Abstract
Purpose:
To evaluate efficacy and safety of veliparib combined with carboplatin/paclitaxel in patients with advanced human epidermal growth factor receptor 2 (HER2)-negative, germline BRCA (gBRCA)-associated breast cancer defined by hormone receptor (HR) and gBRCA1/2 mutation status.
Patients and Methods:
In this phase-3, double-blind, placebo-controlled trial, patients (N = 509) with advanced HER2-negative breast cancer and gBRCA1/2 mutations were randomized 2:1 to receive veliparib plus carboplatin/paclitaxel or placebo plus carboplatin/paclitaxel. Patients who discontinued chemotherapy prior to disease progression continued receiving blinded veliparib/placebo monotherapy. The primary endpoint was investigator-assessed progression-free survival (PFS). Subgroup analyses of PFS stratified by HR and gBRCA1/2 mutation status were prespecified.
Results:
In the intention-to-treat population, there were similar proportions of patients with gBRCA1 versus gBRCA2 mutations (51% vs 49%) and HR+ disease versus triple-negative breast cancer (TNBC) (52% vs 48%). Median PFS was longer in the veliparib arm compared with the placebo arm for all subgroups (HR+: 13.0 vs 12.5 months, hazard ratio (95% confidence interval (CI)): 0.69 (0.52, 0.93), p = 0.013; TNBC: 16.6 vs 14.1 months, hazard ratio (95% CI): 0.72 (0.52, 1.00), p = 0.052; gBRCA1: 14.2 vs 12.6 months, hazard ratio (95% CI): 0.75 (0.55, 1.03), p = 0.073; gBRCA2: 14.6 vs 12.6 months, hazard ratio (95% CI): 0.69 (0.50, 0.95); p = 0.021). Benefit was durable, with improved PFS rates at 2 years (HR+, 27.5% vs 15.3%; TNBC, 40.4% vs 25.0%) and 3 years (HR+, 17.5% vs 8.6%; TNBC, 35.3% vs 13.0%) in all subgroups. gBRCA status (BRCA1 vs BRCA2) did not substantially affect the carboplatin/paclitaxel ± veliparib toxicity profile.
Conclusion:
Veliparib plus carboplatin/paclitaxel resulted in durable benefit in subgroups defined by HR status or by gBRCA1 versus gBRCA2 mutation. Overall, addition of veliparib to carboplatin/paclitaxel was tolerable, and there were no clinically meaningful differences in adverse events between the gBRCA1 versus gBRCA2 and HR+ versus TNBC subgroups.
Trial Registration:
NCT02163694, https://clinicaltrials.gov/ct2/show/NCT02163694
Introduction
Germline mutations in breast cancer susceptibility genes gBRCA1 and gBRCA2 are estimated to occur in approximately 1–5% of all breast cancers. 1 The BRCA1 and BRCA2 proteins function in the repair of double-stranded DNA breaks through homologous recombination. 2 Therapeutic agents that inhibit DNA synthesis or repair mechanisms, including platinum-containing compounds and poly(adenosine diphosphate-ribose) polymerase inhibitors (PARPis), have shown efficacy for treating gBRCA1/2 mutation-associated breast cancers.3–6 Greater chemotherapy-associated acute hematologic toxicity has been reported in patients with breast cancer harboring gBRCA1 mutations compared with gBRCA2 mutations or wild-type BRCA1/2. 7
There is a strong association between gBRCA mutation status and hormone receptor (HR) status. gBRCA1 mutations are primarily associated with triple-negative breast cancer (TNBC),8,9 while gBRCA2 mutations are associated with estrogen receptor (ER)- or progesterone receptor (PgR)-positive breast cancer (HR+). 10 HR status is a primary determinant of treatment strategy. In the advanced stage, patients with HR+ breast cancer may be eligible to receive hormonal therapy with or without cyclin-dependent kinase 4/6 inhibitors prior to receiving chemotherapy. Treatment options for patients with TNBC are generally limited to chemotherapy, 11 although more recently, there is emerging evidence to support the addition of immune checkpoint inhibitors to chemotherapy in a subset of patients.12,13 TNBC is an aggressive form of disease. While the estimated 5-year survival rate for patients with metastatic breast cancer is 28.1%, it is only 11.5% for patients with metastatic TNBC. 14
Veliparib (ABT-888) is a potent, selective, orally bioavailable inhibitor of PARP1 and PARP2 that has been shown to enhance the activity of platinum-based chemotherapy in preclinical studies. 15 In phase-1 and -2 trials, veliparib has demonstrated antitumor activity while maintaining a tolerable safety profile in patients with gBRCA mutation-associated advanced breast cancer, either as a single agent or combined with carboplatin/paclitaxel.16–18 BROCADE3 was the first phase-3 trial to assess a PARPi-platinum chemotherapy combination regimen in patients with human epidermal growth factor receptor 2-negative (HER2–) gBRCA-associated breast cancer. In this study, the combination of veliparib with carboplatin/paclitaxel was generally well-tolerated and significantly improved progression-free survival (PFS) compared with carboplatin/paclitaxel alone (14.5 months vs 12.6 months, respectively; hazard ratio (95% confidence interval (CI)): 0.71 (0.57, 0.88); p = 0.002). Importantly, this effect was durable, with an increased proportion of patients in the veliparib arm alive and progression free at 3 years (25.7%) compared with patients in the placebo arm (10.7%). 19
The BROCADE3 study enrolled patients with either gBRCA1 or gBRCA2 mutations, and either HR+ disease or TNBC. The subgroup analysis presented here evaluates the efficacy and safety of veliparib plus carboplatin/paclitaxel in prespecified subgroups of patients classified by HR and gBRCA1/2 mutation status.
