Abstract
Background:
Cabozantinib monotherapy is approved for the treatment of several types of solid tumors. Investigation into the use of cabozantinib combined with other therapies is increasing. To understand the evidence in this area, we performed a systematic review of cabozantinib combination therapy for the treatment of solid tumors in adults.
Methods:
This study was designed in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses, and the protocol was registered with PROSPERO (CRD42020144680). On 9 October 2020, we searched for clinical trials and observational studies of cabozantinib as part of a combination therapy for solid tumors using Embase, MEDLINE, and Cochrane databases, and by screening relevant congress abstracts. Eligible studies reported clinical or safety outcomes, or biomarker data. Randomized and observational studies with a sample size of fewer than 25 and studies of cabozantinib monotherapy were excluded. For each study, quality was assessed using National Institute for Health and Care Excellence methodology, and the study characteristics were described qualitatively. This study was funded by Ipsen.
Results:
Of 2421 citations identified, 32 articles were included (6 with results from randomized studies, 24 with results from non-randomized phase I or II studies, and 2 with results from both). The most commonly studied tumor types were metastatic urothelial carcinoma/genitourinary tumors and castration-resistant prostate cancer (CRPC). Findings from randomized studies suggested that cabozantinib combined with other therapies may lead to better progression-free survival than some current standards of care in renal cell carcinoma, CRPC, and non-small-cell lung cancer. The most common adverse events were hypertension, diarrhea, and fatigue.
Conclusion:
This review demonstrates the promising efficacy outcomes of cabozantinib combined with other therapies, and a safety profile similar to cabozantinib alone. However, the findings are limited by the fact that most of the identified studies were reported as congress abstracts only. More evidence from randomized trials is needed to explore cabozantinib as a combination therapy further.
Keywords
Introduction
Cabozantinib, approved as monotherapy for the treatment of several types of solid tumors, is the only available multitargeted vascular endothelial growth factor receptor (VEGFR) tyrosine kinase inhibitor (TKI) that targets VEGFR (VEGF-1, VEGFR-2, and VEGFR-3) and also MET and AXL. 1 By targeting VEGFR, cabozantinib inhibits tumor blood vessel growth (angiogenesis) and promotes the vascular normalization of the tumor. 2 Simultaneous inhibition of abnormal MET signaling, which is associated with cell invasion, metastasis, tumor proliferation, and angiogenesis, and AXL signaling, which is linked with increased proliferation, invasion, and metastasis, allows cabozantinib to target multiple parallel key pathways involved in tumor vascularization and growth.3–5 Cabozantinib also targets other specific receptor tyrosine kinases involved in tumorigenesis, including RET, KIT, FLT3, ROS1, MER, TYRO3, TRKB, and TIE-2.6–8 The efficacy of cabozantinib monotherapy has been demonstrated in several solid tumor types, including renal cell carcinoma (RCC),9,10 hepatocellular carcinoma (HCC) 11 and medullary thyroid cancer 12 based on data from phase II and III randomized controlled trials (RCTs). Based on the results from the COSMIC-311 study, 13 the Committee for Medicinal Products for Human Use recently recommended the use of cabozantinib in differentiated thyroid carcinoma (DTC) and a decision is pending in the European Union (EU). Evidence from these trials has supported the approval of cabozantinib in these indications, under specific conditions, in the EU14,15 and in the United States.16,17
Cabozantinib also has the potential to be effective in combination with other therapies. Inhibition of tumor angiogenesis and vascular normalization of the tumor allows for a synergistic effect when combined with immunotherapy because, in a normalized vascular network, it favors infiltration and accumulation of immune effector cells within the tumor.2,18 Given that it has multiple targets, cabozantinib may lead to additional synergistic effects with immunotherapy beyond those of other TKIs. For example, it blocks intracellular signaling pathways that drive epithelial–mesenchymal transition, which is associated with an immunosuppressive state and reduced sensitivity to checkpoint inhibitors. 19 Furthermore, cabozantinib reduces the number of immunosuppressive cells, such as myeloid-derived suppressor cells and regulatory T cells, and increases the number of peripheral and tumor-infiltrating cytotoxic cluster of differentiation 8 (CD8)+ T cells.20,21 In addition, a recent in vitro study showed that cabozantinib triggered immunogenic cell death in prostate cancer cells and directly modulated dendritic cells, suggesting an immunostimulatory role. 22
Mouse models have been used to investigate the combination of cabozantinib with anti-programmed cell death protein 1 (anti-PD1) therapy, a type of immunotherapy, in HCC. Results showed that the combination led to a higher objective response rate (ORR) than cabozantinib alone. 23 This was explained by the enhanced effect of both therapies working to counteract tumor-induced immunosuppression: cabozantinib by acting as an anti-angiogenic agent, and anti-PD1 by acting as a checkpoint inhibitor. 23 A recent study using machine-learning-based technology investigated the potential synergistic effects of cabozantinib and anti-PD1 therapy in the treatment of metastatic RCC (mRCC) and reported that cabozantinib may enhance the impact of anti-PD1 therapy on immunosurveillance via its actions on the innate and adaptive immune system, and that anti-PD1 therapy may enhance the anti-angiogenic and pro-apoptotic effects of cabozantinib via action on angiogenesis and T-cell cytotoxicity. 24
In patients with metastatic urothelial carcinoma (mUC) treated with cabozantinib, reductions in the number of regulatory T cells and myeloid-derived suppressor cells were observed after treatment and were associated with clinical responses 21 ; similar associations have been demonstrated in patients with RCC treated with cabozantinib. 25 In addition, cabozantinib has been shown to increase the number of CD8+ T cells in patients with RCC. 26 One study of patients with mRCC demonstrated that patients treated with cabozantinib following immune checkpoint blockade (ICB) had higher response rates than patients who received ICB before treatment with cabozantinib. 27 The US Food and Drug Administration and European Medicines Agency have recently approved cabozantinib plus nivolumab for the first-line treatment of patients with advanced RCC (aRCC), based on the evidence of a significant improvement in progression-free survival (PFS; primary) and overall survival (OS; secondary) with the combination compared with sunitinib in previously untreated patients with clear-cell aRCC. 28
In addition to immunotherapy, there is some suggestion of cabozantinib having synergistic activity with glutaminase inhibitors, which reduce glutamine metabolism that is upregulated in some tumors and supports tumor angiogenesis.3,29,30 There is also evidence that the activity of androgen deprivation therapy is likely to be enhanced by concomitant inhibition of angiogenesis with cabozantinib. 31 Furthermore, combination treatment with cabozantinib may serve to block the development of MET-driven resistance observed with some therapies, such as endothelial growth factor receptor (EGFR) inhibitors,32–35 and the resistance to VEGFR inhibition with other TKIs driven by additional targets of cabozantinib, such as AXL and fibroblast growth factor receptor. 36
To consolidate the available evidence in this area, we performed a systematic literature review (SLR) to identify the published clinical and observational data examining cabozantinib therapy in combination with other therapies for the treatment of solid tumors in adults. From these studies, we aimed to assess the evidence on the clinical efficacy and safety profile of cabozantinib as part of a combination therapy for solid tumors.
Materials and methods
Search strategy
Published studies relating to the use of cabozantinib as part of a combination therapy for the treatment of solid tumors were identified through a systematic search. MEDLINE In-Process & Other Non-Indexed Citations and Ovid MEDLINE (1946–present), Embase (1974–present), and the Cochrane Library were searched on 9 October 2020 using the Ovid platform (see Supplemental Resource 1 for search terms used in Embase). The protocol for the SLR is registered with the International Prospective Register of Systematic Reviews (PROSPERO; registration name: An SLR of cabozantinib for the treatment of solid tumors; registration number: CRD42020144680). 37
Supplementary searches
The bibliographies of studies identified in the electronic searches were reviewed to identify the additional relevant references. Congress abstracts were searched from 1 January 2016 to 9 October 2020. The congresses included were American Society of Clinical Oncology (ASCO), ASCO Gastrointestinal Cancers Symposium, ASCO Genitourinary Cancers Symposium, European Society for Medical Oncology, and American Association for Cancer Research. Prior to submission of the article, searches were conducted for the full publication of studies already included as abstracts. If the full manuscript was identified later, both the congress abstract and full article are cited in the review; the data presented are from the more recent full article.
