Abstract
Cutaneous melanoma continues to increase in incidence and poses a significant mortality risk. Surgical excision of melanoma in its early stages is often curative. However, patients with resected stages IIB-IV are considered at high risk for relapse and death from melanoma where systemic adjuvant therapy is indicated. The long-studied high-dose interferon-α was shown to improve relapse-free survival (RFS) and overall survival (OS) but is no longer in use. Adjuvant therapy with ipilimumab at 10 mg/kg (ipi10) demonstrated significant RFS and OS improvements but at a high cost in terms of toxicity, while adjuvant ipilimumab 3 mg/kg was shown to be equally effective and less toxic. More recently, the adjuvant therapy for resected stages III-IV melanoma in clinical practice has changed in favor of nivolumab, pembrolizumab, and BRAF-MEK inhibitors dabrafenib plus trametinib (for BRAF mutant melanoma) based on significant improvements in RFS as compared to ipi10 (nivolumab and pembrolizumab) and placebo (dabrafenib plus trametinib). For resected stages IIB-IIC melanoma, pembrolizumab achieved regulatory approval in the United States based on significant RFS benefits. In this article, we review completed and ongoing phase III adjuvant therapy trials. We also briefly discuss neoadjuvant therapy for locoregionally advanced melanoma. Finally, we explore recent studies on predictive and prognostic melanoma biomarkers in the adjuvant setting.
Introduction
The incidence of melanoma has been steadily rising worldwide over the past few decades. The American Cancer Society estimates that in 2022, there will be 97,920 new cases of melanoma (70% stage I, 11% stage II, 7.5% stage III, and 2.5–4% stage 4) and 7650 melanoma-related deaths in the United States.1–4 Patients with surgically resectable melanoma who are considered at high risk for disease relapse and death include patients with stages IIB-C melanoma with a deep primary melanoma with or without ulceration and risk of relapse of at least 35% and stages IIIA-D based on regional lymphatic metastases.2,5 Among this latter group, patients with clinically/radiologically detectable stages IIIB-D melanoma and bulky locoregional advanced disease have a risk of relapse that approaches 90% with surgery alone and may benefit from neoadjuvant therapy.
Systemic adjuvant therapy targets micrometastatic disease that is believed to be the source of future melanoma relapse and potential death from melanoma. The ultimate goals of adjuvant therapy are to reduce the risks of recurrence and death in high-risk patients despite definitive surgical management. Patients with resected melanoma appear to be more susceptible to immunologic interventions with evidence of Th1 immune polarization against melanoma as compared to patients with more advanced metastatic disease characterized by a more immunosuppressive microenvironment. 6 This has supported the testing of immunotherapy in the adjuvant setting in melanoma, starting with high-dose-interferon (IFN)-α2b (HDI). HDI was the first US Food and Drug Administration (FDA)-approved adjuvant therapy for resectable high-risk melanoma in 1995. In the meantime, strides were made in developing cancer immunotherapy and targeted therapy which provided excellent opportunities to treat metastatic melanoma. Those advances were translated into the adjuvant setting in phase III trials testing programmed death-1 (PD-1) blockade with pembrolizumab and nivolumab in addition to dabrafenib plus trametinib for BRAF mutant melanoma, leading to their regulatory approval and broad use. In this review, we discuss approved adjuvant therapy options for patients with melanoma.
Adjuvant immunotherapy
Cytokine immunotherapy: IFN-α
IFN-α was the mainstay of adjuvant therapy in the United States before the development of checkpoint inhibitor immunotherapy. IFN-α is known to have immunomodulatory, differentiation-inducing, antiproliferative, antiangiogenic, and proapoptotic effects. 7 It has been shown to induce signal transducer and activator of transcription (STAT)1 upregulation while causing STAT3 downregulation that may reverse T-cell signaling defects, resulting in T-cell and dendritic cell infiltration into nodal metastases. Kirkwood et al. 8 and the Eastern Cooperative Oncology Group (ECOG) conducted four multicenter, randomized trials using high-dose IFN-α (HDI) for patients with surgically resected American Joint Committee on Cancer (AJCC) 6th edition stages IIB and III melanoma. Altogether, these trials accrued 1916 patients, including three phase III trials where patients were randomized to receive HDI versus observation (E1684 and E1690) or the ganglioside GM2/keyhole limpet hemocyanin vaccine (GMK) (E1694).9–12 In these studies, HDI showed improvements in relapse-free survival (RFS) in all the three trials and overall survival (OS) in two trials (E1684 and E1694). However, factors related to treatment administration frequency and high toxicity limited the uptake of this adjuvant treatment in the community. 4
Adjuvant biochemotherapy
Biochemotherapy (BCT) is a treatment that combines cytotoxic drugs with interleukin (IL)-2 and IFN-α to boost the effectiveness of chemotherapy alone. The South West Oncology Group conducted the S0008 study, 13 which compared adjuvant therapy with the E1684 HDI regimen to a 9-week BCT regimen that included three cycles of cisplatin, vinblastine, and dacarbazine, as well as low doses of IL-2 and IFN-α. There was an improvement in RFS in favor of BCT at a median follow-up of 7.2 years [median RFS of 4.0 versus 1.9 years; hazard ratio (HR): 0.75, 95% confidence interval (CI): 0.58–0.97]. However, no difference in OS rates was observed. In addition, BCT had a higher toxicity rate, with 76% and 64% of grades 3 and 4 adverse events (AEs), respectively.
