Abstract
Colorectal cancer (CRC) is a major contributor to cancer-associated morbidity worldwide and over one-third of CRC is located in the rectum. Neoadjuvant chemoradiotherapy (nCRT) followed by surgical resection is commonly applied to treat locally advanced rectal cancer (LARC). In this review, we summarize current and novel concepts of neoadjuvant therapy for LARC such as total neoadjuvant therapy and describe how these developments impact treatment response. Moreover, as response to nCRT is highly divergent in rectal cancers, we discuss the role of potential predictive biomarkers. We review recent advances in biomarker discovery, from a clinical as well as a histopathological and molecular perspective. Furthermore, the role of emerging predictive biomarkers derived from the tumor environment such as immune cell composition and gut microbiome is presented. Finally, we describe how different tumor models such as patient-derived cancer organoids are used to identify novel predictive biomarkers for chemoradiotherapy (CRT) in rectal cancer.
Keywords
Introduction
Colorectal cancer (CRC) is the third most frequently diagnosed cancer entity worldwide and a leading cause of cancer-related mortality. 1 Rectal cancers account for over one-third of CRC and are frequently diagnosed at a locally advanced stage, which is commonly defined as Union for International Cancer Control (UICC) T3/T4 stage and node negative or positive disease.2,3 Neoadjuvant chemoradiotherapy (nCRT) followed by surgical resection is a standard treatment for locally advanced rectal cancer (LARC) that is located in the middle or lower rectum. 3 The benefits of nCRT comprise a better local disease control and a higher sphincter preservation rate in tumors of the lower rectum compared to postoperative chemoradiotherapy (CRT) in patients with LARC. 4 Pathologic complete response (pCR) is defined as the absence of viable tumor cells in the rectal wall and lymph nodes upon histological examination of the resected specimen.5,6 It is a frequently used surrogate endpoint to evaluate response to neoadjuvant treatment in LARC, as pCR correlates with a significant reduction of local recurrences and an improved overall survival (OS). 7 In contrast, clinical complete response (cCR) refers to the absence of residual tumors, ulcerations, or rectal wall irregularities on both clinical and radiological assessment. It is used as a surrogate marker for pCR in clinical trials that assess treatment response of LARC to nCRT.8,9 Observations from many clinical studies demonstrate that the response to nCRT is highly variable in LARC.6,10 While approximately 20–30% of patients with rectal cancers achieve either pCR or cCR with conventional nCRT, there is a significant proportion of tumors that do not respond to nCRT.11–13 Since nCRT can cause specific, treatment-associated toxicities,14,15 it is important to select the intensity of the neoadjuvant therapy based on the potential benefit for the patient. Therefore, identifying biomarkers that predict response to nCRT is an important clinical challenge in the management of LARC. In this review, we present current and novel approaches of neoadjuvant therapy for LARC and their impact on tumor response. Recent advances in the discovery of predictive biomarkers for nCRT of rectal cancer are outlined and critically discussed, including clinical, histopathological, and molecular markers. Furthermore, we describe how different rectal cancer models including cancer organoids can be used to identify novel predictive biomarkers for CRT response.