To identify the ongoing and planned trials for cabozantinib as part of a combination therapy, searches of ClinicalTrials.gov were performed in November 2020 to identify ‘not yet recruiting’, ‘recruiting’, ‘enrolling’, and ‘active’ studies. The status of these trials was checked prior to final submission of the article (February 2022).
Study selection and data collection
Citations identified by the searches were screened against prespecified criteria in accordance with the 2009 Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. 38 During screening, abstracts and titles were reviewed by a single reviewer against the inclusion/exclusion criteria to identify the relevant studies, and any uncertainties were resolved by a second reviewer. Inclusion and exclusion criteria are presented in Table 1.
Eligibility criteria for studies identified by an SLR.
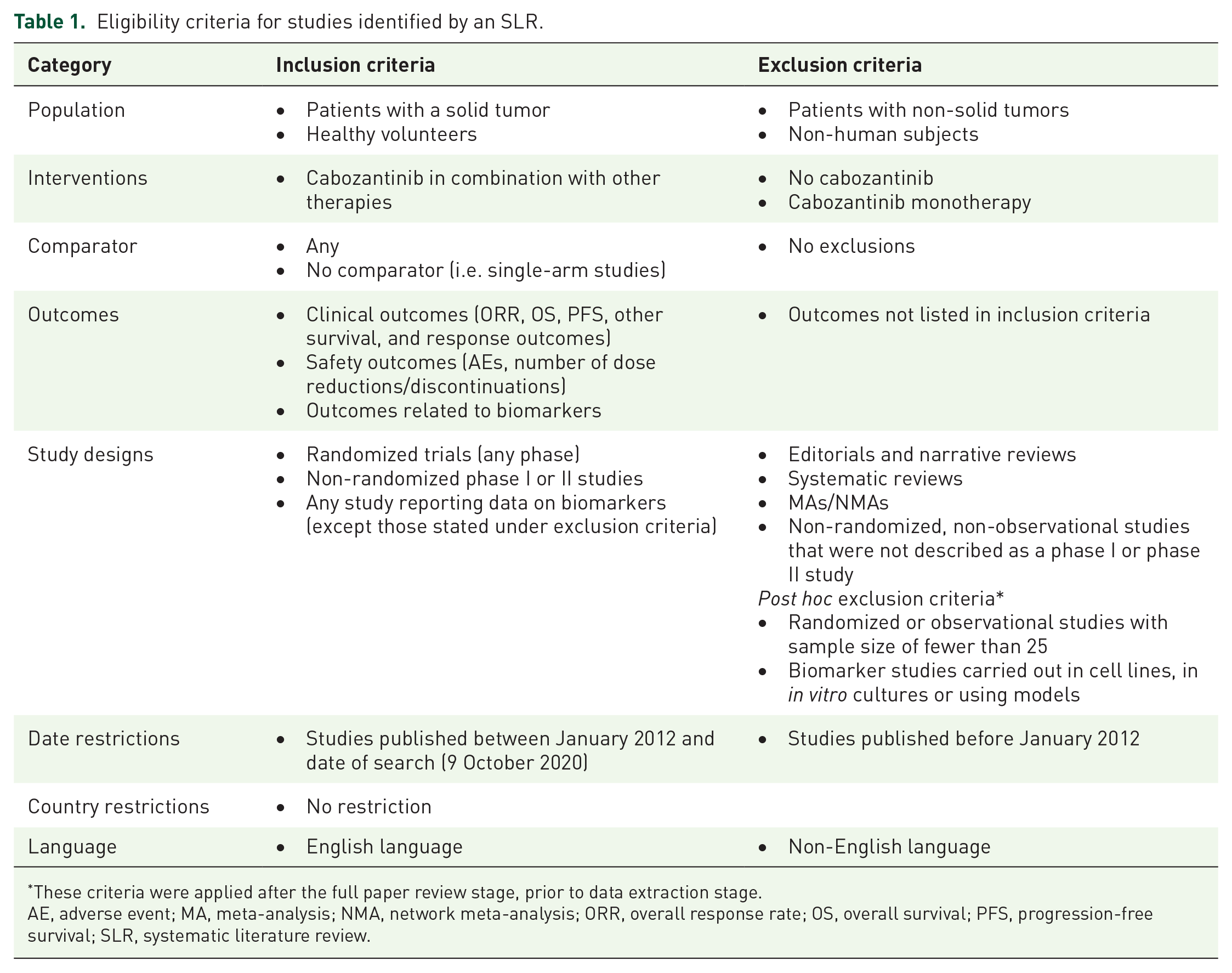
These criteria were applied after the full paper review stage, prior to data extraction stage.
AE, adverse event; MA, meta-analysis; NMA, network meta-analysis; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; SLR, systematic literature review.
For the data extraction stage, study population data and key results relating to efficacy, safety, and biomarkers were extracted manually for each included study into an Excel worksheet.
Results
Overview of evidence
Results of SLR
The electronic searches identified 2421 citations (excluding duplicates), which were then screened. Of these, 386 were selected for full paper review. In addition, 19 articles were considered relevant from the congress searches, and three articles were identified from bibliographies of studies identified in the electronic searches. After full paper review and review of congress abstracts and additional articles, 376 articles were excluded; 58 of these were excluded according to post hoc exclusion criteria (i.e. RCTs with a sample size of fewer than 25, and biomarker studies carried out in cell lines, in vitro cultures or in vivo models). The final number of studies considered relevant was 32 (Figure 1).

PRISMA diagram of included and excluded studies in the SLR.
Characteristics of included studies
Of the 32 included articles, there were five articles reporting findings from RCTs, one reporting findings from a randomized discontinuation trial, 24 reporting findings from non-randomized phase I or II studies and two reporting results from both a non-randomized phase I study and a randomized phase II study. The characteristics of all included studies are presented in Table 2. In terms of quality assessment, none of the 32 references had the highest rating for internal validity. Poorer ratings were predominantly owing to a lack of study details reported in congress abstracts (23 of the included references were abstracts, and nine were articles).
Study characteristics and quality assessment of included studies.
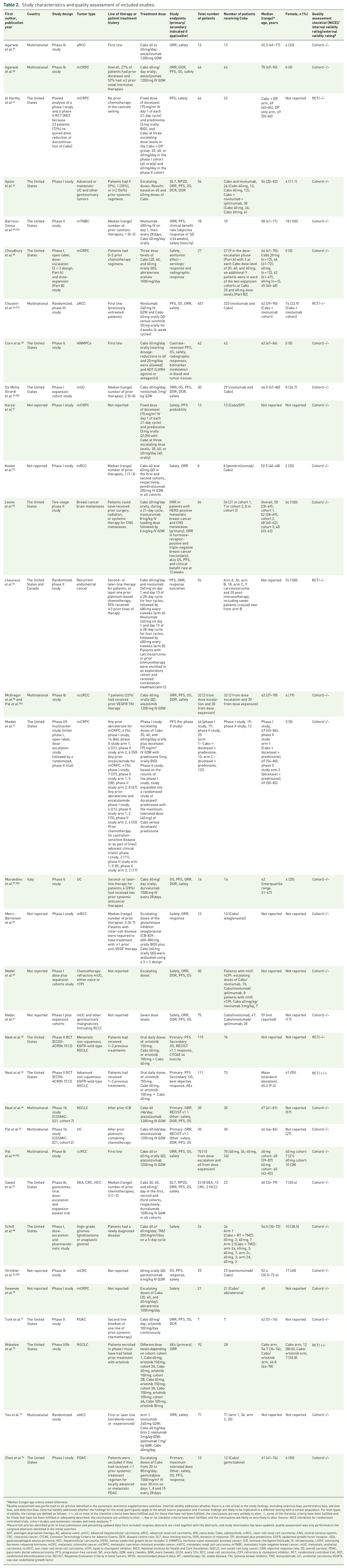
Median (range) age unless stated otherwise.
Quality assessment was performed on all articles identified in the systematic and initial supplementary searches. Internal validity addresses whether there is a risk of bias in the study findings, including selection bias, performance bias, attrition bias, and detection bias. External validity addresses whether the findings for the study participants apply to the whole source population and if similar findings are likely to be replicated in a different setting with a similar population. For both types of validity, the ratings are defined as follows: ++, all or most of the checklist criteria have been fulfilled and, for those that have not been fulfilled, the conclusions are very unlikely to alter; +, some of the checklist criteria have been fulfilled and, for those that have not been fulfilled or adequately described, the conclusions are unlikely to alter; −, few or no checklist criteria have been fulfilled, and the conclusions are likely or very likely to alter. Source: NICE checklists for randomized controlled trials, cohort studies and systematic reviews and meta-analyses. 75
Recent full articles identified prior to final submission and presenting updated data from included congress abstracts are cited together with the abstracts, and study information has been updated; quality assessment was only performed on the congress abstracts identified in the initial searches.