Anti-cytotoxic T-lymphocyte-associated antigen immunotherapy: Ipilimumab
Ipilimumab is a human monoclonal antibody that blocks cytotoxic T-lymphocyte-associated antigen 4 and potentiates antitumor immune responses to treat advanced melanoma. Ipilimumab 3 mg/kg received US FDA approval in 2011 for metastatic melanoma following the MDX-1020 randomized, double-blind phase III trial. In this study, Hodi et al. reported significant OS benefits with ipilimumab 3 mg/kg (ipi3) and the combination of ipilimumab plus peptide vaccine compared to peptide vaccine alone. 14 Another study investigating ipilimumab at 10 mg/kg (ipi10) plus dacarbazine compared with dacarbazine alone also showed significant survival benefits. 15
EORTC 18071 compared adjuvant therapy with ipi10 versus placebo in patients with completely resected AJCC 7th edition stage III melanoma at high risk for recurrence. This trial first reported in 2015 that after 2.74 years of median follow-up, the median RFS was significantly better in the ipilimumab group (26.1 months) compared to the placebo group (17.1 months), 16 leading to the regulatory approval of ipi10 as adjuvant therapy for stage III melanoma. An updated report published in 2016, after a median follow-up of 5.3 years, confirmed the significant improvement in RFS in the ipilimumab group versus placebo (RFSipilimumab = 40.8%, RFSplacebo = 30.3%), as well as increased OS and distant metastasis-free survival (DMFS) in favor of patients treated with ipilimumab (OSipilimumab = 65.4%, OSplacebo = 54.4%; DMFSipilimumab = 48.3, DMFSplacebo = 38.9). 17 However, grade 3 or 4 AEs were also experienced by patients who received ipilimumab (54.1%) as compared to placebo (26.2%). In 2020, Tarhini et al. 18 published the results of the phase III trial, ECOG-ACRIN E1609, that evaluated the safety and efficacy of ipi3 and ipi10 versus HDI for the treatment of resected high-risk melanoma. 19 This study met its primary endpoint of demonstrating significant improvement in OS with ipi3 versus HD-IFN-α2b. Moreover, the lower dose of ipi3 was significantly less toxic than ipi10 as treatment-related AEs of grade 3 or more occurred in 37% of patients receiving ipi3 and 58% in those receiving ipi10 and led to treatment discontinuation in 35% and 54% of patients, respectively. Furthermore, there were no significant differences in RFS or OS between ipi3 and ipi10 study arms.
Anti-PD-1 receptor immunotherapy: Nivolumab and pembrolizumab
In 2017 and 2019, nivolumab and pembrolizumab, two anti-PD-1 immunotherapy agents, were approved by the US FDA for the adjuvant treatment of patients with melanoma with involvement of lymph nodes. While nivolumab also received approval for treatment of patients with metastatic disease who have undergone complete resection. This was the result of two major studies, CheckMate-238 20 and EORTC1325/KEYNOTE-054. 21
In the CheckMate-238 trial, 906 patients with completely resected AJCC 7th edition stages IIIB/C or stage IV melanoma were randomly assigned 1:1 to receive either ipi10 or nivolumab 3 mg/kg. After a median of 18 months of follow-up, the 4-year RFS rate was higher in the nivolumab group was 51.7% (95% CI: 46.8–56.3) compared to 41.2% (95% CI: 36.4–45.9) in the ipilimumab group. The study reported a median RFS of 52.4 months in the nivolumab group and 24.1 months in the ipilimumab group [stratified HR: 0.71 (95% CI: 0.60–0.86); p = 0.0003]. Moreover, the toxicity was significantly lower in the nivolumab group compared to the ipilimumab group (Occurrence of grades 3 and 4 AEs: nivolumab group = 14.4%, ipilimumab group = 45.9%; Treatment discontinuation due to any AEs: nivolumab group = in 9.7% of patients, ipilimumab group = in 42.6% of the patients). Ascierto et al. published a 4-year follow-up study in 2020 that reported sustained RFS benefits in the nivolumab group (51.7%) compared to the ipilimumab group (41.2%). 22 The OS was similar between the two groups (OSnivolumab = 77.9%; OSipilimumab = 76.6%, p = 0.31). Finally, no difference was observed in late-emergent adverse effects between the two groups.
