Abstract
Background:
Trastuzumab combined with cisplatin and fluoropyrimidines, either capecitabine or 5-fluorouracile (XP/FP), is the standard first-line treatment for advanced, HER2-positive, gastric cancer patients based on the ToGA trial. Despite the lack of phase III trials, many clinicians administer trastuzumab with alternative regimens. One meta-analysis suggests that substituting cisplatin for oxaliplatin might lead to greater efficacy and less toxicity.
Methods:
594 patients with HER2-positive gastroesophageal adenocarcinoma were recruited from the AGAMENON-SEOM registry. The objective was to evaluate the external validity of clinical trials with chemotherapy and trastuzumab.
Results:
The regimens used in at least 5% of the patients were XP (27%), oxaliplatin and capecitabine (CAPOX) (26%), oxaliplatin and 5-fluorouracil (FOLFOX) (14%), FP (14%), triplet with anthracycline/docetaxel (7%), and carboplatin-FU (5%). Median exposure to trastuzumab was longer with FOLFOX (11.4 months, 95% CI, 9.1–21.0) versus ToGA regimens (7.5, 6.4–8.5), p < 0.001. Patients with HER2-IHC 3+ cancers had higher response rates than those with IHC 2+/FISH+, odds-ratio 1.97 (95% CI, 1.25–3.09). The results achieved with CAPOX–trastuzumab were comparable to those attained with ToGA regimens. FOLFOX–trastuzumab was superior to ToGA schemes in terms of overall survival (OS), with a greater magnitude of effect in IHC 2+/FISH+ tumors (HR 0.47, 0.24–0.92) compared with IHC 3+ (HR 0.69, 0.49–0.96), and in diffuse (HR 0.37, 0.20–0.69) versus intestinal-type tumors (HR 0.76, 0.54–1.06).
Conclusion:
We have updated the external validity of clinical trials with trastuzumab in first-line treatment of gastric cancer. Our data confirm the comparable outcomes of ToGA regimens and CAPOX–trastuzumab in clinical practice and point toward a possible benefit of FOLFOX–trastuzumab, contingent on the subtypes typically less sensitive to trastuzumab, to be confirmed in clinical trials.
Introduction
Advanced gastric cancer often exhibits chromosomal anomalies that can be acted upon through receptor tyrosine kinase (RTK)-directed therapies. 1 Overexpression of human epidermal growth factor receptor 2 (HER2) comprises one of the most recurring alterations (9–36%).2,3 In 2010, the phase III randomized controlled trial (RCT) Trastuzumab for Gastric Cancer (ToGA) demonstrated the therapeutic effect of adding trastuzumab to six cycles of cisplatin and fluoropyrimidine (capecitabine, fluorouracil) in patients with cancer of the stomach or of the gastroesophageal junction with HER2 amplification or overexpression (HER2+). 4 Patients who received trastuzumab reached longer overall survival (OS), 13.8 versus 11.0 months, [hazard ratio (HR) 0.74, 95% confidence interval (CI), 0.60–0.91] and impairment of their quality of life delayed. 5 In the subgroup with immunohistochemistry (IHC) 2+ scoring and fluorescence in situ hybridization (FISH)-positive tumors or IHC 3+ tumors, OS was 16.0 months.
The European Medicines Agency approval of this combination in 2010, notwithstanding many questions remaining unanswered, including optimal chemotherapy duration or the most suitable backbone for it. Thus, several subsequent trials have suggested that bi-weekly oxaliplatin-based regimens are better tolerated.6,7 Instead of administering cisplatin- and fluoropyrimidine-based treatments (ToGA regimens), many clinicians tend to incorporate trastuzumab to the standard first-line regimen in their department and tailored to the patient’s specific characteristics (e.g. CAPOX, FOLFOX, or triplets). Similarly, there is no consensus in clinical guidelines with respect to chemotherapy regimen, duration, or the best maintenance strategy. While the National Comprehensive Cancer Network (NCCN) endorses the addition of trastuzumab to any chemotherapy regimen, ESMO guidelines recommend using cisplatin and fluoropyrimidine in HER2+ cancers.8,9
A meta-analysis with 557 patients from 15 prospective or retrospective studies indirectly compared OS of regimens alternative to ToGA. 10 Ter Veer et al. concluded that oxaliplatin-based dual-agent schemes (FOLFOX or CAPOX) associated with trastuzumab were more efficacious than ToGA regimens (median OS 20.7 versus 16 months, HR 0.75, 95% CI, 0.59–0.99), which they attributed to its more favorable toxicity profile, although it was unclear whether this depended on patient- or cancer-related factors or on treatment duration. 10 Trastuzumab associated with anthracycline or docetaxel-based triplet increased toxicity without any gain in efficacy. Nevertheless, the data in favor of regimens with oxaliplatin derive from three small phase II trials that, all told, amount to 140 individuals, and yield superior survival endpoints to those generally seen in larger RCTs (median OS rates around 20 months).11–13 This leaves the door open to the existence of selection factors that could affect the phase II trial outcomes. 14 Therefore, the indirect comparison between ToGA and the pooled estimates of these small, heterogeneous studies (e.g. different eligibility criteria) implies that this kind of causal inference runs the risk of bias. Among the limitations, this method impedes adjusting for covariates, controlling for selection or recruitment bias; similarly, the effect on the overall response rate (ORR) or progression-free survival (PFS) is unknown. 15 There are currently no RCTs that compare ToGA versus trastuzumab with oxaliplatin-based regimens.