ADT, androgen deprivation therapy; AE, adverse event; aHCC, advanced hepatocellular carcinoma; aRCC, advanced renal cell carcinoma; BID, twice daily; Cabo, cabozantinib; ccRCC, clear-cell renal cell carcinoma; CNS, central nervous system; CRC, colorectal cancer; CTCAE, Common Terminology Criteria for Adverse Events; DCR, disease control rate; DLT, dose-limiting toxicity; DOR, duration of response; DP, docetaxel plus prednisone; EGFR, epidermal growth factor receptor; GEA, gastroesophageal adenocarcinoma; HCC, hepatocellular carcinoma; HER2, human epidermal growth factor receptor 2; HNMPCa, hormone-naive metastatic prostate cancer; ICB, immune checkpoint blockade; IV, intravenously; LHRH, luteinizing hormone-releasing hormone; mCRC, metastatic colorectal cancer; mCRPC, metastatic castration-resistant prostate cancer; mRCC, metastatic renal cell carcinoma; mTNBC, metastatic triple-negative breast cancer; mUC, metastatic urothelial carcinoma; nccRCC, non-clear-cell renal cell carcinoma; nCPI, naive to checkpoint inhibitor; NICE, National Institute for Health and Care Excellence; NSCLC, non-small-cell lung cancer; ORR, objective response rate; OS, overall survival; PDAC, pancreatic ductal adenocarcinoma; PFS, progression-free survival; QD, once daily; Q2W, every 2 weeks; Q3W, every 3 weeks; Q12H, every 12 h; RCC, renal cell carcinoma; rCPI, refractory to checkpoint inhibitor; RCT, randomized controlled trial; RDT, randomized discontinuation trial; RECIST, Response Evaluation Criteria in Solid Tumors; RP2D, recommended phase II dose; RT, radiotherapy; SD, stable disease; TKI, tyrosine kinase inhibitor; TMZ, temozolomide; UC, urothelial carcinoma; VEGFR, vascular endothelial growth factor.
The SLR identified 28 articles that include efficacy outcomes for cabozantinib in combination with another therapy, all but one of which also reported safety outcomes, and four articles that reported safety outcomes only. Disease types studied for efficacy were as follows: RCC (6 articles); non-small-cell lung cancer (NSCLC; 4 articles); mUC (2 articles, 1 with RCC subgroup) or other genitourinary tumors (5 articles); castration-resistant prostate cancer (CRPC; 6 articles); pancreatic ductal adenocarcinoma (PDAC; 2 articles); and metastatic colorectal cancer (mCRC), high-grade gliomas, advanced HCC (aHCC), hormone-naive metastatic prostate cancer, breast cancer brain metastases, metastatic triple-negative breast cancer, and recurrent endometrial cancer (1 article each). In addition, one article covered gastroesophageal adenocarcinoma (GEA), colorectal cancer (CRC), and HCC.
Prior to submission, six recently published full articles on studies included in the SLR as congress abstracts were identified and included in the results.
Efficacy of cabozantinib in combination
Renal cell carcinoma
One phase III RCT investigating the combination of cabozantinib and nivolumab and four phase I/Ib studies assessing the combination of cabozantinib with anti-PD-1/PD-L1 immunotherapies [atezolizumab, pembrolizumab, or nivolumab with or without ipilimumab (anti-CTLA-4 monoclonal antibody)] or a glutaminase inhibitor (telaglenastat) reported favorable efficacy outcomes with cabozantinib combination therapy in patients with RCC.39,46,47,53,55,59,61,66
In the phase III study CheckMate 9ER, patients with previously untreated aRCC [1:1, stratified by International Metastatic RCC Database Consortium (IMDC) risk score, PD-L1 expression and region] were randomly assigned to receive nivolumab in combination with cabozantinib (40 mg/day) (n = 323) or sunitinib (50 mg/day for 4 weeks of each 6-week cycle) (n = 328).46,47 All three efficacy endpoints were met. Nivolumab plus cabozantinib versus sunitinib significantly improved PFS (primary endpoint) by blinded central review [median: 16.6 months versus 8.3 months; hazard ratio (HR) for disease progression or death 0.51; 95% confidence interval (CI): 0.41–0.64; p < 0.001] and OS (probability at 12 months of 85.7% versus 75.6%; HR for death, 0.60; 98.89% CI: 0.40–0.89; p = 0.0010). The ORR (95% CI) was significantly higher in the combination group than in the sunitinib group [55.7% (50.1–61.2%) versus 27.1% (22.4–32.3%); p < 0.0001]; complete response (CR) was achieved in 8.0% and 4.6% of patients, respectively, and the median duration of response (DOR) was 20.2 months and 11.5 months, respectively. In subsequent analysis, with an extended median follow-up for OS of 32.9 (range: 25.4–45.4) months nivolumab plus cabozantinib versus sunitinib improved OS (median 37.7 months versus 34.3; HR, 0.70; 95% CI: 0.55–0.90); in addition, PFS and ORR benefits with N + C were sustained with minimum 2-year follow-up. 76
Three abstracts and one recent full publication from the phase Ib COSMIC-021 study, which is evaluating cabozantinib plus atezolizumab in various solid tumors, reported efficacy outcomes in patients with aRCC. In the dose-escalation stage in patients with treatment-naive aRCC [n = 12; 10 with clear-cell RCC (ccRCC) and 2 with non-clear cell RCC (nccRCC)], an ORR of 67% [1 CR and 7 partial responses (PRs)] was observed with cabozantinib (40 mg/day or 60 mg/day) plus atezolizumab. 39 In a further analysis of treatment-naive patients with ccRCC receiving atezolizumab plus cabozantinib 40 mg/day (n = 34) or 60 mg/day (n = 36), ORRs were 53% (80% CI: 41–65%) and 58% (80% CI: 46–70%), disease control rates (DCRs) were 94% and 92%, and median PFS was 19.5 months and 15.1 months, respectively. 66 In the cohort of 32 patients with nccRCC (two from dose escalation and 30 from dose expansion), of whom seven had received prior VEGFR TKI therapy, cabozantinib 40 mg/day plus atezolizumab was associated with an ORR of 31% (80% CI: 20–44%; all confirmed PRs), a DCR of 94%, and a median DOR of 8.3 months. 55
Three other phase I studies measured ORR in patients with mRCC treated with cabozantinib in combination with another therapy.52,59,61 Of these, one study included patients receiving cabozantinib (40 mg/day) plus nivolumab (n = 7), or cabozantinib (40 mg/day) plus nivolumab and ipilimumab (n = 6). Among all 13 patients, the ORR was 53.9% and the DCR, defined as CR + PR + stable disease (SD), was 100%. 61 Another study evaluated telaglenastat (CB-839, a glutaminase inhibitor) plus cabozantinib (60 mg/day) as a second-line therapy (following treatment with mammalian target of rapamycin inhibitors or one or more anti-VEGFR therapies) in 13 patients with mRCC (clear-cell or papillary histology), and demonstrated an ORR of 42% and a DCR of 100%. 59 In a study of eight patients receiving pembrolizumab and cabozantinib (40 mg/day or 60 mg/day), the ORR was 25%, and the median number of prior therapies was 1 (range: 1–3). 52
Urothelial carcinoma
One single-arm phase II, one phase Ib and one phase I study reported positive efficacy outcomes with the combination of cabozantinib and anti-PD1 immunotherapy (durvalumab, atezolizumab, or nivolumab with or without ipilimumab) in patients with advanced urothelial carcinoma (UC) or mUC.42,49,50,57,58,65
In an interim analysis of a single-arm, phase II study of 16 patients with advanced UC who received cabozantinib (40 mg/day) and durvalumab after platinum-based chemotherapy, objective responses were observed in six patients (37.5%) including two CRs (12.5%) and four PRs; response was ongoing at 8 months in one patient with a RET short variant alteration (P117T).57,58
Results from cohort 2 of the phase Ib COSMIC-021 study, which is investigating cabozantinib plus atezolizumab, showed an ORR of 27% in patients with locally advanced or mUC who had received prior platinum-containing chemotherapy (n = 30). The DCR was 64% and the median (range) PFS was 5.4 (0–17.3) months. 65
