Abstract
Context:
In France, gemcitabine plus nab-paclitaxel (GEM-NAB) is heterogeneously used in metastatic pancreatic cancer due to disparities in its financial accessibility in the institutions.
Objectives:
GEM-NAB conduct a French multi-institutional cost-effectiveness analysis of GEM-NAB versus gemcitabine alone (GEM) as second-line treatment in pancreatic cancer patients.
Design:
All the unresected metastatic pancreatic ductal adenocarcinoma (PDAC) consecutive patients who received GEM-NAB (institution 1) or GEM alone (institutions 2 and 3) as second-line treatment after failure of a 5-fluorouracil based systemic chemotherapy regimen were screened.
Methods:
This study was conducted from the French national healthcare insurance perspective. The primary endpoint was the overall survival (OS) expressed in months, calculated from the date of the first second-line chemotherapy administration to death. Only direct (medical and non-medical) costs have been considered for this analysis. Data were collected retrospectively in one university hospital and two general hospitals.
Results:
The OS was significantly improved in patients receiving GEM-NAB (hazard ratio: 0.54, 95% confidence interval: 0.38–0.77, p = 0.001), with a median OS of 6.2 months (versus 4.1 months in patients receiving GEM alone). Taking into account the cost of GEM-NAB which was afforded by each institution, the incremental cost-effectiveness ratio was €1,449,231 by year of life (€40,256 per patient). In both groups, most of the costs were attributable to readmissions and outpatient chemotherapy administration.
Conclusion:
The issues of the article is based on the trade-off between the benefit in terms of OS of patients treated with GEM-NAB, which is minor (a gain of 2 months of survival, with an accumulated rate of grade ⩾ 3 non-hematological adverse effects) and the additional institutional cost (€25k per year of life for each patient treated). The debate is complex and refers to an ethical component, which is the cost of human life when no other therapeutic alternative is offered to the patient.
Introduction
Pancreatic cancer is the fourth leading cause of cancer-related deaths in Europe, with an estimated 14,000 new cases per year in France.1,2 Its incidence has been constantly increasing over the past decades, from 2- to 3-fold between 1980 and 2018, and pancreatic cancer could become the second cause of cancer-related deaths in Western countries by 2030.3–6 Pancreatic ductal adenocarcinoma (PDAC) is still associated with poor prognosis, with only 10–20% of patients presenting with resectable non-metastatic disease at diagnosis,1,7 and a 5-year overall survival (OS) of less than 5% in metastatic patients treated with chemotherapy.7–9
Systemic chemotherapy remains the gold-standard treatment in patients with metastatic and locally advanced PDAC, 10 and first-line regimens usually consist of either oxaliplatin, irinotecan, fluorouracil, and leucovorin (FOLFIRINOX) or gemcitabine plus nab-paclitaxel (GEM-NAB).11,12 Indeed, both regimens have shown significantly improved OS, progression-free survival, and response rates as compared with gemcitabine (GEM) alone as first-line therapy in patients with metastatic PDAC.11,12 No randomized phase III study neither compared FOLFIRINOX versus GEM-NAB directly, but retrospective studies showed a trend toward greater OS in patients treated with FOLFIRINOX.13,14 Recently, the first results of NALIRIFOX versus GEM-NAB, presented during ASCO congress, reported OS in favor of NALIRIFOX. 15 These data are opposed to other phase III recent trial, stopped for futility, showing a signal in favor of GEM-NAB over FOLFIRINOX regimen. 16
These data do not argue for a reimbursement of GEM-NAB in first line in France. Recommended second-line chemotherapy strategies can strongly vary after failure of FOLFIRINOX, mostly according to patient’s performance status and comorbidities. In some centers, if patients cannot be enrolled in clinical trials, GEM alone or GEM-NAB are widely used in these settings. 10 However, even if generic GEM-NAB is now available in France, it is not covered by the French national healthcare insurance but must be entirely afforded by each institution where it is administrated. Hence, a significant heterogeneity in clinical practices are observed among French centers. Moreover, robust comparative data regarding GEM-NAB benefit over GEM alone as second-line therapy are scarce, and cost-effectiveness analyses are lacking in this setting.