Current concepts of nCRT of rectal cancer
Based on the results of the landmark CAO/ARO/AIO-94 trial, the current standard neoadjuvant treatment of LARC is a conventionally fractionated radiation (usually a total dose of 50.4 Gy in 28 fractions of 1.8 Gy) with concurrent fluoropyrimidine-based chemotherapy.4,16,17 Six to eight weeks after nCRT, the tumor is surgically removed by total mesorectal excision (TME). Results of the randomized CAO/ARO/AIO-94 trial showed a superiority of nCRT over adjuvant CRT with respect to patient compliance, rate of local recurrence, toxicity, and sphincter preservation for cancers located in the lower rectum.4,18 However, OS rate and the occurrence of distant metastasis were not improved by nCRT. As concomitant chemotherapy, either infusional 5-fluorouracil or oral capecitabine is used, and both agents showed similar clinical outcomes.17,19,20
Another standard regimen of neoadjuvant therapy for LARC is a short-course preoperative radiotherapy (SCPRT) with a total of 25 Gy in five fractions of 5 Gy, followed by surgery within 10 days from the first radiation. 3 The Dutch TME trial showed a reduced rate of local recurrences by preoperative SCPRT compared with surgery alone. 21 Either CRT or SCPRT can be performed as neoadjuvant therapy for rectal cancer according to the European Society for Medical Oncology (ESMO) guidelines. 3 Two randomized trials showed no significant difference in local disease control or survival between these two approaches.22,23 However, in case of borderline resectable tumors, CRT rather than SCPRT is recommended due to superior oncological outcomes.3,24
An ongoing effort is to improve the response to nCRT by adding chemotherapeutic and targeted agents to the above-mentioned standard nCRT. Whether the addition of oxaliplatin is beneficial in LARC remains an open question. Both the CAO/ARO/AIO-04 trial as well as the ADORE trial demonstrated an improvement of disease-free survival (DFS) by adding oxaliplatin to fluorouracil-based regimen in patients with LARC.25,26 On the contrary, the FOWARC, PETACC-6, STAR-01, NSABP, and ACCORD-12 trials failed to demonstrate an improved local tumor response (pCR) or long-term survival (DFS or OS) when oxaliplatin was combined with 5-fluorouracil-based CRT.27–30 In a recent meta-analysis of seven randomized clinical trials including 5782 patients comparing oxaliplatin-based
Novel approaches in the neoadjuvant treatment of rectal cancer
Total neoadjuvant therapy
Many ongoing clinical studies are assessing different therapeutic strategies to improve the outcome of nCRT in LARC. One major field of research is total neoadjuvant therapy (TNT), which is defined as neoadjuvant radiotherapy or chemoradiotherapy with additional chemotherapy administered either before (as induction therapy) or after CRT (as consolidation therapy), 7 as illustrated in Figure 1. The original goal of TNT was to improve DFS in patient with LARC and such a benefit was in fact observed in most studies that compared TNT with standard nCRT (see Table 1). Interesting, results of these trials also demonstrate a considerably higher pCR rate in patient who receive TNT, despite differences in study designs.39–43 Therapy-associated toxicities were also more frequently observed in the TNT group but did not result in differences in therapy adherence, surgical management, or rate of postoperative complications.39,44 Furthermore, the CAO/ARO/AIO-12 and organ preservation of rectal adenocarcinoma (OPRA) study investigated different sequences of TNT, namely, induction chemotherapy followed by CRT or consolidation chemotherapy after CRT. In both trials, consolidation treatment was superior in terms of cCR or pCR rate allowing for organ preservation strategies which amounted to about 55–60% in the OPRA trail in the consolidation arm.45,46 In all, the mentioned TNT studies demonstrate that TNT (especially with consolidation chemotherapy) improves DFS rates and substantially increases cCR and pCR rates, allowing for organ preservation and watch-and-wait strategies, which is outlined in the following section, in patients with LARC.39,42

Schematic overview of regimen for standard neoadjuvant therapy and total neoadjuvant therapy for locally advanced rectal cancer (LARC). In conventional neoadjuvant chemoradiotherapy (CRT), patients with LARC are treated with radiation (usually 50.4 Gy in 28 fractions of 1.8 Gy) and concurrent infusional 5-fluorouracil or oral capecitabine, followed by total mesorectal excision (TME). In total neoadjuvant therapy, CRT is either preceded by induction chemotherapy (with, for instance, fluoropyrimidine- and oxaliplatin-based regimens) or short-course preoperative radiotherapy (SCPRT, 5×5 Gy), or followed by consolidation chemotherapy, prior to TME. Adjuvant chemotherapy is recommended by many national guidelines.
Clinical trials that investigate total neoadjuvant therapy in rectal cancer.

CAPOX, capecitabine/oxaliplatin; cCR, clinical complete response; CRT, chemoradiotherapy; CT, chemotherapy; DFS, disease-free survival; DrTF, disease-related treatment failure; FOLFIRINOX, leucovorin/fluorouracil/irinotecan/oxaliplatin; FOLFOX, leucovorin/fluorouracil/oxaliplatin;
Watch-and-wait strategy
The watch-and-wait strategy was initially introduced by Habr-Gama in 2004 and is increasingly gaining attention. In the original study by Habr-Gama, 265 patients with distal, resectable, rectal adenocarcinoma were enrolled and received nCRT.
