Abstract
Background:
The treatment landscape of metastatic renal cell carcinoma (mRCC) has substantially advanced over the last three decades, whereby data from controlled clinical trials indicate significant improvements regarding patients’ overall survival (OS) in highly selected patient cohorts. The aim of this study is to evaluate the impact of potentially game changing drugs on patients’ outcomes by comparing three different historical mRCC treatment eras.
Methods:
In all, 914 mRCC patients who were diagnosed between July 1985 and September 2020 were included into this observational study and assigned to three different treatment eras [‘cytokine’, ‘first-generation tyrosine kinase inhibitors (TKIs)’, and ‘modern TKIs/immunotherapy’] based on the EMA approval dates of sunitinib (July 2006) and nivolumab (June 2015) in mRCC treatment. OS was considered the primary study endpoint. Kaplan–Meier analyses, log-rank tests, and uni- and multivariable Cox regression models were performed.
Results:
OS was significantly longer in patients of the modern TKIs/immunotherapy era (median OS not reached) as compared to the cytokine (2.4 years) and first-generation TKIs era (1.7 years, all
Conclusion:
Significant advances in the systemic medical treatment of mRCC during the recent decade and the introduction of immunotherapy exerted a major impact on patient outcomes in terms of OS in a real-life population.
Introduction
Renal cell carcinoma (RCC) represents the most common cancer type with an origin in the kidney and currently accounts for 3.6% of all malignant tumors in the United States alone, with approximately 76,000 estimated new cases annually. 1 Etiologically, RCC is a highly heterogeneous disease and can be classified into various histological subtypes, the most frequent being clear cell RCC (80–90%), papillary RCC (10–15%), and chromophobe RCC (4–5%), although these subtypes can be further dissected depending on different histological variants and molecular features in future classifications.2,3 Despite significant advancements in diagnostic imaging methods, surgery, and systemic medical treatment modalities over the recent years, RCC patients prognosis remains poor, with up to 30% suffering from metastatic disease at initial diagnosis, and up to 30% of patients developing metastases during the course of the disease.4,5
Today, systemic medical treatment represents the leading therapeutic choice in metastatic RCC (mRCC), and the treatment landscape has changed substantially throughout the last three decades. Until the early 2000s, immune-modulatory therapies with cytokines, including interferons (IFNs) and interleukin-2 (IL-2), represented the only valid standard-of-care approach. 6 However, besides considerable and often treatment-limiting toxicities and side effects, the benefits in survival outcomes for most patients were limited to in general a few months. 7 Consequently, advances in understanding the underlying RCC pathophysiology and angiogenesis as a central mechanism allowed the specific development of antiangiogenic strategies, mainly driven by the so-called first-generation tyrosine kinase inhibitors (TKIs). 8 The prototype of this class of drugs, namely sunitinib, was first approved based on a significant improvement of progression-free survival (PFS) over a treatment with IFN-alpha (IFN-α) in 2006 (improved PFS from 5 to 11 months), thereby marking the beginning of the TKI-based treatment era in advanced/mRCC treatment. 9 Despite improving PFS, patients’ overall survival (OS) has not been significantly improved in this groundbreaking clinical trial by Motzer and colleagues, 10 which was probably caused by a high cross-over rate of more than 60% of patients from the IFN control-arm switching to the antiangiogenic drug. 10 Similarly, an improvement in PFS, but no difference in OS due to a substantial cross-over effect has been demonstrated for the second first-line standard-of-care drug at that time, namely pazopanib. 11 Recently, the introduction of immune checkpoint inhibitors (ICIs) in the treatment of mRCC, including the combination with second- and third-generation TKIs (‘modern’ substances as axitinib, cabozantinib, and lenvatinib) further revolutionized the therapeutic landscape and led to a significant OS benefit in second and first-line randomized clinical trials.12–17
As these data are retrieved from controlled phase III trials, we aimed to evaluate the impact of novel drugs on patients’ survival outcomes in the treatment of mRCC over the last decades in a real-life population by comparing three different historical treatment cohorts: the ‘cytokine era’, the ‘first-generation TKIs era’, and the ‘modern TKIs and immunotherapy era’.