With this premise, this study of the AGAMENON-SEOM registry seeks to evaluate the external validity of clinical trials with trastuzumab and chemotherapy in HER2+ gastroesophageal adenocarcinoma, analyze the results of the different regimens, and probe into associated predictive factors.
Materials and method
Patients and study design
The study population is from the AGAMENON-SEOM gastroesophageal cancer registry, in which 34 Spanish centers currently participate.3,16–25 Eligibility criteria for this analysis include age >18 years; histologically confirmed, inoperable, locally advanced, recurrent, or metastatic adenocarcinoma of the distal esophagus, stomach, or gastroesophageal junction; HER2+ status, and having received at least one cycle of polychemotherapy (two or more cytotoxic drugs) associated with trastuzumab. HER2 status was determined locally at the participating centers by means of IHC scoring and/or FISH as part of regular clinical practice. HER2+ overexpression was defined as a +3 IHC score or +2 accompanied by FISH (FISH+). Those participants who had completed perioperative or adjuvant treatment in the preceding 6 months and those HER2+ cancers not treated with trastuzumab in first-line treatment were excluded.
The database is managed via an online platform (http://www.agamenonstudy.com/) and is equipped with multiple tools and filters to guarantee data reliability and consistency, as well as minimizing missing values; regular telephone and online monitoring was also conducted.
The study was performed in accordance with Good Clinical Practice guidelines and the Declaration of Helsinki. It was approved by Research Ethics Committees of all the Autonomous Communities and participating hospitals, and was classified as a prospective, postmarketing surveillance study by the Spanish Agency of Medicines and Medical Devices (AEMPS), and was not otherwise involved in it. The list of the Research Ethics Committees involved in this project is provided in Supplemental Table S1. All participants still alive at the time of data collection provided written, signed, informed consent.
Treatment
All the subjects received trastuzumab and polychemotherapy in first-line treatment. The chemotherapeutic regimen, dose intensity, and number of cycles were chosen at the investigator’s discretion. A loading dose of trastuzumab (T) of 8 mg/kg of body weight and maintenance dose of 6 mg/kg was administered in the tri-weekly chemotherapy regimens. In bi-weekly schemes, trastuzumab was administered with both weekly (loading dose of 4 mg/kg and maintenance dose of 2 mg/kg), and bi-weekly doses (loading dose of 6 mg/kg and maintenance dose of 4 mg/kg). Trastuzumab was administered until progression or unacceptable toxicity.
Variables and objectives
OS was defined as the time between initiation of first-line treatment until death from any cause, censoring subjects who were event-free at their last follow-up. PFS was defined as the interval of time between commencement of first-line treatment until progression as per RECIST 1.1 criteria, demise, or last follow-up. Other variables were used for descriptive or auxiliary purposes. Relative dose intensity was expressed in percentages and defined as the dose intensity (the amount of drug per unit of time, expressed as mg/m2 weekly) administered with respect to the planned amount for each schedule. Multivariable models included nine variables, selected on the basis of theoretical criteria or experience in earlier studies. 20 These covariates were performance status (ECOG-PS), histological grade, Lauren histological classification subtype, bone metastases, liver tumor burden, number of metastatic sites, neutrophil/lymphocyte ratio, chemotherapy regimen, and HER2 status (IHC 3+ versus IHC 2/FISH+). Toxicity was classified according to Common Terminology Criteria for AEs (CTCAE) v4.0. Another descriptive variable was the reason stated by the clinician for choosing the chemotherapy regimen as one of the following alternatives (inability to swallow, clinical trial, induction therapy prior to surgery for metastases, tumor regression in a symptomatic patient, quality of life, comorbidities, clinician’s experience, or department protocol). Clinician’s experience refers to the oncologist choosing the chemotherapy regimen without taking into account considerations regarding the patient, the tumor, or the hospital protocol, but instead, their own experience.
