Abstract
Aims:
This study aims to evaluate the safety and efficacy of a new neoadjuvant regimen (FOLFOX4 plus hypofractionated tomotherapy) in patients with locally advanced rectal cancer.
Methods:
Patients with stage II–III rectal cancer were treated with the pre-operative chemoradiotherapy regimen comprising FOLFOX4 (two cycles), TomoTherapy (25 Gy in five consecutive fractions, one fraction per day in 5 days on the clinical target volume at the isodose of 95% of the total dose), FOLFOX4 (two cycles), followed by surgery with total mesorectal excision and adjuvant chemotherapy with FOLFOX4 (eight cycles). The primary endpoint was pathological complete response (pCR).
Results:
Fifty-two patients were enrolled and 50 patients were evaluable. A total of 46 (92%) patients completed chemoradiotherapy according to the study protocol and 49 patients underwent surgery. Overall, 12 patients achieved a pCR (24.5%, 95% CI 12.5–36.5). The most common grade 3 or more adverse events were neutropenia and alteration of the alvus. Adverse reactions due to radiotherapy, mainly grade 1–2 dermatitis, tenesmus, urinary dysfunction and pain, were tolerable and fully reversible. The most important surgical complications included infection, anastomotic leakage and fistula, all resolved with conservative treatment.
Conclusion:
FOLFOX and hypofractionated TomoTherapy is effective and safe in patients with locally advanced rectal cancer. Long-term efficacy needs to be further evaluated.
Trial registration
ClinicalTrials.gov identifier: NCT02000050 (registration date: 26 November 2013) https://clinicaltrials.gov/ct2/show/NCT02000050
Introduction
Neoadjuvant concurrent fluoropyrimidine-based chemoradiotherapy (CRT) followed by total mesorectal excision (TME) is considered as the standard treatment for patients with locally advanced rectal cancer (LARC), that is, stage II and III operable disease. 1 With improvements in surgical techniques and in radiotherapy (RT) delivery, the local recurrence rate has dramatically decreased from 25% to around 5–10%. 2 Indeed, the main objective of RT is to reduce the risk of locoregional relapse. These results are achieved both with concomitant CRT (with conventional fractionation) followed by delayed surgery, and hypofractionated (“short-course”) RT (SCRT) followed by early or delayed surgery.3–5 However, distant metastases remain the leading cause of death in patients with rectal cancer, with a 5-year distant relapse rate of about 30%. 2 Research efforts have thus concentrated on improving the efficacy of systemic treatment to reduce the risk of micrometastatic disease and achieve better control of systemic disease. 6 In this context, adjuvant therapy has shown two substantial limitations, that is, a paucity of data, which are largely extrapolated from colon cancer trials or from patients who did not undergo pre-operative CRT, and poor patient compliance, with only 43–74% of patients receiving all planned cycles due to treatment-related toxicity, post-operative morbidity, disease progression, and patient refusal.2,6 This suggests that researchers should focus their attention on the neoadjuvant rather than the post-operative setting.
Although promising neoadjuvant strategies combining fluoropyrimidine-based CRT with other agents, such as platinum derivatives,
7
irinotecan
8
and molecular-targeted agents,9–11 as well as the use of induction chemotherapy, have resulted in exceptional pathological complete response (pCR) rates, they have failed to improve survival in randomized trials. In particular, the use of platinum derivatives was recently investigated in a meta-analysis of 10 randomized controlled trials on a total of 5599 patients.
7
While the combined treatment did not improve overall survival (OS) [hazard ratio (HR) = 0.93, 95% confidence interval (CI) 0.82–1.05,
In recent years a growing trend has emerged toward a “total neoadjuvant therapy” (TNT) approach in LARC in which CRT and chemotherapy are administered prior to surgery to facilitate the full-dose delivery of planned systemic therapy, increase the rate of downstaging and pCR, and pave the way for nonoperative treatment strategies aimed at organ preservation. In this context, neoadjuvant chemotherapy followed by SCRT, 13 and SCRT followed by chemotherapy are currently being investigated.14,15
Under these premises, we evaluated the safety and efficacy of FOLFOX as both induction and consolidation treatment in combination with SCRT in patients with LARC.