Materials and methods
In this observational cohort study, 914 consecutive patients with histologically confirmed mRCC who were diagnosed with metastatic disease between July 1985 and September 2020, and who were treated at the Division of Oncology, Department of Internal Medicine, and/or at the Department of Urology of the Medical University of Graz in Austria, were included.
Clinicopathological data were collected from patients’ electronic medical records, as well as from paperback archives of both participating departments. Documented clinicopathological data included patients’ age, gender, histological RCC subtype, tumor grade, as well as presence or absence of histological tumor necrosis or sarcomatoid differentiation, and patients’ performance status. Assessed laboratory values included hemoglobin, calcium, neutrophil lymphocyte, and platelet counts to be able to calculate the International Metastatic RCC Database Consortium (IMDC) risk score for each individual patient. 18 Patients with no points were considered ‘good risk’, whereas patients with 1–2 and ⩾3 points were considered ‘intermediate’ or ‘poor risk’, respectively. Dates of death were accurately obtained from the Austrian Social Security System database. This study was approved by the local ethics committee (No. 32-573 ex 19/20) of the Medical University of Graz.
Statistical analyses
OS was considered the primary endpoint of our study and defined as the time (in years) from the date of diagnosis of metastatic disease to patients’ death of any cause. Patients were assigned to three different treatment eras according to the date of diagnosed metastatic disease and important hallmarks in the approval of new therapeutic approaches in the treatment of RCC. Patients who had metastatic disease and died before 27th July 2006, which corresponds with the approval date of Sunitinib in the treatment of mRCC by the European Medical Association (EMA), were considered as patients of the ‘cytokine era’. Patients who developed metastases or were still alive after that date and up until 26th June 2015, corresponding to the EMA approval of Nivolumab, were considered to represent the ‘first-line TKIs era’. Patients who were diagnosed with mRCC and patients that were still alive after 26th June 2015, were referred to as the ‘modern TKIs and immunotherapy era’.