Therefore, the aim of this study was to conduct a French multi-institutional cost-effectiveness analysis of GEM-NAB versus GEM alone as second-line treatment in patients with metastatic pancreatic cancer.
Materials and methods
Study population, therapeutic features, and follow-up
The study was retrospectively performed between January 2015 and January 2019 in three French institutions: one University Hospital (Clermont-Ferrand) and two General Hospitals (Le Puy-en-Velay and Vichy). All the unresected metastatic PDAC consecutive patients who received GEM-NAB (Clermont-Ferrand) or GEM alone (Le Puy-en-Velay and Vichy) as second-line treatment after failure of a 5-fluorouracil (5-FU)-based systemic chemotherapy regimen were included. The query was done via our business software Chimio®. Data regarding demographics, clinicopathologic characteristics, therapeutic features, grade ⩾ 3 adverse effects, healthcare costs details, and survival were collected. This study was approved by an independent Institutional Review Board (Comité de Protection des Personnes Sud-Est VI, no. 2020/CE01).
All the cases were discussed at multidisciplinary digestive tumor board meetings. Treatments were tailored individually at the start of every cycles according to patient’s status, complete physical examination, complete blood counts, and blood chemical tests. Patients received either a 60 min intravenous infusion of nab-paclitaxel (125 mg/m2) followed by a 30-min infusion of GEM (1000 mg/m2) on day 1, 8, 15 and every 4 weeks, or an intravenous infusion of GEM (1000 mg/m2) on day 1, 8, 15 every 4 weeks or weekly for 7 of 8 weeks. Treatments were pursued until disease progression, unacceptable level of adverse events, or patient’s treatment refusal. Patients were followed with a complete physical examination and various blood tests at each visit, tumor response was assessed every 3 months with a cross-sectional imaging and they were followed until death.
Cost-effectiveness analysis
This study was conducted according to the guidelines on cost-effectiveness analyses from the French National Authority for Health,17,18 from the French national healthcare insurance perspective. Due to the short reported median survival with GEM-NAB or GEM alone as second-line chemotherapy for metastatic or locally advanced PDAC, ranging from 3 to 12 months,19,20 the model considered a 2-year lifetime time horizon and no discount rates were applied according to the French guidelines.18,21
The primary endpoint was the OS expressed in months, calculated from the date of the first second-line chemotherapy administration to death, all causes of death combined.
Only direct (medical and non-medical) costs have been considered for this analysis. Indeed, indirect costs were deemed insignificant because of the short reported median survival in these patients who mostly already stopped working due to the disease. The direct costs were established by an expert panel including medical oncologists, surgeons, pharmacists, and health economists. Seven relevant cost categories were identified: outpatient chemotherapy administration, prevention and treatment of adverse effects, blood tests, cross-sectional imaging and other exams performed during treatment, home-based cares, readmission, and medical transportation. Costs were expressed in euros (reference year 2020). The CHEERS 2022 methodological standard checklist was used (Supplemental File 1).