Patients and methods
Patient eligibility
Inclusion criteria were: histologically or cytologically confirmed diagnosis of adenocarcinoma of the mid-low rectum (within 12 cm of the anal verge); stage: lowT2N0M0, T2N+M0, T3–4N–/+M0 (N+ = ⩾3 nodes >0.5 cm diameter or ⩾1 node >1 cm diameter); age ⩾18 and ⩽80 years; Eastern Cooperative Oncology Group performance status (ECOG PS) 0–1; normal organ and bone marrow function (leukocytes ⩾3000/µL, absolute neutrophil count ⩾1500/µL, platelets ⩾100,000/µL, total bilirubin ⩽1.5× upper limit of normality (ULN), AST (SGOT)/ALT (SGPT) ⩽2.5× ULN, creatinine ⩽1.5× ULN); written informed consent for participation in the study.
Exclusion criteria were: any chemotherapy or radiotherapy prior to entering the study; acute or sub-acute gastrointestinal occlusion; stage IV disease; participation in another clinical trial with any investigational agent within 30 days prior to study screening; other known malignant neoplastic diseases in the patient’s medical history with a disease-free interval of less than 5 years (except for previously treated basal cell carcinoma of the skin, superficial bladder tumor and
Procedures and treatment plan
Baseline staging included colonoscopy, contrast-enhanced computed tomography (CT) of the chest and abdomen, and contrast-enhanced pelvic magnetic resonance imaging (MRI). Endorectal ultrasound with rigid endoscope was mandatory for patients with a contraindication for MRI.
Patients were treated as follows: two cycles of induction chemotherapy with FOLFOX4 regimen (weeks 1 and 3) were followed by RT (week 5) and two cycles of consolidation FOLFOX4 (weeks 7 and 9). Restaging was scheduled at week 11 and surgery between weeks 12 and 16. After surgery, patients received adjuvant therapy with FOLFOX4 for eight cycles; the maximum interval between surgery and start of adjuvant therapy was 8 weeks (Figure 1).
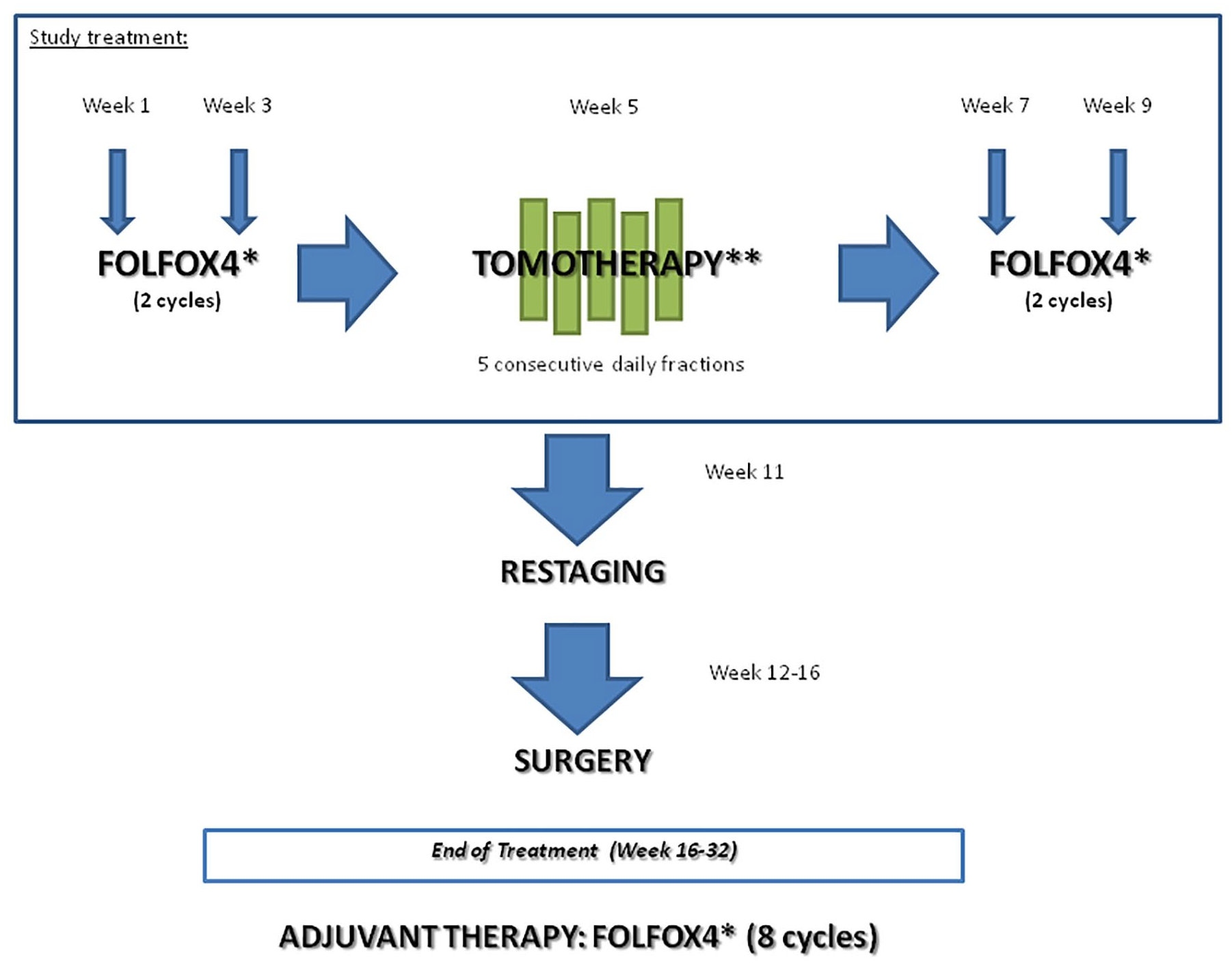
Study treatment. *FOLFOX4: oxaliplatin 85 mg/m² IV: day 1, levofolinate 100 mg/m² IV: day 1–2, 5-fluorouracil 400 mg/m² IV in bolus and 600 mg/m² IV infusion over 22 h: day 1–2; every 2 weeks. **25 Gy in five consecutive fractions, one fraction per day in 5 days, using helical Tomo Therapy or linear accelerator (LINAC) with an intensity modulated RT (IMRT) or a volumetric modulated arc therapy (VMAT) technique.