Materials and methods
Study design, participants, and treatment
BROCADE3 is a phase-3, double-blind, randomized, placebo-controlled study conducted at 147 centers in 36 countries worldwide. The study design and eligibility criteria were previously published. 19 In brief, eligible patients were women and men ⩾18 years of age with HER2-negative metastatic or locally advanced unresectable breast cancer, deleterious or suspected deleterious gBRCA1 or gBRCA2 mutations, and Eastern Cooperative Oncology Group (ECOG) performance status 0–2. Eligible patients received two or fewer prior lines of cytotoxic therapy for metastatic disease and no prior PARPi therapy. gBRCA mutation status was as documented by the central laboratory (Myriad Genetics).
Patients were randomized 2:1 to receive veliparib (120 mg orally twice daily (BID) on days –2 through 5) or a matching placebo combined with carboplatin (area under the concentration-time curve 6 mg/mL/minute intravenously on day 1) and paclitaxel (80 mg/m2 intravenously on days 1, 8, and 15) in 21-day treatment cycles. Randomization was stratified by HR status (ER/PgR+ vs ER/PgR−), prior platinum therapy (yes vs no), and central nervous system metastases (yes vs no).
Veliparib/placebo, carboplatin, or paclitaxel could be reduced in dosage or discontinued independently at the investigator’s discretion. Patients who discontinued chemotherapy in the absence of disease progression could receive blinded veliparib/placebo monotherapy at a dose of 300–400 mg BID (determined by investigator) administered continuously. Patients remained on treatment until disease progression or unacceptable toxicity occurred.
This study was conducted in accordance with the ethical principles that have their origin in the Declaration of Helsinki and are consistent with International Conference on Harmonization Good Clinical Practice guidelines and applicable regulatory requirements. Institutional review boards at each of the participating study sites reviewed and approved the protocol (Advarra Federalwide Assurance no: 00023875). Written informed consent was obtained from each patient before the study. An independent data monitoring committee oversaw patient safety. This study was registered with ClinicalTrials.gov (NCT02163694) on May 19, 2014, and the first patient was enrolled on July 2, 2014.
Assessments
The primary endpoint was investigator-assessed PFS (number of days from randomization to disease progression, or death from any cause within 63 days of last tumor assessment) per Response Evaluation Criteria In Solid Tumors version 1.1 (RECIST v1.1). Secondary endpoints included overall survival (OS; number of days from randomization to death), clinical benefit rate (CBR; progression-free rate at 24 weeks from the Kaplan–Meier curve for time to progression), objective response rate (ORR; proportion of patients who have partial or complete responses (PR or CR) per RECIST v1.1, as assessed by investigator), duration of response (DOR; number of days from documentation of PR or CR to disease progression), and PFS2 (number of days from randomization to disease progression or death on subsequent therapy). Duration of treatment exposure was the number of days a patient was exposed to study treatment. Radiologic tumor assessments were performed at screening and every 9 weeks thereafter until disease progression.
Adverse events (AEs) were monitored throughout the study and graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events, version 4.03. Treatment-emergent adverse events (TEAEs) were those that occurred during treatment or up to 30 days after study drug discontinuation.
Statistical analyses
The cutoff date for efficacy data included in this manuscript was April 5, 2019. Efficacy analyses were performed on the intention-to-treat (ITT) population, consisting of all randomized patients with gBRCA mutation confirmed by the central laboratory. Safety analyses included all patients who received one or more dose of veliparib or placebo. Patients who tested positive for both gBRCA1 and gBRCA2 mutations were excluded from the gBRCA1/2 subgroup analyses. Kaplan–Meier methodology was used to calculate PFS and CBR. The stratified log-rank test and Cox proportional hazards model was used to compare PFS, PFS2, OS, and DOR between treatment arms, and the stratified Cochran–Mantel–Haenszel test was used to compare CBR and ORR between treatment arms. Analyses in the HR+ and TNBC subgroups were stratified by prior platinum use; analyses in the gBRCA1 and gBRCA2 subgroups were stratified by both prior platinum use and HR status. A two-sided p value of ⩽0.05 was considered statistically significant. Analyses were performed using SAS version 9.4.