The primary objective was to perform an external validation of the results of the ToGA RCT and other trials focused on HER2+ patients. 10 Secondary objectives were to assess OS, PFS, ORR, toxicity, subgroup effects, and to ascertain the reason for administering each regimen.
Statistics
Categorical and continuous variables were compared using Pearson’s Chi-squared tests and two-sample Wilcoxon tests, respectively. The Cochran–Mantel–Haenszel test was used to examine the association between categorical variables controlling for stratification factors. The assumption that the odds ratios (OR) are homogeneous across several strata was scrutinized by means of the Breslow–Day test. Time-to-event was analyzed using multivariable Cox proportional hazards (PH) models, comparing average treatment effect of ToGA regimens with others, as well as evaluating the effect conditioned on HER2-IHC (tests of interaction). The non-linear effect of the neutrophil-to-lymphocyte ratio (continuous variable) was assessed by means of a restricted cubic spline with three knots (a kind of smooth function that makes it possible to readily capture non-linear relations). All analyses were performed with the R v4.03 software package, with the rms & ggplot2 libraries.26,27
Results
Patients
The AGAMENON-SEOM registry contains 3018 individuals treated between 2008 and 2020. Within this cohort, 22.3% (n = 675) had HER2+ cancers and 19.6% (n = 594) had received first-line trastuzumab-based chemotherapy. Of the latter, 71% (n = 422) had an IHC 3+ tumor, whereas 29% (n = 172) had ICH 2+/FISH+. The predictive model of HER2 positivity is shown in Supplemental Table S2. HER2 overexpression was associated with Lauren intestinal or mixed cancers and tumors located in the distal esophagus or gastroesophageal junction.
Table 1 displays patients’ baseline characteristics. Most were intestinal cancers, although the presence of a diffuse component in one in four and of signet ring cells in 13% is striking. The primary tumor was located in the stomach in 65.3% of the cases (n = 388); in the gastroesophageal junction in 21.2% (n = 126), and in the esophagus in 13.4% (n = 80). Of the subjects with progression at first-line treatment (n = 535), 63% (n = 337) received a second line, and 18% (n = 96) received a third line.
Baseline characteristics.

CAPOX, capecitabine–oxaliplatin; ECOG-PS, Eastern Cooperative Oncology Group Performance Status; FISH, fluorescence in situ hybridization; FOLFOX, fluorouracil–oxaliplatin; FP, fluorouracil–cisplatin; XP, capecitabine–cisplatin.
Selection of chemotherapy regimen
Different chemotherapy backbones were associated with trastuzumab (Table 1 and Supplemental Table S3), although five of them were administered to 87% in the series: capecitabine and cisplatin (XP) in 27% (n = 163), capecitabine and oxaliplatin (CAPOX) in 26%, fluorouracil (FU) and oxaliplatin (FOLFOX) in 14% (n = 86), FU and cisplatin (FP) in 14% (n = 81), and carboplatin and FU (CarFU) in 5% (n = 30). Alternative ToGA regimens were prescribed in 59% (350/594). The main reasons for choosing a specific scheme was compliance with local protocol and the clinician’s experience (Figure 1); other, less frequent motivations included conversion chemotherapy in unresectable disease, preservation of quality of life, inability to swallow, etc. (Supplemental Figure S1). The characteristics of the subjects treated with the different chemotherapy regimens were comparable except for the older age in the CarFU-T-treated group (median 72 versus 65 years), worse functional status in the FOLFOX-T group (ECOG-PS 2, 25.5 versus 11.8%), and the higher percentage of diffuse subtypes treated with FP-T (34.6 versus 17.6%) (Supplemental Table S4).
