Abstract
Poly(ADP-ribose)polymerase (PARP) inhibitors are targeted therapy for cancers with homologous repair deficiency (HRD). They were first approved for ovarian cancer and have changed current treatment strategies. They have also demonstrated efficacy in HER2-negative metastatic breast cancer and advanced prostate cancer with
Background
Anti-poly(ADP-ribose)polymerase (PARP) therapies have been developed for various solid tumors such as ovarian, breast and prostate cancers, mainly based on
PARP inhibitors have shown efficacy not only in ovarian cancer patients with
Olaparib and other PARP inhibitors have also been evaluated in other solid tumors based on a somatic or germline homologous recombination defect. In the OlympiAD trial, patients with HER2-negative metastatic breast cancer with a germline
Among patients with castration-resistant prostate cancer, Mateo and colleagues found that 88% of the patients with a somatic homologous recombination defect (
As a result, the US FDA has approved olaparib and talazoparib as a treatment for HER2-negative metastatic breast cancer with a
Since the number of indications for PARP inhibitors is increasing, the number of patients requiring genetic counseling and testing is also likely to increase. However, not all these patients have a familial predisposition and/or germline genetic mutation. In ovarian cancer, the prevalence of
This article reviews international guidelines on indications for oncogenetic counseling, considering family predisposition and therapeutic indications, and proposals for new referral systems in ovarian, breast and prostate cancer based on personal or familial history of cancer, type of tumor and PARP inhibitor indications.
Methods
This review was conducted in accordance with PRISMA guidelines. PubMed, Cochrane, Medline and Google Scholar were used to index medical guidelines and publications reporting prevalence of somatic and/or germline mutations in ovarian, breast and prostate cancer, using appropriate search terms. Papers published in either English or French were eligible. The literature search used variations and Boolean connectors of the key terms. An exploratory search was conducted with the various associations of the terms (MESH if possible) ‘genetic counselling’, ‘genetic testing’, ‘breast neoplasms’, ‘prostate neoplasms’, ‘BRCAness’, ‘BRCA1 genes’, ‘BRCA2 genes’, ‘breast cancer, prostate cancer, ovarian cancer, familial’, ‘guideline’, ‘recommendation’, ‘neoplastic syndromes, hereditary’, ‘multi-gene panel’. The websites of associations, colleges and learned societies listing the various recommendations were also examined.
For the selection of guidelines, we considered only the recent and national guidelines or recommendations for oncogenetic care and indications for genetic testing, published in English and/or French until September 2018. First, articles were screened on titles and publication dates, excluding duplicates, surgical, molecular, technical or psychological articles. The different steps are summarized in Figure 1.

Flow chart of the systematic review following PRISMA guidelines.
Results
Twenty-four recommendations for oncogenetic care and indications for genetic testing were examined (Figure 1). The indications for PARP inhibitors are summarized in Table 1.
Indications of PARP inhibitors.

BC, breast cancer; EMA, European Medical Agency; FDA, Food and Drug Administration; OC, ovarian cancer; PC, prostate cancer.
Ovarian cancer
Given the results confirming the efficacy of olaparib, the first FDA-approved PARP inhibitor, most guidelines have extended indications for germline genetic testing at diagnosis to all patients with high-grade serous non-mucinous epithelial ovarian cancer, whatever the patient’s age.4,6,15–28 Some guidelines even suggest extending this indication to other histology types (NICE, GECKO).26,27 These recommendations are reinforced by the SOLO2 results, as patients with endometrioid ovarian cancer were also included in this study. 5 The indications for germline genetic testing in ovarian cancer according to the different guidelines are summarized in Table 2.
Indications for addressing and/or genetic analyses in case of ovarian cancers.

ACOG, American College of Obstetricians and Gynecologists; AGO, Association of Gynecologic Oncology; BC, breast cancer; ESMO, European Society for Medical Oncology; GECKO, Genetics Education Canada – Knowledge Organization; INCA, Institut National du Cancer; NBCG, Norwegian Breast Cancer Group; NCCN, National Comprehensive Cancer Network; NICE, National Institute for Health; OC, ovarian cancer; SEOM, Sociedad Española de Oncología Medica.