Results
Patients
In total, 509 patients with centrally confirmed gBRCA1 or gBRCA2 mutations were randomized in this study (Supplemental Figure S1). 19 Fifty-one percent of patients had gBRCA1 mutations (N = 256), and 49% had gBRCA2 mutations (N = 243). Ten patients with both gBRCA1 and gBRCA2 mutations were excluded from analyses in the gBRCA1 and gBRCA2 subgroups but included in analyses in the HR+ and TNBC subgroups. The study population was also composed of nearly even numbers of patients with HR+ breast cancer (52%; N = 266) and TNBC (48%; N = 243). Proportions of patients with gBRCA1 versus gBRCA2 mutations and HR+ breast cancer versus TNBC were comparable in the veliparib and placebo arms.
Demographic and clinical characteristics of patients characterized by gBRCA mutation status and HR status in the veliparib and placebo arms are shown in Table 1. Patients with gBRCA1 mutations primarily had TNBC (73%), while patients with gBRCA2 mutations primarily had HR+ disease (79%). Higher proportions of patients in the gBRCA1 and TNBC subgroups had received prior platinum therapy, while higher proportions of patients in the gBRCA2 and HR+ subgroups had received prior chemotherapy for metastatic disease. A detailed summary of prior oncology therapies for patients stratified by HR+ or TNBC disease is shown in Supplemental Table S1. Patient characteristics were otherwise generally well-balanced between the gBRCA1 versus gBRCA2 and HR+ versus TNBC subgroups and between treatment arms within each subgroup. In the HR+ subgroup, approximately two-thirds of patients in both treatment arms had received prior endocrine therapy (veliparib: 64.9%, n = 113; placebo: 66.3%, n = 61).
Demographic and clinical characteristics of patients by subgroup in the intention-to-treat population.

BRCA, breast cancer susceptibility gene; C/P, carboplatin plus paclitaxel; CTx, cytotoxic therapy; ECOG, Eastern Cooperative Oncology Group; ER, estrogen receptor; g, germline; HR, hormone receptor; HR+, hormone receptor positive (ER and/or PgR); PgR, progesterone receptor; PS, performance status; TNBC, triple-negative breast cancer; US, United States. +, positive; −, negative.
Values missing from one patient in the gBRCA1 placebo plus C/P arm and one patient in the TNBC placebo plus C/P arm. Percentages calculated on nonmissing values.
gBRCA mutation status as determined by core laboratory. Patients with both BRCA1 and BRCA2 mutations were excluded from analyses in the gBRCA1 and gBRCA2 subgroups but included in analyses in the HR+ and TNBC subgroups.
Efficacy
In the primary analysis, the addition of veliparib to carboplatin/paclitaxel improved investigator-assessed median (m)PFS in the overall study population. Consistent results were observed in the gBRCA1, gBRCA2, HR+, and TNBC subgroups.
Efficacy in subgroups defined by HR expression
In the HR+ subgroup, mPFS was 13.0 months versus 12.5 months in the veliparib and placebo arms, respectively (hazard ratio (95% CI): 0.69 (0.52, 0.93); p = 0.013). In the TNBC subgroup, mPFS was 16.6 months versus 14.1 months (hazard ratio (95% CI): 0.72 (0.52, 1.00); p = 0.052) (Figure 1(a) and (b)). Notably, the benefit of veliparib was remarkably durable, with clear separation after the median as greater proportions of patients in the veliparib arm compared with the placebo arm in both the HR+ and TNBC subgroups remained progression free at 2 years (HR+, 27.5% vs 15.3%; TNBC, 40.4% vs 25.0%) and 3 years (HR+, 17.5% vs 8.6%; TNBC, 35.3% vs 13.0%). ORR and CBR were generally similar between the veliparib and placebo arms within each subgroup (Table 2). However, mDOR was numerically increased in the veliparib arm versus placebo arm in both subgroups (Table 2). There was also a numeric increase in mPFS2 for the veliparib versus placebo arm, and subsequent anticancer therapies that patients received are listed in Supplemental Table S2. In both subgroups, mOS approached 3 years in the veliparib arm, and there was a numeric increase in the veliparib arm compared with the placebo arm (HR+: 32.4 months vs 27.1 months; hazard ratio (95% CI): 0.96 (0.68, 1.36), p = 0.832; TNBC: 35.0 months vs 30.0 months; hazard ratio (95% CI): 0.92 (0.62, 1.36), p = 0.683) (Figure 2(a) and (b)).