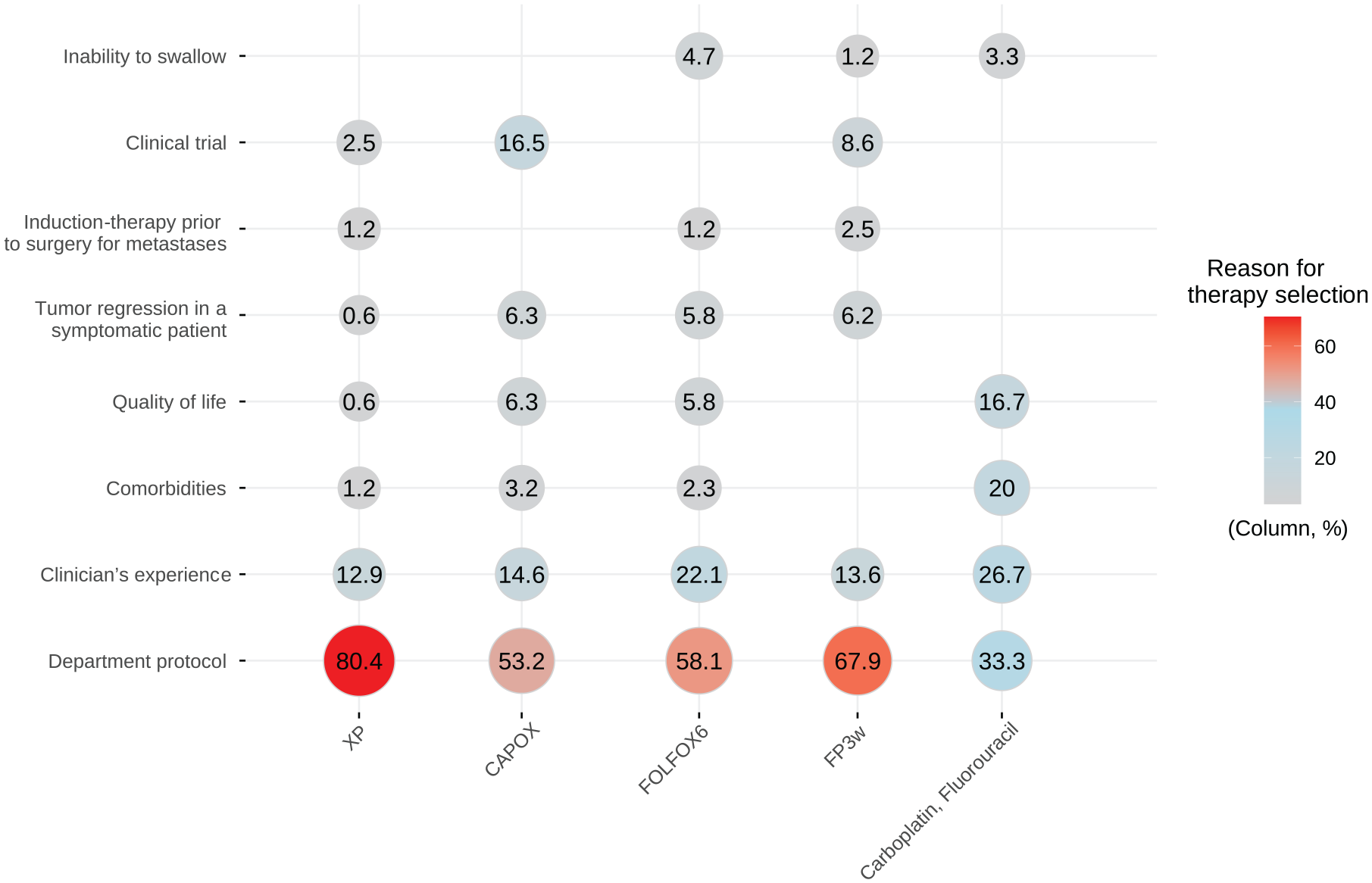
Reasons for therapy selection.
Trastuzumab-based chemotherapy regimens: dosage and administration
Median follow-up in live patients was 62.8 months (95% CI, 42.8–85.1). At the time of analysis (November 2020), 82% had discontinued trastuzumab, with 6.8 months median trastuzumab treatment duration (95% CI, 6.2–7.5). The chemotherapy dosage administered, duration, and criteria for suspension varied depending on the scheme. Supplemental Table S5 illustrates the dosages administered. Maintenance therapies with fluoropyrimidine and trastuzumab were used more frequently following oxaliplatin-based regimens, especially FOLFOX versus ToGA. Thus, median exposure to trastuzumab was more prolonged with FOLFOX (10.6 months, 95% CI, 8.4–20.3) versus CAPOX (7.1 months, 6.1–8.5), FP (7.0 months, 5.5–7.8), or XP (6.8 months, 5.1–7.9) p < 0.001. Furthermore, participants treated with FOLFOX-T received a median of 12 cycles of FU, with tumor progression being the most common cause for withdrawal. In contrast, a median of six cycles of ToGA regimens were administered, with the leading reason for discontinuation being “planned treatment completed” (Supplemental Figure S2).
Safety
Table 2 shows the most common adverse events broken down by treatment type. Most of the toxicities were mild, although 24.2% (n = 136) of the patients were hospitalized for adverse events. Of the subjects treated with XP, 30% were admitted due to toxicity versus around 20% with the remaining regimens.
Treatment-related toxicity in the most frequent regimens.

CTCAE v4.0 grades 2–4 are included.
