Abstract
The standard treatment for non-metastatic muscle-invasive bladder cancer (MIBC) is cisplatin-based neoadjuvant chemotherapy followed by radical cystectomy or trimodality therapy with chemoradiation in select patients. Pathologic complete response (pCR) to neoadjuvant chemotherapy is a reliable predictor of overall and disease-specific survival in MIBC. A pCR rate of 35–40% is attained with neoadjuvant cisplatin-based chemotherapy. With the approval of immune checkpoint inhibitors (ICIs) for the treatment of metastatic urothelial cancer, these agents are now being studied in the neoadjuvant setting for MIBC. We describe the results from clinical trials using single agent ICI, ICI/ICI and ICI/chemotherapy combination therapies in the neoadjuvant setting for MIBC. These single-arm clinical trials have demonstrated safety and pCR comparable to cisplatin-based chemotherapy. Neoadjuvant ICI is a promising approach for cisplatin-ineligible patients, and the role of adding ICIs to cisplatin-based chemotherapy is also being investigated in randomized phase III clinical trials. Ongoing biomarker research to suggest a response to neoadjuvant ICIs will also guide appropriate treatment selection. We also describe the studies using ICIs for adjuvant therapy and in combination with chemoradiation.
Background
Urothelial carcinoma (UC) of the bladder is a common and deadly disease. About 81,400 new cases of bladder cancer were diagnosed in 2020 in the United States alone with an estimated 18,000 deaths/year. 1 Although most UC arises from the bladder, about 10% arise from the upper urinary tract, with a small proportion originating in the urethra. UC is histologically divided into low-grade or high-grade tumors, each associated with distinct genomic alterations and differences in prognosis. 2 Low-grade tumors are almost uniformly non-invasive and have a 5-year survival rate of 95%. 3 In contrast, high-grade tumors often become muscle-invasive [so called muscle-invasive bladder cancer (MIBC)]. MIBC has a propensity to become metastatic, and once metastatic, it is associated with a 5-year survival of only 15%. 4
The most commonly employed standard of care for MIBC is neoadjuvant cisplatin-based chemotherapy (NAC) followed by radical cystectomy with lymph node dissection. NAC reduces tumor bulk and treats micro-metastases, and this approach produces a pathologic complete response (pCR) rate of 35–40%.5,6 Patients who have a pCR with NAC have a significantly higher 5-year survival compared with those with residual disease (85% versus 50%, respectively). 5 However, UC is largely a disease of the elderly, 7 and approximately 30–50% of patients are not cisplatin eligible due to renal dysfunction, Eastern Cooperative Oncology Group performance status ⩾2, or comorbidities such as cardiac dysfunction, neuropathy and hearing loss.8,9 Additionally, since radical cystectomy is associated with high morbidity and mortality, and can negatively impact the quality of life of patients, efforts to incorporate bladder sparing approaches are ongoing with trimodality chemoradiation often utilized. 10
Given the encouraging results of immune checkpoint inhibitors (ICIs) in metastatic UC, and the Food and Drug Administration approval of three anti-programmed death-1 (anti-PD-1) and PD-1 ligand (PD-L1) agents for these patients, these drugs are now being studied in the (neo)adjuvant setting for MIBC. Single-agent ICI and ICI combined with another ICI, standard NAC, and other drugs are being studied as neoadjuvant treatment for MIBC. These treatment options may improve the efficacy of standard NAC and present an option for cisplatin-ineligible patients. In patients who are unable to undergo cystectomy, ICI is being studied in combination with radiation therapy or chemoradiation as a bladder sparing approach. Additionally, in the adjuvant setting, ICIs are being explored in high-risk patients to reduce the chance of distant recurrence.
In this review, we summarize the available data and ongoing trials using ICIs in MIBC in the neoadjuvant and adjuvant settings, discuss their incorporation in bladder-sparing approaches and discuss promising biomarkers to predict response to these novel therapies.
Trials with neoadjuvant immunotherapy for MIBC
Trials using single agent ICI and ICI/ICI combination for neoadjuvant therapy in MIBC have shown encouraging pCR rates and are described below. These pCR rates are comparable to those observed with NAC (35–40%) (Figure 1). However, it is unclear whether the patients responding to chemotherapy also respond to ICI, and the role of the combination needs to be explored further. Additionally, while presence of pCR post-systemic therapy has been consistently shown to correlate with long term survival in chemotherapy-based trials, whether pCR also correlates with overall survival in neoadjuvant immunotherapy-based trials will become clear with long-term outcomes of ongoing trials. 11 Careful attention to the patient population enrolled in each study is needed to avoid biases associated with cross-trial comparison. Additionally, these are single-arm studies, and results from randomized phase III trials comparing ICI against standard treatments are awaited.