50
Of those patients, 71 achieved cCR after nCRT and did not undergo subsequent surgery. Instead, they were closely surveilled. All other patients underwent surgery and 22 of those had a pCR upon pathological examination. Comparison of patients who achieved pCR with those who received close surveillance after cCR showed no significant differences in systemic recurrence, DFS and OS during a mean follow-up of 54.9 months. These results have challenged the standard concept of LARC therapy which required surgical resection of the primary tumor after nCRT and stimulated further studies. To date, accumulating data show that the watch-and-wait approach can lead to an excellent rectal preservation rate after neoadjuvant therapy for LARC.51–56 A large proportion of patients could avoid surgery and of those who experienced local tumor regrowth during follow-up examinations could undergo surgical or endoscopic salvage therapy in most cases. However, evidence in favor of watch-and-wait is primarily based on retrospective or registry data, while evidence from randomized trials is lacking. Furthermore, whether the watch-and-wait approach is noninferior regarding long-term survival compared with surgery is currently unclear. Renehan
Predictive biomarkers for response to nCRT in rectal cancer
Over the past decade, a large spectrum of markers was reported to predict response to nCRT, specifically long-course radiotherapy with concomitant fluoropyrimidines in LARC. These markers include clinical features such as the tumor stage according to the TNM classification, tumor size, and location within the rectum. In addition, a number of histopathological markers were identified, ranging from tumor-intrinsic features such as tumor budding, grade of differentiation to the altered levels of marker proteins. At the molecular level, markers including gene mutations, microRNAs (miRNAs), and epigenetic changes were found to be predictive. More recently, factors derived from the tumor environment such as the immune cell composition or the gut microbiome were reported to have predictive value. A summary of markers is presented in Figure 2. Selected biomarkers are described and critically discussed in the following section.
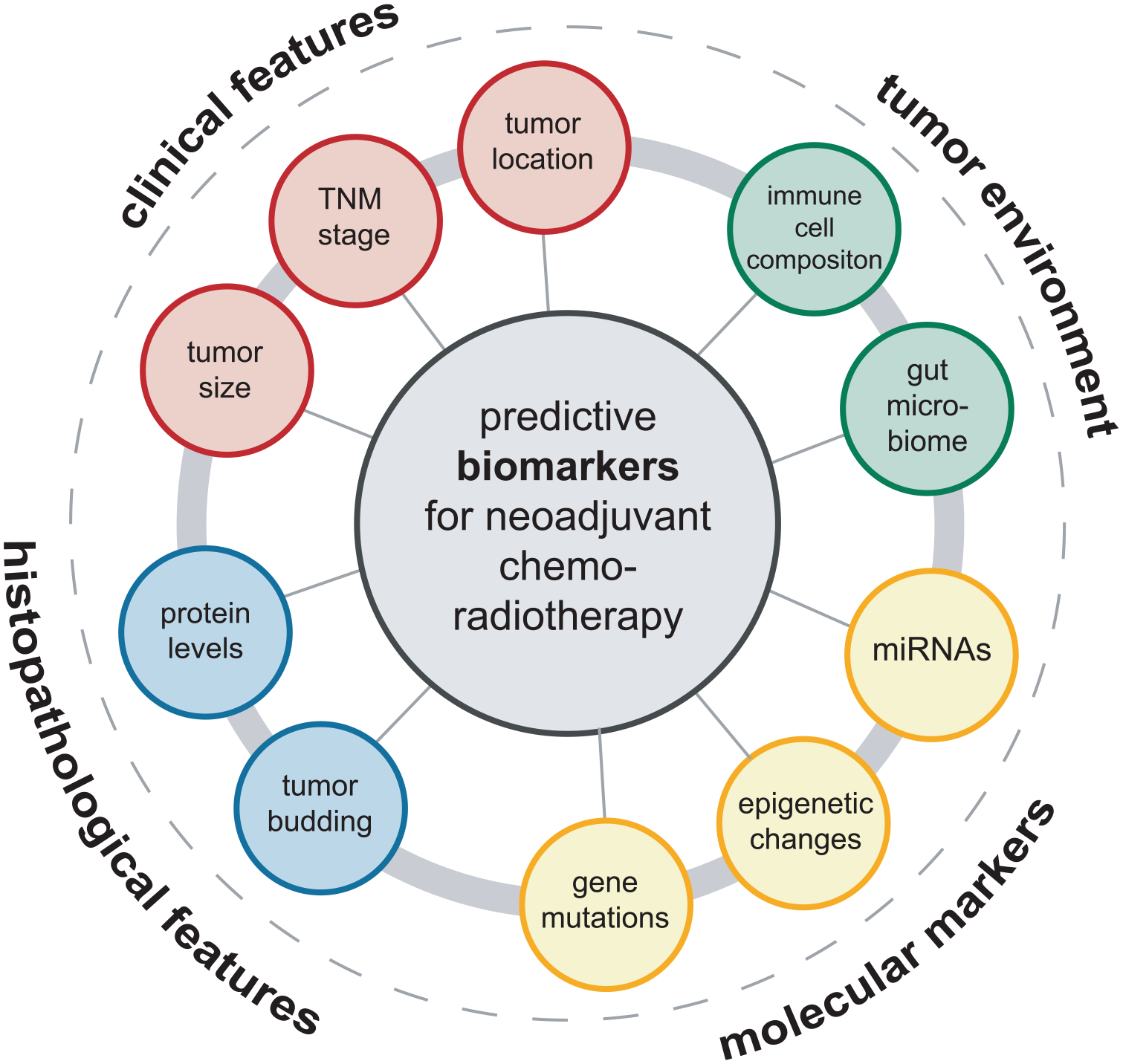
Overview of biomarkers that predict response to neoadjuvant chemoradiotherapy in locally advanced rectal cancer. A spectrum of clinical, histopathological, molecular, and tumor environment-derived factors can influence and predict the local response to neoadjuvant radiochemotherapy in locally advanced rectal cancer.