Because nab-paclitaxel is still not included in a French public diagnosis related group and must be entirely afforded by each institution, the analysis was performed from the French national healthcare insurance perspective, thus excluding its cost. Its impact on the cost-effectiveness ratio has been analyzed separately. The exact number of units per patient has been determined according to the posology and multiplied by its unit purchase price. Unit prices of drugs were obtained from wholesale price lists from official tariffs (http://www.ameli.fr). The cost of chemotherapy sessions was taken from the ‘national cost study’ (https://www.atih.sante.fr/) with a homogeneous patient group code entitled ‘Chemotherapy for tumor, in sessions’ and procedure code number 28Z07Z (April 2019), that is, a cost of €383. The costs of biological screening were taken from the French nomenclature of acts of medical biology (version 53, April 2019). The costs of imaging examinations were taken from the Common Classification of Medical Acts (version 58, July 2019). The costs of home care were based on the general classification system for the professional activities of doctors, dental surgeons (version dated 1 July 2019). The costs of readmissions were calculated using an analytical query based on the hospital accounting reprocessing system, carried out by the financial departments of each hospital to obtain an average cost per department and per year. Travel costs were calculated by kilometric rate, applicable to the distance traveled from the place of departure to the place of arrival, after deduction of the first 3 km included in the departmental flat rate (http://www.ameli.fr). The choice was made to retain only nab-paclitaxel as additional cost of treatment, GEM being dispensed in the two groups. Even if the dose of GEM is slightly different between the groups, its cost is not significant compared to nab-paclitaxel’s one, and is not decisive in the therapeutic decision.
The result is calculated in the form of an incremental cost-effectiveness ratio expressed in euros per life-year gained, calculated according to the following formula 22 : difference in cost between the two groups divided by the difference in median OS between the two groups multiplied by 12 (to have an annual indicator).
A deterministic univariate sensitivity analysis was simultaneously conducted to test the robustness of the results, in order to limit the effects of uncertainty on the economic model. This analysis consisted of varying all the cost item amounts by 20%, in other words by increasing or decreasing them by 20%, to see if the results found differed according to these changes.
Statistical analysis
Statistical analyses were performed using Stata software (version 15; StataCorp, College Station, TX, USA). All tests were two-sided, with an alpha level set at 5%. Categorical data are presented as the number of patients and associated percentages, and continuous data as mean ± standard deviation or median [25th; 75th percentiles]. Baseline comparisons of GEM-NAB and GEM groups were made by the Chi-squared test or the Fisher’s exact test for categorical variables, and by the Student’s t test or the Mann–Whitney U test for continuous variables. Censored data (OS) were estimated using the Kaplan–Meier method, and the groups were compared by Cox model, considering the institution as a random effect. Factors associated with OS were also studied with Cox model: (i) univariate analysis, and (ii) analysis adjusted for the treatment received (GEM-NAB or GEM). The results are expressed as hazard ratios (HR) and 95% confidence interval (CI). The patients were also compared according to their OS (⩽3 versus 3–10 versus ⩾10 months) with ordinal mixed effect logistic regressions. The proportion of each cost category was calculated for each patient and compared between the two groups using linear mixed models, considering the institution as a random effect. Logarithmic transformations were applied when necessary to achieve a normal distribution.
Results
Patient’s characteristics, treatments features, and adverse events
Seventy-two consecutive patients received GEM-NAB or GEM alone as second-line treatments after failure of a 5-FU-based systemic chemotherapy regimen for unresected metastatic PDAC between January 2015 and January 2019, in three institutions. Thirty-six patients were included in each group. Age, sex, comorbidities, Eastern Cooperative Oncology Group (ECOG) performance status, location of PDAC, metastatic sites, and type of previous 5-FU-based first-line chemotherapy regimen were comparable between the two groups. More patients received full dose of chemotherapy in the GEM group than in the GEM-NAB group (75.0% versus 50.0%, p = 0.03). The data concerning adverse events could be recorded for all the patients. No treatment-related deaths were observed. The occurrence of grade ⩾ 3 hematological adverse events was comparable between the two groups. However, grade ⩾ 3 non-hematological adverse events seemed more frequent in patients treated with GEM-NAB, especially sensory neuropathies (19.4% versus 0.0%, p = 0.01). Patient’s characteristics, treatments features, and chemotherapy adverse events are summarized in Table 1.
Patient’s characteristics, treatment features, and chemotherapy adverse effects of patients who received gemcitabine plus nab-paclitaxel or gemcitabine alone as second-line treatments after failure of a 5-FU-based systemic chemotherapy regimen for unresected metastatic PDAC.