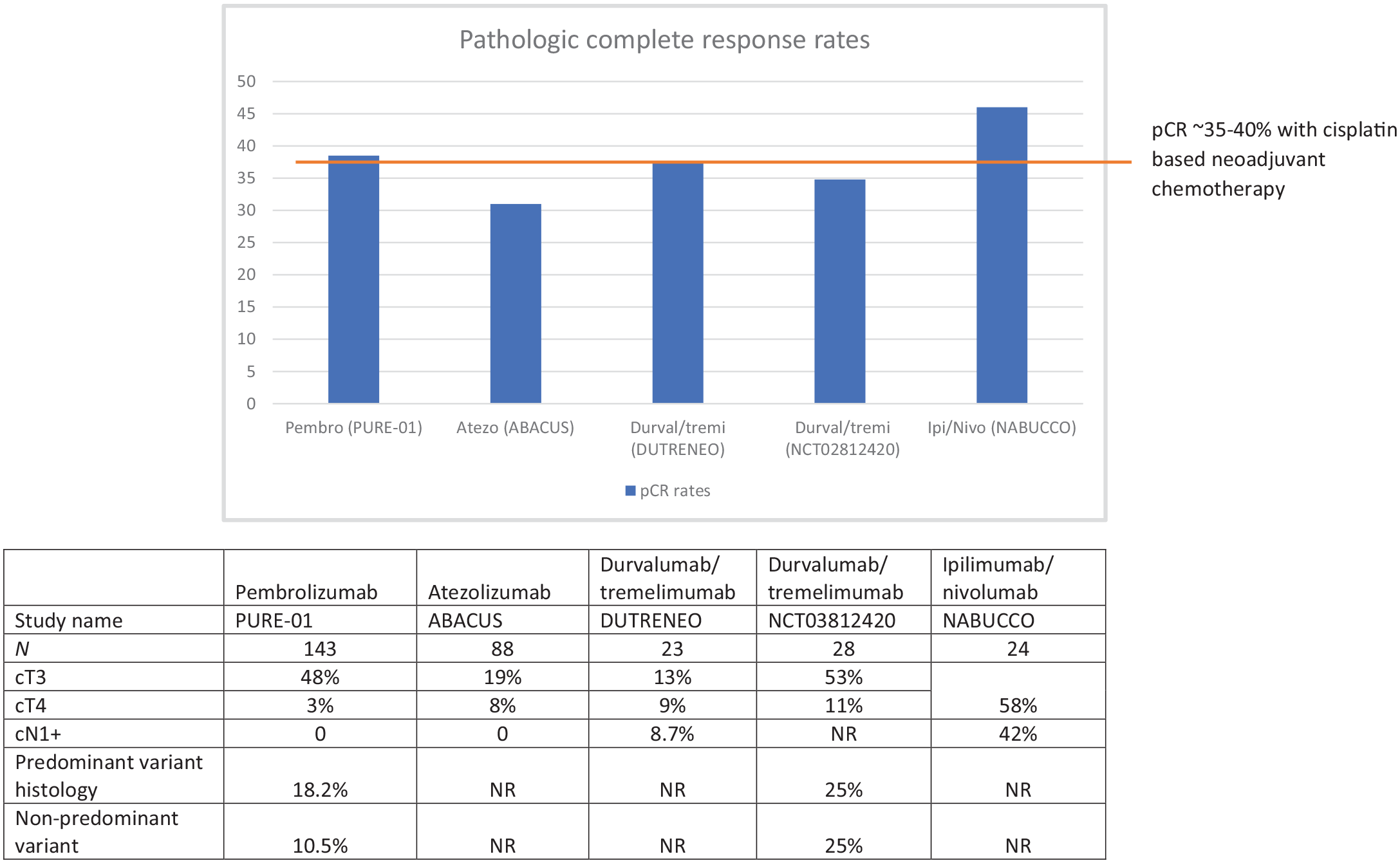
Pathologic complete response (pCR) and patient characteristics in neoadjuvant immunotherapy trials in muscle-invasive bladder cancer.