Clinical and histological predictors of response to neoadjuvant therapy
Clinical features
A number of clinical features were shown to predict treatment response to nCRT in LARC, including tumor size, clinical stage, and distance of the tumor from the anal verge (DTAV). Pretreatment tumor size was found to be associated with treatment response in LARC in several retrospective studies. In a cohort of 138 patients with LARC and locally unresectable T1–T2 tumors, Bitterman
Tumor stage, as determined by the TNM system, is a more comprehensive predictive marker. 62 For the T stage, several studies comprising large patient cohorts found a lower pCR rate in patients with cT4 LARC.63,64 Accordingly, patients with cT1-2 tumors were more likely to exhibit cCR/pCR after nCRT in another large patient series. 8 These observations are confirmed by data from the American College of Surgeons’ National Cancer database which comprised a total of 23,747 patients with LARC treated with nCRT. 65 Nodal status was also found to be predictive for tumor response as patients with cN2 stage LARC were found to have a significantly lower pCR rate after nCRT. 64 Accordingly, a series of studies demonstrated that clinical node-positivity at diagnosis (cN+) was significantly associated with a lower pCR or cCR rate.57,58
The association between DTAV and treatment response to nCRT remains controversial. In a retrospective study comprising 173 LARC patients, a DTAV <5 cm was significantly associated with favorable pathological response (defined as ypT0-1N0).
66
Similarly, a positive correlation of DTAV <3 cm with complete response was reported by other investigators.
57
However, these findings could not be confirmed by other studies,59,67 and even opposite results were described. For instance, Restivo
Histopathological markers
Histopathological features of rectal cancer have been widely investigated for their potential to predict response to nCRT. Mucinous rectal adenocarcinomas (defined by the presence of more than 50% mucin content in the tumor specimen) were associated with a poorer response to nCRT.64,70 Mucinous adenocarcinomas had more advanced tumor stages and a lower proliferative activity compared with nonmucinous tumors, possibly explaining the different response to nCRT.71,72 Tumor differentiation was also reported to be a predictive factor. Patients with well-differentiated tumors, as determined in pretreatment tumor biopsies, were found to more likely achieve complete response to nCRT.59,73,74 Another predictive marker is tumor budding, which is characterized by the presence of isolated single tumor cells or clusters of up to four tumor cells at the tumor invasive front. 75 Tumor budding was found in approximately 20% of pretreatment rectal cancer tissues and shown to be a significant predictor of poor response to nCRT in LARC as well as a negative prognostic factor. 76
In addition, the prognostic value of tumor invasion-related histopathological factors has been widely demonstrated in rectal cancer, including lymphovascular invasion (LVI) and perineural invasion (PNI).77–79 The presence of LVI and/or PNI after CRT was associated with worse survival in RC patients.80,81 Furthermore, the rate of distant recurrence was higher in RC patients after nCRT and TME if LVI and PNI were observed.82,83 However, there is considerably less data supporting a predictive role of LVI and PNI for treatment response. In a retrospective analysis, Agarwal
Protein-based markers
The correlation between protein levels of specific genes and response to nCRT in rectal cancer has been extensively investigated by immunohistochemistry analysis of single candidate proteins and by explorative, proteomic approaches. The identified protein biomarkers are involved in diverse biological processes, such as DNA repair,
87
oncogenic signaling,
88
or apoptosis.
