cabozantinib monotherapy in CRPC were identified.
Evidence from nonrandomized phase I and II clinical studies: Two phase II studies evaluated patients with mCRPC. One included 144 patients who had received at least one previous docetaxel-containing regimen and no more than three previous chemotherapy regimens. The primary endpoint was bone scan response (not reported here), and further endpoints included OS. Median (95% CI) OS was 12.1 (9.4–14.3) months in the cabozantinib 100 mg cohort (n = 93) and 9.1 (8.0–12.9) months in the cabozantinib 40 mg cohort (n = 51). 73 In another study in patients with progressive CRPC and bone metastases who were naive to chemotherapy, 17/22 patients (77%) receiving cabozantinib 60 mg QD had PFS at 12 weeks (primary endpoint; see Supplemental Resource 2 for details of statistical power). 74
Other tumor types
Evidence from randomized studies: An RDT studied cabozantinib 100 mg QD in 526 patients with nine different tumor types, including CRPC, gastric/gastroesophageal junction adenocarcinoma, HCC, metastatic breast cancer, melanoma, NSCLC, small-cell lung cancer, ovarian cancer and pancreatic adenocarcinoma. Primary endpoints were ORR in the 12-week lead-in phase and PFS after randomization. Overall, ORR and median PFS were 0–22% and 2.4–6.9 months, respectively. 75 In a subgroup analysis of only patients with melanoma (n = 77), 66% of whom had at least one line of prior systemic therapy, ORR was 5% and median (95% CI) PFS was 4.1 (1.8–upper limit NR) months. The HR for PFS (cabozantinib versus placebo) was 0.59 and was not statistically significant (p = 0.284). 76 In another subgroup analysis of patients with ovarian cancer (n = 70) who had no more than three prior standard chemotherapy regimens, ORR (95% CI) was 21% (13–32%). After randomization, median PFS was 5.9 months in the cabozantinib arm and 1.4 months in the placebo arm (statistical significance not reported). 77 A randomized phase II trial compared cabozantinib with chemotherapy (temozolomide or dacarbazine) in 46 patients with metastatic uveal melanoma. The primary endpoint was to evaluate whether cabozantinib could improve PFS at 4 months (PFS4) from 15% (previously described for temozolomide) to 40% with cabozantinib. PFS4 was not found to differ between the two groups, at 32% with cabozantinib and 27% with chemotherapy. Furthermore, there were no significant differences in PFS or OS for cabozantinib versus chemotherapy (median PFS: 60 days versus 59 days; median OS: 6.4 months versus 7.3 months). 78
Evidence from real-world studies: No real-world studies of cabozantinib monotherapy in other tumor types were identified.
Evidence from nonrandomized phase I and II clinical studies: In all, 18 phase II studies and one phase I study evaluated patients with various types of tumor.79–96 In all but one of these studies, 79 the cabozantinib dose was 60 mg. In one study in 50 patients with relapsed or refractory metastatic urothelial carcinoma (mUC), the primary endpoint, ORR (95% CI), was 19% (9–34%). Median (95% CI) OS and PFS were 8.1 (5.2–10.3) months and 3.7 (3–6) months, respectively. 80 Another study evaluated 36 patients with breast cancer with brain metastases, with a median of three prior lines of therapy for metastatic disease. After treatment with cabozantinib 60 mg, the primary endpoint (central nervous system ORR) was 5% in cohort 1 [human epidermal growth factor receptor 2 (HER2)+], 14% in cohort 2 (hormone receptor+ HER2–), and 0% in cohort 3 (triple negative). 81 In a study of metastatic triple-negative breast cancer only (n = 35), the primary endpoint, ORR (95% CI), was 9% (2–26%) in patients with up to three prior chemotherapeutic regimens. 