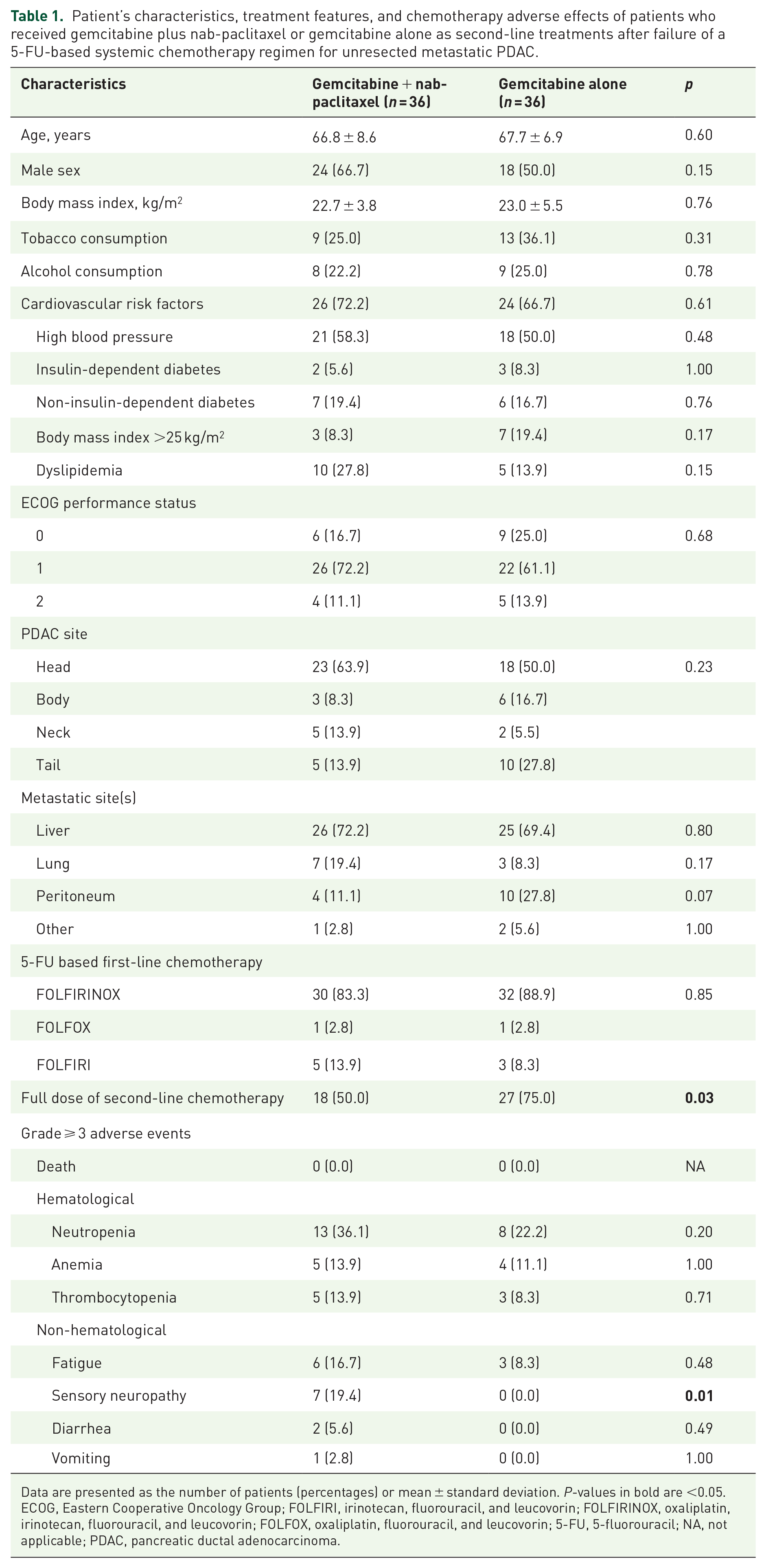
Data are presented as the number of patients (percentages) or mean ± standard deviation. P-values in bold are <0.05.