All clinicopathological parameters in the three treatment era groups were compared using analysis of variance and chi-squared tests as appropriate. The Kaplan–Meier method was used to plot survival functions to the three groups and compared by log-rank tests. To account for missing data, multiple imputation using chained equations was used for all variables with missing data (histology, sarcomatoid differentiation, tumor necrosis, systemic treatment, IMDC risk groups, and tumor grade) with 40 (
Results
In our analysis, 914 mRCC patients were included, of which 311 (34%) patients were categorized as referring to the cytokine era, 311 (34%) patients to the first-generation TKIs era, and 292 (32%) were assigned to the modern TKIs/immunotherapy era. See Table 1 regarding the baseline characteristics of the three cohorts studied. Overall, 495 (54.2%) patients received no systemic anticancer therapy at all, most of which (
Descriptive characteristics (summary table) of the study population.
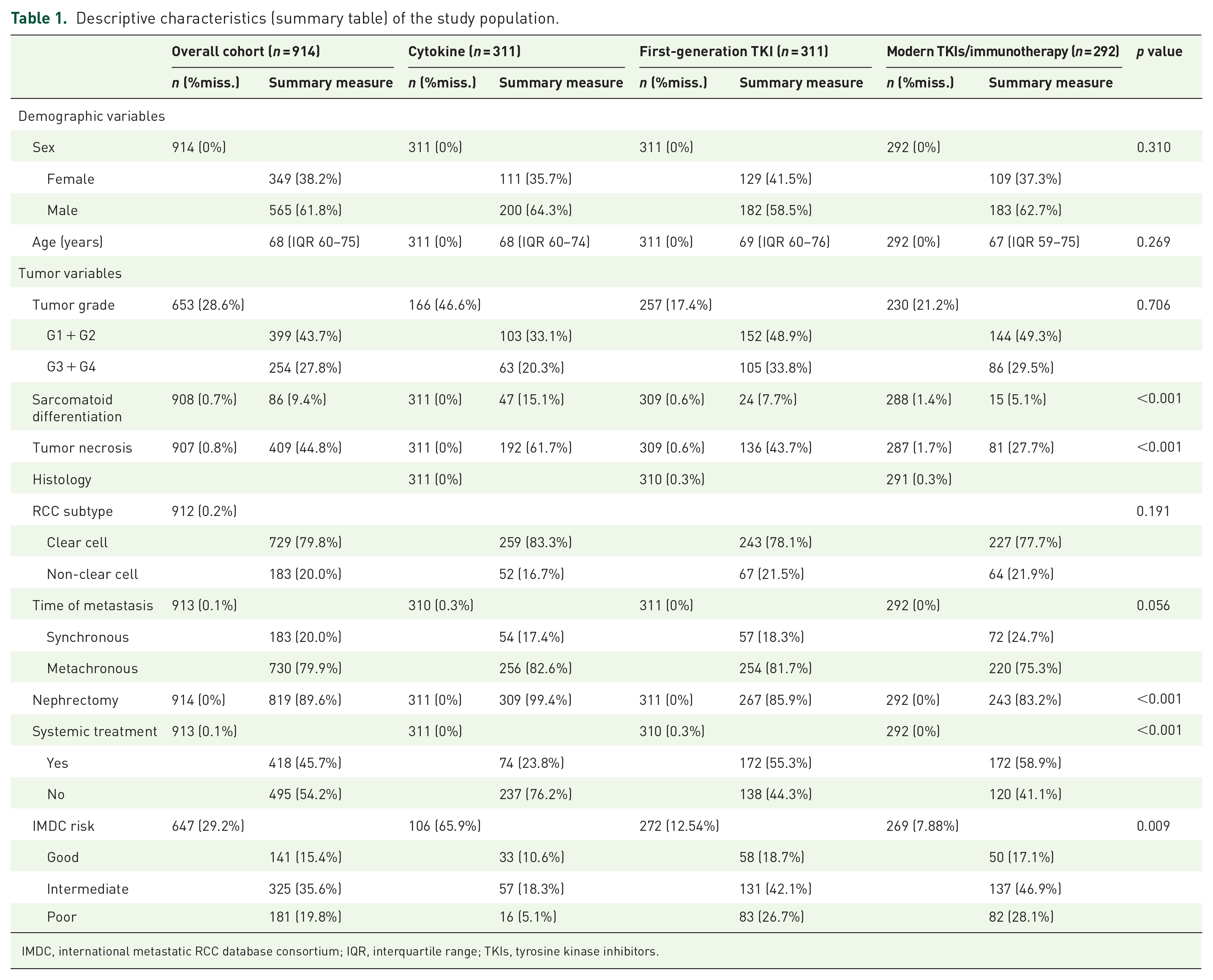
IMDC, international metastatic RCC database consortium; IQR, interquartile range; TKIs, tyrosine kinase inhibitors.
There were no statistically significant differences in the distributions of age, sex, histology, and tumor grade between the patient cohorts (
Survival outcomes in the overall patient cohort
Median OS in the overall study population (including patients who did not receive systemic medical treatment) was 2.6 (95% CI: 2.3–2.9) years. In the IFN era, median OS was 2.4 (95% CI: 1.9–2.8) years, whereas it was 1.7 (95% CI: 1.4–2.1) years in the first-generation TKIs era and was not reached in the modern TKIs/immunotherapy era. There were 151, 300, and 174 reported deaths in the cytokine, first-generation TKI, and TKIs/immunotherapy era, respectively. OS was significantly longer in patients of the modern TKIs/immunotherapy era than it was in the cytokine (log-rank

The Kaplan–Meier curves showing OS for cytokine, first-generation TKIs, and modern TKIs/immunotherapy era in the overall patient cohort (cytokine
In the univariable Cox proportional hazard analysis, including both patients with and without systemic tumor therapies, patients of the modern TKIs/immunotherapy era had a significantly better prognosis, while there was no difference in the first-generation TKIs era cohort. Multivariable cox analyses adjusted for age, nephrectomy, histologic subtype, sarcomatoid differentiation, histologic tumor necrosis, systemic
Uni- and multivariate Cox regression models regarding OS.

CI, confidence interval; HR, hazard ratio; IMDC, international metastatic RCC database consortium; TKIs, tyrosine kinase inhibitors. Statistically significant p-values are highlighted in bold.