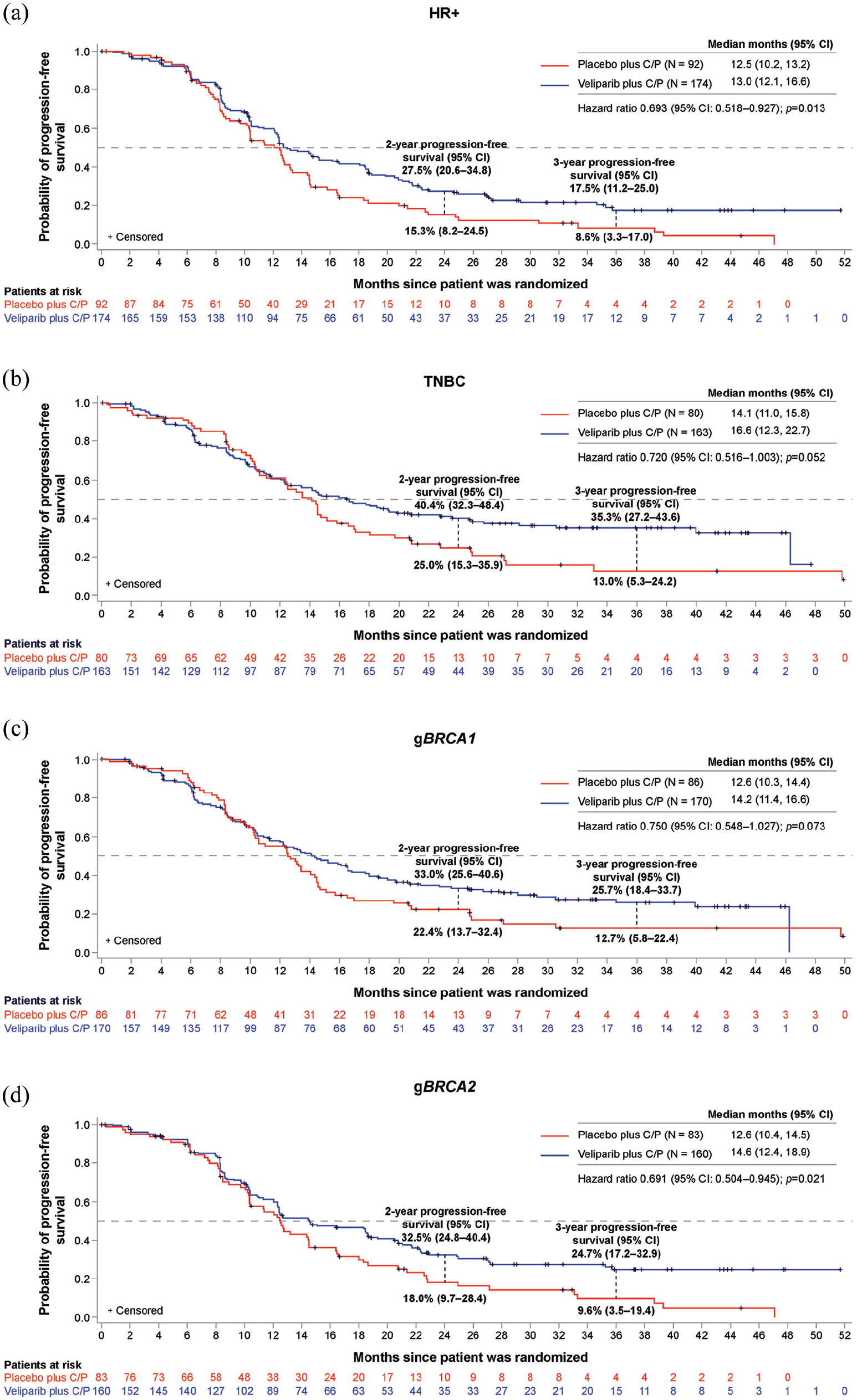
Kaplan–Meier estimates of investigator-assessed progression-free survival in HR+ (a), TNBC (b), gBRCA1 (c), and gBRCA2 (d) subgroups. Distributions were estimated by means of the Kaplan–Meier method.

Kaplan–Meier estimates of investigator-assessed overall survival in HR+ (a), TNBC (b), gBRCA1 (c), and gBRCA2 (d) subgroups. Distributions were estimated by means of the Kaplan-Meier method.
Additional efficacy endpoints by subgroup in the intention-to-treat population.

BRCA, breast cancer susceptibility gene; CBR, clinical benefit rate; CI, confidence interval; C/P, carboplatin plus paclitaxel; CR, complete response; DOR, duration of overall response; g, germline; ER, estrogen receptor; HR+, hormone receptor positive (ER and/or PgR); ORR, objective response rate; PFS2, progression-free survival on subsequent therapy; PgR, progesterone receptor; PR, partial response; TNBC, triple-negative breast cancer.
Data are per investigator assessment. Patients with both gBRCA1 and gBRCA2 mutations were excluded from analyses in the gBRCA1 and gBRCA2 subgroups but included in analyses in the HR+ and TNBC subgroups.
CR plus PR; includes patients with at least one measurable lesion at baseline.
95% CI of ORR is from the exact binomial distribution.
Rate and 95% CI are from Kaplan–Meier estimates.