ECOG, Eastern Cooperative Oncology Group; FOLFIRI, irinotecan, fluorouracil, and leucovorin; FOLFIRINOX, oxaliplatin, irinotecan, fluorouracil, and leucovorin; FOLFOX, oxaliplatin, fluorouracil, and leucovorin; 5-FU, 5-fluorouracil; NA, not applicable; PDAC, pancreatic ductal adenocarcinoma.
OS analysis
The OS was significantly improved in patients receiving GEM-NAB (HR: 0.54, 95% CI: 0.38–0.77, p = 0.001), with a median OS of 6.2 months (versus 4.1 months in patients receiving GEM alone) (Figure 1).

Overall survival in patients who received gemcitabine plus nab-paclitaxel or gemcitabine alone as second-line treatments after failure of a 5-FU-based systemic chemotherapy regimen for unresected metastatic PDAC.
Regardless the type of second-line chemotherapy regimen, factors associated with longer OS were male sex, ECOG performance status equal to zero, absence of insulin-dependent diabetes, presence of dyslipidemia, and absence of peritoneal metastases (Table 2). After adjustment for the treatment received (GEM-NAB or GEM), factors associated with longer OS were ECOG performance status equal to zero, absence of insulin-dependent diabetes, and presence of dyslipidemia.
Univariate and adjusted analysis of factors associated with overall survival in patients who received gemcitabine plus nab-paclitaxel or gemcitabine alone as second-line treatments after failure of a 5-FU based systemic chemotherapy regimen for unresected metastatic PDAC.

Analysis adjusted for the treatment received (gemcitabine plus nab-paclitaxel or gemcitabine alone). P-values in bold are <0.05.
CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; 5-FU, 5-fluorouracil; HR, hazard ratio; PDAC, pancreatic ductal adenocarcinoma; Ref., reference.
When comparing patients according to their OS (⩽3 versus 3–10 versus ⩾10 months), ECOG ⩾ 1 and the administration of GEM alone are significantly associated with a shorter OS (Supplemental File 2).
Health costs analysis
The overall health costs were €796,349 and €671,364 for the GEM-NAB and the GEM groups, respectively. Thus, an additional cost of €124,985 (€3472 per patient in average) was observed in the GEM-NAB group. Considering the additional cost of nab-paclitaxel itself, this additional would have been of €284,849 (€7913 per patient in average). For both GEM-NAB and GEM groups, most of these costs were attributable to readmissions (56.5% and 55.9% of the overall costs, respectively), and to outpatient chemotherapy administration (19.4% and 18.9% of the overall costs, respectively) (Figure 2). More precisely, the median cost of readmissions per patient was €9149 (1986; 18,088) in the GEM-NAB group and €6830 (4538; 11,799) in the GEM group, and the median cost of outpatient chemotherapy administration per patient was €3447 (1724; 6320) and €2873 (1915; 4022), respectively. The others costs (total and per patient) by category are presented in Table 3.

Proportion of each cost category to the total cost of each group.
Total costs and costs per patient (in euros).

The additional cost of nab-paclitaxel (in the GEM-NAB group) is not included. Costs per patient are presented as median (25th; 75th percentiles).
GEM, gemcitabine alone; GEM-NAB, gemcitabine plus nab-paclitaxel.
Considering the costs for each patient, the median proportions of the costs related to readmissions were comparable between patients who received GEM-NAB and patients who received GEM alone [41% (21; 72) versus 51% (36; 75), respectively, p = 0.34]. The same was observed regarding the median proportions of the costs related to outpatient chemotherapy administration: 28% (12; 49) versus 21% (8; 35), respectively (p = 0.35). However, the costs attributable to cross-sectional imaging and blood tests performed during treatment were significantly higher in patients receiving GEM-NAB: 3% (1; 7) versus 1% (0; 3) (p = 0.008), and 3% (2; 4) versus 2% (1; 3) (p = 0.04), respectively. These results are summarized in Figure 3.