Survival outcomes stratified by IMDC risk groups
The IMDC risk score was available in 647 patients, of which 141 (21.8%), 325 (50.2%), and 181 (28.0%) patients were classified as ‘good’, ‘intermediate’, and ‘poor’ risk according to their IMDC scores, respectively. Overall, median OS was 4.0 years in patients in the ‘good’ prognosis group, 2.3 years in the ‘intermediate’ prognosis group, and 1.5 years in the ‘poor’ risk group, which represented statistically significant differences at a
In the ‘good’ risk group, both patients in the modern TKIs/immunotherapy era had a significantly better OS than patients in the first-generation TKIs (log-rank

The Kaplan–Meier curves showing OS for cytokine, first-generation TKIs, and modern TKIs/immunotherapy era in IMDC good risk group patients (cytokine
As for patients with ‘intermediate’ risk RCC, the modern TKIs/immunotherapy era showed significantly better survival as indicated by the Kaplan–Meier curves than the first-generation TKIs (log-rank
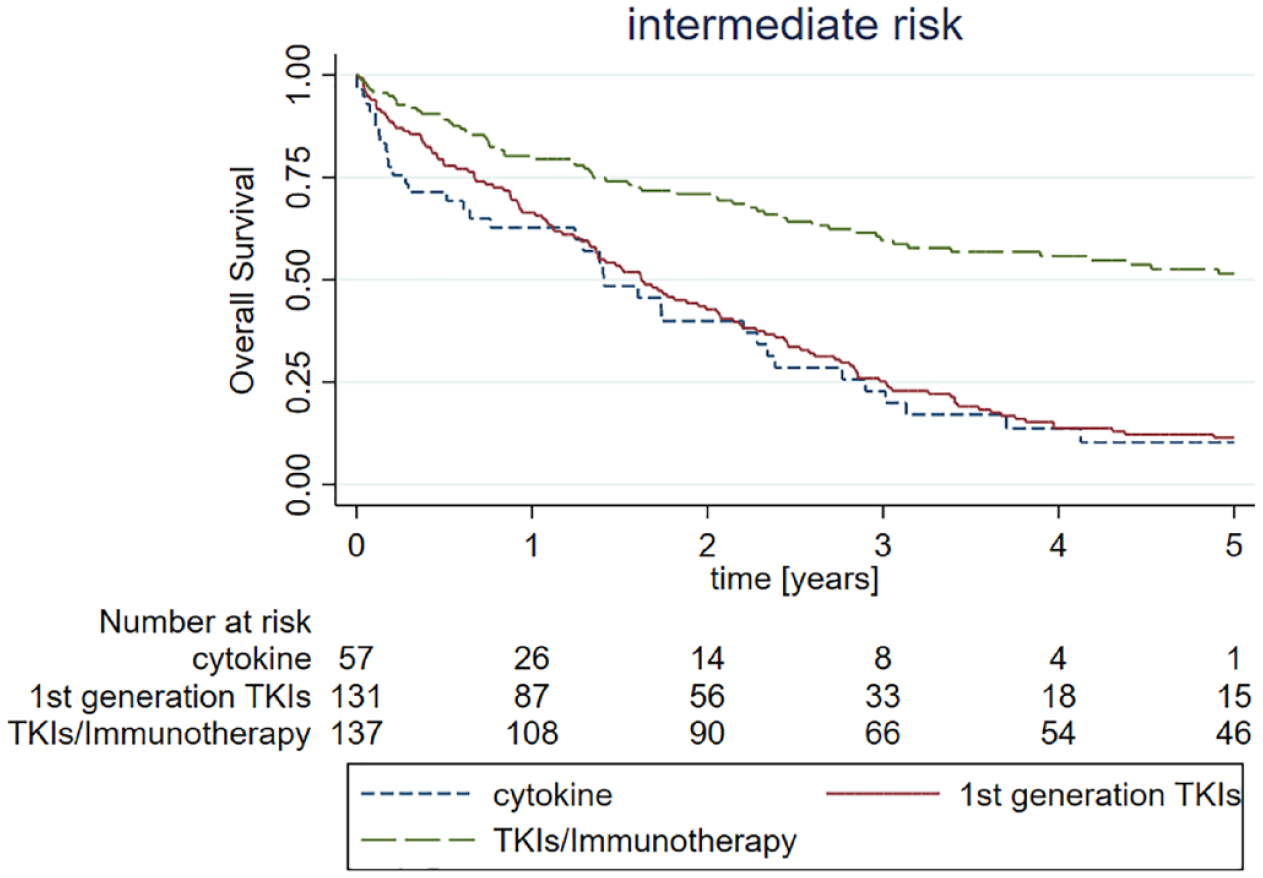
The Kaplan–Meier curves showing OS for cytokine, first-generation TKIs, and modern TKIs/immunotherapy era in IMDC intermediate risk group patients (cytokine
In patients defined as ‘poor’ risk according to their IMDC score, patients of the modern TKIs/immunotherapy era showed again a significantly better OS than patients of the first-generation TKIs era (log-rank

The Kaplan–Meier curves showing OS for cytokine, first-generation TKIs, and modern TKIs/immunotherapy era in IMDC poor risk group patients (cytokine
Discussion
The systemic medical treatment landscape of mRCC has dramatically changed over the past decades, whereby immunotherapy represented the latest breakthrough in first- and second-line settings within the last 5 years.
