Abstract
Background:
The efficacy and safety of poly (ADP-ribose) polymerase inhibitors (PARPis) in the Chinese real-world setting have not been well characterized.
Design:
This is a retrospective analysis of PARPis efficacy in metastatic breast cancer (MBC) patients with homologous recombination repair (HRR) gene pathogenic variants (PVs).
Objectives:
We aimed to evaluate the efficacy and toxicities of PARPis in real-world MBC patients.
Methods:
Patients who received PARPi for MBC at the National Cancer Center and two other centers between January 1, 2019, and December 31, 2024, were consecutively included. The primary endpoint was progression-free survival (PFS). Univariable and multivariable Cox proportional hazard models were used to evaluate the predictive impact of clinicopathologic characteristics on PFS.
Results:
In total, 62 MBC patients treated with olaparib (N = 55), talazoparib (N = 4), pamiparib (N = 2), and fluzoparib (N = 1) were enrolled. The median PFS (mPFS) in all patients was 6.0 months (95% confidence interval: 4.1–7.9). mPFS in the germline BRCA1 (gBRCA1; N = 19), gBRCA2 (N = 30), gBRCA (N = 4), somatic BRCA2 (sBRCA2; N = 1), gPALB2 (N = 4), and other HRR gene (N = 4) PVs carriers were 3.7, 8.0, 2.8, 2.7, 5.3, and 7.1 months, respectively (p = 0.334). In multivariate analysis, ⩽40 years old (hazard ratio (HR): 2.281, p = 0.008), third-line or later therapy (HR: 2.429, p = 0.019), and prior platinum-based treatment (HR: 2.172, p = 0.014) were independently associated with shorter PFS. The incidence of adverse events (AEs) of all grades was 62.5% (35/56). The most common AEs in all grades were anemia (30.4%), nausea (21.4%), and leukopenia (17.9%). Hematologic toxicity was the most common grade ⩾3 AEs.
Conclusion:
PARPis showed promising PFS and tolerable toxicity in the real-world treatment of Chinese MBC patients with HRR-related gene mutations.
Introduction
Homologous recombination repair (HRR) is one of the most important mechanisms of DNA damage repair (DDR). Homologous recombination repair deficiency (HRD) results in genomic instability, which is associated with a significantly increased risk of breast cancer. BRCA1 and BRCA2 are tumor suppressor genes encoding two key proteins involved in the HRR pathway that rely on the undamaged chromosome as the template for error-free damage reversal and have a crucial role in repairing double-strand breaks (DSBs).1,2 BRCA1/2 mutations (BRCA1/2m) confer an average cumulative risk of breast cancer by age 70 of approximately 57% for BRCA1m carriers, and 49% for BRCA2m carriers. 3 BRCA1/2m carriers are more likely to be initially diagnosed at a more advanced stage and to experience worse cancer-related outcomes.4–6 Approximately 3%–5% of patients with breast cancer carry germline (g) pathogenic variants (PVs) in BRCA1 (gBRCA1) or BRCA2 (gBRCA2) in Western countries.7,8 In China, the overall prevalence of gBRCA1/2 PVs was 6.0% in the breast cancer patients, 2.4% in BRCA1, and 3.7% in BRCA2.9,10 Patients carrying gBRCA1 variants have a higher incidence of triple negative breast cancer (TNBC), while carriers of gBRCA2 variants tend to develop hormone receptor (HR)-positive breast cancer. 11 Apart from BRCA, studies have found that mutations in other HRR-related genes, such as PALB2, ATM, and CHEK2, are also associated with an increased risk of breast cancer.12–14
Poly (adenosine diphosphate (ADP)-ribose) polymerase (PARP) proteins are a family of nuclear enzymes involved in multiple DDR processes. PARP inhibitor (PARPi) blocks PARP1’s enzymatic activity by competing with NAD+ binding, and traps PARP1 and PARP2 at DNA damage sites, converting transient single-stranded breaks into replication-associated DSBs.15–17 In individuals with gBRCA1/2 alterations, PARP inhibition leads to the impairment of alternative DDR pathways and thus to irreparable genomic damage and cell death.18,19 PARPis have been extensively integrated into clinical management strategies for breast cancer. For gBRCA PV carriers with early-stage high-risk breast cancer, 1 year of adjuvant treatment with olaparib following primary treatment continued to improve overall survival (OS) compared with placebo. 20 In metastatic breast cancer (MBC), olaparib improved progression-free survival (PFS; hazard ratio (HR) = 0.58, 95% confidence interval (CI): 0.43–0.80, p < 0.001), objective response rate (ORR; 59.9% vs 28.8%) and had a more favorable safety profile compared with the treatment of physician’s choice (TPC) in patients with gBRCA1/2 PVs pretreated with up to two lines of chemotherapy in OlympiAD trial. 