Pembrolizumab
PURE-01 was a phase II trial in which patients with cT2-T4aN0 MIBC received three cycles of neoadjuvant pembrolizumab followed by cystectomy.12,13 Of 143 intent-to-treat patients, 102 patients had pure UC histology, 15 patients had mixed histology with predominant UC component and 26 had predominant variant histology. The clinical stage was T3–4 in 73 (51%) patients.12,14 The primary endpoint of this study, pCR, was met at 37% [95% confidence interval (CI) 28–46] and ⩽pT1 disease was achieved in 55% (95% CI 46–65) of patients. In patients with predominant variant histology, the pT0 rate was 16%. 12 The squamous cell carcinoma variant and lymphoepithelioma like variant achieved a response of pT ⩽1 in 86% and 67% respectively.
The study enrolled patients irrespective of cisplatin eligibility although >90% were cisplatin eligible. 13 During the study, 13 patients received cisplatin chemotherapy post-pembrolizumab and prior to cystectomy and nine patients received adjuvant cisplatin-based chemotherapy after cystectomy. Eight patients did not receive radical cystectomy due to systemic progression or patient refusal. 14 The secondary endpoint of the study, event-free survival (EFS), was defined as the development of metastasis, death, inability to undergo cystectomy due to treatment-related adverse effects or unresectable disease, or need to initiate chemotherapy prior to cystectomy. 14 The median EFS was not reached in the intention to treat (ITT) cohort and the 12- and 24-month EFS was 84.5% and 71.1%, respectively. 14 In patients with ypT0 disease, the 24-month EFS was 96.1% whereas in non-complete response patients the 24-month EFS was 59.6%, suggesting that ypT0N0 response may be a good surrogate of EFS within ICI trials, as it is with cisplatin-based chemotherapy. 14
Atezolizumab
As compared with PURE-01, in ABACUS, cisplatin-ineligible patients with the cT2–T4N0 disease received two cycles of neoadjuvant atezolizumab prior to cystectomy. Of 88 patients assessable for the primary endpoint, 24 patients had a cT3–T4 disease. The study met its primary endpoint of pCR with 31% of patients experiencing pCR at the time of cystectomy. 15 One-year relapse-free survival was 79%. Eight patients did not receive cystectomy, of which three patients had treatment-related adverse effects. Interestingly, 17% of patients had grade 3–4 surgical complications, most commonly wound dehiscence. 15
Durvalumab
In BLASST-2, cisplatin-ineligible patients with the cT2–T4N0 disease received three cycles of neoadjuvant durvalumab in cohort 1. In cohort 2, patients receive neoadjuvant durvalumab with oleclumab, an anti-CD73 antibody. Preliminary results of cohort 1 were reported at GU ASCO 2020. The primary endpoint of feasibility was met as all 10 patients in cohort 1 completed three doses of durvalumab without any dose-limiting toxicities. 16 A pT0 rate of 12.5% was reported.
Durvalumab/tremelimumab
Neoadjuvant durvalumab and tremelimumab (anti-CTLA4) combination therapy were studied in NCT02812420, in which cisplatin-ineligible patients with localized high-risk MIBC received durvalumab 1500 mg/kg and tremelimumab 75 mg/kg every 4 weeks for two cycles before cystectomy.17,18 Patients with variant histology and concomitant high-grade upper tract UC were also included. At baseline, 43% and 11% of patients in this study had cT3 and cT4 disease, respectively, and 25% of patients had UC with variant histology.17,18 The primary endpoint of this study was safety. Most patients experienced an immune-related adverse event (AE) of any grade. The study did not exceed its safety or futility rules with six patients experiencing grade or higher immune-related AEs. Two patients had grade 3 immune-related hepatitis/colitis and had a delay in cystectomy. Among the 24 patients who received cystectomy, 37.5% of patients had pCR and 58% of patients had downstaging of disease.