89
A selection of predictive, tissue-based protein markers is summarized in Table 2. For instance, Yu
Selection of protein-based predictive markers for response to nCRT in rectal cancer.

ALDOB, aldolase B; CA9, carbonic anhydrase 9; CCR6, C-C motif chemokine receptor 6; COX2, cyclooxygenase 2; DUOX2, dual oxidase 2; FAK, focal adhesion kinase; FOX, forkhead box; LARC, locally advanced rectal cancer; MRP3, multidrug resistance–associated protein 3; nCRT, neoadjuvant chemoradiotherapy; pCR, pathological complete response; PLGF, placental growth factor; PLK1, polo-like kinase 1; RAD18, RAD18 E3 Ubiquitin Protein Ligase; SDF-1α, stromal cell-derived factor 1α; TCF-4, T-cell factor 4; TRG, tumor regression grade; VRK1: vaccinia related kinase 1; VRK2: vaccinia related kinase 2.
Tumor environment
Immune microenvironment
The tumor microenvironment in solid tumors is highly complex and consists of tumor, immune, and stroma cells that interact with the extracellular matrix.
107
The composition of the tumor microenvironment and the subtle interactions between its components determine cancer development and progression.
107
The immune cell component of the microenvironment can elicit both tumor-inhibiting and tumor-promoting effects: while cytotoxic T cells and natural killer cells can cause cytolysis of tumor cells, regulatory T cells and M2 macrophages are immunosuppressive and can support tumor survival.
108
The composition of immune cells in the tumor microenvironment of CRC was shown to have prognostic and predictive values.
109
To characterize the immune microenvironment in CRC, an immunoscore was developed based on cellular densities of CD3(+) and CD8(+) lymphocytes in the tumor center and at the invasive margin.110,111 Accumulating evidence suggests that alterations in immune cell composition in the tumor microenvironment might influence the response to nCRT. In LARC, high immunoscores in pretreatment tumor tissues are associated with a higher tumor downstaging rate (partial and complete response) after nCRT.
112
To further characterize the subtypes of tumor-infiltrating lymphocytes (TILs), several studies measured the densities of CD8+ and CD4+ TILs in rectal cancer samples before nCRT.113–117 These studies consistently found that high CD8+ TIL levels were associated with a favorable response to nCRT. Furthermore, many of those studies also observed high CD4+ TIL density to be a favorable predictive marker.113,115 Interestingly, the pretreatment CD8+ TIL count also showed a prognostic value, as it was associated with a superior 5-year-DFS and OS rate.117,118 Regulatory T cells mediate peripheral immune tolerance in the tumor microenvironment, thus playing an important role in suppressing antitumor immunity.
119
To investigate if regulatory T cells can impact response to nCRT, McCoy
Programmed cell death-ligand 1 (PD-L1) pathway mediates immune escape and is a potent target for anticancer immunotherapy.
124
Interestingly, multiple studies reported that PD-L1 expression and T-cell infiltration were increased after nCRT in LARC.117,125,126 Hecht
Cytokines and chemokines
Systemic cytokine and chemokine levels indicate inflammatory processes which play an important role in CRC progression. 128 Thus, serum levels of specific cytokines have been studied in the context of rectal cancer treatment. Interleukin-6 (IL-6) is a pro-inflammatory cytokine, and serum levels of IL-6 were determined in patients at different time points during nCRT. IL-6 levels tended to be lower in patients who achieved complete response. 129 In another study, levels of soluble CD40L, CCL-5, and a set of cytokines were analyzed during the course of nCRT. A decrease in soluble CD40L levels was associated with a favorable response, while higher post-CRT levels of IL-6 were associated with nonresponse. 130 However, in another large study that analyzed levels of a set of blood-based markers including IL-6 and IL-8 (interleukin-8), only IL-8 could predict response to nCRT. 131
Extracellular matrix
The composition of the extracellular matrix can vary in rectal cancers, and altered levels of specific components are associated with specific responses to nCRT in LARC. Jayne
Gut microbiome
Over the last decade, accumulating data from microbial metagenomics studies suggest a link between the composition of the gut microbiome and specific diseases, including cancer.135,136 The gut microbiome is not only linked to intestinal tumorigenesis
137
but also evolves during cancer therapy such as chemoradiation.138–140 Conversely, the gut microbiota may modulate response to cancer treatment by a diverse set of potential mechanisms, including direct enzymatic degradation and metabolism of drugs by specific bacterial species, alteration of bacterial diversity resulting in local tissue inflammation, and modulation of tumor immune response.141,142 The effect of the microbiome on response to radiotherapy is less extensively investigated, but studies in preclinical models support an underlying immunomodulatory effect of gut bacteria.140,143 Recently, clinical studies investigating the predictive value of the gut microbiome for response to nCRT of LARC have been reported. The largest prospective, longitudinal study to date compared fecal samples of 45 responders and 38 nonresponders with LARC before and after nCRT.