One phase I dose-escalation study compared outcomes in 54 patients with mUC and other metastatic genitourinary tumors following treatment with cabozantinib plus nivolumab, and cabozantinib plus nivolumab and ipilimumab. 42 The median (95% CI) PFS and OS were 5.1 (3.5–6.9) months and 12.6 (6.9–18.8) months, respectively. In the 49 patients evaluable for tumor response, ORR (95% CI) was 30.6% (18.3–45.4%), and four patients (8.2%) had a CR. ORR was numerically higher in patients (n = 23) who received cabozantinib plus nivolumab than in those (n = 26) who received cabozantinib plus nivolumab and ipilimumab (39.1% versus 23.1%). Among those with evaluable responses, the DCR (95% CI) was 77.6% (63.4–88.2%) and the median (95% CI) DOR was 21.0 (5.4–24.1) months. DCR was 82.6% in patients who received cabozantinib plus nivolumab and 73.1% in those who received cabozantinib plus nivolumab and ipilimumab. In the expansion study, involving 29 patients with mUC who received prior ICB treatment with cabozantinib 40 mg/day and nivolumab, the median (95% CI) PFS and OS were 3.6 (2.1–5.5) months and 10.4 (5.8–19.5) months, respectively; ORR was 13.8% (one patient with CR, three patients with PR).49,50
Gynecological cancer
In one randomized phase II study of cabozantinib (40 mg/day) and nivolumab (n = 36) versus nivolumab (n = 18) in women with recurrent endometrial cancer, median (95% CI) PFS was significantly longer in those receiving cabozantinib plus nivolumab than in those receiving nivolumab alone [5.3 (3.5–9.5) months versus 1.9 (1.6–3.8) months; log-rank p = 0.07]. Analysis of the tumor microenvironment to identify predictive immune biomarkers of response is ongoing in this study. 54
Prostate cancer
A phase II study reported promising clinical activity with the combination of cabozantinib and androgen deprivation therapy in patients with hormone-naive metastatic prostate cancer. 48 Two phase I/II studies demonstrated a clinical benefit of the combination with cabozantinib and prednisone (DP) compared with DP alone in patients with metastatic CRPC (mCRPC).31,41,51 In addition, a phase Ib study showed clinically relevant activity with the combination of cabozantinib and atezolizumab in patients with mCRPC. 40
Evaluation of cabozantinib (60 mg/day) plus androgen deprivation therapy in a phase II study of 62 patients with hormone-naive metastatic prostate cancer (median follow-up: 31.2 months) showed that median (95% CI) PFS was 16.1 (14.6–22.7) months, whereas median OS was not reached. 48
A pooled analysis of a phase I study and a randomized phase II study reported outcomes in 44 patients with mCRPC with no prior chemotherapy, who had received docetaxel plus DP or a combination of cabozantinib (20–60 mg/day) and DP. Patients in each arm had the same median age (69 years), but those in the cabozantinib combination arm (n = 32) had lower median prostate-specific antigen than patients in the DP arm (n = 12) (74.8 ng/mL versus 309.5 ng/mL). Patients in the cabozantinib combination arm had a longer median (95% CI) PFS [13.6 (8.31–21.0) months] than those in the DP arm [6.6 (2.9–10.4) months; p value not reported]. 41 In the original phase I study, which included 13 patients receiving a combination of cabozantinib and DP, the probability of PFS was 90% at 6 months and 67.5% at 8 months. 51
In another phase I/II study, 19 patients with mCRPC received escalating doses of cabozantinib (20, 40, and 60 mg/day) plus DP in the phase I part of the study, with a median time to progression (TTP) and OS of 13.6 months and 16.3 months, respectively. In the randomized phase II expansion part of the study, which was terminated early owing to poor accrual, comparison of cabozantinib plus DP (n = 13) with DP (n = 12) showed a median TTP of 21.0 months versus 6.6 months (p = 0.035) and a median OS of 23.8 months versus 15.6 months (p = 0.072), respectively. 31
Interim results from cohort 6 of the phase Ib COSMIC-021 study, which is evaluating cabozantinib (40 mg/day) plus atezolizumab in patients with a range of solid tumors, including 44 patients with mCRPC, demonstrated an ORR of 32% (2 CRs and 12 PRs). 40 Recently reported results for the expanded cohort 6 of 132 mCRPC patients with a median (range) follow-up of 15.2 months showed ORR by investigator among all patients per RECIST 1.1 was 23%, ORR by independent review (BIRC) was 15% and DCR was 84% by investigator, and 81% by BIRC (Blinded Independent Central Review). 77
Gastrointestinal cancers
Five studies reported outcomes in patients with gastrointestinal cancers, including three that showed encouraging clinical activity with: cabozantinib in combination with either nivolumab or durvalumab in patients with HCC; cabozantinib plus durvalumab in patients with advanced GEA and CRC; and cabozantinib plus panitumumab in patients with mCRC.32,67,69,71,73,74
In the phase I/II CheckMate 040 study, patients with aHCC were randomized to receive cabozantinib (40 mg/day) and nivolumab, or cabozantinib plus nivolumab and ipilimumab. In the cabozantinib and nivolumab arm, ORR was 17% (6 PRs), DCR was 81%, and median PFS was 5.5 months, whereas, in the cabozantinib plus nivolumab and ipilimumab arm, ORR was 26% (9 PRs), DCR was 83%, and median PFS was 6.8 months. Median OS was not reached in either arm. 73
In a phase Ib study of 25 patients with chemotherapy refractory KRAS wild-type mCRC, outcomes were measured after treatment with cabozantinib (60 mg/day) and panitumumab.32,69 Median (95% CI) OS and PFS were 12.1 (7.5–14.3) months and 3.7 (2.3–7.1) months, respectively. Of the 25 patients treated, 13 had received prior anti-epidermal growth factor receptor (anti-EGFR) therapy, and four patients (16%) had a confirmed PR. 32
Cabozantinib (20, 40, and 60 mg/day) in combination with durvalumab was assessed in patients with advanced GEA, CRC, and HCC in the phase Ib CAMILLA study. Preliminary analysis of the 19 patients with evaluable responses showed an ORR of 21% (four PRs: two in patients with GEA and two in patients with CRC), a clinical benefit rate of 84% and a median time to progressive disease (PD) of 16 weeks (range: 8–40+ weeks). 67 In recently report results on the phase II CRC cohort of the 36 patients, cabozantinib plus durvalumab demonstrated the promising efficacy with an ORR of 27.6% (8/29), DCR of 86.2% (25/29), median PFS of 4.4 months, and a median OS of 9.1 months. 78
A phase I study of 26 patients with advanced PDAC and no more than one prior systemic treatment regimen assessed cabozantinib treatment (escalating doses: 20–80 mg) in combination with gemcitabine. Of the eight patients evaluable, three had PR, three had SD, and two had PD; the median (95% CI) OS and PFS were 10.1 (3.6–20.6) months and 4.7 (1.4–9.7) months, respectively. 74 Further investigation of this combination was not recommended, however, owing to safety issues.
Finally, cabozantinib (40 mg/day) and erlotinib in patients with metastatic PDAC (with EGFR and c-Met overexpression) that had progressed while patients had been receiving at least one chemotherapy regimen was evaluated in a phase II study. Overall, 43 patients were screened, and seven patients were enrolled and treated; however, all patients had clinical and/or radiographic progression in the 1–2 months after initiating treatment, and the trial was stopped owing to futility. 71
Non-small-cell lung cancer
One RCT and one phase Ib/II study investigated the efficacy outcomes with cabozantinib plus erlotinib in patients with NSCLC but reported inconsistent results; one phase Ib study reported the encouraging efficacy outcomes with the combination of cabozantinib and atezolizumab in patients with NSCLC.