1 including cancer of the peritoneum and fallopian tubes should be considered a part of the spectrum of the hereditary breast and OC syndrome.
2 indication for referral and not for testing.
4 close relative: defined as a first-degree relative (mother, sister, daughter) or second-degree relative (grandmother, granddaughter, aunt, niece).
Nowadays, a genetic consultation is recommended for all patients with high-grade non-mucinous epithelial ovarian cancer at initial diagnosis, in order to facilitate genetic testing for germline and somatic
In some countries such as France, for patients without prior oncogenetic testing and with a platinum-sensitive recurrence, rapid-access genetic testing pathways have been developed to decrease the time to consultation (<3 months) and have been validated by the French National Cancer Institute.
25
The indication for tumor testing has also been included in the guidelines in addition to germline testing. This somatic testing is done at the same time as the first germline genetic test, or can be prescribed for patients with prior negative germline genetic testing. For patients with platinum-sensitive recurrence and without prior germline screening, tumor and germline testing are recommended at the same time. For patients with germline-negative screening, tumor testing is mandatory. With the new results of the SOLO1 study confirming the efficacy of olaparib as first-line maintenance treatment among patients with
Breast cancer
The recommendations are presented in Table 3.
Indications for addressing and/or genetic analyses in the case of breast cancers.

ACOG, American College of Obstetricians and Gynecologists; AGO, Association of Gynecologic Oncology; ASCO, American Society of Clinical Oncology; BC, breast cancer; BeSHG, Belgian Society for Human Genetics; ESMO, European Society for Medical Oncology; GECKO, Genetics Education Canada – Knowledge Organization; INCA, Institut National du Cancer; NBCG, Norwegian Breast Cancer Group; NCCN, National Comprehensive Cancer Network; NICE, National Institute for Health; OC, ovarian cancer; PC, prostate cancer; SEOM, Sociedad Española de Oncología Medica; SIGN, Scottish Intercollegiate Guidelines Network.
1 including cancer of the peritoneum and fallopian tubes should be considered a part of the spectrum of the hereditary breast and OC syndrome.
2 indication for referral and not for testing.
3 close blood relatives include first-, second-, and third-degree relatives on same side of family (NCCN).
4 close relative: defined as a first-degree relative (mother, sister, daughter) or second-degree relative (grandmother, granddaughter, aunt, niece).
There are some small differences between guidelines but they generally recommend genetic counseling for patients with breast cancer at an early age, or in the event of triple-negative synchronous or metachronous bilateral breast cancer, or combined with ovarian cancer, or a family history of breast cancer, male breast cancer, or ovarian cancer.18–20,23,26–30 In addition, Ashkenazi, Icelandic or French-Canadian heritage are also risk factors for
Unlike ovarian cancer, there have been no recent modifications of the guidelines concerning the indications for oncogenetic counseling in association with indications for PARP inhibitors. However, the FDA has recently approved olaparib for patients with HER2-negative metastatic breast cancer and a
Prostate cancer
Few guidelines are available regarding oncogenetic counseling for familial risks of prostate cancer (Table 4). Germline mutations affect fewer than 3% of prostate cancer patients. Only patients with early (<55 years) and/or aggressive prostate cancer (Gleason ⩾ 7), associated with an evocative familial history of breast cancer (early, triple-negative, bilateral, multiple), ovarian cancer or other Gleason ⩾ 7 prostate cancers are referred to oncogenetic counseling (especially in the USA and Australia).20,21,29,33
Indications of addressing and/or genetic analyses in case of prostate cancers.