The KEYNOTE-054 study evaluated the effect of pembrolizumab 200 mg or placebo on RFS of patients with resected high-risk AJCC 7th edition stage III melanoma. In this study, pembrolizumab was superior to placebo with a 3.5-year RFS rate of 59.8% (95% CI: 55.3–64.1) in the pembrolizumab group and 41.4% (37.0–45.8) in the placebo group [HR: 0.59 [95% CI: 0.49–0.70]). 23 In addition, in this trial, grades 3–5 AEs were generally manageable, but one treatment-related death occurred in the pembrolizumab group. However, Bottomley et al. 24 reported quality of life (QOL) outcomes results where pembrolizumab did not lead to a clinically significant decrease in health-related QOL when compared to placebo. Table 1 summarizes key trials that had an impact on clinical practice.
Completed adjuvant phase III trials involving immune checkpoint inhibitors and targeted therapy (for BRAF mutant melanoma).
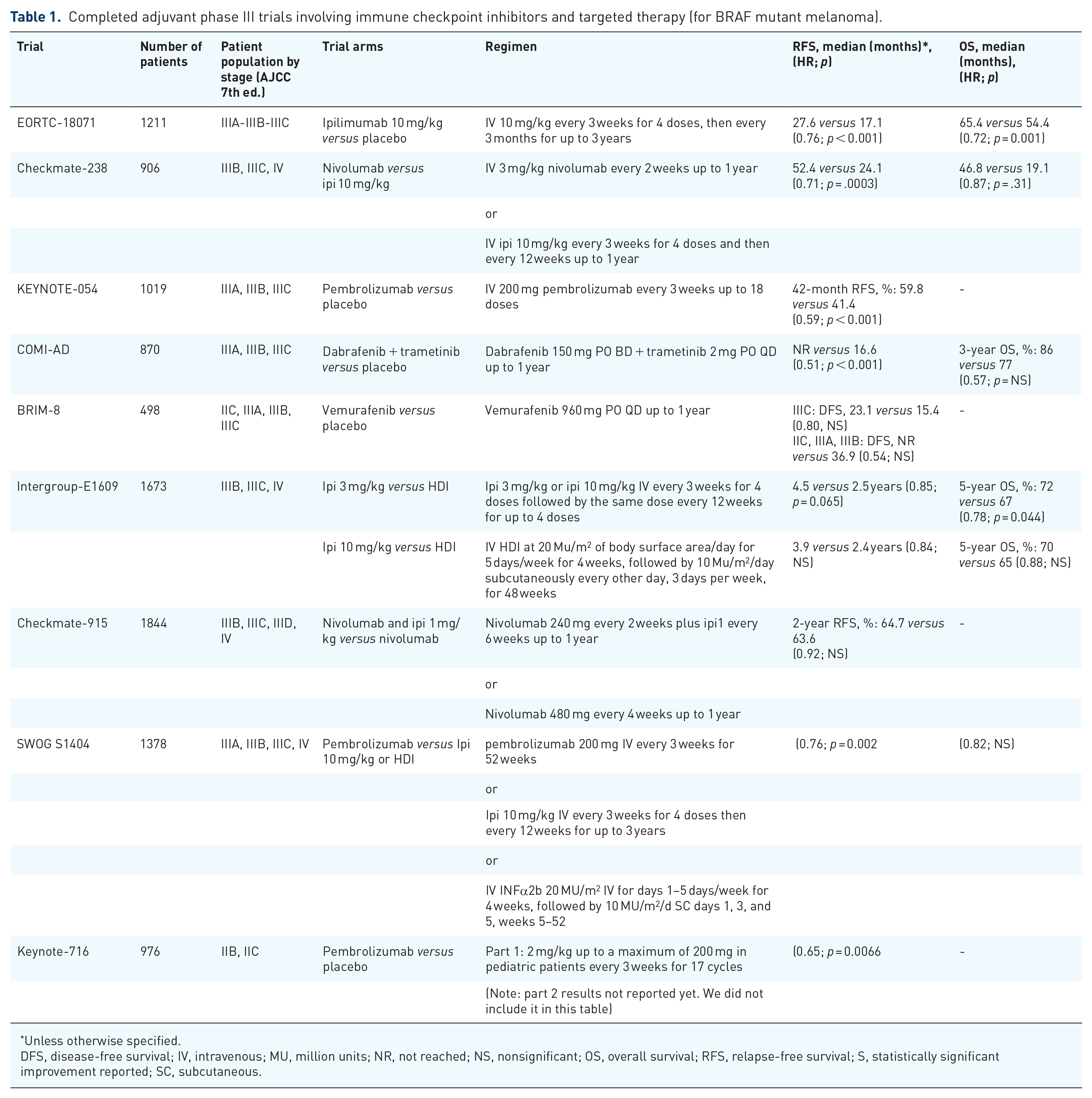
Unless otherwise specified.
DFS, disease-free survival; IV, intravenous; MU, million units; NR, not reached; NS, nonsignificant; OS, overall survival; RFS, relapse-free survival; S, statistically significant improvement reported; SC, subcutaneous.