19
Whether real-life mRCC patient cohorts reflect the significant differences in OS found in controlled clinical trials is poorly understood and defined yet. In our observational study, we report the advances in survival outcomes in the treatment of mRCC over the last three decades in a large real-life population (
In our study cohort, OS has significantly improved during the last decades and was substantially longer in patients living in the modern TKIs/immunotherapy era cohort, as compared to the two other historical patient cohorts. Furthermore, the observed differences in survival were consistent across all three IMDC risk groups. Since data on survival benefit over different treatment eras are mostly based on controlled phase III trials, our study adds an important insight on the real-world impact of novel drugs in RCC treatment over a timespan of more than three decades. Considering the high treatment costs of novel treatment strategies such as ICIs, reporting outcomes in a real-world setting outside of highly selected phase III study cohorts is crucial to ultimately justify higher healthcare expenses. Furthermore, our analysis revealed IMDC risk groups, patient’s age, and tumor grade as significant predictors of survival.
As mentioned above, the mRCC treatment landscape has significantly changed since the 1990s, evolving to a broader spectrum of approved drugs with increased efficiency and fewer adverse side effects. In 1992, IL-2 was approved as the first systemic medical treatment option for mRCC patients.20,21 Although the use of cytokine immunotherapies, such as IL-2 or IFN-α revealed reasonable response rates in a certain amount of patients, it is out of use nowadays due to unfavorable outcomes and significant (and sometimes life threatening) toxicities in comparison to first-generation TKIs. 22 In 2006, the approval of sunitinib initiated the therapeutic era of first-generation TKIs. These mainly orally administrated multi-targeting drugs against vascular endothelial growth factor (VEGF) receptors 1, 2, and 3, c-Kit, and platelet-derived growth factor (PDGF) receptors, prevent the induction of hypoxia-inducible factor genes (e.g. VEGF, PDGF) as tumor progression determinants. 23 For almost one whole decade, first-generation TKIs have been the mainstay for the first- and higher treatment lines in advanced or mRCC.
Nonetheless, the introduction of the ‘programmed cell death 1’ (PD-1) inhibitor nivolumab in the second-line setting, as well as in combination with the ‘cytotoxic T-lymphocyte-associated Protein 4’ (CTLA4) inhibitor ipilimumab in the first-line treatment of intermediate and poor risk mRCC revolutionized the systemic medical treatment of mRCC. ICIs targeting immune checkpoint molecules, such as PD-1, PD-L1, or CTLA4, promote antitumor immune responses through effector T-cell proliferation and activation. 24 Since these findings, various ICIs and novel combinations with next-generation TKIs have expanded the possible treatment options in the palliative first-line setting of ‘good’, ‘intermediate’, and ‘poor’ risk RCC patients.12,16,17,25
Each different treatment era came along with a distinctive toxicity profile of the respective therapy used at that time. On the one hand, especially IFN treatment had significant side effects 22 ; thus, a large proportion of patients were considered unfit for systemic treatments and consequently treated with best-supportive care approaches from the initial diagnosis of metastatic disease. On the other hand, modern therapies such as TKIs and immunotherapies showed improved and manageable toxicity profiles; therefore, patients, who had been denied the chance of palliative systemic therapy in the cytokine era, might have been considered fit for various treatments today.26,27 To avoid a selection of patients with a more favorable prognosis only due to their PS in the cytokine era, we included both patients who did and did not receive systemic treatments across all three historical cohorts. However, when considering only patients who received treatment in a subgroup analysis, the obtained results were similar.