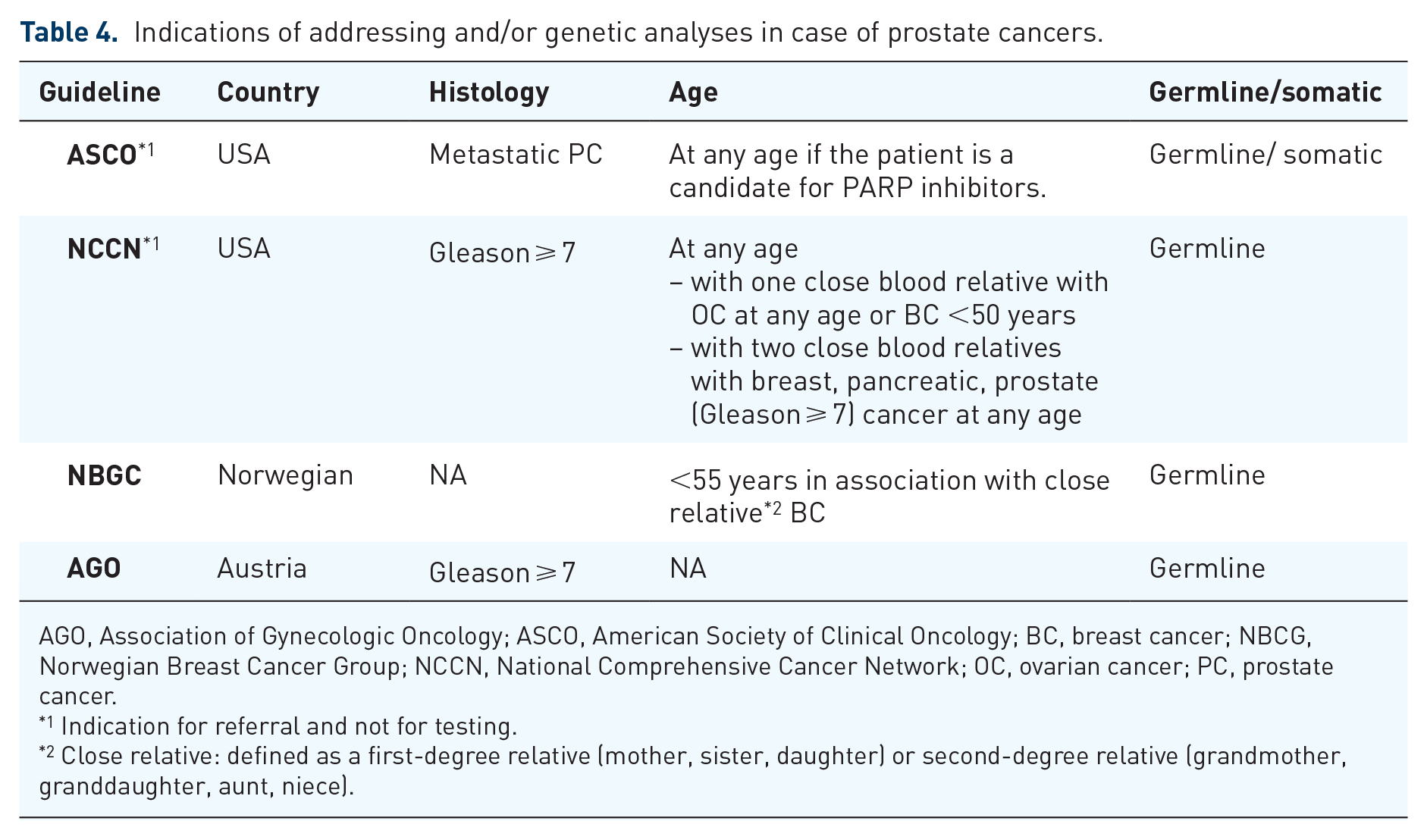
AGO, Association of Gynecologic Oncology; ASCO, American Society of Clinical Oncology; BC, breast cancer; NBCG, Norwegian Breast Cancer Group; NCCN, National Comprehensive Cancer Network; OC, ovarian cancer; PC, prostate cancer.
1 Indication for referral and not for testing.
2 Close relative: defined as a first-degree relative (mother, sister, daughter) or second-degree relative (grandmother, granddaughter, aunt, niece).
PARP inhibitors are showing very encouraging preliminary results in prostate cancer and phase III trials are ongoing.13,34 As in breast cancer, tumor tests have not been included in the current guidelines for genetic testing. Based on this efficacy data, the FDA has approved olaparib monotherapy for patients with metastatic castration-resistant prostate cancer (mCRPC) with germline or somatic
Discussion and proposals
With PARP inhibitors approved in ovarian, breast and prostate cancers, the classical care route consisting of an initial germline genetic test after genetic counseling is no longer efficient. New care pathways need to be developed with early tumor testing, based on predisposing risk factors, and a new approach to genetic counseling notably for patients without a family history of cancer. For patients with a family history, an initial genetic consultation before any germline testing remains mandatory.