Combination immunotherapy: Nivolumab plus ipilimumab
The CheckMate 067 study compared the effects of the combination of nivolumab 1 mg/kg plus ipi3 with either nivolumab 3 mg/kg or ipi3 alone in unresectable AJCC 7th edition stage III or stage IV melanoma. Nivolumab plus ipilimumab combination was associated with a significant prolongation of in RFS and OS compared to the control arm of ipilimumab.25,26 However, combination therapy was associated with a significantly higher rate of grade 3 or 4 AEs.
The results of the CheckMate 915 study investigated the benefits of adjuvant combination immunotherapy in surgically resected AJCC 7th edition stages IIIB/C/D or stage IV melanoma. Patients were randomly assigned to receive either nivolumab 240 mg every 2 weeks in combination with ipilimumab 1 mg/kg every 6 weeks or nivolumab 480 mg every 4 weeks for up to a year. Results were presented at the AACR annual meeting 27 in April 2021 and ASCO annual meeting 28 in May 2021. After 2 years of follow-up, the adjuvant immunotherapy combination did not provide significant benefits in RFS or DMFS compared to nivolumab alone. Additionally, the nivolumab plus ipilimumab combination was more toxic than nivolumab alone, with grade 3 or 4 treatment-related AEs of 33% and 13%, respectively.
Adjuvant immunotherapy therapy for stages IIB and IIC melanoma
Patients with AJCC 7th edition stages IIB and IIC melanoma are considered to have a high risk of relapse that justified inclusion in the early adjuvant trials testing HDI.9,29 HDI has been used on a limited basis as adjuvant therapy for these patients, and observation has been the mainstay of management after surgery for most stage II melanoma patients. Keynote 716 was a phase III clinical trial that investigated the use of pembrolizumab as adjuvant therapy in patients with resected stages IIB and IIC melanoma. This trial reported an 18-month RFS rate of 86% (95% CI: 82–89) in the pembrolizumab group compared to 77% (95% CI: 73–81) in the placebo group (HR: 0.60, 95% CI: 0.45–0.79). 30 Median RFS was not reached in either group. Treatment-related events of any grade occurred in 80% of patients treated with pembrolizumab, with treatment-related endocrine disorders occurring in 24% of these patients. The rate of grades 3 and 4 treatment-related AEs was reported in 16% of patients in the pembrolizumab group versus 4% in the placebo group. Based on data generated so far from this study, the FDA-approved adjuvant immunotherapy with pembrolizumab for high-risk stages IIB-C melanoma in 2021. 31
Adjuvant targeted therapy
Dabrafenib and trametinib
BRAF V600 mutation is present in 50% of cutaneous melanomas, 32 and combination therapy with the BRAF inhibitor dabrafenib and the MEK inhibitor trametinib has significant efficacy benefits including OS in patients with unresectable or metastatic melanoma with BRAF V600E or V600K mutations.33–35 Based on these results, Long et al. 36 reported the COMI-AD trial that tested the benefits of adjuvant dabrafenib and trametinib in patients with stage III melanoma with BRAF V600E or V600K mutations after complete surgical resection. The estimated 3-year rate of RFS was significantly higher in the combination therapy group compared to the placebo group (RFScombination = 58%, RFSplacebo = 39%, p < 0.001) as was the 3-year OS (OScombination = 86%, OSplacebo = 77 %, p = 0.0006). The DMFS and freedom from relapse were also higher. Finally, the safety profile of dabrafenib plus trametinib was similar to what was reported in patients with metastatic melanoma. After 5 years, the RFS and DMFS were persistently higher in the dabrafenib plus trametinib group versus placebo. An estimated cure rate analysis also supported potential OS benefits. 37
Based on the results of the studies with adjuvant immunotherapy and targeted therapy, the current practice for operable high-risk melanoma favors adjuvant therapy with nivolumab, pembrolizumab, and dabrafenib plus trametinib.
Vemurafenib
BRIM-8 was a phase III study that tested vemurafenib versus placebo as adjuvant therapy in melanoma patients with AJCC 7th edition stages IIC-IIIA-IIIB (cohort 1) and stage IIIC (cohort 2) with BRAF V600-mutated melanoma. 38 The study followed a hierarchical analytic approach in testing the primary endpoint of disease-free survival (DFS) separately in each cohort, with cohort 2 being prespecified before cohort 1. The study enrolled 184 patients in cohort 2 and 314 in cohort 1. After a median study follow-up of 33.5 months, patients with stage IIIC (cohort 2) had a median DFS of 23.1 months with vemurafenib as compared to 15.4 months with placebo, HR: 0.80, 95% CI (0.54–1.18; p = 0.26). Patients with stages IIC-IIIA-IIIB had a median DFS that was not reached in the vemurafenib arm as compared to 36.9 months in the placebo arm, HR: 0.54, 95% CI: (0.37–0.78; p = 0.0010). This outcome in cohort 1 was not considered significant owing to the prespecified hierarchical prerequisite.
Neoadjuvant therapy
Neoadjuvant systemic therapy in the form of induction therapy prior to definitive surgery has recently made significant strides in the management of locoregionally advanced melanoma. In this section, we provide a brief summary of some of the main neoadjuvant trials in melanoma.