Neoadjuvant durvalumab/tremelimumab was compared with standard cisplatin-based chemotherapy in the phase II DUTRENEO study (NCT03472274). In this study, 61 patients were classified based on an 18 gene inflammation signature into ‘hot’ and ‘cold’ tumors. Patients with cold tumor received chemotherapy and hot tumors were randomized to chemotherapy or durvalumab/tremelimumab. Among the hot tumors (n = 45), no significant difference was seen in patients receiving chemotherapy or ICI combination (pCR 36.4% versus 34.8%, respectively). Interestingly, the pCR was 68.6% in cold tumors (n = 16) receiving cisplatin-based chemotherapy, which is higher than in historic NAC trials, suggesting a role to study the inflammation signature in predicting NAC response. 19
Ipilimumab/nivolumab
The NABUCCO trial (NCT03387761) was a phase I trial that included 24 cisplatin-ineligible patients with cT3–T4a, N1–N3 disease patients who received neoadjuvant ipilimumab 3 mg/kg on day 1, ipilimumab 3 mg/kg with nivolumab 1 mg/kg on day 21 and nivolumab 3 mg/kg on day 42 prior to surgery. 20 The clinical trial included a high risk population comprising 58% with cT3–T4 disease and 42% with lymph node positive disease prior to treatment. The primary endpoint of this study, the feasibility of surgical resection within 12 weeks of combination immunotherapy, was met at a 96% resection rate. An impressive pCR rate of 46% and downstaging to non-muscle invasive disease in 58% was noted in this high-risk patient population. 20
Ongoing neoadjuvant trials for cisplatin-ineligible patients
Ongoing clinical trials using neoadjuvant ICI with no available results are presented in Table 1. A study testing neoadjuvant nivolumab and ipilimumab/nivolumab in cisplatin-ineligible MIBC is ongoing (NCT03520491). NKTR-214, a CD122-preferential IL2 pathway agonist, is being studied with nivolumab in a phase III randomized trial against nivolumab and standard of care (NCT04209114). A randomized phase III clinical trial studying a combination of pembrolizumab with enfortumab vedotin (EV), an antibody drug conjugate targeting Nectin-4, is also ongoing (EV-303/KEYNOTE-905). In this study, cisplatin-ineligible MIBC patients are randomized to perioperative pembrolizumab versus perioperative pembrolizumab/EV combination versus cystectomy alone. This combination is also being tested in the MIBC cohorts of a phase II study, EV-103 (NCT03288545).
Ongoing trials on neoadjuvant immunotherapy in muscle-invasive bladder cancer with pending results.

BCG, Bacillus Calmette-Guerin; ddMVAC, dose-dense methotrexate, vinblastine, adriamycin, cisplatin; EFS, event-free survival; ITT, intention to treat; MFS, metastasis-free survival; NR, not reported; pCR, pathologic complete response; PD-L1, programmed death-1 ligand.
In EV-303 (NCT03924895), patients in the pembrolizumab arm receive three cycles of pre-operative and 14 cycles of post-operative pembrolizumab. Similarly, patients in the EV + pembrolizumab arm receive three cycles of pre-operative therapy and six cycles of post-operative EV and pembrolizumab and then eight cycles of pembrolizumab. Several other ongoing trials including, NCT03924856, NCT04700124, NCT03661320, NCT03558087, NCT03732677, and NCT03406650, in cisplatin-eligible patients use perioperative ICI. The role of pre-operative and peri-operative ICI remains to be explored in randomized control trials. Data from PURE-01 showed that patients with ypN+ disease had a 24-month recurrence-free survival of 39.3% compared with 96% and 79% for ypT0N0 and ypT2-4N0 patients, respectively. This, and promising data from adjuvant trials, indicates a role for adjuvant ICI in high-risk patients. However, treatment with peri-operative ICI or EV in all patients, regardless of their pathologic response to neoadjuvant therapy, may lead to unnecessary toxicity and it remains to be explored further to guide future trial design.
Trials with neoadjuvant immuno-chemotherapy for MIBC
In metastatic UC, the combination of gemcitabine/cisplatin and ICI has had three negative studies with encouraging safety data from randomized phase III studies: KN-361, 21 IMVigor130, 22 and DANUBE. 23 Nevertheless, chemotherapy/ICI combinations are now being studied in neoadjuvant treatment of MIBC. Available data from the clinical trials presented below have demonstrated the safety of these combinations. However, similar to the ICI trials, the combination of single-arm chemotherapy/ICI combination studies have demonstrated pCR rates comparable to NAC alone (Figure 2), and data from randomized trials comparing combination therapy with NAC alone are awaited (NCT03661320, NCT03924856, NCT03732677) (Table 1).

Pathologic complete response (pCR) and patient characteristics in neoadjuvant immunotherapy/chemotherapy combination trials in muscle-invasive bladder cancer.
Neoadjuvant gemcitabine-based chemotherapy with pembrolizumab was studied in GU14-188 (NCT02365766), in which cT2–4 MIBC patients received four cycles of gemcitabine/cisplatin if cisplatin eligible (cohort 1) or gemcitabine if cisplatin ineligible (cohort 2) with five cycles of neoadjuvant pembrolizumab. Preliminary results from 43 patients in cohort 1 and 37 patients in cohort 2 have been reported.24,25 The primary endpoint of these studies was pathologic non-muscle invasive rate at the time of cystectomy. Non-muscle invasive rates of 61% and 51.6% were achieved in the two cohorts, respectively. Similarly, pCR rates of 44% and 45.2% were reported in the two cohorts, respectively.