CAPOX, capecitabine–oxaliplatin; FOLFOX, fluorouracil–oxaliplatin; FP, fluorouracil–cisplatin; XP, capecitabine–cisplatin.
Overall, ToGA schemes exhibited the same hematological toxicity profile as in the original study, except for more anemia of any grade (XP-T 71%, FP-T 65%). Non-hematological toxicity was likewise, comparable, although it depended on the specific regimen. XP-T correlated with more grade 3–4 diarrhea compared with FP-T (XP-T 11% versus FP-T 2%), but less stomatitis (XP-T 2% versus FP-T 11%) (Table 2).
Head-to-head comparisons of cisplatin- and oxiplatin-based regimens revealed that the former associated less grade 3–4 diarrhea (XP-T 4.9%, FP-T 2.5% versus CAPOX-T 10.8%, FOLFOX-T 9.3%) and more grade 3–4 thrombosis (XP-T 8.6%, FP-T 8.6% versus CAPOX-T 2.5%, FOLFOX-T 3.5%). Capecitabine-containing schemes had less grade 3–4 stomatitis (XP-T 3%, CAPOX-T 2%, versus FP-T 11% versus FOLFOX-T 9%) and more palmar-plantar erythrodysesthesia (CAPOX-T 3%, XP-T 2%, versus FP-T or FOLFOX-T 0%) relative to those based on FU. Toxicity-related mortality was uncommon (n = 5, 0.8%) and only occurred in capecitabine-containing regimens (CAPOX-T 1.3%, XP-T 1.8%) (Supplemental Figure S3).
Efficacy
At approximately 3 months of treatment initiation, 437/594 (74%) had measurable and response-evaluable disease. In these cases, the investigator-rated RECIST 1.1 ORR at 3 months revealed: complete response, 1.1% (n = 5); partial response, 55.8% (n = 244); stable disease, 23.1% (n = 101); and tumor progression, 19.9% (n = 87). Individuals with HER2-IHC 3+ cancers had a higher ORR, with common OR 1.97 (95% CI, 1.25–3.09) (Mantel–Haenszel χ2 = 8.060, df = 1, p = 0.004). This association is homogenous regardless of the scheme used (Breslow–Day χ2 = 1.062, df = 4, p = 0.900), with no evidence that the ORR differs based on the regimen (χ2 = 3.977, df = 4, p = 0.409). Figure 2 shows a slope plot with ORR stratified according to HER2 and scheme.

Slope plots with RECIST categories for HER2 ICH 3+ versus ICH 2+/FISH+.
At the time of analysis, 500 fatal events had been recorded, with median OS of 14.0 months (95% CI, 12.9–15), and 535 progression events, with median PFS of 7.6 (7.0–8.2) months. Figure 3 illustrates the Kaplan–Meier curves stratified by chemotherapy scheme.
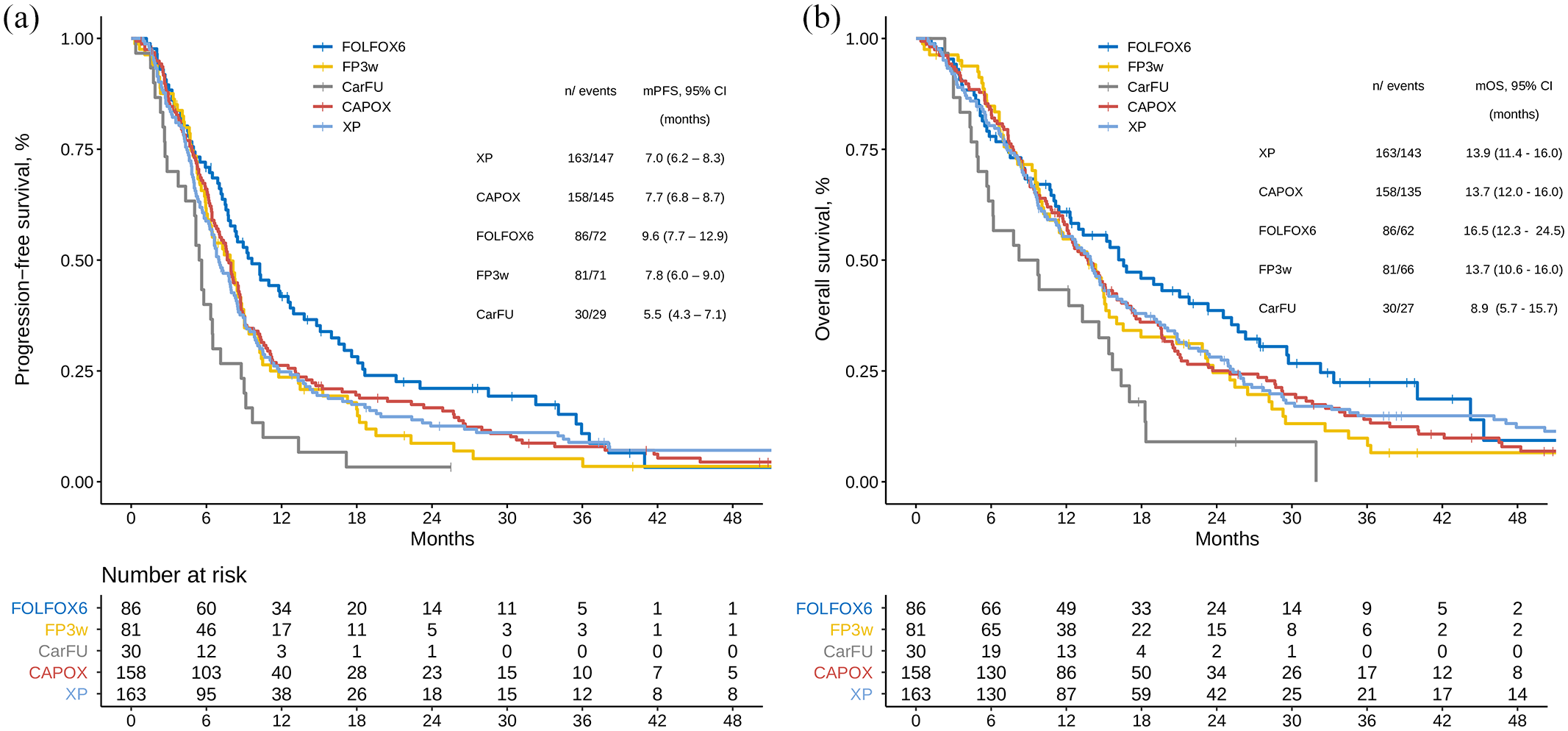
Survival functions stratified by type of treatment. (a) Progression-free survival. (b) Overall survival.