82 In a study of 52 patients with hormone-receptor-positive breast cancer with breast metastases (who had received at least one prior line of hormonal therapy or chemotherapy for metastatic disease), cabozantinib had an initial dosage of 100 mg QD, but this was reduced to 60 mg QD after the first seven patients. The bone scan response rate (primary endpoint) was 38%; 26 patients (50%) had SD as measured by Response Evaluation Criteria in Solid Tumors. Median (90% CI) OS and PFS were 19.6 (18.0–26.8) months and 4.3 (2.8–5.5) months, respectively. 83 In one study of treatment of metastatic collecting-duct carcinoma in the first-line setting, response (primary endpoint) was partial in 2/9 patients (22%), and there were SD and PD in 2/9 patients (22%) and 3/9 patients (33%), respectively. 84 Two studies assessed patients with any number of prior therapies. The first study included patients with grade 1‒2 carcinoid (n = 41) or pancreatic neuroendocrine tumors (n = 20), 85 and reported an ORR (95% CI; primary endpoint) of 15% (7–28%) and 15% (5–36%), respectively. The second study included patients with RET-rearranged lung cancers (n = 26) and reported an ORR (95% CI) of 28% (12–49%). 86 One study, which also measured response as the primary endpoint, evaluated patients with unresectable metastatic pheochromocytomas and paragangliomas; results showed that 6/15 patients (40%) had PR. 87 PFS was the primary endpoint in four studies that evaluated patients with recurrent/metastatic endometrial cancer (n = 102), 88 cholangiocarcinoma (n = 19), 89 uterine carcinosarcoma (n = 15), 90 and gastrointestinal stromal tumor (n = 50). 91 Prior to the studies, patients had progressed after one or two lines of therapy.88–91 Median PFS was 1.8–5.5 months. One study, in 44 patients with metastatic colorectal cancer, had 12-week PFS rate as the primary endpoint, which was 34%. 92
A retrospective analysis of phase II data measured OS in 108 patients with recurrent glioblastoma, in whom antiangiogenic therapy had not previously failed. After treatment with cabozantinib 100 mg or 140 mg QD, median OS was 11.0 months. 79
A phase II study evaluated patients with advanced Ewing sarcomas and osteosarcomas, many of whom had received two or more prior lines of therapy (Ewing sarcomas: 67%; osteosarcomas: 40%). In patients with osteosarcoma (n = 45), ORR was 12%, with median (95% CI) PFS of 6.7 (5.4–7.9) months. In patients with Ewing sarcoma (n = 45), ORR was 26%, with median (95% CI) PFS of 4.4 (3.7–5.6) months. 93
A small phase II study in eight patients with advanced Merkel cell carcinoma was closed prematurely, partly due to lack of response (best response was SD in one patient). Next-generation sequencing did not detect any genetic alteration in either MET or VEGFR2. 94 Another phase II study, in 25 patients with salivary gland cancer, reported ORRs of 6% for those with adenoid cystic carcinoma, 20% for those with salivary duct carcinoma, and 0% for those with other salivary gland cancers; median (95% CI) PFS was 12.6 (6.8–18.4) months, 9.0 months (insufficient events for 95% CI) and 6.9 (0–15.2) months, respectively. 95 Finally, a phase I study of cabozantinib in patients with advanced or metastatic solid tumors included an expansion cohort of patients with NSCLC (cabozantinib was dosed at 60 mg in the expansion cohort). Objective responses were seen in 4/23 patients (17%) in the dose-escalation cohorts and 4/20 patients (20%) in the NSCLC expansion cohort. 96
Safety of cabozantinib monotherapy
Renal cell carcinoma
Evidence from randomized studies: In two RCTs, the proportion of patients who experienced grade 3–4 adverse events (AEs) was 67–68%13,28; the most common grade 3–4 AEs with cabozantinib 60 mg treatment were hypertension (15–28%), diarrhea (10–11%), fatigue (6–9%), and palmar–plantar erythrodysesthesia syndrome (PPES; 8%).13,28 Overall, 9–20% of patients discontinued treatment owing to AEs and 46–60% required a dose reduction.13,28 All safety findings of the METEOR trial13,21,22,24,25,27,30,97,98 and the CABOSUN trial28,29 are presented in Table 2.