In ovarian cancer, oncogenetic counseling is still recommended at diagnosis, whatever the patient’s age. Some oncology teams have specific care pathways for these patients with rapid-access genetic testing and pre-counseling telephone interviews, in order to have genetic test results at the time of recurrence. The limited number of patients and the high risk of family predisposition (around 15–25%) have made it possible to continue to refer patients for rapid-access genetic testing and an oncogenetic consultation if they require treatment promptly.
35
However, care pathways are evolving quickly. With the recent results of the SOLO1 study, underlining the potential benefit of maintenance olaparib after first-line platinum chemotherapy in
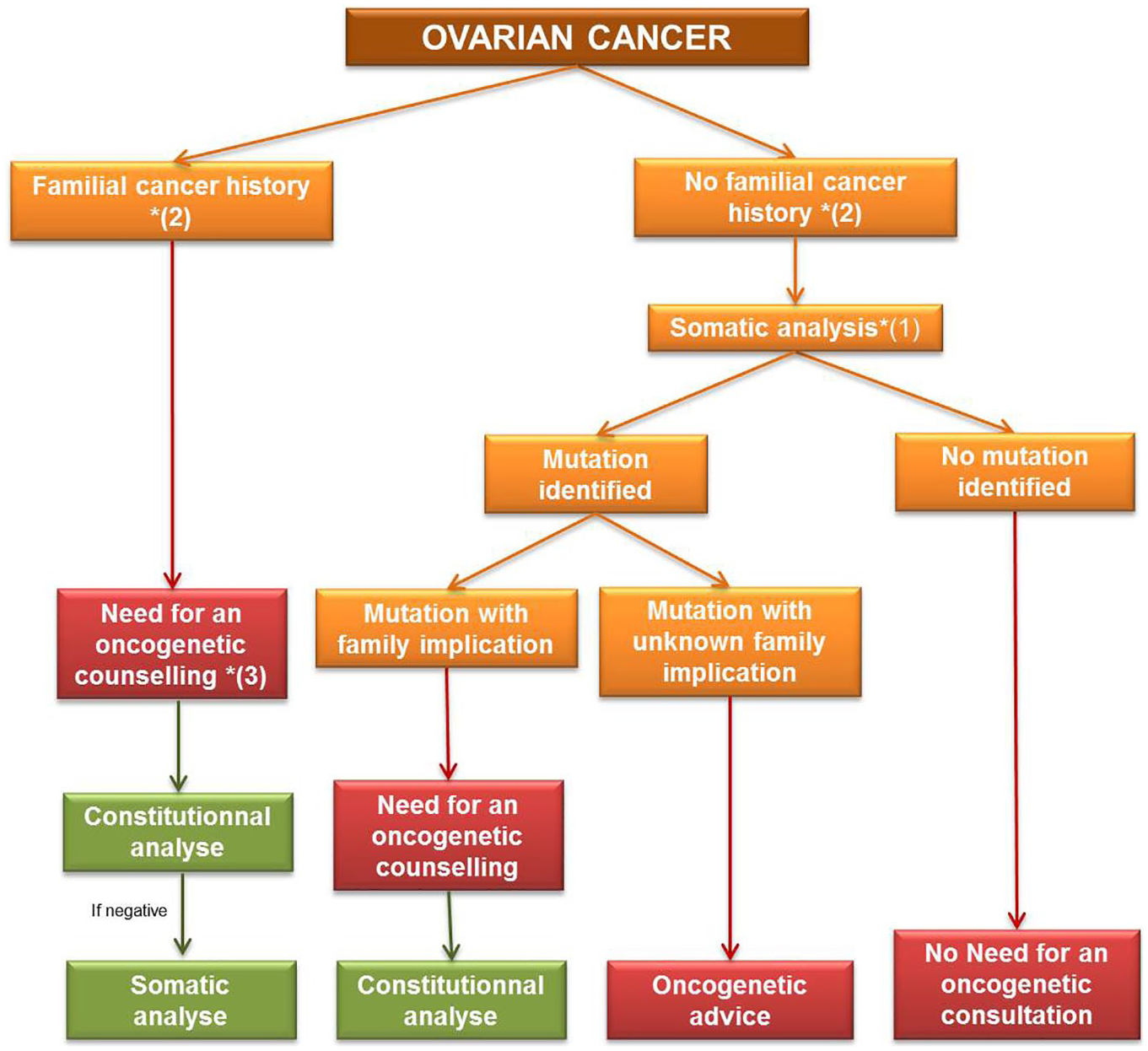
Proposal of care pathway for patients with ovarian cancers at initial diagnosis.