In this clinical practice study, FOLFOX-T reached a median PFS of 9.6 months (7.7–12.9) and OS of 16.5 months (12.3–24.5), comparable to the IHC 3+ or IHC 2+/FISH+ subgroup in the reference RCT. Subjects treated with CarFU-T achieved the lowest medians with PFS of 5.5 months (4.3–7.1) and OS of 8.9 months (5.7–15.7). ToGA regimens displayed the lowest median OS compared with the original RCT: 13.7 months (95% CI, 10.6–16.0) for FP-T and 13.9 months (11.4–16.0) for XP-T. Median OS was comparable in our study for individuals treated with CAPOX-T or ToGA regimens (Figures 3 and 4). PFS data were likewise consistent. The numbers are small, yet a signal in favor of additional benefit with triplets cannot be ruled out (Supplemental Table S6).

Cox proportional hazards model for overall survival.
Using the multivariable Cox PH model, HER2-IHC 2+/FISH+ cancers correlated with worse PFS (HR 1.22, 95% CI, 1.00–1.49) and OS (HR 1.20, 0.98–1.47). As for average treatment effects, the Cox model generates the hypothesis that FOLFOX-T might be superior to ToGA regimens with HR for PFS and OS of 0.60 (95% CI, 0.45–0.79) (p < 0.0003) and 0.63 (0.47–0.85) (p = 0.002), respectively. In a sensitivity analysis, the addition of the variable “reason for treatment choice” (e.g. clinician’s experience, department protocol, etc.) did not modify these estimations (see Supplemental Figure S4). The inclusion of the secular year as a confounding factor did not substantially modify these estimations (datum not shown).
No differences were detected between CAPOX-T and ToGA schemes with HR for PFS and demise of 0.88 (95% CI, 0.71–1.10) and 0.94 (0.75–1.18), respectively (Figure 4 and Supplemental Figure S5). Other Cox model estimates for OS are also displayed in Supplemental Table S7. The non-linear effect of the neutrophil-to-lymphocyte ratio on OS is illustrated in Supplemental Figure S6. Concerning conditional effects, FOLFOX-T displayed a greater magnitude of effect versus ToGA regimens in cancer patients with diffuse (HR 0.37, 95% CI, 0.20–0.69) versus intestinal (HR 0.76, 0.54–1.06) [p(int) = 0.047] subtypes. The same tendency was seen with respect to HER2 overexpression intensity, with HR for deaths of 0.47 (95% CI, 0.24–0.92) in IHC2+/FISH+ versus 0.69 (0.49–0.96) in ICH3+, although the CI were broad [p(int) = 0.317]. No subgroup effects were noted for hepatic tumor burden, bone metastases, histological grade, or for CAPOX-T versus ToGA regimens.