Evidence from real-world studies: In all, 11 real-world studies that reported safety data relating to cabozantinib in patients with RCC were identified. Six studies reported the overall proportions of patients with grade 3–4 AEs, ranging from 15% to 49% of patients,35,44,46,47,55,99 which were lower than those reported in RCTs. Four real-world studies reported the most common grade 3–4AEs35,44,47,53,54: hypertension (4–7%),44,47,53,54 diarrhea (3–10%),35,44,47,53 fatigue (2–23%),35,47,53,54 and PPES (or hand–foot skin reaction; 2–12%)35,44,47,53,54 that were all seen at broadly similar levels to those reported in RCTs. Other grade 3–4 AEs reported in the real-world studies in more than 2% of patients in at least one study were as follows: deep venous thrombosis or pulmonary embolism (4–13%)44,54; asthenia (7%) 44 ; proteinuria (including nephrotic range; 1–7%)53,54; mucosal inflammation or mucositis (3–5%)35,44; increased aspartate aminotransferase (AST), alanine aminotransferase (ALT) or transaminitis (1–5%)44,47,53; rash (4%) 47 ; anemia (0–3%)44,47; and pneumonitis (1–2%).44,47 Nine studies reported the proportions of patients who discontinued treatment owing to AEs, which ranged from 4% to 16%35,41,44,46,47,53,55,99,100 and were broadly similar to those reported in RCTs. The proportions of patients who required dose reductions owing to AEs were reported in nine studies, which ranged from 23% to 69%35,38,44,46,47,53–55,100 and were consistent with those seen in the RCTs.
In a retrospective analysis (n = 65), results showed that the rate of AEs was greater in patients who received a reduced starting dose of cabozantinib than in those who received a starting dose of 60 mg (95% versus 66%). 38 Another study observed a rate of dose reductions of 57% in 21 patients receiving cabozantinib 60 mg. 54
A European real-world study of 337 patients with advanced RCC (CASSIOPE) found that AE-related dose modifications were similar for patients initiating cabozantinib at the recommended 60 mg and for those initiating at any dose (dose reductions in 46% versus 39%; discontinuations in 11% versus 10%). 46
In two analyses (n = 91–96) of Italian real-world data for treatment beyond the first-line setting, rate of grade 3–4 AEs after treatment with cabozantinib 60 mg was 21–36%44,99 (most common AEs were asthenia and diarrhea), and rate of dose reduction was 42%. 44
In a UK study (n = 128), the most common grade 3–4 AEs following treatment with cabozantinib (dose not reported) were fatigue (11%) and diarrhea (9%), and 55% of patients required a dose reduction. 35 In a North American observational study, 86 patients with metastatic clear-cell RCC received cabozantinib following progression while undergoing ICB; dose reductions for AEs occurred in 45% of patients [most commonly fatigue (27%) and PPES (16%)]. 55
Fatigue was also the most common AE (52%) in a study of patients with non-clear-cell RCC; the most common grade 3 AEs were skin toxicity (including PPES: 4%), hypertension (4%), and diarrhea (3%). 53
In a Polish real-world retrospective analysis of 115 patients with metastatic RCC who had received prior VEGF-targeted therapy, the most common grade 3–4 AEs with cabozantinib 60 mg were fatigue (23%), PPES (12%), and diarrhea (10%). Only 4% of patients discontinued treatment owing to AEs. 47 An evaluation of cabozantinib in 91 patients with metastatic RCC in a real-world setting in France reported that 35% of patients had more than 10% weight loss and that 11% experienced more than 20% weight loss. 41 Fatigue was the most common AE (80%) in a US retrospective chart review of 35 patients with metastatic RCC, followed by gastrointestinal AEs (54%), PPES (26%), and hypertension (23%). Four patients (12%) discontinued owing to AEs. 100