Proportion of each cost category to the total cost of each patient.
Cost-effectiveness analysis of GEM-NAB chemotherapy
Considering all the patients receiving GEM-NAB during the study period, the incremental cost-effectiveness ratio was €724,615 by year of life (€20,128 per patient in average) since the first second-line chemotherapy administration. Taking into account the cost of nab-paclitaxel, which was afforded by each institution, this incremental cost-effectiveness ratio was €1,449,231 by year of life (€40,256 per patient in average).
To test the robustness of these results, a deterministic univariate sensitivity analysis was conducted, considering consensual uncertainty values with 20% upper and lower bounds for each cost categories (Figure 4). In the most favorable settings, the incremental cost-effectiveness ratio was €638,176 by year of life (€17,727 per patient in average) since the first second-line chemotherapy administration, so the ratio and cost per patient are consistent with the first calculation.
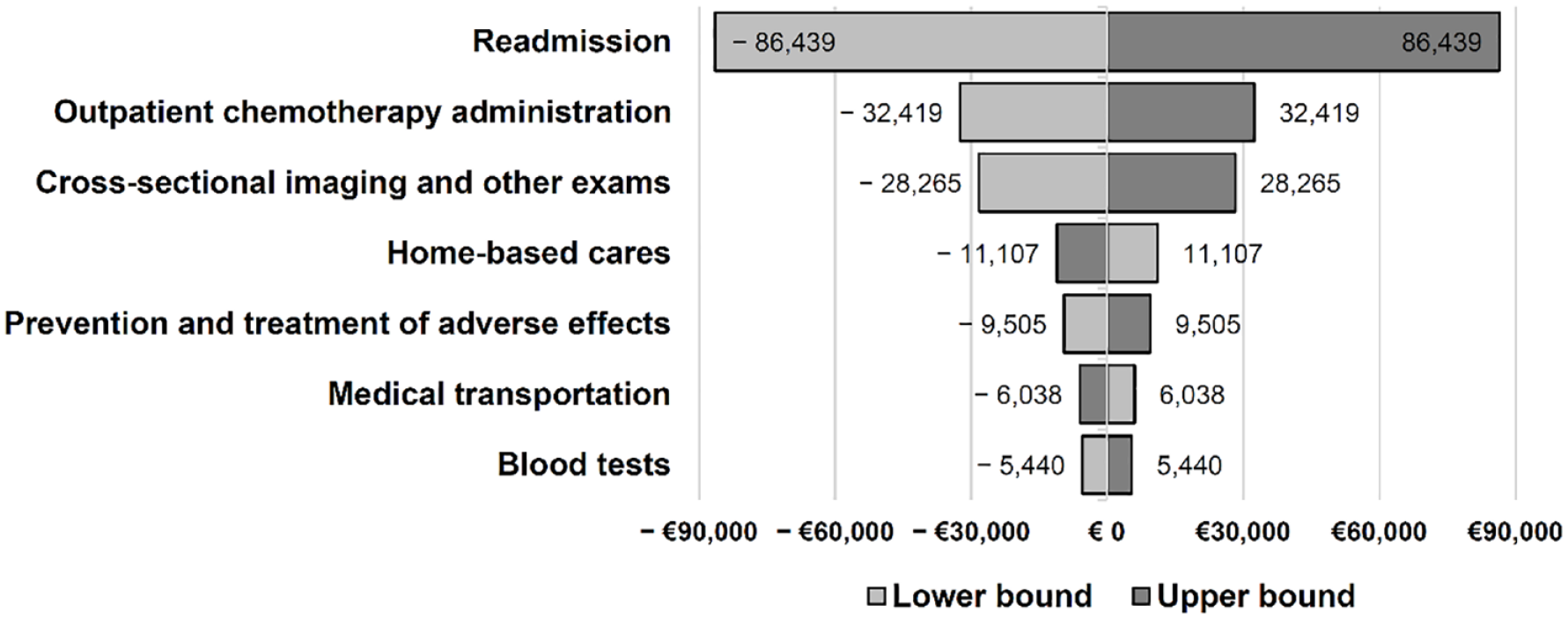
Sensitivity analysis of the cost-effectiveness ratio.