Induction, consolidation and adjuvant FOLFOX4 consisted of intravenous infusion
All patients received pelvic RT. Subjects were immobilized with a belly board device in a prone position for CT simulation and treatment. A full bladder at the time of planning and daily treatment were required. The gross tumor volume encompassing all visible primary tumors and the clinical target volume (CTV), including at least 3 cm of healthy rectum above the tumor, the entire mesorectum and obturator, presacral, and internal iliac lymph nodes (plus external iliac lymph nodes in cT4 patients and patients with positive obturator lymph nodes), was contoured. The identified organs at risk (OARs) were: bilateral femoral head, bladder and peritoneal cavity. Optional OARs were: penile bulb, prostate and uterus. The planning target volume (PTV) was generated with a 3 mm isotropic expansion around the CTV.
RT treatment was delivered using helical TomoTherapy or by linear accelerator (LINAC) with an intensity modulated RT (IMRT) or volumetric modulated arc therapy (VMAT) technique. Daily image-guided RT (IGRT) using cone beam CT (kV or MV) was systematically applied. The protocol provided the prescription to the PTV of 25 Gy in five consecutive fractions, one fraction per day in 5 days.
Restaging at week 11 included the same procedures used for baseline staging. Surgery was performed according to the principles of TME. Patients who achieved a clinical complete response were recommended to receive TME because of the high risk of recurrence, while local excision and “wait and see” approaches were not recommended in this study.
Post-surgical morbidity was assessed 30–60 days after surgery, before starting adjuvant chemotherapy. Patients were followed up to assess disease status at 6 months and 12 months following surgery, with a CT scan mandated at 12 months. Investigations and follow-up beyond 12 months were done as per institutional standards.