Evidence from nonrandomized phase I and II clinical studies: In a phase I study of 25 patients with clear-cell RCC with refractory tumors or tumors that progressed following standard therapies, the most common grade 3 AEs were hypophosphatemia [n = 10 (40%)], fatigue [n = 5 (20%)], and hyponatremia [n = 5 (20%)] after treatment with cabozantinib 140 mg QD. There were three reports of grade 4 AEs, including pulmonary embolism, peritoneal hemorrhage, and a mental status change (not related to cabozantinib). 56 A phase II, open-label, single-arm study of cabozantinib (60 mg QD) in Japanese patients (n = 35) with advanced RCC who had received at least one prior TKI reported a safety profile consistent with those reported in other studies. Hypertension was the most common AE of grade 3 or above (11%), followed by PPES, diarrhea, proteinuria, abnormal hepatic function, decrease appetite, and fatigue (all 9%). Two patients (6%) discontinued owing to AEs. 57
Hepatocellular carcinoma
Evidence from randomized studies: The phase III CELESTIAL trial of cabozantinib (tablets, 60 mg) in patients with advanced HCC following prior sorafenib therapy reported grade 3–4 AEs in 68% of the safety population (n = 467). 14 The most common grade 3–4 AEs were PPES (17%), hypertension (16%), increased AST (12%), fatigue (10%), and diarrhea (10%). 14 In a retrospective analysis of the AEs observed in the CELESTIAL trial, results showed that the development of any grade PPES or hypertension of grade 3 or above was associated with prolonged OS and PFS. 33 A retrospective analysis showed that cabozantinib may have a manageable safety profile in patients in CELESTIAL who had deterioration of liver function to Child–Pugh B by study week 8. In the Child–Pugh B subgroup, versus the overall population, there were similar rates of dose reductions (61% versus 62%) and discontinuations due to AEs (18% versus 16%). Rates of grade 3–4 PPES (17% versus 8%) and hypertension (16% versus 8%) were higher in the Child–Pugh B subgroup than in the overall population. 59 Further safety findings from the CELESTIAL study are presented in Table 2.
Evidence from real-world studies: One real-world study of cabozantinib in HCC was identified. This Italian study in patients with unresectable HCC (n = 52) reported that the most common treatment-related grade 3–4 AEs were fatigue (10%), hand–foot skin reaction (8%), hypertension (6%), and increased ALT (6%). 62 These were similar to those reported in the CELESTIAL trial, 14 but the RCT reports the overall event rate rather than the treatment-related events. Permanent dose reductions from 60 to 40 mg were required in 46% of patients, and from 60 to 20 mg in 17%. Permanent discontinuation for intolerance occurred in 17% of patients. 62 Overall, the rates of dose reductions and discontinuations are in line with those reported in the CELESTIAL trial (62% and 16%, respectively). 14
Evidence from nonrandomized phase I and II clinical studies: In a phase II study (n = 41), the most common AEs of grade 3 or above following cabozantinib 100 mg QD were diarrhea (20%), PPES (termed as hand-foot syndrome; 15%), thrombocytopenia (15%), hypertension (10%), and increased AST (10%). 63
Medullary thyroid cancer
Evidence from randomized studies: One RCT (cabozantinib arm, n = 219) identified diarrhea PPES and fatigue as the most common grade 3–4 AEs with cabozantinib (capsule,140 mg). Further safety findings from the EXAM study are presented in Table 2.12,31,65,67,101
Evidence from real-world studies/nonrandomized phase I and II clinical studies: No real-world studies or phase I or II studies of cabozantinib monotherapy in MTC were identified.
Differentiated thyroid cancer
Evidence from randomized studies/real-world studies: No randomized studies or real-world studies of cabozantinib monotherapy in DTC were identified. As noted earlier, the literature searches for this review were conducted before the results of the COSMIC-311 trial were published. For completeness, however, the key safety results of COSMIC-311 are included in the Discussion. 9
Evidence from nonrandomized phase I and II clinical studies: In a phase I study (n = 15) by Cabanillas et al., 69 rates of discontinuation and dose reduction were 7% and 93%, respectively, with cabozantinib 140 mg. In a subsequent phase II study (n = 25), 70 the rate of dose reduction was 56% following a starting dose of 60 mg. With cabozantinib 140 mg, the most common AEs (grade 3 or above) were hyponatremia (27%), and diarrhea and increased lipase (both 20%). With cabozantinib 60 mg, the most common AEs (grade 3) were hypophosphatemia (16%), and fatigue, weight loss, lipase or amylase elevation, and neutropenia (all 12%).