While the combination of gemcitabine/cisplatin + pembrolizumab was considered safe in NCT02365766, in NCT02690558 combining gemcitabine/cisplatin with pembrolizumab led to a protocol amendment due to significant toxicity noted in the lead-in phase. In the lead-in phase, six patients received pembrolizumab two weeks prior to full dose gemcitabine/cisplatin (cisplatin 70 mg/m2, gemcitabine 1000 mg/m2). This led to excessive myelosuppression and acute kidney injury leading to removal of the lead-in dose. In the subsequent patients, split dose gemcitabine/cisplatin was administered with pembrolizumab for four cycles. The primary endpoint of non-muscle invasive rate in 39 treated patients was 56% with 36% achieving pCR. 26 Higher rates of grade 3/4 cytopenias and febrile neutropenia were noted in this study, but due to a small number of patients, definitive conclusions cannot be drawn.
The combination of gemcitabine/cisplatin with nivolumab has been tested in BLASST-1, a phase II single arm study in cT2–4 MIBC. 27 The study included 41 patients, with predominantly cT2 stage (90%). The primary endpoint of the non-muscle invasive rate was 65.8% with 34% pCR reported. Grade 3–4 toxicities were seen in 20% of patients, the most common being thrombocytopenia, neutropenia, and renal insufficiency. 27
Neoadjuvant gemcitabine/cisplatin with nivolumab has also been studied with the goal of selective bladder sparing in NCT03558087 (HCRN GU 16-257). In this study, patients received repeat transurethral bladder tumor (TURBT) after four cycles of neoadjuvant combination therapy. Patients with the presence of cTa or cT0 disease, negative urine cytology, and no evidence of disease on magnetic resonance imaging preserved their bladder and received maintenance nivolumab for 4 months. If TURBT showed cTis, cT1, or MIBC, patients proceeded to cystectomy. The primary outcome of the study was to determine clinical complete response (CCR), which was defined as 2-year metastasis-free survival in patients pursuing bladder preservation or pCR in patients undergoing cystectomy. 28 In the ITT population, CCR was 48% (95% CI 36–61). Of 76 patients enrolled in the study, 31 patients had CCR and 30 patients pursued surveillance. Among the 30 patients pursuing surveillance, at a median follow-up of 13.7 months, six patients recurred: three with non-muscle invasive bladder cancer, two with MIBC, and one with metastatic disease. However, bladder-intact recurrence-free survival was possible and longer follow-up is needed to assess the durability of responses.
In SAKK 06/17 (NCT03406650), a single-arm phase II study, neoadjuvant gemcitabine/cisplatin was combined with perioperative durvalumab in 53 patients with cT2–T4N0–1 MIBC. 29 Three cycles of neoadjuvant durvalumab and 10 cycles of adjuvant durvalumab were administered. The co-primary endpoints in this study was EFS at 2 years. In this study with the high-risk population (17% with cN1 and 31% with cT3–T4 disease), a pCR of 34% (95% CI 21.5–48.3) was reported. Of note, about one-third of the patients did not receive adjuvant durvalumab. 29
Ongoing neoadjuvant trials for cisplatin-eligible patients
Ongoing clinical trials using neoadjuvant ICI with no available results are presented in Table 1. In addition to gemcitabine/cisplatin, ddMVAC (dose-dense methotrexate, vinblastin, adriamycin, cisplatin) has also been tested in combination with ICI in the neoadjuvant setting for MIBC. NCT03549715 is a randomized phase II trial in which patients with MIBC received four cycles of neoadjuvant ddMVAC with two cycles of durvalumab alone or in combination with tremelimumab. 30 The primary endpoint in the 12 patient run-in phase, dose-limiting toxicity up to 3 months after cystectomy, was presented and indicated that the combination is safe. Other ongoing trials combining ddMVAC with ICI include NCT04383743 using pembrolizumab, NCT03674424 using avelumab, and RETAIN-2 (NCT04506554) using nivolumab. RETAIN-2 uses a novel adaptive design in which patients with cT2–T3N0 MIBC receive three cycles of nivolumab and ddMVAC followed by repeat TURBT. Patients with mutations in ATM, ERCC2, or RB1 who have no clinical disease after neoadjuvant therapy have an option of active surveillance. Patients with the residual disease receive standard of care therapy.