Statistical considerations
This was an open-label, single-arm, multicenter, phase II study to evaluate the safety and the proportion of pCR in patients with LARC treated with an innovative CRT scheme. The study consisted of two steps: Step A to identify the safety of the CRT regimen after two cycles of FOLFOX4, and Step B to identify the pCR rate at the end of the neoadjuvant treatment. Secondary endpoints were pathological downstaging, the rate of R0 resection, the sphincter-saving resection rate, median progression-free survival (PFS) and OS.
The sample size for Step A was calculated assuming that the probability of registering a toxicity involving CRT discontinuation with the new treatment was less than 45%. Six patients were to be evaluated for toxicity: if ⩽1 case of toxicity involving CRT discontinuation was observed, the treatment could be considered safe with a probability >90%; if two or more cases of toxicity involving RT discontinuation were observed, the study would be stopped and another kind of CRT schedule would be designed. If the CRT treatment was considered safe, the study would continue to Step B and the patients enrolled in the first step would be also evaluated in this second step.
The sample size of Step B was calculated considering the hypothesis of an increase in the proportion of pCR of at least 15% with the new treatment. Considering P0 as the expected proportion of pCR patients and P1 as the proportion of pCR patients with the new CRT treatment, 50 patients were considered a sufficient number to show an increase in the proportion of pCR patients from P0 = 10% to P1 = 25% (alpha = 0.1, one-side test, and power of 90%). The treatment could be considered active if at least seven out of the 50 patients enrolled achieved a pCR.
Efficacy and toxicity analyses were performed on all patients with a baseline assessment of disease who received two cycles of treatment of FOLFOX4 and at least 1 day of RT. Toxicity was assessed as per the National Cancer Institute’s Common Terminology Criteria for Adverse Events (CTCAE version 4.0) at the end of each chemotherapy cycle and at the end of SCRT. The pCR was defined as no residual cancer cells found at pathological examination after radical surgery, both at the primary tumor site and at regional lymph nodes. The system used for tumor regression grade assessment was that recommended by the American Joint Committee on Cancer
The study was performed in accordance with the principles of Good Clinical Practice and the ethical standards laid down in the 1964 Declaration of Helsinki. The protocol was approved by the local Ethics Committee (Comitato Etico Area Vasta Romagna n. I5/424 on 15 September 2010) and written informed consent was obtained from each patient. This trial is registered on ClinicalTrials.gov NCT02000050 (Eudract number 2013-000770-30).
Results
Patient characteristics
From October 2013 to May 2017, 52 patients with stage II–III adenocarcinoma of the mid-low rectum were prospectively enrolled onto the phase II trial in two Italian centers. Two patients were considered unevaluable: one patient was excluded due to eligibility criteria violation (stage IV disease) and one received only one cycle of chemotherapy and no RT. The characteristics of the remaining 50 evaluable patients are shown in Table 1.
Baseline patient demographics and clinical characteristics.