In the analyses of a phase II RCT of patients who received cabozantinib (60 mg/day) (n = 39), cabozantinib (40 mg/day) plus erlotinib (n = 37), or erlotinib alone (n = 39) in a second- or third-line setting (with no prior erlotinib or MET TKI therapy),62,63 the primary endpoint was PFS, with 91% power to detect an HR of 0.5. Cabozantinib and cabozantinib plus erlotinib both had significantly better PFS than erlotinib alone [HRs 0.39 (80% CI: 0.27–0.55) and 0.37 (80% CI: 0.25–0.53), respectively]. 63 Cabozantinib groups also demonstrated significantly better OS (secondary endpoint) than erlotinib alone. In a phase Ib/II study of cabozantinib (40–100 mg/day) with or without erlotinib in 64 patients with no prior VEGFR TKI therapy, the ORR was 6.7% (90% CI: 0.3–27.9%) in the cabozantinib group and 0% in the combination group. 72
Results from cohort 7 of the phase Ib COSMIC-021 study, which is investigating cabozantinib plus atezolizumab, showed an ORR of 23% in patients with metastatic non-squamous NSCLC (n = 30). The time to response (range) was 1.4 (1–3) months, the median (range) DOR was 5.6 (2.6–6.9) months, and the DCR was 83%. 64
Breast cancer
One phase II study investigated the efficacy outcomes with the combination of cabozantinib and nivolumab in patients with metastatic triple-negative breast cancer, and a phase II study assessed cabozantinib alone and in combination with trastuzumab in heavily pretreated patients with breast cancer and brain metastases.43,44,53
In the phase II, single-arm study of cabozantinib (40 mg/day) plus nivolumab in patients with metastatic triple-negative breast cancer who had received 0–3 prior cytotoxic therapies, only one of the first 18 patients had a PR (ORR, 6%; 95% CI: 0–27%), and the study was closed to further accrual; the primary endpoint was not met.43,44 The median (95% CI) PFS was 3.6 (1.9–6.9) months. In all, 14 patients (78%) had SD and two patients (11%) had PD as best response. The clinical benefit rate was 17% (95% CI: 4–41%). In the single-arm, phase II study of 36 patients [median (range) prior lines of therapy: 3 (1–9)] with breast cancer and brain metastases, patients with human epidermal growth factor receptor 2 (HER2)-positive disease (n = 21; cohort 1) received cabozantinib (60 mg/day) in combination with trastuzumab, and those with hormone-receptor-positive/HER2-negative (n = 7; cohort 2) and triple-negative disease (n = 8; cohort 3) received cabozantinib (60 mg/day) alone. Central nervous system ORR was 5% in cohort 1 (primary outcome), and 14% and 0% in cohorts 2 and 3, respectively (secondary outcomes). The authors concluded that cabozantinib had insufficient activity in these patients. 53
Safety of cabozantinib in combination
Cabozantinib plus nivolumab
Safety outcomes with the combination of cabozantinib and nivolumab were reported in six studies: a phase III study in patients with aRCC; a phase I study in patients with advanced or mUC or other genitourinary tumors; a phase I expansion study in patients with mUC; a phase II study in patients with recurrent endometrial cancer; a phase I/II study in patients with aHCC; and a phase II study in patients with metastatic triple-negative breast cancer (Table 3).42,43,46,47,49,50,54,73
Safety outcomes with cabozantinib combination therapies.

ADT, androgen deprivation therapy; AE, adverse event; aHCC, advanced hepatocellular carcinoma; ALT, alanine aminotransferase; aRCC, advanced renal cell carcinoma; AST, aspartate aminotransferase; BID, twice daily; Cabo, cabozantinib; ccRCC, clear-cell renal cell carcinoma; CNS, central nervous system; CRC, colorectal cancer; DLT, dose-limiting toxicity; EGFR, epidermal growth factor receptor; GEA, gastroesophageal adenocarcinoma; HCC, hepatocellular carcinoma; HNMPCa, hormone-naive metastatic prostate cancer; ICB, immune checkpoint blockade; IV, intravenously; LHRH, luteinizing hormone-releasing hormone; mCRC, metastatic colorectal cancer; mCRPC, metastatic castration-resistant prostate cancer; mRCC, metastatic renal cell carcinoma; mTNBC, metastatic triple-negative breast cancer; mUC, metastatic urothelial carcinoma; nccRCC, non-clear-cell renal cell carcinoma; NSCLC, non-small-cell lung cancer; PDAC, pancreatic ductal adenocarcinoma; PPES, palmar–plantar erythrodysesthesia syndrome; QD, once daily; Q2W, every 2 weeks; Q3W, every 3 weeks; Q12H, every 12 hours; RCC, renal cell carcinoma; RT, radiotherapy; TEAE, treatment-emergent adverse event; TKI, tyrosine kinase inhibitor; TMZ, temozolomide; TRAE, treatment-related adverse event; UC, urothelial carcinoma; VEGFR, vascular endothelial growth factor.
In the phase III CheckMate 9ER study of nivolumab (240 mg every 2 weeks) plus cabozantinib (40 mg/day) versus sunitinib (50 mg/day for 4 weeks in 6-week cycles) in previously untreated patients with clear-cell aRCC, 96.6% of patients receiving nivolumab plus cabozantinib and 93.1% of those receiving sunitinib experienced treatment-related adverse events (AEs); grade 3 treatment-related AEs occurred in 60.6% and 50.9% of patients, respectively. Treatment-related AEs led to discontinuation of nivolumab in 5.6%, cabozantinib in 6.6%, and sunitinib in 8.8% of patients. One and two treatment-related deaths were reported in the nivolumab plus cabozantinib and sunitinib groups, respectively.46,47 In recently reported analysis of patients with a 2-year follow-up, no new safety signals emerged with extended follow-up in either arm among all treated patients. 76
In a Phase I study involving patients with mUC and other metastatic genitourinary tumors (cabozantinib 40 mg/day and escalating doses of other treatments) grade 3–4 treatment-related AEs with cabozantinib plus nivolumab and with cabozantinib plus nivolumab and ipilimumab, respectively, occurred in 75% and 87% of patients, and in 42% and 71% of patients in the CheckMate 040 aHCC study.42,73 Among the patients with aHCC, these AEs led to discontinuation in 3% of patients receiving cabozantinib plus nivolumab and 20% of those treated with cabozantinib plus nivolumab and ipilimumab.
In a study of 18 patients with metastatic triple-negative breast cancer and 0–3 prior cytotoxic therapies, all-cause AEs occurred in 100% of patients and grade 3–4 AEs in 83% of patients. 43
The most commonly reported AEs and treatment-related AEs with the combination of cabozantinib and nivolumab included diarrhea, palmar–plantar erythrodysesthesia (PPES), hypertension, fatigue, and elevated liver enzyme levels (Table 3). Diarrhea related to cabozantinib and nivolumab combination therapy was recorded in 56.9% of patients with aRCC and 47.2% of patients with recurrent endometrial cancer; a grade 3 or above treatment-related AE of diarrhea with cabozantinib plus nivolumab was reported for 5.6% of patients with aRCC.46,54 A grade 3 or above or grade 3–4 treatment-related AE of hypertension with cabozantinib plus nivolumab was reported in 10.9% of patients with aRCC and in 21% (cabozantinib plus nivolumab) and 10% (cabozantinib plus nivolumab and ipilimumab) of patients with advanced or mUC and other genitourinary tumors.42,46,50 In patients with aRCC, the incidence of treatment-related PPES with cabozantinib plus nivolumab was 38.1% overall (any grade) and the incidence of grade 3 or above treatment-related PPES was 7.5%; 17% of patients with metastatic triple-negative breast cancer had a grade 3–4 AE of PPES.43,46 Treatment-related elevations in liver enzyme levels with cabozantinib and nivolumab were found in 44.4% of patients with recurrent endometrial cancer, and grade 3–4 AEs of elevated aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels were reported in 17% and 11% of patients, respectively, in the metastatic triple-negative breast cancer study.43,54
In addition to hypertension, grade 3–4 treatment-related AEs in patients with advanced or mUC and other genitourinary tumors who were treated with cabozantinib plus nivolumab or cabozantinib plus nivolumab and ipilimumab included fatigue (17% and 10%, respectively) and thromboembolic events (4% and 10%, respectively); grade 3–4 immune-related AEs included hepatitis (0% and 13%, respectively) and colitis (0% and 7%, respectively). The recommended phase II dosage was cabozantinib 40 mg/day and nivolumab 3 mg/kg for cabozantinib plus nivolumab, and cabozantinib 40 mg/day, nivolumab 3 mg/kg, and ipilimumab 1 mg/kg for cabozantinib plus nivolumab and ipilimumab. 42 In the expansion study, in 30 patients with mUC with PD following ICB, 52% treated with cabozantinib (40 mg/day) plus nivolumab had a grade 3 treatment-related AE and 3% a grade 4 treatment-related AE; the most common were fatigue (10%), hypophosphatemia (10%), lymphocyte count decrease (10%), thromboembolic event (10%), hypertension (7%), diarrhea (7%), and increased amylase (7%).49,50 The management of overlapping toxicities of combination cabozantinib and nivolumab using the 9ER phase III trial, the phase 1 combination study, and published guidelines is discussed in a recently published review. 79
Cabozantinib plus atezolizumab
Six congress abstracts presenting safety data from the phase Ib COSMIC-021 study of cabozantinib plus atezolizumab in patients with RCC, UC, mCRPC, and NSCLC were retrieved from the searches, and a recently published manuscript later identified.39,40,55,56,64,65,66
Three of the six congress abstracts and the full manuscript reported safety outcomes in patients with aRCC. In the dose-escalation stage in patients with treatment-naive aRCC, 11/12 patients (92%) experienced grade 3 AEs, including hypertension (three patients and two patients in the 40 mg and 60 mg dose groups, respectively), hypophosphatemia (one patient in each dose group) and diarrhea (two patients, both in the 60 mg dose group). There were nine dose reductions (75%) due to AEs. 39 Among all patients with ccRCC enrolled in the study (treatment-naive with the exception of two patients from the dose-escalation stage), grade 3–4 treatment-related AEs were reported in 71% and 67% of patients in the 40 mg and 60 mg dose groups, respectively, the most common being hypertension (24% and 14%, respectively), diarrhea (9% and 19%, respectively), hypophosphatemia (15% and 3%, respectively), and increased ALT (3% and 14%, respectively). 56 In the cohort of 32 patients with nccRCC who received cabozantinib 40 mg/day plus atezolizumab, treatment-related grade 3–4 AEs were reported in 38% of patients, most commonly hypophosphatemia (13%). 56
Grade 3–4 treatment-related AEs with cabozantinib (40 mg/day) plus atezolizumab occurred in 57% of patients with locally advanced or mUC who had received prior platinum-containing chemotherapy (n = 30) and in 47% of patients with NSCLC (n = 30).64,65 In the patients with NSCLC, 3% of patients had grade 5 treatment-related AEs of myocarditis and pneumonitis.