21 In the phase III EMBRACA trial, talazoparib improved PFS (HR = 0.54, 95% CI: 0.41–0.71, p < 0.001) and ORR (62.6% vs 27.2%) compared with TPC in carriers of gBRCA1/2 PVs.22,23 Although more limited in data, pamiparib and fluzoparib have shown early promise in treatment efficacy, warranting further evaluation.24,25 In addition to gBRCA mutations, PARPis have also demonstrated therapeutic potential in tumors harboring mutations in other HRR-related genes. TBCRC-048 further focused on the efficacy of olaparib in MBC patients with a germline or somatic (s) mutation in HRR pathway genes other than BRCA1/2 or in patients with somatic BRCA (sBRCA) mutation. The ORR was 33% in the germline non-BRCA cohort and 31% in the somatic mutation cohort. Patients with gPALB2 mutations experienced an ORR of 82% and a median PFS (mPFS) of 13.3 months, while those with sBRCA1/2 mutations had an ORR of 50% and an mPFS of 6.3 months. TBCRC-048 provides compelling evidence that olaparib is effective beyond BRCA-mutated tumors, particularly in gPALB2 mutant MBC, thereby expanding the population likely to benefit from PARPis and underscoring the importance of comprehensive HRR gene testing in guiding therapy.
Although previous studies have demonstrated the therapeutic potential of PARPis in MBC, most clinical trials have focused on early lines of treatment. Whether patients receiving later-line therapies in real-world settings can benefit from PARPis remains unclear. Moreover, variations in genetic background, somatic mutation profiles, and real-world treatment practices in China may result in distinct efficacy and safety outcomes compared to global trial populations. The data regarding the effectiveness and adverse events (AEs) of PARP inhibitors in Chinese patients remain limited. Therefore, this study aims to evaluate the real-world efficacy and safety of PARPis in Chinese MBC patients through a multicenter retrospective analysis, to provide clinical guidance for their use in late-stage treatment.
Methods
Study design and data collection
Our retrospective study consecutively included patients who received PARPi for MBC at the Cancer Hospital Chinese Academy of Medical Sciences (National Cancer Center (China)), SanHuan Cancer Hospital, and Beijing Chaoyang Huanxing Cancer Hospital between January 1, 2019, and December 31, 2024. Patients ⩾18 years old with cytological or histologically confirmed diagnosis of MBC who received at least one cycle of PARPi with imaging efficacy evaluation were included in our study. All patients included should have complete clinicopathological profiles before treatment, including, but not limited to, demographics, tumor characteristics, and clinical outcomes. Patients receiving PARPi during the adjuvant treatment or clinical trials, and those coexisting with other indications such as ovarian cancer and fallopian tube carcinoma, were excluded. The study protocol was approved by the ethical committee of the Cancer Hospital, Chinese Academy of Medical Sciences (Approval No. 25/238-5184). The requirement for individual informed consent was waived by the committee because of the retrospective nature of the study. The reporting of this study conforms to the STROBE statement 26 (Supplemental Material).
Assessment
The primary endpoint of the analysis was PFS in the overall study population. PFS was defined as the time from initiation of PARPi treatment until documented disease progression or death from any cause. The treatment response was evaluated according to Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. Secondary endpoints included ORR, disease control rate (DCR), and safety. AEs were graded according to the Common Terminology Criteria for Adverse Events v5.0 (CTCAE version 5.0). The ORR was calculated as the proportion of patients who achieved a complete response (CR) or partial response (PR). The DCR was calculated as the proportion of patients who achieved a CR, PR, or stable disease (SD). Follow-up was censored on April 10, 2025. Patient characteristics were extracted from the medical records. Patient information was collected, including demographics, pathological characteristics, previous treatment regimens, and imaging assessment. HR-positive status is determined by the expression of estrogen receptor (ER) and/or progesterone receptor (PR), with tumors considered HR-positive when ⩾1% of the tumor cell nuclei show positive staining for ER and/or PR. 27 HER2-positive is defined as immunohistochemistry (IHC) 3+ or IHC 2+ with positive fluorescence in situ hybridization (FISH), while HER2-negative is defined as IHC 0, 1+, or IHC 2+ with negative FISH results. 28 Platinum-sensitive patients were defined as those with PFS exceeding 6 months following platinum-based treatment. For patients with bilateral breast cancer, pathological characteristics were recorded based on their status prior to the initiation of PARPi treatment. In the classification of mutation types, patients harboring both somatic and germline mutations were primarily categorized according to their germline alterations.