Moschos et al. first investigated the role of HDI as a 4-week regimen preoperatively in the neoadjuvant setting in patients with locoregionally advanced stages III B-C melanoma. Pathologic complete response (pCR) was observed in 15% of the 20 patients enrolled in the study. 39
Subsequently, Tarhini et al. 40 conducted two separate trials testing ipilimumab as monotherapy and in combination with HDI. 41 The first study revealed a response rate of about 10% including major pathologic responses but no pCRs. Ipi10 leads to significant immunomodulating effects on regulatory T cells, myeloid-derived suppressor cells, and effector T cells that were evident in the circulation and tumor microenvironment including evidence of massive infiltration of CD8+ T cells into tumor when tested at 6 weeks. The second study looked at safety, efficacy, and impact on the T-cell repertoire of neoadjuvant ipi3 or ipi10 in combination with HDI in locally/regionally advanced melanoma. Thirty-two percent of patients had pCR, and in these patients, the fraction of T cells was significantly higher when measured in the primary melanoma. Higher tumor T-cell clonality in primary tumor and more so following neoadjuvant therapy was significantly associated with improved RFS.
In 2019, Huang et al. 42 studied neoadjuvant anti-PD-1 with pembrolizumab at 200 mg given for one dose only in patients with resectable stages III/IV melanoma. A pCR of 19% was observed in patients who received treatment. Neoadjuvant pembrolizumab plus HDI combination was studied by Najjar and Tarhini et al. in patients with resectable stages III/IV melanoma. It resulted in a higher pCR rate of 43%. However, the combination was associated with a higher toxicity rate attributed to HDI use. 43
Recent trials also tested combination neoadjuvant immunotherapy with ipilimumab plus nivolumab. Three major studies combined nivolumab 1 or 3 mg/kg with ipi1 or ipi3 with variable numbers of cycles and duration of treatments. OpACIN phase Ib trial tested a combination of ipi3 and nivolumab 1 mg/kg as either four courses post-surgery (adjuvant arm) or two courses pre-surgery and two courses post-surgery (neoadjuvant arm) in patients with palpable stage III melanoma. 44 This trial had encouraging results with 78% of pathologic response. However, a high rate of grades 3 and 4 treatment-related AEs were reported. Therefore, while the results of this trial in terms of treatment efficacy were very encouraging, the safety profile required testing alternative dosing schedules to decrease the toxicity. Amaria et al. 45 compared nivolumab monotherapy to nivolumab plus ipilimumab combination. Patients who received nivolumab monotherapy had low toxicity rates but also modest responses. Different combinations resulted in different efficiency and toxicity profiles, hereby, the neo-OpACIN phase II trial was designed to compare three different combinations of ipilimumab plus nivolumab neoadjuvant treatment regimens. It was concluded that two cycles of ipilimumab 1 mg/kg plus nivolumab 3 mg/kg is a tolerable and efficacious neoadjuvant dosing schedule, with 77% of patients achieving a pCR and grades 3 and 4 immune-related AE rate of 20%. In addition, the PRADO study confirmed the results of the Neo-OpACIN trial, and interestingly, by basing the decision of therapeutic lymph node dissection (TLND) on the pathologic resposne status following neoadjuvant therapy, the number of patients requiring TLND the pathologic resposne status decreased by 60%. 46
In a more recent study by Amaria et al. 47 the combination of neoadjuvant and adjuvant nivolumab with anti-LAG3 antibody relatlimab resulted in a high pCR rate of 59% and lower toxicity (26% Gr 3/4 AEs) in patients with resectable clinical stage III melanoma. Neoadjuvant immunotherapy with Talimogene Laherparepvec was also investigated as monotherapy in resectable stages IIIB-IVM1a melanoma. 48 Furthermore, neoadjuvant targeted therapy for BRAF mutant melanoma has shown significant promise including trials testing dabrafenib and trametinib in patients with resectable stages IIIB-C, BRAF(V600) mutation-positive melanoma as reported in two phase II trials with pCR rates of 49–58%.49,50
Altogether, these studies support significant potential benefits for neoadjuvant immunotherapy and targeted therapy in locoregionally advanced operable melanoma and justify further investigations utilizing emerging active agents in melanoma, especially in combination. A number of randomized neoadjuvant trials are ongoing.
Optimization of adjuvant therapy
Optimal outcome measures
One unsolved question is whether adjuvant anti-PD-1 immunotherapy improves OS in all patients with resected high-risk melanoma. This question is complicated by the lack of improvement in OS in trials comparing anti-PD-1 antibodies to active controls where salvage patters involving effective agents that may impact OS are expected to confound OS analyses. CheckMate-238, for example, compared nivolumab to ipilimumab and found that nivolumab improved RFS but not OS (20). Pembrolizumab was also found to improve RFS but not OS in the S1404 study, which compared it to ipilimumab and IFN-α. 51 However, the anticipated increase in OS may have more implications for lower-risk patient populations who are candidates for adjuvant therapy. The question arises as to whether adjuvant treatments that only improve RFS without significantly improving OS can be used at the time of relapse to spare lower risk patients AEs and costs of systemic therapy. That is, treating late (at the time of recurrence) versus early (following surgery). However, at this time RFS continues to be the optimal outcome measure for adjuvant trials understanding that an OS endpoint can be confounded by salvage therapy. In addition, we recommend that OS should continue to be considered as a co-primary endpoint. Future phase III studies should allow crossover to be built into the study design that may help shed more light into the OS question which is ultimately the main outcome.