The most common treatment-related AEs (any grade) with cabozantinib and atezolizumab combination therapy were asthenia (37%), increased transaminases (23%), and mucosal inflammation (20%) in patients with UC, and fatigue (57%), nausea (48%), PPES (32%), and vomiting (32%) in patients with mCRPC.40,65 In the UC and mCRPC studies, respectively, treatment-related decreased appetite was reported in 23% and 45% of patients, and treatment-related diarrhea in 27% and 39% of patients.
Cabozantinib plus other immunotherapies
Two studies reported the safety profile of cabozantinib and durvalumab combination therapy: one study in patients with advanced UC and the second study in patients with advanced GEA, CRC, or HCC.47,58,67 Single studies were found investigating the safety of cabozantinib and pembrolizumab in patients with mRCC; cabozantinib plus panitumumab in patients with mCRC; and cabozantinib plus trastuzumab in patients with breast cancer and brain metastases (Table 3).32,52,53,69
In a phase I study of cabozantinib and pembrolizumab in patients with mRCC who had a median of one prior therapy, dose reductions were not required in the cabozantinib 40 mg dose cohort (n = 5), whereas dose reductions were needed for one patient in the 60 mg dose cohort (n = 3). Grade 3 AEs included reversible posterior leukoencephalopathy syndrome, hypertension, anorexia, and confusion (occurring in one patient each). 52
In the two studies (phase Ib and phase II studies) of cabozantinib plus durvalumab, diarrhea was one of the most common grade 1–2 treatment-related AEs, occurring in 31.3% of patients with advanced UC after platinum chemotherapy (n = 16; cabozantinib 40 mg/day) and 26% of patients with GEA, CRC, or HCC.58,67 Other common grade 1–2 treatment-related AEs with this combination therapy included fatigue (43.8%) and dysphonia (31.3%) in patients with UC, and fatigue (83%), abnormal liver function tests (39%), and anorexia (26%) in patients with GEA, CRC, or HCC. Three of the patients with GEA, CRC, or HCC developed grade 3 treatment-related hypertension, hyperthyroidism, and thrombocytopenia and a thromboembolic event (one patient each), all outside the dose-limiting toxicity (DLT) window.
In a phase Ib study of 25 patients with mCRC receiving cabozantinib (60 mg/day) and panitumumab, there were no grade 5 treatment-related AEs and one grade 4 treatment-related AE (hypertension).32,69 The most common grade 2–4 treatment-related AEs were acneiform rash (64%), fatigue (48%), and diarrhea (48%). Five patients (20%) discontinued treatment owing to toxicity.
Results from a phase II study of cabozantinib (60 mg/day) alone or in combination with trastuzumab in patients with breast cancer with brain metastases showed that the most common grade 3–4 AEs included elevations in lipase (11%), AST (8%), ALT (6%), hyponatremia (8%), and hypertension (6%). 53
Cabozantinib plus erlotinib
Safety data on the combination of cabozantinib and erlotinib were available from three studies: one phase II study in patients with pancreatic adenocarcinoma and one phase II RCT and one phase Ib study in patients with NSCLC (Table 3).63,71,72
In the phase II study evaluating cabozantinib (40 mg/day) and erlotinib in EGFR- and c-Met-expressing pancreatic adenocarcinoma following at least one chemotherapy regimen, the most common any-grade AEs attributable to cabozantinib and erlotinib included diarrhea (71%), AST increase (43%), fatigue (43%), nausea (43%), and rash (43%). 71
The RCT randomized patients to receive cabozantinib (60 mg/day) (n = 39) or cabozantinib (40 mg/day) plus erlotinib (n = 37) in a second- or third-line setting.62,63 The rate of grade 3 AEs was similar in the monotherapy and combination groups (70% and 72%, respectively). 63 The rate of planned or unplanned dose modification was also similar (95% in the monotherapy group and 97% in the combination group). Grade 3 or 4 hypertension, oral mucositis, and thromboembolic events were more common in the monotherapy group than in the combination group (25% versus 3%, 10% versus 3% and 8% versus 5%, respectively), whereas diarrhea was more common in the combination group (28%) than in the monotherapy group (8%). 63 Wakelee et al. conducted a phase Ib/II study of cabozantinib (40–100 mg) with or without erlotinib in patients with no prior VEGFR TKI therapy. Dose interruption or reduction occurred in 67% of patients receiving cabozantinib only and in 69% of patients receiving cabozantinib and erlotinib. In the combination group, the most common grade 3–4 AEs were diarrhea [n = 4 (31%)] and dehydration [n = 3 (23%)]. In the monotherapy group, the most common AEs were hypertension, hyponatremia, and fatigue [n = 2 (13%) each]. 72
Cabozantinib plus telaglenastat
Safety outcomes were reported in a phase I study of cabozantinib in combination with telaglenastat as second- or later-line therapy in patients with mRCC (n = 13). The grade 3 AEs that occurred were diarrhea, hypertension, decrease in platelet count, and hallucination (one event each). 59 In primary analysis from CANTATA, rates of AEs were similar between cabozantinib plus telaglenastat and cabozantinib plus placebo arms, with grade 3-4 AEs occurred in 71% and 79% of patients with mRCC, respectively. 80
Cabozantinib plus hormone therapy
One phase II study evaluated the safety of the combination of cabozantinib and androgen ablation in patients with hormone-naive metastatic prostate cancer, and a dose-finding study investigated the safety of cabozantinib and abiraterone combination therapy in patients with mCRPC.45,48,70
In the phase II study of cabozantinib (starting dosage: 60 mg/day) and androgen ablation in 62 patients with hormone-naive metastatic prostate cancer, the most common grade 3 AEs were hypertension (19%), diarrhea (6%), and thromboembolic events (6%), and dose reductions occurred in 85% of patients. 48
In the initial analysis of the phase I dose-finding study (n = 21) of cabozantinib (20, 40, or 60 mg/day) plus abiraterone in patients with progressive mCRPC (before or after chemotherapy), six patients had grade 3 AEs, which included diarrhea, anemia, and increased AST/ALT in the 20 mg cohort, and hypertension, low phosphate, and increased lipase in the 40 mg cohort. 70 In the final analysis of this phase I study including 27 patients treated with cabozantinib plus abiraterone, the most common treatment-related grade 3 AEs were infection and hypophosphatemia [n = 3 (11%) each]. Some grade 4 AEs occurred, but these were not treatment related. Dose reductions due to toxicity occurred in 3/12 patients receiving cabozantinib 20 mg, 8/12 patients receiving 40 mg, and 3/3 patients receiving 60 mg. 45
Cabozantinib plus chemotherapy
Three studies reported safety outcomes with cabozantinib in combination with three different chemotherapy drugs: a phase I/II study of cabozantinib plus docetaxel in patients with mCRPC; a phase I study of cabozantinib plus gemcitabine in patients with PDAC; and a phase I study of cabozantinib plus temozolomide in patients with high-grade gliomas (Table 3).31,68,74
Among patients with mCRPC who received cabozantinib 40 mg with docetaxel in a phase I/II study,31,41,51 DLTs were neutropenic fever and PPES, and there was one death due to a thromboembolic event. In addition, grade 3–4 myelosuppression, hypophosphatemia, and neuropathy were observed in at least three patients. 31
A phase I study in 12 patients with advanced PDAC who had received no more than one prior systemic treatment regimen showed that, following treatment with cabozantinib (escalating doses: 20–80 mg) and gemcitabine, 7/12 patients (64%) discontinued therapy owing to toxicity. The most common grade 3 AEs were neutropenia and AST and/or ALT elevation (5/11 patients each) and thrombocytopenia (2/11 patients); there were no grade 4 AEs. 74
In another phase I study, patients with newly diagnosed high-grade gliomas were treated with cabozantinib (40 or 60 mg/day), either concurrent with temozolomide followed by radiotherapy or following completion of radiotherapy and at least one cycle of temozolomide. Dose modifications occurred in 80.8% of patients, mainly owing to the occurrence of thrombocytopenia [n = 11 (42.3%)] and neutropenia [n = 5 (19.2%)]. 68
Ongoing studies