HRR gene testing and circulating tumor DNA
All HRR gene alterations relevant to the application of PARPi therapy were extracted from patients’ medical records. A subset of patients underwent genetic testing at the Department of Pathology, Chinese Academy of Medical Sciences. For these individuals, germline DNA was obtained from either peripheral blood or saliva samples. Ten patients included in this study underwent peripheral plasma circulating tumor DNA (ctDNA) testing during real-world treatment prior to PARP inhibitor therapy. All ctDNA analyses in our study were performed using a standardized assay from the same commercial testing institution. Peripheral blood (10 mL) was collected from each patient. Targeted sequencing of 1021 genes that are frequently mutated in breast cancer and other solid tumors was conducted. Circulating DNA was initially extracted from plasma using the QIAamp Circulating Nucleic Acid Kit, and then purified with the DNeasy Blood and Tissue Kit. Samples with greater than 1 µg DNA amount and without obvious degradation were considered qualified. 29 The list of HRR-related genes is shown in Supplemental Table 1. 30 Given that all patients with available ctDNA results in our study received PARPi due to either germline or somatic BRCA2 mutations, HRR-related gene co-mutation was defined as the presence of mutations in any HRR-related gene other than BRCA2.
Statistical analysis
Patient characteristics and treatment administration were summarized by descriptive statistics and were compared via the χ2 test, Fisher’s exact test, or Mann–Whitney U test. PFS was analyzed via the Kaplan–Meier method with the log-rank test between characteristic groups. Univariable and multivariable Cox proportional hazard models were used to evaluate the prognostic impact of baseline clinicopathologic characteristics on PFS. Variables presented p < 0.1 in univariable analysis or strongly associated with efficacy in the clinical setting were included in the multivariable analysis. SPSS 26.0 (IBM, Armonk, NY, USA) and GraphPad Prism 9 (GraphPad Software Inc., San Diego, CA, USA) were used for statistical analysis. Two-sided p < 0.05 was considered to indicate statistical significance.
Results
Patients’ characteristics
In total, 210 patients with breast cancer prescribed PARPi from January 1, 2019, to December 31, 2024, was screened. Sixty-two patients were ultimately enrolled in the analyses. As of April 10, 2025, the median follow-up duration was 11.3 months. Details of the study flowchart are shown in Figure 1. The median age at the initial diagnosis of breast cancer was 51 years (range, 24–56). Thirty-seven patients were HR-positive/human epidermal growth factor receptor 2-negative (HR+/HER2−), and 25 were TNBC. PARPis were administered at a median of the third line of systemic therapy. As for the types of PARPis, 55 received olaparib, 4 received talazoparib, 2 received pamiparib, and 1 received fluzoparib. Fifty-four patients were carriers of BRCA PVs (gBRCA2: N = 30, gBRCA1: N = 19, both gBRCA1 and gBRCA2: N = 1; gBRCA without clearly documented specific mutation types: N = 3; sBRCA2: N = 1), four patients carried gPALB2 PVs, and four with other HRR genes mutations ((gRAD51C: N = 1, gBARD1: N = 1, sCHEK1: N = 1, and sCDK12: N = 1). Twenty-eight (45.2%) had previously received platinum-based treatment, of whom 16 were classified as platinum sensitive. Details of patient characteristics are shown in Table 1.

Study flowchart.
Patients’ characteristics.