Predictive and prognostic biomarkers
Multiple therapeutic options are currently available for advanced melanoma. For BRAF mutant melanoma, the existence of the activating mutation predicts benefits from BRAF-MEK targeted therapy. However, for immunotherapy, reliable clinical grade biomarkers are yet to be determined.
Serum LDH and S100B have been shown to correlate with prognosis in metastatic melanoma and patients with the high-risk resected disease. Elevated levels may be associated with the development of melanoma recurrence and could trigger imaging studies if not recently obtained.18,52 For instance, Tarhini et al. 18 reported that baseline S100b levels were significantly associated with RFS [HR: 1.70 (95% CI: 1.21–1.92; p < 0.001)] and OS [HR: 1.44 (95% CI: 1.06–1.95; p = 0.0210)], with higher values signifying a worse prognosis.
In the context of the COMBI-AD clinical trial, an IFN-gamma (IFN-γ) gene expression signature along with the tumor mutational burden (TMB) was proposed as potential predictive biomarkers of RFS benefits in BRAF-V600 mutant stage III melanoma. 53 IFN-γ gene expression signature higher than the median was associated with higher RFS in patients treated with dabrafenib plus trametinib group (HR: 0.41, 95% CI: 0.27–0.61, p < 0.0001) as well as in the placebo group (HR: 0.54, 95% CI: 0.39–0.75 p = 0.0002). In the same study, TMB was independently associated with better RFS in the placebo group (HR: 0.56, 95% CI: 0.37–0.85, p = 0.0056). However, no significant association was found between TMB and RFS in patients receiving the dabrafenib–trametinib combination (0.83, 95% CI: 0.53–1.32, p = 0.44). In patients receiving dabrafenib plus trametinib therapy, a low TMB was significantly associated with higher RFS (HR: 0.49, 95% CI: 0.35–0.68, p < 0.0001), in contrast to patients with high TMB, where no significant RFS benefit was seen in patients receiving therapy when compared to placebo (HR: 0.75, 95% CI: 0.44–1.26, p = 0.27). The greatest benefit with dabrafenib plus trametinib over placebo was seen in those with low TMB and high IFN-γ tumors (HR: 0.31, p = 0.0002). The combination of TMB and IFN-γ can therefore identify patients who might benefit from therapy. 18
Lee et al. 54 studied the utility of circulating tumor DNA (ctDNA) in stages IIIB-C-D melanoma patients not receiving systemic adjuvant therapy. Patients with a detectable ctDNA had a median RFS of 6.2 months compared to 13.9 months in patients with undetectable ctDNA (HR: 1.59, 95% CI: 1.0–2.52, p = 0.027). An interesting finding is the ability for ctDNA to stratify the outcome of patients within the AJCC stage groups, despite the absence of association between ctDNA levels and stage groups classification. The study did not find a significant difference in melanoma-specific survival (MSS) at stage IIIB between patients with detectable and undetectable ctDNA (HR: 2.0, 95% CI: 0.74–5.43, p = 0.09). In contrast, a significant difference in MSS between patients with detectable and undetectable ctDNA in stage IIIC (HR: 1.7, 95% CI: 0.95–3.11, p = 0.05) and stage IIID (HR: 6.4, 95% CI: 1.63–25.04, p < 0.01). This indicates the value of ctDNA with a more advanced stage III stage groups, and the possibility to determine prognosis of patients within the specific stage groups based on ctDNA levels.
Another study randomly selected patients with resected high-risk stages II and III melanoma with confirmed BRAF or NRAS mutation from the AVAST-M trial that tested adjuvant therapy with bevacizumab. 55 Patients with detectable ctDNA following surgery had a worse disease-free interval (HR: 3.12, 95% CI: 1.79–5.47, p < 0.0001) as compared to those with undetectable ctDNA. The same pattern was noted for OS in patients with detectable ctDNA (HR: 2.63, 95% CI: 1.40–4.96, p = 0.003).