At the time of the searches, 67 studies of cabozantinib in a combination therapy were identified on ClinicalTrials.gov that are either active, recruiting or preparing for recruitment (Table 4). Tumor types studied include RCC (16), NSCLC (6), genitourinary (not RCC) (4), prostate (5), breast cancer (3), HCC (3), neuroendocrine tumors (3), solid tumors (3), muscle or soft tissue sarcoma (4), head and neck squamous cell cancer (2), melanoma (2), thyroid cancer (2), UC (2), gastrointestinal (2), gynecologic cancers (2), liver cancer (2), melanoma (2), and myeloma (1). Seven studies include various types of tumors. Among the ongoing studies are: the six phase III studies PDIGREE, COSMIC-313 in mRCC (cabozantinib plus nivolumab and ipilimumab versus nivolumab and ipilimumab), COSMIC-312 in aHCC (cabozantinib plus atezolizumab versus cabozantinib monotherapy versus sorafenib), CONTACT-01 in NSCLC, CONTACT-02 in mCRPC, and CONTACT-03 in aRCC; and the seven phase II studies ARCADIA and NICARAGUA including patients with advanced UC, the CANTATA study in mRCC, and CABATEN in patients with advanced and progressive neoplasms of the endocrine system, LOLA, NCT04079712, and NCT04197310 in patients with neuroendocrine tumors. An expansion of the COSMIC-021 phase I study (NCT03170960) is ongoing in multiple tumor types.
Ongoing studies of cabozantinib in combination with other therapies.

Registry was searched in November 2020. The status of all trials was then checked prior to final submission in February 2022; any further updates are detailed in the footnotes below.
Status updated to active, not recruiting.
Now completed.
Subsequently withdrawn.
Status updated to recruiting.
Status updated to suspended (for scheduled interim monitoring); ¶Status updated to terminated (owing to lack of efficacy).
Status updated to terminated (unexpected response to treatment in phase I).
CRPC, castration-resistant prostate cancer; NR, not recruiting.
At the time of final submission of the article, five of the studies identified in this search had completed. Published results from four of these studies are included in this review; data from the fifth study, the CANTATA study in mRCC, are reviewed in the discussion section. Four studies had been withdrawn or terminated since the searches were performed, leaving a total of 56 studies ongoing at the time of submission of this article (Table 4).
Discussion
This SLR was designed to capture the evidence of the efficacy and safety of cabozantinib in combination with other therapies for the treatment of solid tumors. To our knowledge, this is the first SLR to summarize the evidence of cabozantinib in combination, and it complements a separate manuscript in which we discuss studies of cabozantinib as monotherapy identified from the same SLR Moher et al. 81 Searching from 2012 onwards, 32 articles were identified that report findings for cabozantinib in combination with immunotherapy, chemotherapy and other targeted agents in patients with a range of cancers, including RCC, NSCLC, prostate cancer, breast cancer, gastrointestinal cancers, and UC and other genitourinary cancers. Overall, studies demonstrate promising findings for cabozantinib as part of a combination therapy.
The anti-angiogenic and immunosuppressive effects of cabozantinib, which lead to a potential synergistic effect with other therapies, especially immunotherapy, make cabozantinib an attractive candidate for combination therapy. 19 The immunosuppressive effects have been demonstrated in patient correlative studies of cabozantinib monotherapy in mUC 21 and RCC,25,26 and the synergistic potential of cabozantinib plus immunotherapy has been shown in preclinical mouse models of HCC. 23 This enhanced efficacy could be achieved in a range of disease types. Although our SLR shows that the evidence of cabozantinib in combination is relatively limited at present (32 articles published from 2012 until 2020), our supplementary searches indicate that there are many ongoing or planned studies in indications for which cabozantinib is approved as monotherapy, as well as in unapproved tumor types, and this is a research topic of much interest.
In the current review, we identified the studies of patients with RCC, HCC, UC, CRPC, CRC, pancreatic cancer, prostate cancer, GEA, NSCLC, breast cancer, endometrial cancer, and gliomas. Cabozantinib has been most studied in patients with RCC; there is strong evidence supporting the efficacy of cabozantinib monotherapy in RCC from the METEOR9,82–86 and CABOSUN10,87 trials. This review highlights that RCC is also well studied in terms of the use of cabozantinib as part of a combination therapy, including cabozantinib plus anti-PD1/anti-PD-L1 immunotherapies [pembrolizumab, atezolizumab, or nivolumab with or without ipilimumab (anti-CTLA-4 monoclonal antibody)39,52,61] and glutaminase inhibitors (telaglenastat). 59 The available evidence suggests promising results for all combinations, with an ORR of 25% for pembrolizumab; 52 ORR and DCR of 53% and 94%, respectively, in patients with ccRCC treated with atezolizumab;39,56 ORR and DCR of 54% and 100%, respectively, for cabozantinib plus nivolumab, or cabozantinib plus nivolumab and ipilimumab; 61 and ORR and DCR of 42% and 100%, respectively, for telaglenastat. 59
This review includes evidence identified by systemic bibliometric searches and subsequent searches for full publication of data presented in the retrieved congress abstracts. In addition, for the phase I study of cabozantinib plus nivolumab versus cabozantinib plus nivolumab and ipilimumab in patients with metastatic genitourinary cancers, a report of the final data for the expansion cohorts was presented at the genitourinary symposium 2021, which reported updated ORRs for the 33 patients with UC of 42.2% and for the 16 patients with RCC of 62.5% (efficacy results are not reported by therapy for the expansion cohorts). Overall, grade 3–4 treatment-related AEs occurred in 84% of patients treated with cabozantinib plus nivolumab versus 80% of patients treated with cabozantinib plus nivolumab and ipilimumab. 88
Several ongoing studies aim to investigate the cabozantinib combination therapy in RCC further. Initial results from the CANTATA study suggest that a combination of cabozantinib plus telaglenastat offers no PFS benefit compared with cabozantinib alone in patients with mRCC or aRCC who have received one to two prior anticancer systemic therapies. 89 An expansion stage of the COSMIC-021 study is underway to investigate cabozantinib plus atezolizumab in tumor types beyond RCC. This expansion includes 1732 patients in 24 cohorts and will include 12 tumor types: RCC, UC, NSCLC, CRPC, triple-negative breast cancer, ovarian cancer, endometrial cancer, HCC, gastric or gastroesophageal junction adenocarcinoma, colorectal adenocarcinoma, head and neck cancer and DTC. Interim efficacy and safety results are positive from patients with mCRPC (cohort 6) 40 and aRCC (cohort 10) 55 and NSCLC (cohort 7). 64 In addition, based on the positive outcomes from the original COSMIC-021 study, a phase III trial (NCT04338269), designed to evaluate the efficacy of cabozantinib plus atezolizumab versus cabozantinib in patients with aRCC after immunotherapy failure, is currently active. Also, several phase III RCTs (PDIGREE and COSMIC-313 trials) are investigating the doublet and triplet combinations of cabozantinib plus nivolumab, and cabozantinib plus nivolumab and ipilimumab further, following the positive findings from a phase I study. The phase I study by Apolo et al. was the first to report on these doublet and triplet combinations in patients with mRCC, carcinoma, and urothelial and rare tumors, such as bladder squamous cell carcinoma, urachal adenocarcinoma, and penile cancer;42,90 the study resulted in recommended phase II and expansion cohort doses of 40 mg (cabozantinib) and 3 mg/kg (nivolumab), with or without ipilimumab 1 mg/kg. The Alliance for Clinical Trials in Oncology 91 is supporting the PDIGREE trial, in which patients with mRCC will be treated in the first-line setting with nivolumab and ipilimumab for 3 months, then those with CR will continue nivolumab monotherapy, those with PD will be changed to cabozantinib monotherapy and those with PR or SD will be randomized to nivolumab monotherapy or cabozantinib plus nivolumab. 92 In COSMIC-313, cabozantinib plus nivolumab and ipilimumab will be compared with nivolumab and ipilimumab in patients with previously untreated mRCC with intermediate or poor risk, according to IMDC categorization. 93 COSMIC-313 will be the first RCT in aRCC, with nivolumab and ipilimumab as the standard of care comparator. CONTACT-03 is an ongoing phase III study of cabozantinib plus atezolizumab versus cabozantinib monotherapy in patients with RCC.