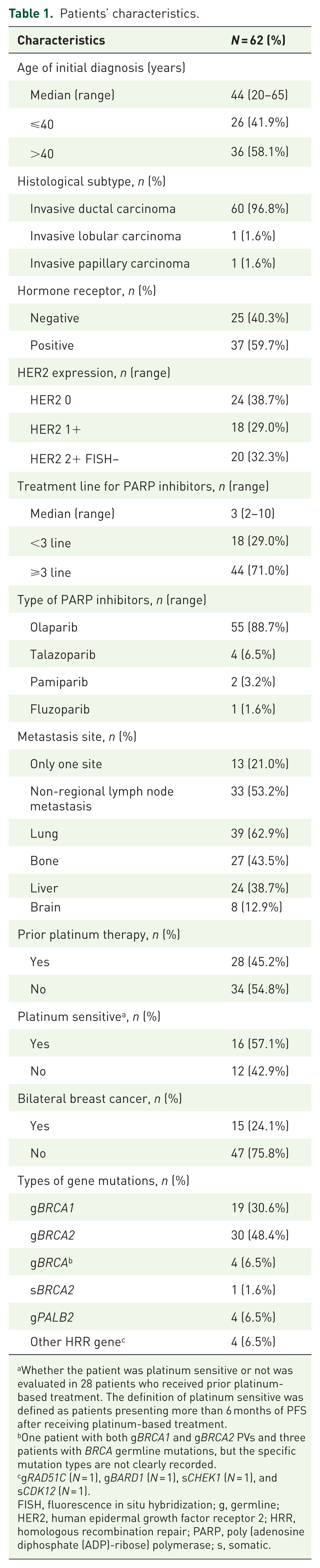
Whether the patient was platinum sensitive or not was evaluated in 28 patients who received prior platinum-based treatment. The definition of platinum sensitive was defined as patients presenting more than 6 months of PFS after receiving platinum-based treatment.
One patient with both gBRCA1 and gBRCA2 PVs and three patients with BRCA germline mutations, but the specific mutation types are not clearly recorded.
gRAD51C (N = 1), gBARD1 (N = 1), sCHEK1 (N = 1), and sCDK12 (N = 1).
FISH, fluorescence in situ hybridization; g, germline; HER2, human epidermal growth factor receptor 2; HRR, homologous recombination repair; PARP, poly (adenosine diphosphate (ADP)-ribose) polymerase; s, somatic.
Efficacy
As of the data cutoff, 15 patients (24.2%) achieved PR, 33 (53.2%) had SD, and 14 (22.6%) experienced PD as their best overall response. The ORR was 24.2% and DCR was 77.4% in all patients (Figures 2(a) and (b) and 3(a)). There were no significant differences in ORR and DCR across subgroup analyses based on different clinicopathological characteristics (Supplemental Table 2). The mPFS in the overall cohort was 6.0 months (95% CI: 4.1–7.9; Figure 3(b)). Among patients harboring germline PVs in BRCA1 and BRCA2, the mPFS was 3.7 months (95% CI: 1.4–6.1) and 8.0 months (95% CI: 4.3–11.7), respectively (p = 0.801; HR = 0.920, 95% CI: 0.459–1.844; Figure 3(c)). The mPFS in patients treated with olaparib and talazoparib was 5.4 months (95% CI: 2.8–8.1) and 5.2 months (95% CI: 0.0–12.8), respectively (p = 0.621, HR = 1.296, 95% CI: 0.460–3.652; Figure 3(d)). Univariate analysis using Cox proportional hazards model was performed on subgroups of clinicopathological factors and treatment information. Patients who were first diagnosed with breast cancer under the age of 40 showed worse PFS compared to those over 40 years old (mPFS: 4.7 vs 7.1, HR = 1.853, 95% CI: 1.030–3.332, p = 0.036), No statistically significant differences in the lines of prior therapy or other baseline clinical characteristics were observed between the two groups (Supplemental Table 3). Patients who received PARPi as third-line or later therapy had significantly worse PFS compared to those treated in earlier lines (5.4 vs 7.9 months, HR = 2.196, 95% CI: 1.057–4.566, p = 0.030; Figure 3(e) and (f)). PFS was numerically shorter among patients who had received prior platinum-based treatment compared to those who had not (5.2 vs 7.3 months, HR = 1.655, 95% CI: 0.935–2.965, p = 0.079). No significant differences were observed in PFS among TNBC and HR+/HER2− patients (5.4 vs 6.3 months, HR = 1.269, 95% CI: 0.703–2.291, p = 0.428; Supplemental Table 4), as well as other factors, such as HER2 expression, platinum-sensitive, bilateral breast cancer, gene mutation subtypes, and PARPi types. Multivariate analysis demonstrated that age (⩽40 vs >40; HR = 2.281, 95% CI: 1.242–4.189, p = 0.008), prior platinum-based treatment (Yes vs No; HR = 2.172, 95% CI: 1.173–4.020, p = 0.014), and the number of treatment line (⩾3 line vs <3 line; HR = 2.429, 95% CI: 1.156–5.103, p = 0.019) were independent predictive factors for PFS (Table 2).

Tumor responses of patients in the overall study cohort. (a) Waterfall plot summarizing maximum change in tumor size from baseline assessed according to RECIST (version 1.1). The dotted line at −30% represents partial response. (b) Swimmer plot summarizing Progression events and duration of response according to RECIST (version 1.1).