Discussion
In keeping with the ToGA study, trastuzumab plus cisplatin and capecitabine/fluorouracil is standard first-line treatment for HER2+ advanced gastric or gastroesophageal junction adenocarcinoma. Nevertheless, clinicians assiduously use alternative trastuzumab-associated regimens. 8 Ter Veer et al. 28 compared ToGA regimens against alternative chemotherapy schemes administered in phase II and observational studies. 10 Despite the authors’ conclusion that oxaliplatin-based dual-agent schemes were preferable to ToGA regimens, the methodology was susceptible to confounding bias with consistency among the studies being contravened and in the presence of problems of external validity. 29
With this study, we have evaluated the efficacy and safety of trastuzumab-based chemotherapy regimens utilized in the real-world AGAMENON-SEOM registry, and arrived at four main conclusions. First, the ToGA regimens used in the AGAMENON-SEOM registry achieved worse survival outcomes compared with the IHC 3+ or IHC 2+/FISH+ subgroup of the original RCT. The inclusion of non-Asian patients or the greater frailty of the individuals of this sample (ECOG-PS 2 in 14% or >2 comorbidities in 31%) might account for this discrepancy. Moreover, each specific scheme (XP-T versus FP-T) has a slightly different tolerance profile; particularly of note is the 30% of hospitalizations due to toxicity associated with XP-T in our study.
Second, in this series, CAPOX-T behaves similarly to the ToGA regimens, despite its different safety profile (e.g. less thrombosis and stomatitis). As for survival endpoints, our results with CAPOX-T are on a par with those reported by Rivera et al. 30 in a Western population. Nevertheless, they are more discreet than those found in both the IHC 3+ or IHC 2+/FISH+ subgroup from the ToGA RCT, as well as in the two Asian, phase II studies that reported median PFS and OS of approximately 9 and 20 months, respectively11,12 and comprise the basis of the meta-analysis by ter Veer. 10 Consequently, these data similarly fail to replicate the Western population of the AGAMENON-SEOM registry, or point toward a selection bias in favor of patients with better prognoses in both phase II clinical trials.11,12
Third, our data support the hypothesis that, in daily practice, the average treatment effect of FOLFOX-T might be superior to ToGA regimens. This hypothesis should be tested in a RCT, although it is in line with the result reported by Soulareu in a phase II trial with a Western population and incorporated into the meta-analysis by ter Veer.10,13 Tratuzumab dosing in bi-weekly schedules is different, albeit there are data that suggest that these dosages are capable of reaching the minimum concentrations associated with inhibition of tumor growth. 31 One possible cause for this advantage in the AGAMENON registry is that by using ToGA or CAPOX-T regimens, there was a tendency to assume that the treatment had a pre-defined duration as considered for the CF or XP regimens, while with FOLFOX-T, FU may have been managed differently, which facilitated maintenance together with trastuzumab. Optimal treatment duration has not been well defined in advanced gastric cancer.21,32 In the ToGA RCT, individuals without progression after six cycles discontinued chemotherapy and maintained trastuzumab until progression. 4 However, this strategy may not be the best for some patient profiles (e.g. those having a high tumor burden, without toxicity). 21
Beyond the average treatment effects, our study suggests that subgroup effects could be particularly relevant in choosing the optimum backbone of chemotherapy based on ORR and on the trend in survival endpoints. Thus, should this hypothesis that FOLFOX-T might be more efficacious than ToGA regimens be confirmed, it would be especially pertinent in populations who benefit less from this strategy, such as diffuse tumors and those that are HER2+ on IHQ2+/FISH+. 33 The rest of the literature is compatible with this hypothesis. In one pre-planned sub-analysis of the ToGA RCT, cisplatin, fluoropyrimidine, and trastuzumab were seen to have a greater magnitude of treatment effect in IHC 3+/FISH+ (HR 0.58, 95% CI, 0.41–0.81) versus IHC 2+/FISH+ (HR 0.75, 0.51–1.11) tumors. 4 The analysis of biomarkers from the GATSBY RCT of trastuzumab emtansine in second line yielded similar outcomes. 34 In parallel, there are data in the literature that speak to a slight superiority of oxaliplatin dual-agent schemes versus cisplatin in tumors not selected by HER2.6,7 For example, one meta-analysis detected a discreet benefit for oxaliplatin (HR for death 0.88, 95% CI, 0.78–0.99), which, in addition, was tolerated better in bi-weekly regimens. 6 In light of this evidence, one possible explanation for our results is that the smaller targeted effect of ToGA regimens in tumors a priori less sensitive to trastuzumab is offset by the greater cytotoxic spectrum of oxaliplatin in these groups. 4 On the other hand, in cancers that are more sensitive to trastuzumab (intestinal, IHC 3+), trastuzumab’s activity might dilute the lesser efficacy of chemotherapy not based on oxaliplatin. This is also in line with various studies that report that trastuzumab is less effective on tumors with heterogenous HER2 expression, diffuse histopathology, or other molecular alterations that provide resistance.35–37
These considerations may be relevant when choosing treatment schemes in future trials with immunotherapy, since, although the evidence is still limited, preliminary data indicate that oxaliplatin would also be more immunogenic than other platins, given its greater capacity to induce immunogenic cell death. 38 Finally, CarFU-T combinations were the least active, even after controlling for confounding factors. Data with triple-agent regimens are still scarce.