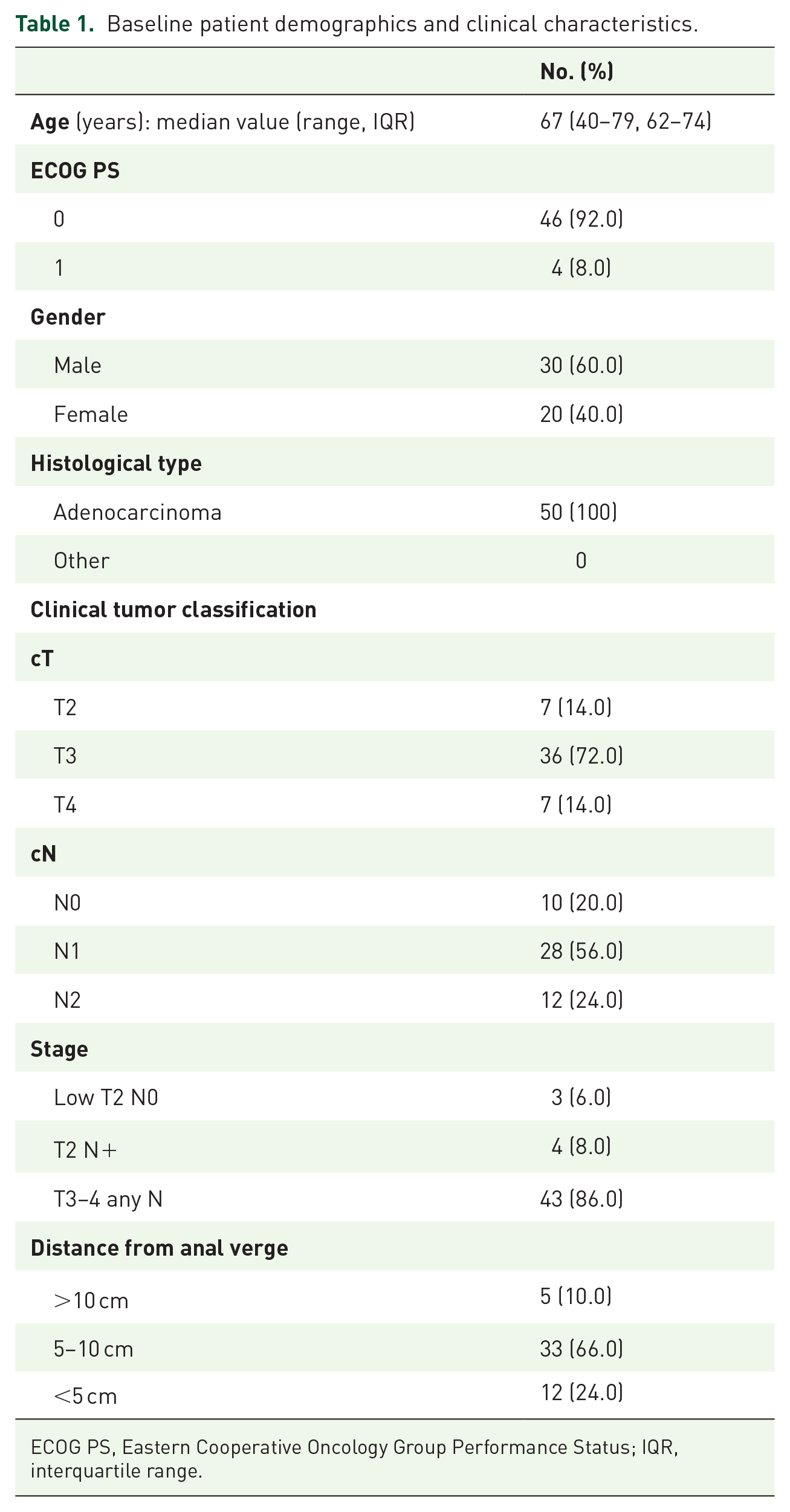
ECOG PS, Eastern Cooperative Oncology Group Performance Status; IQR, interquartile range.
Median age at the time of diagnosis was 67 years (range 40–79); 40% of patients were female, and ECOG PS was zero in 46 patients (92%). There were 10 (20%) patients with stage II disease and 40 (80%) with stage III disease. Seven (14%) patients had clinical T4 and 12 (24%) clinical N2 tumors. Distance from anal verge was less than 5 cm in 12 (24%) patients.
Treatment
A total of 196 cycles of induction and consolidation chemotherapy were administered. Forty-six patients received four cycles of chemotherapy (two induction and two consolidation), and all 50 patients underwent full-dose RT. Therefore, 46 (92%) patients completed CRT according to the study protocol. Four (8%) received only three cycles: three patients experienced persistent hematological toxicity after the first cycle of consolidation treatment, while one refused to undergo the last treatment cycle. Median dose intensity of chemotherapy was 96.4%, 95.2% and 95.8% for oxaliplatin, bolus infusion 5-fluorouracil and continuous infusion 5-fluorouracil, respectively. The start of consolidation chemotherapy was significantly delayed in two patients due to toxicity (one case of hospitalization due to intestinal obstruction and one of persistent hematological toxicity).