Another phase II study (n = 35), in the first-line setting, found that the most common treatment-related AEs were hyperglycemia, diarrhea, fatigue/malaise, and weight loss, following treatment with cabozantinib 60 mg. 68
Castration-resistant prostate cancer
Evidence from randomized studies: In two RCTs that evaluated 61 and 682 patients treated with cabozantinib 60 mg, the most common grade 3–4 AEs were found to be hypertension and fatigue. Further safety findings of COMET-117 and COMET-216,71 are presented in Table 2.
In a phase II RDT that evaluated 171 patients with mCRPC who received cabozantinib 100 mg and one or no prior standard chemotherapy regimens, fatigue [n = 27 (16%)] and hypertension [n = 21 (12%)] were the most common grade 3 AEs. The rate of discontinuation during the lead-in stage was 12%. 72
Evidence from real-world studies: No real-world studies of cabozantinib monotherapy in CRPC were identified.
Evidence from nonrandomized phase I and II clinical studies: Three phase II studies (n = 19, 51, and 144) examined AEs in patients with mCRPC receiving cabozantinib. Common AEs (grade 3 or above) were fatigue (14% 73 ) and hypertension (13% 102 ) in those receiving 40 mg; venous thromboembolism (23%) and diarrhea (14%) 74 in those receiving 60 mg; and fatigue (27%) in those receiving 100 mg. 73 One study reported the rate of discontinuation, which was 25% in the 100 mg cohort and 18% in the 40 mg cohort. 73
Other tumor types
Evidence from randomized studies: In an RDT of cabozantinib 100 mg QD of various tumor types, the rates of dose reduction were 74% (390/526 patients) overall, 75 29% in metastatic melanoma (22/77 patients), 76 and 37% in ovarian cancer (26/70). 77 Fatigue and diarrhea were among the most common grade 3–4 AEs. In a randomized phase II trial of cabozantinib in patients with mUC, grade 3–4 AEs were thromboembolic events (13%) and hypertension (20%). 78
Evidence from real-world studies: No real-world studies of cabozantinib monotherapy in other tumor types were identified.
Evidence from nonrandomized phase I and II clinical studies: In all, 16 phase II studies evaluated the safety of cabozantinib 60 mg in various tumor types.80–83,85–95,103 The rate of dose reductions ranged from 34% 82 to 87% 87 in seven studies that reported these data. The most common grade 3–4 AEs included hypertension (⩽36% in 10 studies),80–83,85,86,88,90,91,95 diarrhea (⩽26% in seven studies),80,81,83,85,90,91,95 and increased lipase (⩽15% in five studies).81,82,86,91,93
In a phase I study of patients with advanced or metastatic solid tumors, including an expansion cohort of patients with NSCLC, the most common grade 3–4 AEs were gamma glutamyl transferase increase (17%), hypertension (13%), and lymphopenia (13%) in the dose-escalation cohort, and hypertension (30%), neutropenia (25%), and ALT increase, PPES, hypophosphatemia, and dyspnea (all 15%) in the NSCLC expansion cohort. The recommended phase II dose for cabozantinib was 60 mg. 96
Biomarkers for response to cabozantinib
Of 114 articles identified from the SLR, 54 report findings related to biomarkers. The biomarkers identified include RET mutational status (seven studies),12,64–67,101,104 presence of bone metastases (five studies),28,40,66,105,106 MET expression level (five studies),18,19,29,107,108 RAS mutational status (four studies),67,92,101,104 circulating tumor cells (two studies),73,102 and alpha-fetoprotein (three studies).109–111 Six studies investigated tumor characteristics, including diameter and volume.97,112–115 The remaining studies included various biomarkers, such as plasma and cell biomarkers.34,47,53,78,81,82,91,94,96,103,116–127