Efficacy in subgroups defined by gBRCA mutation
In the gBRCA1 subgroup, investigator-assessed mPFS was 14.2 months versus 12.6 months in the veliparib and placebo arms, respectively (hazard ratio (95% CI): 0.75 (0.55, 1.03); p = 0.073). In the gBRCA2 subgroup, mPFS was 14.6 months versus 12.6 months (hazard ratio (95% CI): 0.69 (0.50, 0.95); p = 0.021) (Figure 1(c) and (d)). The durability of veliparib treatment was also evident when analyzing patient subgroups defined by gBRCA mutation status, with more patients alive and progression free in the veliparib arm compared with the placebo arm at both 2 years (BRCA1, 33.0% vs 22.4%; BRCA2, 32.5% vs 18.0%) and 3 years (BRCA1, 25.7% vs 12.7%; BRCA2, 24.7% vs 9.6%). ORR and CBR were high and comparable between treatment arms of both subgroups (Table 2). mDOR and PFS2 were numerically increased for the veliparib arm versus placebo arm in each subgroup (Table 2). mOS was greater than 2 years for both treatment arms in both subgroups. In the gBRCA2 subgroup, mOS approached 3 years for the veliparib arm and was numerically increased compared with the placebo arm (Figure 2(c) and (d)).
Safety
The mean number of cycles of carboplatin and of paclitaxel was generally comparable regardless of gBRCA mutation status, HR status, or treatment arm (Table 3). Mean number of days of study drug therapy was higher in the veliparib arm compared with the placebo arm in each subgroup. There were also numeric increases in the mean number of days of therapy for the TNBC subgroup compared with the HR+ subgroup within each treatment arm. Within treatment arms and subgroups, 32.9–43.2% of patients discontinued carboplatin and paclitaxel prior to progression and received blinded study drug monotherapy. Primary reasons for discontinuation of carboplatin or paclitaxel stratified by HR+ disease and TNBC are listed in Supplemental Table S3. The mean duration of blinded monotherapy was longer in the veliparib arm compared with the placebo arm (Table 3). Mean duration of blinded monotherapy was generally longer for the gBRCA1 and TNBC subgroups compared with the gBRCA2 and HR+ subgroups, respectively.
Exposure summary by subgroup.
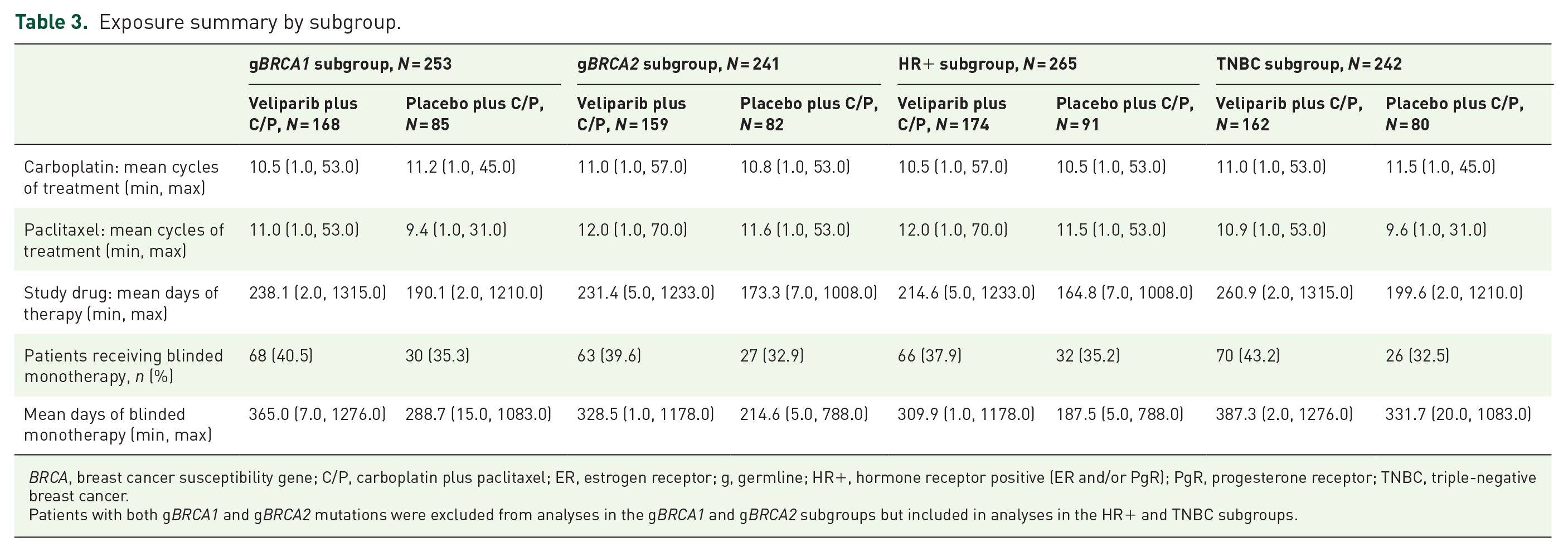
BRCA, breast cancer susceptibility gene; C/P, carboplatin plus paclitaxel; ER, estrogen receptor; g, germline; HR+, hormone receptor positive (ER and/or PgR); PgR, progesterone receptor; TNBC, triple-negative breast cancer.
Patients with both gBRCA1 and gBRCA2 mutations were excluded from analyses in the gBRCA1 and gBRCA2 subgroups but included in analyses in the HR+ and TNBC subgroups.