Adjuvant immunotherapy
Several phase III clinical trials are ongoing which explore the role of adjuvant ICI in high-risk MIBC and upper tract UC, post cystectomy or nephroureterectomy, respectively.
IMVigor010 was a phase III open-label randomized trial that included 809 patients with pT2–4a, or pN+ who received NAC, or pT3–4a or pN+ who did not receive NAC. 31 Patients were randomized to adjuvant atezolizumab for 1 year versus observation. The study did not meet its primary endpoint of disease-free survival (DFS) [19.4 months versus 16.6 months, hazard ratio (HR) = 0.89; 95% CI 0.74–1.08; p = 0.24]. Overall survival follow-up is immature and ongoing. Biomarkers such as PD-L1 and tumor mutation burden (TMB) did not identify patients benefiting from atezolizumab versus observation in the ITT population. However, in a pre-specified exploratory analysis, the presence of circulating tumor DNA (ctDNA) prior to treatment and at cycle 3 was indicative of treatment benefit. Patients with a positive ctDNA assay had a significant DFS and overall survival (OS) improvement from adjuvant atezolizumab. In the ctDNA positive patients, the median DFS was 5.9 versus 4.4 months (HR 0.58; 95% CI 0.43, 0.79; p = 0.0024) and the median OS was 25.8 versus 15.8 months (HR 0.59; 95% CI 0.41, 0.86; p = 0.0059), suggesting a benefit of adjuvant atezolizumab versus observation in this population. The ctDNA negative patients had a low risk of relapse and did not have improved outcomes with atezolizumab compared with observation, suggesting that they could be spared adjuvant therapy.32,33 Patients in the atezolizumab arm who cleared ctDNA had a superior DFS compared with those who remained positive for ctDNA (HR 0.26; 95% CI 0.12–0.56, p = 0.0014). The role of sequential ctDNA testing in the adjuvant setting is further explored in IMVigor011 (NCT04660344).
Nivolumab was also tested in the adjuvant setting in CheckMate274, a randomized phase III trial comparing adjuvant nivolumab versus placebo in 709 patients with high-risk MIBC or upper tract urothelial cancer. In this study neoadjuvant chemotherapy was allowed in patients eligible to receive it. The primary endpoint of the study was DFS in ITT and PD-L1+ patients.34,35 The study met its primary endpoint with an improvement in DFS in the ITT (21 months versus 10.9 months) and PDL1+ patients (NR versus 10.8 mo). However, an overall survival benefit has not yet been shown and raises the question of whether DFS benefit is enough to warrant the use of adjuvant immunotherapy.
Why CheckMate274 was a positive trial and IMVigor010 was negative is a matter of speculation. CheckMate274 was a placebo controlled trial, whereas in IMVigor010 the control arm was best supportive care. In the latter study, patients with more aggressive disease may have dropped out after being randomized to the best supportive care arm, leading to better performance of the control arm compared with CheckMate274. Adjuvant pembrolizumab is being studied in phase III AMBASSADOR trials and the results are awaited. While DFS was the primary endpoint of IMVigor010 and CheckMate274 studies, the co-primary endpoints of AMBASSADOR are DFS and OS, which will provide valuable information. All trials included a fix proportion of upper tract UC patients and ICI was administered for 1 year.
Immunotherapy with definitive chemoradiation
Trimodality bladder preserving therapy with TURBT, radio-sensitizing chemotherapy, and definitive radiation therapy is commonly used in patients who are unable or refuse to undergo cystectomy. However, this approach has about a 10-year disease-specific survival rate of about 59% with a 29% risk of salvage cystectomy at 5 years. 10 Strategies to include ICI with these approaches are ongoing to reduce recurrence and improve survival (Table 2).
Ongoing trials on immunotherapy with radiation therapy in muscle-invasive bladder cancer with pending results.

Patients may have received neoadjuvant cisplatin-based chemotherapy.
chemoRT, chemoradiation therapy; DFS, disease-free survival; EFS, event free survival; MMC, mitomycin C; MTD, maximum tolerated dose; NR, not reported; pCR, pathologic complete response.