For UC, published findings show that cabozantinib plus nivolumab is clinically active and well tolerated in heavily pretreated patients with progressive mUC following ICB. 49 In another study, initial data show encouraging clinical activity with an acceptable safety profile in patients with advanced UC treated with cabozantinib in combination with durvalumab. 57 Following the phase I findings on cabozantinib plus nivolumab reported by Apolo et al., 90 a cooperative group study run by the Alliance for Clinical Trials in Oncology initiated enrollment in April 2019 for the phase II ICONIC study (NCT03866382) to investigate cabozantinib plus nivolumab and ipilimumab in rare genitourinary tumors. 94 In a trial currently in development, the same group will also investigate cabozantinib plus pembrolizumab versus pembrolizumab after first-line chemotherapy in patients with mUC. In terms of other combinations, the PemCab study (NCT03534804, estimated completion date September 2023) 95 is a non-randomized phase II study that will evaluate cabozantinib plus pembrolizumab in patients with mUC who are ineligible for cisplatin.
For endometrial cancer, a published randomized phase II trial, comparing the combination of cabozantinib and nivolumab versus nivolumab in recurrent endometrial cancer, has reported that cabozantinib plus nivolumab demonstrated improved PFS compared with nivolumab in heavily pretreated women with recurrent endometrial cancer. 54
For prostate cancer, two studies investigated the triple combination therapy of cabozantinib plus DP in patients with CRPC and found positive results.41,51 One of these was a randomized phase II study, which showed that cabozantinib plus DP was associated with longer PFS than DP in patients with CRPC. 41 In the expansion of the COSMIC-021 trial, which originally investigated cabozantinib and atezolizumab in RCC, preliminary data on the CRPC cohort have been encouraging, leading to the decision to expand this cohort further. 96 A phase III trial (CONTACT-02) is also currently recruiting to investigate cabozantinib plus atezolizumab in patients with CRPC. Cabozantinib plus androgen deprivation therapy has also demonstrated favorable results in patients with hormone-I metastatic prostate cancer; candidate prognostic and predictive markers of cabozantinib benefit were identified and have provided insights for rational therapy combinations. 48
At the time of the searches for the current review, two studies were identified that had presented findings in HCC.67,73 Results from the CheckMate 040 trial showed that cabozantinib plus nivolumab and ipilimumab led to higher investigator-assessed ORR than cabozantinib plus nivolumab alone (26% versus 17%) in patients with advanced liver cancer. Results also favored the triple combination arm for DCR and PFS. 73 HCC is also being studied in the randomized phase III COSMIC-312 trial. The primary endpoint of PFS in the first 372 patients randomized to receive cabozantinib plus atezolizumab or sorafenib was met: the combination therapy significantly improved PFS compared with sorafenib monotherapy (median PFS, 6.8 months versus 4.2 months; HR 0.63; 95% CI: 0.44–0.91; p = 0.0012). 97 However, in a prespecified interim analysis of the second primary endpoint, OS in all randomized patients, there was not a statistically significant benefit of cabozantinib and atezolizumab combination therapy versus sorafenib monotherapy (HR 0.90; 96% CI: 0.69–1.18; p = 0.438). Final OS analysis, announce in March 2022, reported neither improvement nor detriment in OS for cabozantinib in combination with atezolizumab versus sorafenib. 98
In pancreatic cancer, one phase I study evaluated the combination of cabozantinib with gemcitabine, which is a standard chemotherapy for advanced PDAC, and cabozantinib; 74 the authors did not recommend further exploration of this combination because of DLTs. 74 In CRC, one phase Ib study reported promising safety and efficacy findings for cabozantinib in combination with the EGFR inhibitor panitumumab.32,69
For NSCLC, there were mixed results for the combination of cabozantinib and erlotinib,63,72 but promising findings were reported for cabozantinib plus atezolizumab in patients with advanced NSCLC who had progressed after prior ICB in the expansion of the COSMIC-021 study. 64 A phase III trial (CONTACT-01) is currently recruiting to investigate cabozantinib plus atezolizumab versus docetaxel monotherapy in patients with CRPC.
For breast cancer, there was a lack of evidence for the benefits of cabozantinib combination therapy from the two available studies, one assessing combination with nivolumab in patients with metastatic triple-negative breast cancer and the other investigating combination with trastuzumab in heavily pretreated patients with breast cancer and brain metastases.43,44,53
Several ongoing studies aimed at evaluating the combination of cabozantinib with other therapies will increase evidence on the observed safety profiles of different combinations in various tumor types. In the current review, the most common AEs reported across all included studies were diarrhea, hypertension, fatigue, and lipase elevation, which are also commonly observed with cabozantinib monotherapy. AEs appeared to be manageable with dose reductions, although the range of patients requiring dose reductions while receiving cabozantinib 40–100 mg varied widely from 0% to 100%.
This SLR is a comprehensive review of cabozantinib as part of a combination therapy for the treatment of solid tumors. Inclusion criteria were broad, with no restriction on disease type and all phases of clinical trial included. Only nine published manuscripts were identified in the systematic searches, eight of which reported non-randomized phase I and II studies. Most of the studies identified in the searches were reported as congress abstracts only. For six of these studies, published full articles were identified later and the relevant data updated. Evidence from randomized trials was identified in five tumor types: aRCC, CRPC, NSCLC, aHCC, and endometrial cancer. Numerous planned and ongoing trials across a range of disease areas indicate that this area of research continues to be of interest. These include the aforementioned studies CONTACT-01, CONTACT-02, CONTACT-03, COSMIC-021, COSMIC-312/313, and PDIGREE. The published evidence in this review, together with emerging findings from ongoing trials, will strengthen our understanding of the potential benefits to patients using cabozantinib in combination.
In conclusion, research is increasing for the use of cabozantinib as part of a combination therapy to treat solid tumors. The current review identified evidence from phase I, II, and III trials, demonstrating promising response to treatment and manageable safety profiles. Further evidence from randomized phase III trials is expected in the coming years, across a range of disease areas. The ongoing studies will expand our understanding of the potential benefits of cabozantinib combination therapy in the near future.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221108691 – Supplemental material for Cabozantinib combination therapy for the treatment of solid tumors: a systematic review
Supplemental material, sj-docx-1-tam-10.1177_17588359221108691 for Cabozantinib combination therapy for the treatment of solid tumors: a systematic review by Daniel Castellano, Andrea B. Apolo, Camillo Porta, Jaume Capdevila, Santiago Viteri, Cristina Rodriguez-Antona, Lidia Martin and Pablo Maroto in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