Breast and prostate cancer affect a larger number of patients, so PARP inhibitor indications in these types of tumors will concern more patients. These patients also present a lower risk of family predisposition than ovarian cancer patients. In fact, the risk of presenting a family predisposition is approximately 1–2% in prostate cancer and 5–7% in breast cancer.11,13 On the other hand, only 3% of breast cancer patients present only a somatic mutation of
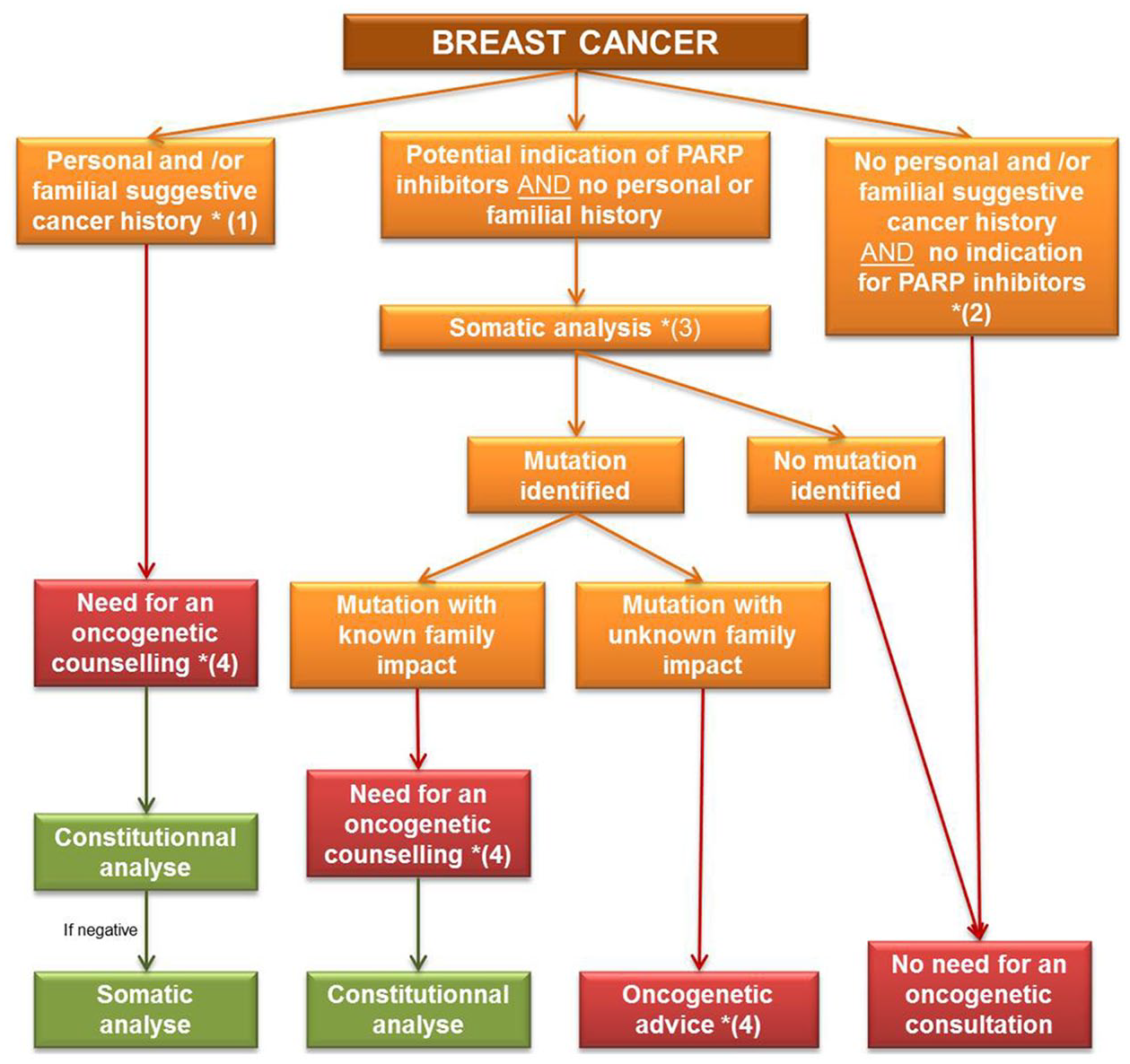
Proposal of care pathway for patients with breast cancers.