Efficacy of the overall cohort and different subgroups. (a) ORR and DCR for overall patients. (b) PFS for overall patients. (c) PFS for gBRCA1 or gBRCA2 PV carriers. (d) PFS for patients treated with olaparib or talazoparib. (e) PFS for patients who received PARPi as third-line or later therapy or treated in earlier lines. (f) PFS for patients who were first diagnosed with breast cancer after the age of 40 years or younger than 40 years.
Univariate and multivariate Cox proportional hazard regression analysis of predictors of PFS in metastatic breast cancer patients treated with PARP inhibitor.

Whether the patient was platinum sensitive or not was evaluated in 28 patients who received prior platinum-based treatment. The definition of platinum sensitive was defined as patients presenting more than 6 months of PFS after receiving platinum-based treatment.
One patient with both gBRCA1 and gBRCA2 PVs and three patients with BRCA germline mutations, but the specific mutation types are not clearly recorded.
gRAD51C (n = 1), gBARD1 (n = 1), sCHEK1 (n = 1), and sCDK12 (n = 1).
FISH, fluorescence in situ hybridization; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; HRR, homologous recombination repair; PARP, Poly (adenosine diphosphate (ADP)-ribose) polymerase; PFS, progression-free survival; NA, not available.
Safety
A total of 56 individuals had treatment-related adverse events (TRAEs) documented in their medical records. The incidence of AEs of all grades was 62.5% (35/56) (Table 3). A total of 18 patients (18/56, 32.1%) required dose reduction (14 due to grade 3 or higher TRAEs and 4 on their own initiative). The most common AEs in all grades were anemia (30.4%), nausea (21.4%), and leukopenia (17.9%). Among the grade 1/2 toxicities, nausea was the most common (19.6%), followed by anemia (14.3%), leukopenia (12.5%), and elevated alanine aminotransferase/aspartate aminotransferase (10.7%). Hematologic toxicity (anemia (16.1%), leukopenia (5.4%), and thrombocytopenia (3.6%)) was the most common grade ⩾3 AEs. There was one case of grade 3 Guillain–Barre syndrome. No treatment-related death was registered.
Incidence of TRAEs in patients with AE was recorded and graded using CTCAE v5.0 a (N = 56).

AEs of 56 patients with AE recorded were shown in this table.
AE, adverse event; CTCAE, Common Terminology Criteria for Adverse Events; TRAE, treatment-related adverse event.
Genomic landscape of ctDNA
A total of 10 patients in our study underwent peripheral blood ctDNA testing. The oncoplot of gene alterations for 10 samples is presented in Figure 4(a). All 10 patients with available ctDNA results received PARPis due to BRCA2 mutations (gBRCA2: N = 9; sBRCA2: N = 1). Somatic mutations were detected in nine of these patients. Clinical information for all patients with available ctDNA data is provided in Supplemental Table 5. Somatic BRCA2 mutations were detected in ctDNA from three patients. Among them, one patient carried only a somatic BRCA2 mutation without a germline BRCA2 alteration and received PARPi based on this somatic finding. The other two patients harbored both germline BRCA2 and somatic BRCA2 mutations identified in ctDNA. The remaining seven patients had germline BRCA2 mutations but no somatic BRCA2 alterations detected in ctDNA. The most frequently observed gene mutations were CREBBP, FAT1, MLL, NF1, and SLX4 (30%, 3/10). No gene mutations associated with PARPi efficacy were identified. Seven patients harbored co-mutations in HRR-related genes (Figure 4(b)). The list of HRR-related genes is shown in Supplemental Table 1. Patients harboring co-mutations in HRR-related genes exhibited a numerically shorter PFS compared with those without co-mutations (6.0 vs 10.1 months; HR = 6.032, 95% CI: 0.704–51.646, p = 0.065; Figure 4(c)).

The co-occurrence of HRR-related gene mutations in patients with ctDNA testing. (a) The oncoplot of gene alterations. All 10 patients with available ctDNA results received PARP inhibitors due to BRCA2 mutations (gBRCA2: N = 9; sBRCA2: N = 1). *The patient carried only a somatic BRCA2 mutation without a germline BRCA2 alteration. (b) The co-mutations of HRR-related genes. *Patient 01 carried only a somatic BRCA2 mutation without a germline BRCA2 alteration and received PARP inhibitors due to sBRCA2 mutations. (c) PFS for patients with or without co-mutations of HRR-related genes.