Discussion
IFN-α was extensively studied in melanoma including various dosages, delivery methods, formulations, and treatment durations. In the adjuvant setting, three RCTs (E1684, E1690, and E1694) tested HDI and showed significant improvements in RFS (three trials) and OS (two trials). The EORTC 18991 trial found that pegylated IFN (PegIFN) reduced relapse probability while not affecting OS. Ipilimumab at a high dosage of 10 mg/kg (ipi10) was later proven to improve RFS and OS, although at a high toxicity cost (EORTC 18071). North American Intergroup E1609 evaluated ipi3 or ipi10 versus HDI and found that ipi3 had significant OS improvement over HDI and less toxicity as compared to ipi10. This trial reported significant improvement in OS compared to an active control regimen for the first time in melanoma adjuvant treatment supported the use of adjuvant ipi3 over HDI. In cases where adjuvant therapy with ipilimumab is an option, such as patients who recur and continue to have operable disease following adjuvant anti-PD-1, ipi3 has an advantage over the approved dosage of ipi10 as supported by E1609 with similar efficacy and less toxicity in favor of ipi3. Based on significant RFS benefits, the standard of management has lately shifted in favor of the anti-PD-1 antibodies nivolumab (CheckMate-238; versus ipi10) and pembrolizumab (KEYNOTE-054; versus placebo). Similarly, dabrafenib with trametinib significantly improved RFS in subjects with BRAF mutant melanoma (COMBI-AD; versus placebo). The clinical benefits were similar when comparing studies that examined anti-PD-1 or dabrafenib plus trametinib in patients with BRAF-mutated melanoma, with no clear advantage for either. The choice of therapy may be based on the patient’s preference for delivery routes and toxicity profiles. A recent Bayesian network meta-analysis examined RFS DMFS and OS in adjuvant trials that tested dabrafenib plus trametinib, nivolumab, pembrolizumab ipilimumab, vemurafenib, chemotherapy, and IFN-α. 56 The analysis revealed that targeted therapy with dabrafenib plus trametinib and anti-PD-1 checkpoint inhibitors had similar efficacy. However, recent trial, DREAMseq, studying patients with BRAFV600-mutant unresectable stage III-IV melanoma, studied two sequencing approaches involving immunotherapy with ipilimumab and nivolumab followed by dabrafenib plus trametinib at disease progression and vice versa. The sequence starting with immunotherapy was associated with a higher OS at 72% (95% CI: 62–81%) compared to 52% (95% CI: 42–62%) for patients receiving targeted therapy with dabrafenib plus trametinib as first-line therapy. 57 Whether this study will impact the choice of adjuvant therapy in patients with BRAF mutant melanoma remains to be answered. However, it may justify a similarly sequencing study in the adjuvant setting.
It is relevant to note that completed clinical trials leading to regulatory approvals utilized previous editions of the AJCC staging system, including the 7th edition for the most recent trials. However, currently in clinical practice the 8th edition is in use. This is important since early 30% of patients with AJCC7 stage III melanoma would be reclassified in a higher stage III group by AJCC8 versus 7% in lower stage group.58,59 For example, when it comes to adjuvant therapy patients with stage IIIA and sentinel lymph metastatic burden of <1 mm were excluded from COMBI-AD and KeyNote-054 based on their AJCC7 stage, while some of these patients (T3a) would be staged as IIIB in AJCC8. 2 These subtle differences in staging should be discussed with patients as applicable to each case and pros and cons of systemic adjuvant therapy should be discussed based on their potential eligibility to published trials inclusion criteria. For patients with stages IIB-IIC melanoma, a careful discussion of the benefits and risks needs to take place with the patients taking into account the toxicities and risks and systemic adjuvant therapy and weighing the option of treating early versus late (at the time of melanoma relapse).
Adjuvant clinical trials currently underway are detailed in Table 2. Among these trials is the S1801 (NCT03698019) which is a large phase II randomized study comparing adjuvant versus neoadjuvant pembrolizumab for clinically detectable and operable AJCC 7th edition stages III-IV high-risk melanoma. To note, PIVOT-12 was recently terminated after disappointing results from the PIVOT IO-001 trial which failed to reach primary endpoints of PFS, objective response rate and OS. 60 Both trials intended to investigate a nivolumab plus bempegaldesleukin combination, with PIVOT-12 testing the combination as adjuvant therapy in patients with completely resected stages IIIA-D or IV melanoma.
Ongoing adjuvant therapy clinical trials.