Proposal of care pathway for patients with prostatic cancers.
Although the indications for referral are being optimized, the number of oncogenetic consultations is likely to increase and some teams have already proposed training programs in which health professionals (oncologists, nurses, etc.) discuss how the indications for genetic testing and the turnaround time for consultations can be improved.15,37–39 As reported by George and colleagues, new genetic testing pathways have been proposed.
15
In this study, genetic counseling is performed by a trained oncologist prior to germline testing. This organization has shortened waiting times for results and reduced the required resources, with good feedback from patients and medical teams. Likewise, ENGAGE (Evaluating a Streamlined Onco-genetic
Another emerging approach is telephone interviews or telemedicine before face-to-face genetic counseling and testing.37,40,41 Patients can be better selected for oncogenetic consultations and the initial information can be given to patients by trained caregivers during telephone interviews or face-to-face consultations. In a French study, a first telephone interview was conducted by genetic counselors and/or physicians to identify the familial risk requiring a complete genetic work-up. 37 The study showed that pre-counseling telephone interviews were cost-effective as they did not lead to consultations in 39% of cases due to the absence of a significant medical history or by designating a more appropriate index case. In a Swedish study, telephone interviews were conducted by a well-trained nurse and a physician to replace the initial part of face-to-face counseling sessions. 40 Participants reported a high satisfaction rate regardless of whether counseling was conducted by telephone or in person.
A US team offered telephone counseling instead of usual care to healthy women with a risk of a
To save time and help oncogeneticists, genetic counselors can take on some of the physicians’ tasks and deliver genetic counseling.
Even if new care pathways need to be developed in this theranostic era, somatic testing still has limitations. Tumor samples have to be available and in sufficient quantity. There are other specific problems such as tumor heterogeneity and the characterization of driver or passenger mutations. There are also technical limitations, as with Formalin-Fixed Paraffin-Embedded (FFPE) samples, which can present altered DNA, or chemical pre-treatments which increase genomic instability. These limitations reinforce the need for a genetic consultation.
In this context, oncogenetic consultations are a key aspect of the healthcare system and are required for patients with germline and somatic mutations and/or a family history and/or unavailability for tumor testing. During these consultations, potential risks and benefits of genetic testing, the probability of finding a mutation, and the implications for the individual and the family are detailed. They also make it possible to explain variants of uncertain significance or a negative result. This requires time for genetic testing, expertise and follow up. Oncogenetic consultations allow results to be delivered within the family and to organize preventive family care. The disclosure of a deleterious mutation is a sensitive moment and requires medical and psychological follow up for patients and their families. These new proposed care pathways need to be evaluated in terms of efficiency and satisfaction of medical staff, patients and their families.
In conclusion, the advent of new cancer therapies such as PARP inhibitors is increasing the demand for genetic counseling. The emergence of this personalized medicine is leading to the systematic molecular characterization of advanced tumors for therapeutic purposes. Genetic testing and counseling pathways need to adapt to this theranostic purpose.
Footnotes
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Conflict of interest statement
Prof Joly’s COI are: TESARO GSK Roche Clovis and Astra Zeneca.