An overview of AEs by subgroup is provided in Supplemental Table S4. The most common AEs reported were hematologic. Neutropenia was the most common any-grade AE (⩾88% of patients in each treatment arm by subgroup), but frequencies were comparable between the veliparib and placebo arms in all subgroups. Within each subgroup, the frequencies of any-grade anemia and of thrombocytopenia were ⩾5% higher in the veliparib arm compared with the placebo arm. Gastrointestinal AEs of nausea and diarrhea (predominantly grade 1/2) occurred at higher frequencies in the veliparib arm of each subgroup.
Summary of treatment-emergent serious adverse events and adverse events of special interest.

AE, adverse event; BRCA, breast cancer susceptibility gene; C/P, carboplatin plus paclitaxel; ER, estrogen receptor; g, germline; HR+, hormone receptor positive (ER and/or PgR); Pbo, placebo; PgR, progesterone receptor; TNBC, triple-negative breast cancer; Vel, veliparib.
Patients with both gBRCA1 and gBRCA2 mutations were excluded from analyses in the gBRCA1 and gBRCA2 subgroups but included in analyses in the HR+ and TNBC subgroups. Adverse events were reported using preferred terms within a System Organ Class (SOC) according to the Medical Dictionary for Regulatory Activities (MedDRA) and graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events v4.03.
The overall toxicity profile was generally comparable between the HR+ and TNBC subgroups. Differences in frequencies of common TEAEs between the HR+ and TNBC subgroups were not directionally consistent. In the HR+ subgroup, AEs not related to progression that led to veliparib/placebo discontinuation occurred in 14 patients (8%) in the veliparib arm and three patients (3%) in the placebo arm (Table 4). Comparable proportions of patients in the TNBC subgroup experienced AEs not related to progression leading to veliparib/placebo discontinuation (veliparib: 17 patients (10%); placebo: six patients (8%)). Frequencies of serious adverse events (SAEs) were also comparable in the veliparib (HR+: 59 patients (34%); TNBC: 56 patients (35%)) and placebo (HR+: 27 patients (30%); TNBC: 22 patients (28%)) arms (Table 4).
Frequencies of any-grade anemia and of thrombocytopenia were, at most, slightly higher in the gBRCA1 subgroup compared with the gBRCA2 subgroup, although these events were common in both subgroups (Supplemental Table S4). In the veliparib arm, any-grade anemia was reported for 139 patients (83%) in the gBRCA1 subgroup and 122 patients (77%) in the gBRCA2 subgroup. In the placebo arm, any-grade anemia was reported for 64 patients (75%) in the gBRCA1 subgroup and 52 patients (63%) in the gBRCA2 subgroup. A consistent increase in grade 3 or higher anemia events within treatment arms was not observed between the gBRCA1 versus gBRCA2 subgroups. In the veliparib arm, any-grade thrombocytopenia was reported for 140 patients (83%) in the gBRCA1 subgroup and 124 patients (78%) in the gBRCA2 subgroup. In the placebo arm, any-grade thrombocytopenia was reported for 66 patients (78%) in the gBRCA1 subgroup and 54 patients (66%) in the gBRCA2 subgroup. Increased frequencies of grade 3 or higher thrombocytopenia were also observed in the gBRCA1 versus gBRCA2 subgroup in the veliparib (72 patients (43%) and 59 patients (37%), respectively) and placebo (30 patients (35%) and 18 patients (22%), respectively) arms. This increased frequency in the gBRCA1 subgroup compared with the gBRCA2 subgroup was not consistently observed for other common AEs. Higher frequencies (⩾5% difference) of patients in both arms of the gBRCA2 subgroup experienced any-grade nausea, constipation, fatigue, peripheral sensory neuropathy, back pain, and dyspnea compared with the gBRCA1 subgroup. AEs not related to progression leading to discontinuation of veliparib/placebo occurred in comparable proportions of patients in the gBRCA1 (veliparib: 16 patients (10%); placebo: 5 patients (6%)) and gBRCA2 (veliparib: 15 patients (9%); placebo: 4 patients (5%)) subgroups. Frequencies of SAEs were also comparable for the gBRCA1 (veliparib: 58 patients (35%); placebo: 26 patients (31%))and gBRCA2 (veliparib: 56 patients (35%); placebo: 22 patients (27%)) subgroups (Table 4).
Discussion
In the primary BROCADE3 analysis, the addition of veliparib to carboplatin/paclitaxel prolonged PFS in patients with advanced breast cancer. This study evaluated the combination regimen in a biomarker-selected population defined by the presence of a gBRCA1/2 mutation. In contrast to the use of gBRCA1/2 mutation status to select patients in this and other PARPi trials,5,6 many other therapies for advanced breast cancer have been evaluated specifically in patients with either HR+ disease or TNBC. The differences in prognosis and available therapies for patients with HR+ disease compared with TNBC highlight the need to understand the safety and efficacy of the veliparib regimen in each of these subgroups. Because patients with either a gBRCA1 or gBRCA2 mutation were enrolled in this trial, a better understanding of how these biomarker differences among enrolled patients impact safety and efficacy of the treatment regimen is also of interest.