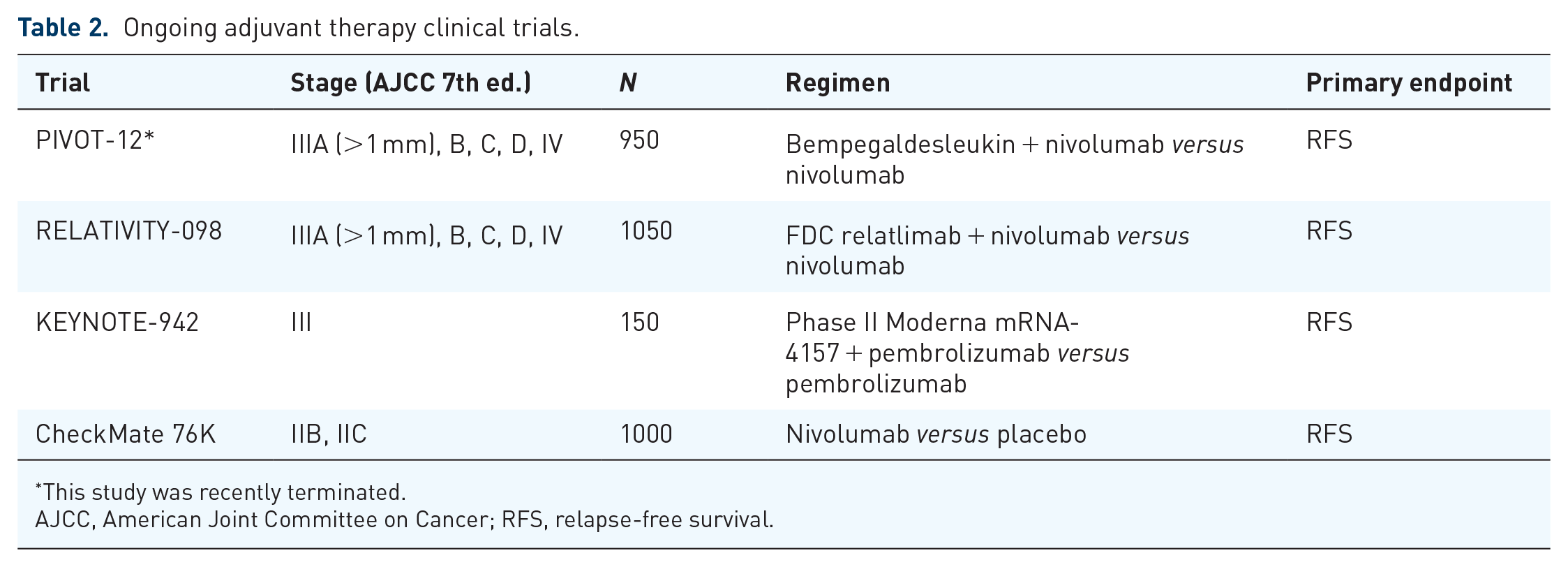
This study was recently terminated.
AJCC, American Joint Committee on Cancer; RFS, relapse-free survival.
Another promising progress is seen with the neoadjuvant approach, which allows for the assessment of clinical and radiologic tumor responses and pathologic responses. Neoadjuvant therapy may also enhance the clinical outcomes for patients with locoregionally advanced operable melanoma. Neoadjuvant therapy utilizing immune checkpoint inhibitors and BRAF-MEK inhibitors has demonstrated promising data in early trials that have started to impact the clinical practice, and phase III testing is currently underway. Both approaches lead to high rates of pCRs, but as reported by Menzies et al. 61 pCRs following immunotherapy may be more durable. In this pooled analysis from six neoadjuvant trials, pCR was significantly associated with improved RFS and OS in patients with stage III resectable melanoma. Patients who had a pCR with neoadjuvant therapy had superior RFS compared to those who did not achieve a pCR, with RFS 89% versus 50% at 2 years, (p < 0.001). 61 In addition, attainment of pCR was shown to be particularly important in targeted therapy as a pathological partial response (pPR) was correlated with similar outcomes as those seen in a pathological no response (pNR). In contrast, patients with near pCR or pPR with immunotherapy had a similar RFS as those with pCR, while those with pNR had poor RFS. Moreover, among patients who underwent immunotherapy and had pCR, near pCR or pPR, relapses were rarely seen with a reported 2-year RFS of 96%.
There is a need for prognostic and predictive biomarkers that may allow us to treat those who destined to relapse and those who predicted to benefit from adjuvant systemic therapy while saving others unwanted toxicities and costs. However, biomarker studies investigated to date are limited by the small sample size of the studies and the retrospective designs of the analyses. Recent research identified TMB, IFN-γ signature expression, and circulating cell-free tumor DNA as potential biomarkers that may utility with adjuvant therapy. Relying on a set of biomarkers will allow better stratification of patients, with high-risk patients set to receive more aggressive treatments early-on while evading the use of highly toxic regimens in patients with a lower risk of relapse. The need for biomarkers that can better define the risk of relapse and death is especially relevant for earlier stages including IIB, IIC, and IIIA where the risk of treatment can be more concerning taking into account the toxicity profiles of systemic adjuvant therapy and the cost to patients and the society.
Conclusion
The choice of adjuvant therapy in resected cutaneous melanoma patients depends on tumor stage, BRAF mutation status, and risk of recurrence. High-risk patients are treated with adjuvant anti-PD-1 immunotherapy or targeted therapy. While adjuvant ipilimumab has favorable survival benefits, it is associated with high toxicity rates. Moreover, anti-PD-1 antibodies, nivolumab and pembrolizumab, have significant benefits over ipilimumab both in terms of RFS and safety profile. Adjuvant therapy with pembrolizumab use is also approved for resected stages IIB and IIC melanoma based on recent results from the Keynote-716 trial. Targeted therapy with dabrafenib plus trametinib has been shown to be an effective adjuvant therapy option for patients with BRAF V600E/K mutant melanoma. Moreover, ongoing trials show a promising role for neoadjuvant therapy utilizing immunotherapeutic and targeted therapy approaches. Future trials should integrate the investigation of predictive and prognostic biomarkers into the clinical trial designs.
Footnotes
Acknowledgements
None.