Discussion
PARPis, particularly olaparib and talazoparib, have profoundly reshaped treatment paradigms for germline BRCA1/2-mutated HER2-negative MBC, offering a targeted and mechanism-based treatment approach. While the efficacy and safety profiles of PARPis have been well characterized in several large-scale randomized clinical trials, the majority of existing evidence is derived from Western populations. Although the pivotal OlympiAD and EMBRACA studies included Asian patients, these subpopulations accounted for only a small minority of the overall cohorts (approximately 10%–30%). Moreover, in real-world clinical practice in China, PARPis are often administered in later lines of therapy due to limited drug accessibility, delayed genetic testing, and high treatment costs. Data regarding the real-world efficacy and tolerability in Chinese patients remain limited and inconclusive. Given potential differences in genetic background, treatment accessibility, and clinical practice patterns, it is imperative to evaluate the performance of PARPis in a Chinese clinical setting. Accordingly, our study aims to retrospectively assess the real-world efficacy and safety of PARPis in Chinese MBC patients, thereby providing supportive evidence to inform clinical decision-making and optimize the therapeutic value of PARPis in this specific population.
Olaparib and talazoparib have demonstrated superior efficacy over chemotherapy in gBRCA-mutant MBC, with improved PFS and ORR confirmed across multiple phase III trials.21–23,31 Additional data from Japanese cohorts confirmed an ORR of 57.9 % and mPFS of 7.2 months in patients with advanced disease. 32 Real-world evidence has also echoed these results, reporting mPFS of 7–8 months and ORR around 50% among gBRCA-mutated patient.33,34 Our study included 62 Chinese patients in a multicenter with MBC who received PARPi therapy in a real-world clinical setting. The results showed an mPFS of 6.0 months, with an ORR of 24.2% and a DCR of 77.4%. Our findings suggest that PARPis are effective in Chinese patients with MBC carrying HRR-related mutations. Although the mPFS observed in our study was numerically lower than the 7–9 months reported in previous trials, this difference may be attributed to the more heavily pretreated population in our cohort. Notably, in pivotal trials such as OlympiAD and EMBRACA, patients had received no more than two prior lines of therapy for advanced disease, whereas the median number of prior treatment lines in our study was 3. This reflects real-world treatment patterns in China, where off-label application, limited drug accessibility, financial constraints, and delays in germline BRCA testing often result in later-line use of PARP inhibitors. During the study period, PARPis were prescribed off-label and had not yet been included in the national reimbursement list for patients with BRCA-mutant MBC. This regulatory and financial limitation significantly constrained physicians’ treatment choices, resulting in markedly reduced clinical adoption of PARPis. Thus, chemotherapy remained the predominant front-line treatment option in routine practice. These factors underscore the scarcity of real-world data on PARPis efficacy and safety in Chinese patients. Our findings indicate that patients treated with PARPis in the third-line or later setting exhibited a shorter PFS. Previous studies have also reported a decline in ORR with an increasing number of prior treatment lines, 35 suggesting that the number of prior treatment lines may be a critical determinant of PARPi efficacy.
Predictive factors for the efficacy of PARP inhibitors have long been a focus of clinical interest. Previous studies have demonstrated a significant association between the clinical benefit rate and the platinum-free interval among platinum-sensitive, resistant, and refractory subgroups, with CBRs of 69%, 45%, and 23%, respectively, in patients with BRCA1/2-mutated ovarian cancer treated with olaparib. 36 However, in our study, platinum sensitivity was not significantly associated with the efficacy of PARPis. Moreover, prior platinum-based treatment was identified as an independent negative predictor of PARPis efficacy. This may be attributed to the fact that patients who had previously received platinum-based therapy in our cohort were treated at more advanced lines of therapy. Furthermore, prior platinum-based therapy can induce BRCA reversion mutations, enabling tumors to regain HRR capacity and thereby develop cross-resistance to PARPis. 37 Nevertheless, given the retrospective nature of our study and the potential for confounding bias, these findings warrant validation in larger prospective cohorts. In addition, our findings suggest that younger breast cancer patients experienced poorer efficacy with PARPis. This indicates that, although BRCA mutations are more frequently observed in younger patients, the clinical benefit derived from PARPis in this population may be limited. Although no studies have directly compared the clinicopathologic characteristics and efficacy of PARPis between younger and older patients with advanced BRCA-mutated breast cancer, several studies have reported significant differences in molecular classifications and genomic alterations according to age at diagnosis. Women <40 years demonstrated significantly higher mutation and amplification burdens compared with those aged 40–60 years or >60 years, and these genomic differences were associated with shorter PFS, recurrence-free survival, and disease-specific survival