144
Molecular predictors of response to neoadjuvant therapy
Gene mutations
Activating mutations of
Mutations in
DNA methylation
DNA methylation is an epigenetic process that affects cytosines in CpG-rich promoters and thereby modulates transcriptional activity of genes.
157
Increased methylation of CpG island, also described as CIMP, is a prevalent biological feature of CRC.
158
CpG island methylator phenotype (CIMP) status has been assessed as a predictive biomarker for treatment response to nCRT in LARC, but the results vary depending on the method applied. Jo
MicroRNA
miRNAs are short noncoding RNAs, sized from 19 to 25 nucleotides, that posttranscriptionally regulate the expression of target genes and are functionally involved in many biological processes in rectal cancer.
163
Several differentially expressed miRNAs in rectal cancer were reported as biomarkers that can predict response to nCRT.164,165 For instance, expression level of miR-21, a well-characterized miRNA, was found to discriminate between complete responders and noncomplete responders in rectal cancer.166,167 However, while one study demonstrated that overexpression of miR-21-5p was associated with complete regression after nCRT,
166
another study found the opposite.
167
Similarly, Eriksen
Liquid biopsies
Liquid biopsy is a powerful diagnostic tool to monitor changes in tumor genetics. In comparison with tissue biopsies, liquid biopsy is noninvasive and can be collected serially, thereby facilitating a real-time assessment of the mutational landscape.
173
Circulating tumor DNA (ctDNA) has been extensively studied as a diagnostic tool in patients with LARC, but the data regarding its predictive value remain controversial.
174
Several studies investigated ctDNA levels at three time points: pretreatment/baseline, post-CRT, and postsurgery.175,176 Murahashi
Tumor models to investigate response to radiochemotherapy in rectal cancer
The identification and subsequent functional characterization of predictive biomarkers in rectal cancer requires modeling of the disease and the therapy response

Tumor model systems to study treatment response in rectal cancer. Cancer cell lines, tumor organoids, and patient-derived mouse xenograft models can be used to study treatment response. Advantages and disadvantages of each model system are described.
Cancer cell lines are the most commonly used model of CRC, as they are easy to cultivate and amenable to most genetic manipulations. Hence, they have been extensively used to study the impact of candidate genes and cellular processes on radiosensitivity. CRC cell lines were used to decipher the role of oncogenic signaling in response to CRT, including Wnt, RAS, and PI3K signaling.182–184 Furthermore, the role of many novel genes that modulate radiosensitivity were identified and characterized in CRC cell lines, including COASY,
184
XPO1,
185
CRBP1,
186
or inducible nitric oxide synthase,
187
among others. While providing interesting insights into the mechanistic function of these genes for radiosensitivity, a major drawback is that these potential biomarkers have not been confirmed in independent clinical cohorts. The impact of different miRNAs on radiosensitivity in rectal cancer was also elucidated in CRC cell line models, thereby identifying transcriptional targets that mediate the phenotypic effects of the miRNAs.188,189 Finally, CRC cell lines were used for drug screens to identify novel radiosensitizing compounds.
190
One limitation of most cell line models is that they originate from cancer tissues of the colon, and only a very limited number of rectal cancer cell lines are used in experimental studies so far. Furthermore, primarily rectal cancer is genetically heterogeneous and different single cell clones from the same tumor can respond differently to radiotherapy due to their genetic diversity and differential activation of oncogenic pathways.