Discussion
After failure of oxaliplatin- and/or irinotecan-based chemotherapy regimens as first-line treatment in patients with metastatic PDAC, administration of GEM-NAB as a second second-line chemotherapy is an option. Several economic studies have been done in first line: Lazzaro et al. 23 performed a cost-effectiveness analysis of GEM-NAB compared with GEM in unresectable pancreatic cancer from the perspective of Italian payers. They reported that GEM-NAB is more cost-effective than GEM alone. Carrato et al. 24 and Gharaibeh et al. 25 reported similar results. In contrast, from the perspective of US healthcare payers, Gharaibeh et al. 25 reported that GEM-NAB is less cost-effective than GEM. Kurimoto et al. 26 performed trial-based cost-effectiveness analysis in Japan. They reported that S-1 (TS-1) and GEM-NAB was more cost-effective than both GEM-NAB and FOLFIRINOX. Nevertheless, this analysis used a shorter time horizon and used median survival time instead of quality-adjusted life years as the outcome of effectiveness. Robust comparative data regarding cost-effectiveness analyses are lacking in PDAC patients.27,28
In this study, a significant improved OS was observed in patients receiving GEM-NAB as second-line therapy. Grade ⩾ 3 non-hematological adverse events were more frequent in patients treated with GEM-NAB. From the French national healthcare insurance perspective, the cost-effectiveness analysis shown an incremental cost-effectiveness ratio of €20,128 by year of life for each patient treated with GEM-NAB since the first second-line chemotherapy administration, which is increased to €40,256 by year of life for each patient when taking into account the cost of nab-paclitaxel itself. Without counting the additional cost of the nab-paclitaxel itself, extra costs were mainly attributable to more blood tests and cross-sectional imaging performed in patients treated with GEM-NAB, probably due to the management of more adverse events in these patients.
To the best of our knowledge, this is the first cost-effectiveness study carried out on patients who received either GEM-NAB or GEM as a second-line chemotherapy for metastatic PDAC after failure of an oxaliplatin- and/or irinotecan-based first-line regimen. It was performed on comparable groups, including comprehensive data collection and deterministic sensitivity analyses. In carrying out this study, however, the authors were confronted with limitations specific to retrospective medico-economic studies. First, it was difficult to obtain a population corresponding to the ‘GEM/GEM-NAB’ inclusion criteria for second-line post-5-fluorouracil treatment. Second, it is always more difficult to retrieve economic data retrospectively than with clinical data, which are often well documented in patients’ medical records. The person who carried out the data collection spent a great deal of time collecting data. Finally, because of the retrospective nature of the study, it was impossible to work on quality of life.