Forty-nine (98%) patients underwent surgery after completing treatment, whereas one experienced early systemic progression after the end of CRT and died without receiving further therapy. Surgery consisted in anterior resection of the rectum in 42 (85.7%) patients and abdominoperineal resection in 7 (14.3%). The median interval between the end of RT and surgery was 70 days (range 25–114, IQR 63–80). The median interval between the end of chemotherapy and surgery was 41 days (range 1–78, IQR 36–50).
Of the 49 evaluable patients who underwent surgery, 43 (87.8%) received adjuvant therapy, 36 with FOLFOX4 for eight cycles every 2 weeks (as per protocol), and seven with fluoropyrimidine only (de Gramont schedule or oral capecitabine).
Toxicity
A safety evaluation was performed after the first six patients were enrolled in Step A. No cases of toxicity requiring RT discontinuation were observed, and the study thus continued to Step B. All 50 patients were evaluable for toxicity after neoadjuvant therapy. Treatment was generally well tolerated and the most common adverse events are listed in Table 2. Both hematological and non-hematological toxicities were as expected. There were 20 cases of grade 3–4 neutropenia and one case of febrile neutropenia, but no cases of grade 3–4 anemia or thrombocytopenia. The most frequently reported non-hematological adverse events were grade 1–2 nausea or vomiting, grade 1–3 diarrhea or constipation, and grade 1–3 fever and fatigue. Only one patient experienced grade 3 peripheral neuropathy, which was completely reversible. Adverse reactions due to RT, mainly grade 1–2 dermatitis, tenesmus, urinary dysfunction and pain, were tolerable and fully reversible.
Toxicity during neoadjuvant CRT and surgical complications (any adverse event).

CRT, chemoradiotherapy.
The median duration of hospitalization after surgery was 12 days (range 8–43). Early and delayed post-surgical complications, observed in 22 (44.9%) and six (12.2%) patients, respectively, are reported in Table 2. The main complications included infection and anastomotic leakage and fistula, all managed conservatively.
Efficacy
Overall, 12 patients achieved a pCR (24.5%, 95% CI 12.5–36.5) and pathological downstaging was achieved in 37 (75.5%, 95% CI 63.5–87.6). The rate of R0 resection was 100% as all patients who underwent surgery had negative margins, both distally and circumferentially. The sphincter-saving resection rate was 85.7% (42 patients) (95% CI 75.1–96.3). The median PFS and OS had not been reached after a median follow-up of 48.7 months (range 4.1–62.3). One- and 2-year OS rates were 98% (95% CI 94–100) and 94% (95% CI 86–100), respectively (Figure 2A and B).

(A) Progression-free survival (PFS) of 50 patients with locally advanced rectal cancer. Vertical bars represent 95% confidence interval of survival probability at 1 and 2 years; and (B) Overall survival (OS) of 50 patients with locally advanced rectal cancer. Vertical bars represent 95% confidence interval of survival probability at 1 and 2 years.
Discussion
The present trial analyzed the safety and efficacy of a new neoadjuvant treatment schedule for patients with LARC based on the administration of SCRT in association with both induction and consolidation FOLFOX. Randomized trials of SCRT
Increasing interest is being shown in the TNT strategy, but a longer follow-up is needed to determine whether this approach translates into improved survival. Indeed, the escalation of neoadjuvant (rather than post-operative) therapy has several goals: improved chemotherapy compliance, reduced toxicity, increased pathological response (to allow sphincter-preserving surgery and pave the way for nonoperative management), and treatment of micrometastases to increase survival.
2
Specifically, two paradigms are emerging: chemotherapy followed by CRT, and CRT followed by chemotherapy,
2
that is, induction and consolidation treatment, respectively. Induction chemotherapy is especially attractive for patients with high-risk disease such as node-positive or large bulky tumors. It also permits drug delivery to the primary tumor before its vasculature is disrupted by radiation or surgery.
6
Conversely, consolidation therapy aims to counteract the risk of systemic disease progression when there is a longer interval between CRT and surgery and to enhance locoregional tumor regression.