outcomes.38,39 This also suggests that younger patients with BRCA-mutated breast cancer may exhibit more aggressive tumor biology, higher tumor burden, and greater genomic instability, which could contribute to poorer treatment outcomes, even though these tumors are intrinsically sensitive to DNA-damaging agents. Moreover, no significant difference in the efficacy of PARPis was observed between TNBC and HR+/HER2− patients, which is consistent with the findings of the OlympiAD and EMBRACA studies. Previous studies analyzing the tumor biological features and prognosis of young breast cancer patients carrying BRCA mutations have similarly shown no significant difference in 8-year OS between HR-positive and HR-negative subtypes. HR positivity did not appear to confer a favorable prognostic value in these patients, particularly among those with luminal A-like tumors or harboring BRCA2 PVs. 40 A recent network meta-analysis confirmed that olaparib and talazoparib have similar efficacy and safety in BRCA-mutated MBC, with no significant differences in PFS, OS, ORR, or discontinuation rates. 41 Our study also found no significant difference in efficacy among different PARPis. However, the majority of patients in our cohort received olaparib, while the number of patients treated with other PARPis was relatively small. Therefore, these findings should be confirmed in future studies. Moreover, in our study, factors such as HR status, HER2 status, and whether the patient had bilateral primary breast cancer were not significantly associated with PFS.
As key members of the HRR pathway, gBRCA1/2m remains the most important predictor of PARPi efficacy. 42 However, homologous recombination is a multifactorial process involving many different proteins beyond BRCA1 and BRCA2. Genomic alterations affecting genes encoding for these proteins may impair HRR and become a target of synthetic lethality for PARPis as well as other DDR-targeting agents. 43 Moreover, somatic BRCA1 and BRCA2 genetic alterations are estimated in approximately 2.5% of sporadic tumors, 44 raising the question of whether patients with sBRCAm or non-BRCA HRR-related gene alterations may also derive clinical benefit from PARPis. 45 TBCRC-048 highlighted the difficulty in studying the predictive effect of rare gene alterations. High tumor or plasma ctDNA homologous recombination deficiency (HRD) scores correlated with positive response outcomes, particularly among PALB2-mutated tumors.46,47 In a retrospective study, patients with sBRCA and gPALB2 derive similar benefit from PARPi as those with gBRCA alterations (gBRCA vs sBRCA/gPALB2, PFS: 6.3 vs 5.4 months, HR = 1.37, 95% CI: 0.77–2.43; OS: 16.2 vs 21.2 months, HR = 1.45, 95% CI: 0.74–2.86). 48 A real-world study conducted in Mayo Clinic also found no significant difference in time-to-treatment-failure between gBRCA1, gBRCA2, or gPALB2 PV carriers with MBC. In addition, a novel immunofluorescence-based assay measuring RAD51 foci has been shown to identify homologous recombination proficient tumors that are misclassified as HRD by mutational signatures, and to demonstrate a stronger correlation with PARPi efficacy. 49 In our study, patients with gPALB2 mutations, other HRR gene alterations, as well as somatic BRCA mutations were still able to derive clinical benefit from PARPis treatment. Although the number of such patients in our cohort was limited, our data may provide preliminary numerical evidence suggesting that PARPis could be considered as a therapeutic option for these subgroups.
Studies have explored the association between co-occurring genetic alterations and the clinical activity of PARPi in BRCA-mutant cancers, but their correlation remains inconsistent. A small retrospective study reported that PARPi appeared more effective in patients with BRCA2mut/SPOPmut than in those with BRCA2mut/SPOPwt metastatic castration-resistant prostate cancer. 50 Conversely, loss-of-function mutations in HRR-related genes such as TP53BP1, RIF1, or PAXIP1 have been observed in BRCA1-, BRCA2-, or PALB2-mutant breast cancer patients with acquired PARPi resistance, though the underlying mechanisms remain unclear. 51 In addition, mutations in other HRR-related genes have also been found to be associated with PARPi efficacy. SLX4 is a DNA repair protein that preserves genomic stability through multiple DNA damage response pathways. When recruited to sites of DNA damage, SLX4 interacts with PARP1 and undergoes PARylation. This process can be inhibited by PARPi treatment, suggesting a functional interplay between these two proteins. 52 In vitro studies further demonstrated that SLX4 deficiency is associated with a higher sensitivity to PARPi, 53 although this correlation lacks robust clinical validation. In our study, while no single-gene alteration was found to be directly associated with PARPis efficacy, patients harboring co-mutations in HRR-related genes exhibited a trend toward shorter PFS compared with those without such co-mutations. The underlying mechanism for this observation remains unclear. However, a previous study reported that the HR defect caused by BRCA1 deficiency can be reversed through concomitant loss of 53BP1 (TP53BP1), a key HRR-related gene involved in the non-homologous end-joining pathway. 54 This finding suggests a potential mechanism by which co-mutations in HRR pathway genes may compromise the efficacy of PARPis in patients with BRCA-mutant tumors. However, given our limited sample size, these results warrant confirmation in larger prospective studies.