One approach being used is to add ICI to chemoradiation. NCT02662062 is an ongoing study in which patients commence pembrolizumab concurrently with cisplatin and radiation therapy and continue until the 12-week cystoscopy and assessment. Initial results in 10 patients confirmed the feasibility and by week 24, 90% of patients achieved a complete cystoscopic response. 36
Pembrolizumab with gemcitabine/radiation therapy is also being studied in NCT02621151, with results recently reported at ASCO 2021. 37 In this study, 54 patients who could not receive cisplatin-based neoadjuvant chemotherapy and were not cystectomy candidates received one cycle of pembrolizumab → maximal TURBT → three cycles of pembrolizumab with twice weekly gemcitabine and hypofractionated radiation therapy. The primary endpoint was 2-year bladder intact DFS. At 15.5 month follow-up, the 1-year bladder intact DFS was 89%. In the 48 patient efficacy cohort, 35% patients had a grade 3 AE including urinary tract infection (8%), diarrhea (4%), colitis (4%), bladder pain (4%), neutropenia (2%), and thrombocytopenia (2%).
Atezolizumab with chemoradiation therapy (chemoRT) is being studied in a randomized clinical trial (NRG/SWOG 1806, NCT03775265). Initial safety data from 73 patients showed manageable toxicities. There was no increase in grade 3 gastrointestinal toxicity and although there were more hematologic toxicities and urinary tract infections in the investigational arm, they were not considered to be immune related. 38
Induction chemotherapy followed by durvalumab/chemoRT+ adjuvant durvalumab versus chemoRT combination is being evaluated in the ongoing EA8185 (NCT04216290) clinical trial for lymph node positive patients.
Another approach employed, especially in chemotherapy-ineligible patients, is to combine ICI with radiation therapy without the use of sensitizing chemotherapy. Durvalumab with radiation is being studied in the phase Ib/II DUART study (NCT02891161) in which cT2–4N0–2 patients who are either unable to receive cisplatin-based chemotherapy or cystectomy received durvalumab concurrently with radiation followed by 1 year of adjuvant durvalumab. Among the 21 patients evaluable for response, the co-primary endpoints of progression free survival (PFS) at 1 year and disease control rates were 73% and 70%, respectively. 39
Nivolumab with radiation therapy is being studied in the NUTRA trial (NCT03421652), in which cisplatin-ineligible MIBC patients receiving nivolumab are commenced with radiation therapy and continued for a maximum of 6 months. Among 17 patients with available safety data, the combination was tolerable with promising efficacy and six of 14 evaluable patients had complete response. 40
In the IMMUNOPRESERVE-SOGUG study, 32 patients with cT2–T4aN0M0 who were not cystectomy candidates received four cycles of durvalumab/tremelimumab with radiation therapy followed by a repeat TURBT. 41 Patients with either residual or relapsed MIBC were offered salvage cystectomy. The primary endpoint of this study was CCR as assessed by lack of MIBC at post-treatment tumor site biopsy. At a median follow-up of 12.7 months, CCR (cT0) was 78% in 28 assessable patients and 12-month DFS rate was 76%. Grade 3 or 4 AEs related to therapy were reported in 31% patients, the most frequent being gastrointestinal toxicity (12.5%), acute kidney failure (6%), and hepatitis (6%).
Taken together, these early data indicate that ICI with radiation therapy or chemoRT may be safe with manageable adverse effects. While long-term data will be more informative, preliminary results have suggested promising survival and maybe a good option for frail patients who are unable to receive NAC or cystectomy. Data from KEYNOTE-992, a phase III trial comparing chemoRT with or without pembrolizumab, are awaited.
Biomarkers in neoadjuvant immunotherapy trials
Several studies are underway to develop clinically useful biomarkers that can predict the therapeutic responses to neoadjuvant ICI in MIBC. Here we summarize current efforts to develop novel biomarkers.