191
Thus, while cell lines are a suitable model to characterize the function of single genes, recapitulating the
One strategy to overcome this challenge is the development of patient-derived tumor xenografts (PDX). In this approach, cells from tumor tissues are implanted subcutaneously or orthotopically into immunodeficient rodent lines such as athymic nude mice. A multitude of evidence demonstrates that PDX preserves the histological architecture and genetic characteristics of the original tumors. 192 Hence, several studies have been performed with colon cancer PDX models to assess drug response and drug resistance, as well as to discover new therapeutic targets and predictive biomarkers.193,194 However, only one study used PDX models of primary rectal cancer tissue to investigate the response to 5-FU-based CRT. This study demonstrated that PDX models could reproduce the heterogeneous response of primary tumors to nCRT. 195 Despite this finding, PDX models have several limitations. For instance, the impact of the immune system on therapy response is not adequately modeled in immunocompromised host animals. In addition, experiments with PDX are time-consuming and resource-intensive, limiting their use for large-scale screening experiments.
A novel approach to modeling CRC is the use of patient-derived organoids (PDOs). To establish PDO lines, cancer stem cells are isolated from tumor biopsies, seeded in base matrix, and cultured with defined culture medium containing specific growth factors.196–198 Under these conditions, an outgrowth of a self-organizing, three-dimensional mini-organ, termed organoid, can be achieved
Conclusion
Over the last decade, significant progress has been made in the treatment of LARC by the introduction and refinement of nCRT. 38 Accumulating evidence from prospective clinical trials demonstrates that a fraction of patients can achieve pCR and therefore might not require tumor resection. 5 Therefore, current efforts aim to improve the treatment response by adding an intensified chemotherapy regimen to nCRT (‘total neoadjuvant therapy’). Results from advanced phase clinical trials indicate that TNT can further increase the fraction of patients with clinical or pathological complete response and improve DFS. However, intensifying neoadjuvant therapy is inevitably associated with increased risk for and severity of adverse effects. Due to inter- and intratumoral heterogeneity, some tumors will not respond completely to the intensified neoadjuvant treatment. 204 Therefore, predictive biomarkers are needed to optimize patient selection for intensified neoadjuvant therapy regimens. In this review, we provide a broad overview of biomarkers that predict response to nCRT in LARC. While the predictive value of some markers, such as the tumor stage, has been well characterized, others including the tumor immune microenvironment and the gut microbiome are yet emerging. However, only few of these predictive markers are used in the clinical routine, as their potential utility is limited for several reasons. First, biomarker studies in the field of rectal cancer are conceptually very heterogeneous, with large differences in the selected therapy regimen, radiation dose, inclusion of clinical confounders, and grading system of tumor response. This heterogeneity limits the comparability and robustness of the identified biomarkers. Second, for the majority of predictive markers, validation by independent cohorts is lacking or produced conflicting results. Currently, the predictive value of only a few novel biomarkers could be consistently confirmed, including TILs or specific combination of oncogenic mutations. Third, it is questionable if a single biomarker will be sufficient for response prediction or a combination of several markers is needed to achieve substantial predictive power. To address these shortcomings, future prospective clinical trials that evaluate novel nCRT regimens should be complemented by biomarker discovery programs. These should include the collection of tumor tissues, blood, and stool samples, enabling a comprehensive multi-omics analysis. In this context, an important question is how to rationally select candidate markers for clinical validation. For instance, it is unclear whether biomarkers that were identified in patients who received fluoropyrimidine-based nCRT retain their predictive power when TNT is used instead. Therefore, it is necessary to understand the molecular mechanisms underlying the radiosensitizing effects of individual compounds and to define biomarkers based on this knowledge. This translational approach will be facilitated by the recent development of PDO models which can recapitulate the clinical response of individual patients to nCRT.201–203 Future studies with large panels of organoids that combine response assessment to nCRT and comprehensive molecular characterization will reveal novel molecular modulators of response. In summary, predictive biomarkers will become an important part in the management of LARC. The most important challenge ahead is to validate biomarkers discovered in preclinical or translational studies in well-designed, prospective clinical trials, and to integrate these multidimensional markers in clinically useful scores.
Footnotes
Acknowledgements
We would like to thank Leonhard Bamberg for helpful comments on the manuscript.
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: T.Z. and M.L. were supported by the Clinician Scientist program ‘Interfaces and Interventions in Chronic Complex Conditions’ funded by the DFG (EB 187/8-1). Q.X. was supported by a fellowship of the Chinese Scholarship Council (CSC). M.P.E. was supported by the DFG (GRK2727) and a grant provided by the MERCK Heidelberg Innovation Call (Darmstadt, Germany). N.V. was supported by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation), SFB 1324, project number 331351713.