In our analysis, we chose to assign patients to different treatment eras based on historical drug approval landmarks in the RCC treatment landscape, following the EMA approval dates of the primary agent of a new treatment era. Comparing OS outcomes for the cytokine and first-generation TKIs era, we would have expected an improved OS for TKIs; nevertheless, our analysis did not show a significant difference between both cohorts. These data are in contrast to a Danish wide population-based study including a similar number of patients and demonstrated that targeted therapy resulted in significantly improved treatment rate and OS rate in mRCC patients. 28 This finding might be explained by our means of cutoff selection, as some patients may have received agents from more than one historical treatment era. Patients who received more potent second- or third-generation TKIs29,30 near the end of the TKI era, and survived until the approval of nivolumab in mRCC, crossed over into the modern TKIs/immunotherapy era, resulting in a potential underestimation of the outcomes of the first-generation TKIs era. Another explanation might be that more patients with even worse prognostic features were treated in the first-generation TKIs era due to toxicity profile and multiple treatment options.
Finally, some limitations of our study shall be discussed. First, due to the retrospective single-center study design, selection bias cannot be excluded entirely. Second, the number of treatment lines might also influence survival in different treatment areas, but were not systematically investigated in our study. Third, due to missing electronic data processing and inconsistent paper charts documentation during the 1990s and early 2000s, our study showed a considerable amount of missing data in the cytokine era cohort regarding the parameters defining the IMDC risk groups, as well as in tumor grading. However, multiple imputation models were implemented to account for missing data, to include tumor grading and IMDC risk scores in the multivariable Cox model. In a subgroup analysis stratified for IMDC risk groups, patients of the modern TKIs/immunotherapy era had consistently, and significantly better OS as compared to the first-generation TKIs and cytokine era.
Conclusion
The advances in various treatment modalities of mRCC during the recent decades and the introduction of modern TKIs and immunotherapy exerted a major impact on patient outcomes in a real-life population in Austria.
Supplemental Material
sj-tif-1-tam-10.1177_17588359221134065 – Supplemental material for Improved overall survival of metastatic renal cell carcinoma patients in the era of modern tyrosine kinase inhibitors and immune checkpoint inhibitors: results from a real-life, population-based Austrian study comprising three decades of follow-up
Supplemental material, sj-tif-1-tam-10.1177_17588359221134065 for Improved overall survival of metastatic renal cell carcinoma patients in the era of modern tyrosine kinase inhibitors and immune checkpoint inhibitors: results from a real-life, population-based Austrian study comprising three decades of follow-up by Hannah Fluhrer, Georg C. Hutterer, Sylvia Golbeck, Michael Stidl, Tobias Niedrist, Renate Pichler, Johannes Mischinger, Maximilian Seles, Sebastian Mannweiler, Jasmin Spiegelberg, Thomas Bauernhofer, Philipp J. Jost, Sascha Ahyai, Richard Zigeuner, Martin Pichler and Dominik A. Barth in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tif-2-tam-10.1177_17588359221134065 – Supplemental material for Improved overall survival of metastatic renal cell carcinoma patients in the era of modern tyrosine kinase inhibitors and immune checkpoint inhibitors: results from a real-life, population-based Austrian study comprising three decades of follow-up
Supplemental material, sj-tif-2-tam-10.1177_17588359221134065 for Improved overall survival of metastatic renal cell carcinoma patients in the era of modern tyrosine kinase inhibitors and immune checkpoint inhibitors: results from a real-life, population-based Austrian study comprising three decades of follow-up by Hannah Fluhrer, Georg C. Hutterer, Sylvia Golbeck, Michael Stidl, Tobias Niedrist, Renate Pichler, Johannes Mischinger, Maximilian Seles, Sebastian Mannweiler, Jasmin Spiegelberg, Thomas Bauernhofer, Philipp J. Jost, Sascha Ahyai, Richard Zigeuner, Martin Pichler and Dominik A. Barth in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
None of the contributing authors have any conflicts of interest, including specific financial interests and relationships and affiliations relevant to the subject matter or materials discussed in the manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