RET status was the most commonly investigated biomarker; all seven studies were subgroup analyses of MTC study populations. Patients with positive RET-mutation status (mainly RET M918T) generally experienced greater cabozantinib treatment benefit (prolonged OS and PFS with cabozantinib versus placebo) than patients without RET mutations.12,64,66,67,101,104 In a post hoc analysis of the CABOSUN trial, including 131 patients with RCC in whom MET expression was determined by immunohistochemistry, findings suggested the treatment effect (cabozantinib versus sunitinib) may be stronger in the group with MET overexpression than in the MET-negative group [HR (95% CI) for PFS: 0.32 (0.16–0.63) versus 0.67 (0.37–1.23)], respectively. 29 In another study (n = 90), higher soluble MET (sMET) concentrations were associated with improved PFS in patients with osteosarcoma (7.8 months versus 5·4 months for the sMET < 300.6 ng/mL and =300.6 ng/mL groups, respectively; log-rank p = 0.0167). 93 No association has been observed between MET expression and treatment response in patients with Ewing sarcoma, NSCLC, or urothelial carcinoma.18,19,80
Discussion
In carrying out a thorough systematic review of the literature, we have captured a wealth of evidence from 114 articles reporting on the efficacy, effectiveness, and safety of cabozantinib as a monotherapy for the treatment of solid tumors.
The review captured key efficacy evidence from randomized trials that have supported the approval of cabozantinib for RCC (METEOR and CABOSUN trials), MTC (EXAM trial), and HCC (CELESTIAL trial). Evidence for approved indications is continuing to grow. The phase IV EXAMINER trial (NCT01896479) compared cabozantinib (60 mg tablets and 140 mg capsules) in patients with metastatic MTC. Activity in patients with advanced MTC was shown for both dose regimens, but the 60 mg tablet did not meet the pre-specified non-inferiority criteria for PFS versus the 140 mg capsule (HR 1.24; 95% CI: 0.90–1.70). The safety profile was consistent with that observed previously with cabozantinib the 60 mg tablet versus 140 mg capsules associated with a non-significant lower frequency of Grade ⩾3 AEs (63% versus 72%), dose reductions (69% versus 81%), and discontinuations due to AEs (23% versus 36%).
As well as approved indications, we identified studies of cabozantinib in additional tumor types. Among these, CRPC was the most studied tumor type in randomized trials of cabozantinib; two RCTs (COMET-1 and COMET-2) did not provide supporting evidence for cabozantinib versus prednisone or MP, while an RDT did demonstrate the improved efficacy of cabozantinib versus placebo.16,17,72 Cabozantinib monotherapy in CRPC is being studied further in two ongoing phase II studies. The aim of one randomized study is to compare immediate prostatectomy versus cabozantinib followed by prostatectomy in patients with high-risk prostate cancer (NCT03964337), and the aim of the second single-arm study is to determine the effectiveness of cabozantinib in the treatment of patients with mCRPC (NCT04631744). Based on the 140 active cabozantinib monotherapy studies listed on ClinicalTrials.gov in May 2021, 27 are in indications that are not yet approved, including the recently published COSMIC 311 trial (NCT03690388; estimated study completion, December 2022). 9 COSMIC 311 was a phase III RCT comparing cabozantinib with placebo and focuses on patients with RAI-refractory DTC who progressed during or following treatment with ⩽2 prior VEGFR inhibitors. As is the case for other types of systemic therapy, there are relatively few studies of cabozantinib (and other TKIs) in DTC populations, probably because patients with DTC largely respond well to standard surgical treatment and RAI therapy. 128 The COSMIC 311 results (published after the bibliometric searches were conducted and so not included in the quantitative analysis of the search results) reported significantly prolonged PFS with cabozantinib compared with placebo (HR 0.22, 95% CI: 0.13–0.36; p < 0.0001); the safety profile was manageable and consistent with the known safety profile of cabozantinib. 9
A recent review emphasized that antiangiogenic drugs, such as cabozantinib, are of particular interest in ovarian cancer, in which immunotherapy has achieved only modest results. 129 Our SLR supports the use of antiangiogenic therapy in patients with ovarian cancer, albeit from a single study that reported prolonged PFS with cabozantinib versus placebo (significance not reported) in pretreated patients with ovarian cancer. 77 Additionally, a phase II trial evaluating the efficacy of cabozantinib in the treatment of patients with incurable, refractory, germ cell tumors, including ovarian germ cell tumors, started in May 2021 (NCT04876456).