PARPis lead to the impairment of alternative DDR pathways and thus to irreparable genomic damage and cell death. 18 Therefore, hematopoietic cells with a high proliferative rate are more susceptible to damage, making hematologic toxicity the most common AE with PARPis and a major concern in its clinical application. In the OlympiAD trial, olaparib demonstrated a tolerable safety profile. The most common AE associated with olaparib is nausea (58.0%), followed by anemia (40.0%) and vomiting (29.8%). Olaparib had fewer grade ⩾3 AEs than chemotherapy (36.6% vs 50.5%). The rate of treatment discontinuation due to toxic effects in the olaparib and chemotherapy groups was 4.9% and 7.7%. Dose reduction was most commonly due to anemia in the olaparib group (in 13.7% of patients). 31 Talazoparib was associated with a higher incidence of grade 3–4 hematologic toxicities (mainly anemia), but fewer grade ⩾3 nonhematologic and hepatic toxicities compared to standard chemotherapy, with lower discontinuation rates and manageable safety through dose modifications. 22 Quality-of-life assessments and patient-reported outcomes consistently favored the talazoparib arm, with notable delays in clinically meaningful deterioration. 55 In our study, the overall incidence of AEs was slightly lower than OlympiAD and EMBRACE. Anemia remains the most common AE (30.4%), followed by nausea (21.4%), suggesting that PARPis are generally well tolerated in the Chinese population.
Our study has several limitations. First, the small sample size may have introduced potential small-sample bias. Second, most of the enrolled patients were treated with olaparib, making it difficult to compare the efficacy among different PARPis. Third, due to the limited number of patients with available ctDNA results, we were unable to evaluate its predictive value of PARPis treatment; further validation with larger cohorts is needed. Finally, the efficacy and toxicity data were abstracted from electronic medical records and may not be as comprehensive or accurate as data collected in prospective studies.
Conclusion
This retrospective study evaluated the efficacy and safety of PARPis in Chinese MBC patients harboring HRR-related gene mutations in a real-world clinical setting. PARPis showed favorable efficacy and tolerability. However, treatment efficacy was relatively poorer in younger patients, third-line or later therapy, and patients with prior platinum-based treatment. Patients harboring co-mutations in HRR-related genes may also exhibit poor prognosis. Notably, PARPis also showed therapeutic potential in patients with other germline HRR-related genes. These findings suggest that PARPis may offer a promising treatment option for MBC patients with HRR-related gene mutations. Nevertheless, the predictive factors for PARPis efficacy require further validation in studies with larger sample sizes.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251411096 – Supplemental material for Efficacy and safety of PARP inhibitors in metastatic breast cancer patients with homologous recombination repair pathway gene mutations: a retrospective multicenter real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359251411096 for Efficacy and safety of PARP inhibitors in metastatic breast cancer patients with homologous recombination repair pathway gene mutations: a retrospective multicenter real-world study by Xuenan Peng, Ruixia Song, Ying Fan, Jiayu Wang, Qiao Li, Hongnan Mo, Jiani Wang, Yang Luo, Shanshan Chen, Xiaoying Sun, Jintao Zhang, Pin Zhang, Binghe Xu, Bo Lan and Fei Ma in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251411096 – Supplemental material for Efficacy and safety of PARP inhibitors in metastatic breast cancer patients with homologous recombination repair pathway gene mutations: a retrospective multicenter real-world study
Supplemental material, sj-docx-2-tam-10.1177_17588359251411096 for Efficacy and safety of PARP inhibitors in metastatic breast cancer patients with homologous recombination repair pathway gene mutations: a retrospective multicenter real-world study by Xuenan Peng, Ruixia Song, Ying Fan, Jiayu Wang, Qiao Li, Hongnan Mo, Jiani Wang, Yang Luo, Shanshan Chen, Xiaoying Sun, Jintao Zhang, Pin Zhang, Binghe Xu, Bo Lan and Fei Ma in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