Metastatic TNBC is associated with poorer prognoses than other types of breast cancer, given its aggressive nature and the relative lack of targeted therapeutic options. In the TNBC subgroup of this trial, the mPFS was durable in both arms with a numeric increase for veliparib compared with placebo (16.6 vs 14.1 months; hazard ratio (95% CI): 0.72 (0.52, 1.00); p = 0.052). A considerable proportion of patients in the veliparib arm were alive and progression free at 2 years (veliparib: 40.4%, placebo: 25.0%) and 3 years (35.3%, 13.0%). Notably, the mOS was nearly 3 years in patients with TNBC in the veliparib arm at 35.0 months, numerically increased over 30.0 months in the placebo arm. These data suggest that patients with TNBC contributed to the benefit of the veliparib regimen observed in the primary analysis of the BROCADE3 study. The mOS for patients with metastatic TNBC is approximately 9–12 months with standard chemotherapy. 20 Atezolizumab plus nab-paclitaxel has recently become available for some patients with metastatic TNBC, based on results from the IMpassion130 trial. 12 Within the programmed cell death protein 1 ligand 1+ subgroup in this study, mPFS was 7.5 and 5.0 months for atezolizumab plus nab-paclitaxel versus placebo plus nab-paclitaxel, respectively. mOS in this subgroup was 25.0 months compared with 17.6 months. Additional subgroup analyses suggested similar benefit in patients with and without gBRCA mutations. 21 Pembrolizumab plus chemotherapy has also been studied in patients with metastatic TNBC. 13 mPFS in the ITT population was 7.5 and 5.6 months for pembrolizumab plus chemotherapy versus placebo plus chemotherapy, respectively. The mPFS and mOS observed here suggest that the veliparib plus carboplatin/paclitaxel approach may provide a substantial benefit for patients with gBRCA mutation-associated advanced TNBC. However, it should be noted that paclitaxel may not be the optimal chemotherapy backbone for immunotherapy regimens in patients with gBRCA mutations. Given the demonstrated superiority of platinum chemotherapy compared with taxanes, 4 further data are needed on the efficacy of platinum chemotherapy combinations with new immune checkpoint inhibitors in patients with gBRCA mutations.
First-line treatment options for patients with HR+ breast cancer differ from those for patients with TNBC. According to current guidelines, standard chemotherapy, including taxanes and platinum agents, is preferred for patients with advanced HER2-negative TNBC, while endocrine therapy is preferred for patients with HR+ breast cancer.11,20 However, recommendations for chemotherapy begin to align in these subgroups once patients with HR+ disease are no longer considered candidates for endocrine therapy due to visceral involvement and resistance to hormonal therapies. Prior endocrine therapy was common among patients in the HR+ subgroup in this study, with approximately two-thirds of patients in both treatment arms having received prior endocrine therapy. As in the TNBC subgroup, mPFS of greater than 1 year was observed in both treatment arms of the HR+ subgroup, with a hazard ratio favoring the veliparib arm (13.0 months vs 12.5 months; hazard ratio (95% CI): 0.69 (0.52, 0.93); p = 0.013). Many patients had long-lasting benefit, evidenced by the proportion of patients alive and progression free at 2 years (veliparib: 27.5%, placebo: 15.3%) and 3 years (17.5%, 8.6%). The mOS observed in HR+ patients also contributed to the long durations of OS seen in the whole BROCADE3 study population: 32.4 months in the veliparib arm versus 27.1 months in the placebo arm (hazard ratio (95% CI): 0.96 (0.68, 1.36); p = 0.832). These data suggest a benefit of the veliparib combination regimen for patients with HR+ disease for whom chemotherapy is appropriate. The safety profile of the regimen was comparable in the HR+ and TNBC subgroups and similar to the overall study population. 19
Friedlaender and colleagues 7 previously reported an increase in acute hematologic toxicities from chemotherapy in patients with gBRCA1 compared with gBRCA2 mutations. While we observed slightly increased frequencies of anemia and thrombocytopenia in the gBRCA1 subgroup compared with the gBRCA2 subgroup, the frequencies were high in both arms and differences were not clinically meaningful. The magnitude of PFS benefit observed was comparable between the two subgroups. These data suggest that patients benefited, with comparable toxicity burden whether they harbored a gBRCA1 or gBRCA2 mutation.
While these subgroup analyses were prespecified, the study was not powered to formally test hypotheses in the different subgroups. Furthermore, a portion of patients in each subgroup discontinued carboplatin and paclitaxel prior to progression and received study drug monotherapy. The study was not designed to evaluate benefit of veliparib combination therapy compared with veliparib monotherapy.
PARPi monotherapy is a treatment option for patients with gBRCA1/2 mutations and advanced breast cancer. The subgroup analyses presented here suggest that combining veliparib with carboplatin/paclitaxel provides benefit to patients with gBRCA mutation-associated breast cancer, regardless of whether their tumors are HR+ or triple negative, and without additional toxicity based on the gBRCA gene bearing a mutation.