The remaining tumor types identified in this SLR, which included breast cancer and mUC, were evaluated in single-arm, phase I and II studies only; in breast cancer, ORR was 0–17%.81,82 In mUC, ORR was 19% and median OS was 8.1 months. 80 Across a variety of tumor types, clinical evidence continues to be generated.
This SLR showed that AEs were similar across disease types and were in line with those commonly observed for other TKIs. 130 The most common AEs were hypertension, diarrhea, fatigue, lipase elevation, and PPES. Cases of hyponatremia mainly occurred with higher doses of cabozantinib (140 mg versus 60 mg). 69 AEs were managed well with dosing adjustments as part of standard practice. Even with rates of dose reduction of 46–62% in phase III RCTs (from a starting dose of 60 mg), patients experienced improved efficacy relative to everolimus, sunitinib, and placebo.13,14,28
Evidence of the real-world effectiveness of cancer therapies is vital, because it demonstrates whether findings observed during monitored clinical settings and among selected patient populations can be translated to clinical practice. This review captured several real-world studies of cabozantinib in Europe, North America, Asia, and Australasia for patients with RCC,35,36,38,40,43,44,47,50–55,100 but only two studies were identified that reported the effectiveness/safety data for HCC with cabozantinib as a single agent.61,62 We observed that real-world findings in RCC are generally consistent with the results from RCTs. In RCC, OS, PFS, ORR, and DCR were similar in RCTs (21.2–30.3 months,21,24,28,29 7.4–8.6 months,13,28,29 17–33%,13,21,28,29 and 75–83%,13,28,29 respectively) and real-world studies (9.1–25.4 months,35–43,53–55 5.6–12.5 months,35–38,42,44,45,47,53,54 14–52%,35,37–39,41–44,46,47,53–55 and 50–96%,35,37,39,41–44,46,47,53–55 respectively). Likewise, in HCC, OS, PFS, ORR, and DCR were comparable between the CELESTIAL trial (10.2 months, 14 5.2 months, 14 4% 14 and 64%, 14 respectively) and the real-world studies (7.7–12.9 months,58,59 5.1 months, 62 5% 61 and 59%, 62 respectively). In terms of safety data, the overall percentages of patients with RCC experiencing grade 3–4 AEs were lower in real-world studies (15–49%35,44,46,47,55,99) than in the RCTs (67–68%13,28). The report of the Italian Managed Access Program proposed that the lower tolerability in the real-world studies may be related to bias due to the retrospective nature of the studies, the smaller sample sizes, and the improvements in management of dose and AEs because of better comprehension of the activity of cabozantinib in the clinic. 44 For both RCC and HCC, rates of dose reductions and discontinuations were consistent with clinical trials.
Almost half of the publications included in this review (54 of the 114) reported the results of biomarker analyses. RET mutational status was the most frequently studied biomarker; all studies were in patients with MTC. Other biomarkers that featured in the eligible publications included the following: bone metastases, MET expression levels, RAS mutational status, circulating tumor cells, alpha-fetoprotein, and tumor characteristics. Detailed analysis of the biomarker evidence was beyond the scope of this review, but it is an important area of research, given the potential value of cancer biomarkers in clinical practice. Depending on the type, biomarkers could be used for determining prognosis, monitoring progression of the disease, or measuring response to treatment. 131 More prospective studies are needed to explore predictive biomarkers for tumor response to cabozantinib.