In this study, the unfavorable incremental cost-effectiveness ratio with the addition of nab-paclitaxel to GEM makes this strategy non-cost-effective from the French national healthcare insurance perspective. Therefore, these results support the absence of nab-paclitaxel costs coverage by the French national healthcare insurance, costs which must be entirely afforded by each institution. Nowadays, the great debate is whether the additional cost induced by GEM-NAB (€25k per patient per year of life gained) for the institutions that dispense it is not too substantial in view of the benefits (a gain of 2 months of OS, and an increased rate of non-hematological adverse events of grade ⩾ 3). This debate refers to the concept of the cost of human life, in other words the price that individuals are willing to pay to obtain a reduction in their probability of death and to the notion of the responsibility of the public decision-maker, who before committing the resources of the community must be sure that there is no more efficient therapeutic option. 29 These human life value data are generally unknown and subjective. Several statistical methods have been used to approach them. 30 Among them is the Human Capital method where the value of the individual for the community is related to his contribution to the world’s gross domestic product, which is the most commonly used indicator of national wealth. In practice, this method is highly criticized because it is reductive and induces an underestimation bias. Other solutions such as revealed preference or contingent valuation methods provide more complete estimates of the value of human life, but due to the strong assumptions that weigh on these models (rationality of individuals, complete information, etc.) these solutions pose methodological problems.29,31
To help answer this crucial question, which obviously also implies both human and ethics considerations besides medico-economic aspects in these patients with limited life expectancy and no efficient alternative therapeutics, quality of life assessment should be included in such analysis. Further prospective studies on this topic are thus needed to better precise both cost-utility and cost-effectiveness of the addition of nab-paclitaxel to GEM in this setting, probably using more complete composite criteria of effectiveness, including both survival and quality of life. However, the concept of cost-effectiveness remains unclear in the settings of more efficient but more expensive therapeutics, depending on both individual and collective willingness to pay for a treatment and/or some survival gain.32,33 In addition, other items were missing like the use of granulocyte growth factors, usually being prescribed in primary prevention for GEM-NAB treated patients, which could make worst the cost analysis of GEM-NAB group of patients. Current prospective studies should probably associate such medico-economic approaches in ancillary endpoints, in order to better address these questions. All French healthcare establishments are faced with the same issues when it comes to GEM-NAB treatment, since this is a costly treatment that is not supported by the health insurance system. The results of this study provide useful information for public decision-makers, who must choose to optimize clinical decisions given the limited resources at their disposal. These results can therefore be used by healthcare establishments in the regional area covered by the study, as well as at national level.
Conclusion
Even if this study confirmed an improved OS in patients treated with GEM-NAB compared with GEM for metastatic pancreatic cancer, the unfavorable incremental cost-effectiveness ratio with the addition of GEM-NAB makes this strategy non-cost-effective from the French national healthcare insurance perspective. The additional costs linked to the management of more severe adverse events in these patients mainly explained this result. If considered, the addition of nab-paclitaxel could probably be reserved for patients with ECOG performance status equal to zero without advanced peritoneal metastases. However, further prospective studies are needed to better precise both cost-utility and cost-effectiveness of the addition of nab-paclitaxel, probably using composite criteria of effectiveness, including both survival and quality of life.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241259635 – Supplemental material for French multi-institutional cost-effectiveness analysis of gemcitabine plus nab-paclitaxel versus gemcitabine alone as second-line treatment in metastatic pancreatic cancer patients
Supplemental material, sj-docx-1-tam-10.1177_17588359241259635 for French multi-institutional cost-effectiveness analysis of gemcitabine plus nab-paclitaxel versus gemcitabine alone as second-line treatment in metastatic pancreatic cancer patients by Amaury Demaziere, Charline Mourgues, Céline Lambert, Sophie Trevis, Hélène Bertucat, Isabelle Grange, Denis Pezet, Valérie Sautou, Marine Jary and Johan Gagnière in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359241259635 – Supplemental material for French multi-institutional cost-effectiveness analysis of gemcitabine plus nab-paclitaxel versus gemcitabine alone as second-line treatment in metastatic pancreatic cancer patients
Supplemental material, sj-pdf-2-tam-10.1177_17588359241259635 for French multi-institutional cost-effectiveness analysis of gemcitabine plus nab-paclitaxel versus gemcitabine alone as second-line treatment in metastatic pancreatic cancer patients by Amaury Demaziere, Charline Mourgues, Céline Lambert, Sophie Trevis, Hélène Bertucat, Isabelle Grange, Denis Pezet, Valérie Sautou, Marine Jary and Johan Gagnière in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