Supplemental Material
sj-docx-1-tam-10.1177_17588359211059601 – Supplemental material for Safety and efficacy of veliparib plus carboplatin/paclitaxel in patients with HER2-negative metastatic or locally advanced breast cancer: subgroup analyses by germline BRCA1/2 mutations and hormone receptor status from the phase-3 BROCADE3 trial
Supplemental material, sj-docx-1-tam-10.1177_17588359211059601 for Safety and efficacy of veliparib plus carboplatin/paclitaxel in patients with HER2-negative metastatic or locally advanced breast cancer: subgroup analyses by germline BRCA1/2 mutations and hormone receptor status from the phase-3 BROCADE3 trial by Jean-Pierre Ayoub, Hans Wildiers, Michael Friedlander, Banu K. Arun, Hyo S. Han, Shannon Puhalla, Yaroslav Shparyk, Erik H. Jakobsen, Meijing Wu, Bruce A. Bach, Dai Feng, Christine K. Ratajczak, David Maag and Véronique Diéras in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors thank all the trial investigators, including the late Dr Bella Kaufman, and the patients who participated in this clinical trial. Medical writing support was provided by Thayer Darling, PhD, from Aptitude Health, Atlanta, Georgia, and funded by AbbVie.
Author contributions
J-PA, HW, MF, BKA, HSH, SLP, YS, EHJ, and VD: conceptualization, investigation, data curation, writing—review and editing. MW and DF: conceptualization, formal analysis, methodology, data curation, writing—review and editing. BB, CKR and DM: conceptualization, data curation, writing—review and editing.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JPA: Research funding (institution), AbbVie, Boston Biomedical; consultancy, AstraZeneca, Eisai, Eli Lilly, Novartis, Pfizer, Puma, Roche. HW: Consulting fees and honoraria (institution), Roche, AstraZeneca, Amgen, Lilly, Novartis, AbbVie, Vifor Pharma, Pfizer, Celldex Therapeutics, Janssen-Cilag, TRM Oncology, PUMA Biotechnology, Orion Corporation; unrestricted research grant, Roche; travel support, Roche, Pfizer. MF: Consulting/advisory role, AstraZeneca, MSD, AbbVie, Lilly, GSK, Takeda; speakers’ bureau, AstraZeneca; honoraria, AstraZeneca, MSD, GSK, Lilly, Takeda, Novartis, ACT Genomics; travel/accommodation expenses, AstraZeneca; research funding, BeiGene, AstraZeneca, Novartis. BKA: Research support (institution), AbbVie, PharmaMar, AstraZeneca, Invitae; steering committee (nonpaid), AbbVie. HSH: Research funding (institutions), Arvinas, AbbVie, BMS, Daiichi Pharma, GSK, G1 Therapeutics, Horizon, Karyopharm, Marker Therapeutics, Novartis, Pfizer, Prescient, Seattle Genetics, Zymeworks; speakers’ bureau, Lilly. SLP: Consultant, AbbVie, MedImmune, Celldex, Puma, Pfizer, AstraZeneca, Eisai, NanoString; research funding to institution, AbbVie, Pfizer, Lilly, Novartis, Incyte, Covance-Bayer, AstraZeneca, Genentech, Medivation. YS: Advisory board: Pfizer; speaker: Roche, AstraZeneca, Pfizer; research/clinical studies: AbbVie, Roche, MSD, Boehringer Ingelheim. EHJ: Consulting/advisory role: Pfizer, Roche, Novartis, Eli Lilly. BB: Former employee of AbbVie and may hold AbbVie stock. MW, DF, CKR, and DM: AbbVie employees and may own stock. VD: Consulting/advisory role, Roche/Genentech, Novartis, Lilly, Pfizer, AbbVie, MSD, Daiichi Sankyo, Seattle Genetics, AstraZeneca, Gilead.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AbbVie funded this study and participated in the study design, research, analysis, data collection, interpretation of data, reviewing, and approval of the manuscript. All authors had access to relevant data and participated in the drafting, review, and approval of this manuscript. No honoraria or payments were made for authorship.
Data sharing statement
AbbVie is committed to responsible data sharing regarding the clinical trials we sponsor. This includes access to anonymized, individual, and trial-level data (analysis data sets), as well as other information (e.g., protocols and clinical study reports), as long as the trials are not part of an ongoing or planned regulatory submission. This includes requests for clinical trial data for unlicensed products and indications. These clinical trial data can be requested by any qualified researchers who engage in rigorous, independent scientific research and will be provided following review and approval of a research proposal and statistical analysis plan (SAP) and execution of a data sharing agreement (DSA). Data requests can be submitted at any time, and the data will be accessible for 12 months, with possible extensions considered. For more information on the process, or to submit a request, visit the following link: ![]() .
.
Statement of prior presentation
Data from this study were previously presented at the European Society for Medical Oncology Breast Cancer (ESMO BC) 2020 Annual Congress and the San Antonio Breast Cancer Symposium (SABCS) 2020.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