The strength of this SLR is its comprehensiveness. To our knowledge, given that there was no restriction by disease type, this is the most comprehensive review of cabozantinib monotherapy to date. Although not included in this manuscript, studies of cabozantinib used in combination with other therapies have been discussed in a separate manuscript (citation to be included once available). The main limitation of the current review is the high proportion of abstracts, in which information is often incomplete and for which peer review may have been less stringent. However, inclusion of congress materials allowed us to capture the most up-to-date findings, even if full results have not yet been published, which is important for the fast-moving field of oncology.
In conclusion, in this extensive review of the literature, the efficacy and safety of cabozantinib monotherapy has been demonstrated for the treatment of various types of solid tumor. Real-world effectiveness has been demonstrated for RCC, and initial data are encouraging for HCC, indicating that positive findings from clinical trials do, indeed, translate into tangible benefits for patients in clinical practice. Ultimately, while evidence continues to be generated for various tumors, patient prognosis does not only rely on effective treatments, but also on patient education, consistent monitoring and early detection by healthcare providers, and the application of emerging management strategies evidenced in the literature.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221107112 – Supplemental material for Cabozantinib for the treatment of solid tumors: a systematic review
Supplemental material, sj-docx-1-tam-10.1177_17588359221107112 for Cabozantinib for the treatment of solid tumors: a systematic review by Pablo Maroto, Camillo Porta, Jaume Capdevila, Andrea B. Apolo, Santiago Viteri, Cristina Rodriguez-Antona, Lidia Martin and Daniel Castellano in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359221107112 – Supplemental material for Cabozantinib for the treatment of solid tumors: a systematic review
Supplemental material, sj-docx-2-tam-10.1177_17588359221107112 for Cabozantinib for the treatment of solid tumors: a systematic review by Pablo Maroto, Camillo Porta, Jaume Capdevila, Andrea B. Apolo, Santiago Viteri, Cristina Rodriguez-Antona, Lidia Martin and Daniel Castellano in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Ipsen.
Conflict of interest statement
PM is on advisory boards for Astellas, AstraZeneca, Bayer, Bristol, Ipsen, Janssen, Novartis, Pfizer, and Roche, and has received research grants from Bayer and Roche. CP has an advisory role with AstraZeneca, Bristol Myers Squibb, Eisai, EUSA Pharma, General Electric Healthcare, Ipsen, Janssen, MSD, Novartis and Pfizer; has been a speaker for Bristol Myers Squibb, EUSA Pharma, General Electric Healthcare, Ipsen, and Pfizer; has been a protocol steering committee member for Bristol Myers Squibb, Eisai and EUSA Pharma; and has provided expert testimony for EUSA Pharma and Pfizer. JC has a scientific consultancy role (speaker and advisory roles) for Advanced Accelerator Applications, Amgen, Bayer, Eisai, Exelixis, Ipsen, ITM, Merck Serono, Novartis, Pfizer, Sanofi and Sirlex, and has received research grants from Advanced Accelerator Applications, AstraZeneca, Bayer, Eisai, Novartis, and Pfizer. ABA has no interests to declare. SV reports personal fees and nonfinancial support from Bristol Myers Squibb, personal fees and nonfinancial support from Roche, personal fees from MSD, personal fees from AbbVie, nonfinancial support from OSE PHARMA, and nonfinancial support from Merck. CR-A has no interests to declare. LM is an employee of Ipsen. DC has received institutional research funding from Janssen Oncology; has received travel and accommodation expenses from AstraZeneca (Spain), Bristol Myers Squibb, Pfizer and Roche; and has a consulting or advisory role with Astellas, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol Myers Squibb, Eli Lilly, Ipsen, Janssen Oncology, MSD Oncology, Novartis, Pfizer, Pierre Fabre, Roche/Genentech, and Sanofi.
Availability of data and material
Not applicable.
Code availability
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