PD-L1
Unfortunately, PD-L1 positivity has not reliably predicted pCR in neoadjuvant ICI trials. In PURE-01, pT0 was achieved in 19 patients (54.3%) with PD-L1 combined positive score (CPS) ⩾10% compared with only two patients (13.3%) with CPS <10% (p = 0.011). Likewise, in the NABUCCO trial with neoadjuvant ipilimumab/nivolumab, PDL1+ tumors had a trend towards higher pCR. 20 The patients with CPS score > 10% showed a significantly higher rate of complete response of 73% compared to 33% in PDL-1 negative tumors (p = 0.15) However, in the neoadjuvant durvalumab/tremelimumab (DUTRENEO) and atezolizumab (ABACUS) studies, no correlation was observed between responses and PD-L1 expression in pre-treatment tissue.15,19
TMB
TMB has been significantly associated with the ICI responses in several cancer types including metastatic bladder cancer. 42 In PURE-01 the pT0 endpoint correlated with a significantly higher TMB (odds ratio 1.06; 95% CI 1.01–1.13; p = 0.03). 12 In the NABUCCO trial, tumors with pCR had a trend towards higher TMB. 20 However, such a correlation between TMB and response was not seen in the ABACUS or DUTRENEO studies.15,19
Mutations
The mutations of DNA damage repair (DDR) genes contribute to the significant increase of TMB or neoantigen that might be associated with the response to ICIs. In PURE-01, DDR gene alterations were associated with pT0 and higher TMB, although by adjusting for TMB the association between DDR alterations and CR was weakened. 13 Such a correlation between DDR and response was not seen in the ABACUS or DUTRENEO studies.15,19 In NABUCCO, alterations in DDR genes were more frequently observed in responders compared with non-responders (p = 0.03). 20
FGFR3 alterations did not correlate with response to preoperative pembrolizumab in MIBC. 12 Despite an association of high FGFR3 gene expression with a lower CR rate (p = 0.01), they did not find a correlation between FGFR3 activity or mutation/fusion and CR (p = 0.2 and p = 0.8). 12 In the DUTRENEO study, mutations in KRAS, PIK3CA, PBRM1, EGFR, NRAS, APC2, and FGFR were analyzed but did not correlate with response in the small cohort of patients. 19
Molecular subtypes
In PURE-01, molecular subtypes according to the consensus subtype classification were not associated with CR (p > 0.2), although higher major response rates were seen for basal-squamous and luminal unstable tumors per consensus subtype and claudin-low per genomic subtype classifier (GSC) classification. 43 This observation is consistent with previous studies, where a lack of an association between molecular subtype and response to atezolizumab or cisplatin-based chemotherapy has been reported.15,44 Interestingly, although molecular subtypes could not predict the pathologic responses to ICI, they are associated with PFS. The claudin-low tumors in GSC had the best PFS outcomes with no events in 24 months after pembrolizumab treatment while neuroendocrine-like tumors had the worst survival outcomes. 43 The uncoupling between pathologic responses and PFS outcomes may be due to a limited number of samples and immature OS in the trial.
RNA-based immune signatures
In PURE-01 the Immune190 signature, interferon-gamma, and interferon-alpha signatures were associated significantly with pCR and PFS in multivariable analyses. 43 However, the interferon-gamma signature was not associated with pCR in the neoadjuvant ipilimumab/nivolumab study. 20 In ABACUS, an eight-gene cytotoxic T-cell signature correlated with response. However, in DUTRENEO, an 18-gene tumor inflammation signature did not differ significantly between responder versus non-responder patients. 19 Such discordance between predictive signatures for monotherapy versus combination therapy is likely explained by the fundamental differences in cellular mechanisms between anti-CTLA-4 and anti-PD-1/PD-L1 therapy. Conversely, TGF-b signatures have been previously shown to be associated with resistance to immunotherapy in metastatic UC 42 and also correlated with poor response in ABACUS and NABUCCO trials.15,20
Immune cell infiltration
In the neoadjuvant durvalumab/tremelimumab study, a significant increase in the frequency of ICOS+CD4+ T-cells was observed in post-treatment tumor tissues of responder patients as compared with non-responder patients, consistent with previous studies demonstrating the effect of anti-CTLA4 therapy.18,45 Additionally, a higher density of tertiary lymphoid structures (TLSs) was observed in the pre-treatment tumor tissues of responder patients as compared with non-responder patients. 19 The authors derived a four-gene TLS signature consisting of POU2AF1, LAMP3, CD79A, and MS4A1 and found a significantly higher expression of the four-gene TLS signature in responders (n = 9) as compared with non-responders (n = 7). However, in the neoadjuvant ipilimumab/nivolumab study, no such correlation between TLS and response was noted. 20 Similarly, in this study the presence of CD8+ T-cells or CD8+ T effector signatures did not correlate with pCR. 20
Conclusion
Significant efforts are ongoing in the treatment of MIBC incorporating single agent ICI, ICI/ICI, and ICI/chemotherapy combination therapies in the perioperative setting. These clinical trials have demonstrated the safety of neoadjuvant ICI with encouraging pCR, making them promising treatment options for cisplatin-ineligible patients. The role of ICI/ICI and ICI/chemotherapy in cisplatin-eligible patients will be clearer with awaited results from ongoing randomized phase III clinical trials. Biomarkers such as PD-L1, TMB, and DDR have not consistently correlated with response to ICI and novel biomarkers using RNA-based signatures and immune cell infiltration need to be explored further to guide treatment.