18
A randomized phase II trial (CAO/ARO/AIO-12) comparing the two strategies (induction chemotherapy with three cycles of 5-fluorouracil, leucovorin, and oxaliplatin before fluorouracil/oxaliplatin CRT
Among the strategies proposed, one of the most promising is SCRT followed by consolidation chemotherapy. Given that higher downstaging and increased pCR have been obtained with delayed surgery after SCRT compared with immediate surgery,20,21 some authors advocate a further delay in surgery (12–20 weeks after the start of SCRT) during which time FOLFOX chemotherapy can be administered. In particular, the use of hypofractionated RT followed by consolidation chemotherapy was compared with standard CRT in the Polish 2 trial. 14 No differences were observed in local efficacy, whereas improved OS and lower acute toxicity favored the 5 × 5 Gy schedule with consolidation chemotherapy. 14 Results from the similarly designed and ongoing phase III RAPIDO (NCT01558921) 15 and STELLAR trials (NCT02533271) are eagerly awaited.22,23
The COLORE trial met its primary endpoint with a pCR rate of 24.5%. Some studies have reported that patients achieving a pCR after neoadjuvant CRT may have a better prognosis (lower incidence of local and distant recurrence, higher disease-free survival and OS) compared with patients with residual pathological disease.24–26 One- and 2-year OS rates are noteworthy, suggesting the efficacy of treatment (in particular, induction and consolidation chemotherapy) in preventing the development of metastatic disease.
Growing interest in the potential of nonoperative management for rectal cancer is now overshadowing the increase in pCR rates achieved by the addition of induction or consolidation chemotherapy to standard CRT. 27 Indeed, TNT potentially increases the number of patients eligible for organ preservation or a watch-and-wait approach, 2 as shown in studies administering both induction and consolidation therapy: the addition of three cycles of chemotherapy after CRT (together with an increased RT dose of 54 Gy) improved complete clinical response rates from 27% to 57%, 28 while induction chemotherapy prior to CRT increased the number of complete responses (including both clinical and pathological) from 21% to 36% with respect to standard treatment (pre-operative CRT and adjuvant chemotherapy). 29
In the COLORE trial, toxicity was mild and easily manageable. The majority of patients completed the neoadjuvant treatment as per protocol and underwent surgery within the allotted time. The use of dose modulation techniques for RT permits optimal dose delivery for large tumor volumes or volumes-at-risk of disease involvement, leading to reduced RT-related toxicity and better patient compliance due to a less demanding schedule.
Conclusion
FOLFOX and hypofractionated TomoTherapy is effective and safe in patients with LARC. However, long-term efficacy needs to be further evaluated and our results require validation in a randomized phase III trial to confirm the advantage in survival.
Further research is warranted to determine the best way to integrate chemotherapy and SCRT (induction or consolidation chemotherapy, or both, as in the present trial), to identify the optimal interval between each treatment and timing of surgery, and to study in depth the potential role of nonoperative management. Moreover, consolidation chemotherapy with more aggressive regimens (FOLFOXIRI or XELOX and bevacizumab) or a longer duration is currently under investigation. A tailored approach to treatment, for example, TNT in high-risk patients, identified through imaging and molecular signatures, could also represent a major breakthrough in this setting. Finally, depending on how the concept of pre-operative management evolves, the role of adjuvant therapy may also need to be reviewed (e.g. in patients with residual disease).
Footnotes
Acknowledgements
The authors thank Massimo Giannini for his important contribution to the design of the clinical trial and Gráinne Tierney and Cristiano Verna for editorial assistance.
Author contributions
A.P. and A.R. conceived the idea for the study. Patient data were collected by A.P., A.R., I.G.R., E.P., E.N., E.P., G.G., G.E., A.A., D.C., B.R., M.V., D.B., and F.F. were responsible for data interpretation. E.S. performed the statistical analyses. G.L.F., A.P., A.R. and E.S. drafted the paper. All of the authors read and approved the final version of the manuscript for submission.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Availability of data and material
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