The readers must be mindful that our study has several limitations, the most conspicuous one being the observational design, use of modified or non-conventional regimens, such as CarFU-T, and toxicity being recorded in the course of regular care. Despite attempting to control for the most important confounding factors, residual confounding bias cannot be entirely ruled out. Still, there are no data in this study that point toward a selection bias of patients having a better prognosis among those treated with FOLFOX-T, while this regimen tended to be used more in subjects having a worse ECOG-PS. Another pertinent aspect is that HER2-testing was not centrally analyzed, but rather as part of routine practice in each center. The reader should also be aware that the safety profile was assessed solely for descriptive purposes, without any attempt to match or adjust for covariates. Finally, despite the fact that the study covers individuals treated over the course of 12 years, the sample does not allow for temporal trends to be analyzed. These trends are generally needed to interpret results in the conditions of ever-changing clinical practice.
In conclusion, our data emphasize the diversity of criteria and rationale for the use of trastuzumab-based chemotherapy regimens in everyday clinical practice. This analysis does not support the conclusion of Ter Veer’s meta-analysis that oxaliplatin schemes are more efficacious and safe in general than cisplatin-based regimens, given that in this series of real-world data, the benefit appears to be limited exclusively to FOLFOX-T. Our study generates the hypothesis that this scheme may be more efficacious and convenient than ToGA regimens, whose efficacy is, in turn, comparable to CAPOX-T. If confirmed in RCTs, the incremental benefit of FOLFOX-T could be associated with a pattern of maintaining fluoropyrimidine, and is more relevant in tumor subtypes a priori less sensitive to anti-HER2 therapy. Therefore, these results endorse the future investigation of FOLFOX-T in advanced HER2+ gastroesophageal cancer.
Supplemental Material
sj-docx-1-tam-10.1177_17588359211019672 – Supplemental material for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry
Supplemental material, sj-docx-1-tam-10.1177_17588359211019672 for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry by Paula Jimenez-Fonseca, Alberto Carmona-Bayonas, Alba Martinez-Torron, Maria Alsina, Ana Custodio, Olbia Serra, Diego Cacho Lavin, María Luisa Limón, Tamara Sauri, Flora López, Laura Visa, Mónica Granja, Nieves Martínez Lago, Virginia Arrazubi, Rosario Vidal Tocino, Raquel Hernandez, Gema Aguado, Juana María Cano, Alfonso Martín Carnicero, Monserrat Mangas, Paola Pimentel, Ana Fernández Montes, Ismael Macias Declara, Federico Longo, Avinash Ramchandani, Marta Martín Richard, Alicia Hurtado, Aitor Azkarate, Carolina Hernández Pérez, Raquel Serrano and Javier Gallego in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359211019672 – Supplemental material for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry
Supplemental material, sj-docx-2-tam-10.1177_17588359211019672 for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry by Paula Jimenez-Fonseca, Alberto Carmona-Bayonas, Alba Martinez-Torron, Maria Alsina, Ana Custodio, Olbia Serra, Diego Cacho Lavin, María Luisa Limón, Tamara Sauri, Flora López, Laura Visa, Mónica Granja, Nieves Martínez Lago, Virginia Arrazubi, Rosario Vidal Tocino, Raquel Hernandez, Gema Aguado, Juana María Cano, Alfonso Martín Carnicero, Monserrat Mangas, Paola Pimentel, Ana Fernández Montes, Ismael Macias Declara, Federico Longo, Avinash Ramchandani, Marta Martín Richard, Alicia Hurtado, Aitor Azkarate, Carolina Hernández Pérez, Raquel Serrano and Javier Gallego in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359211019672 – Supplemental material for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry
Supplemental material, sj-docx-3-tam-10.1177_17588359211019672 for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry by Paula Jimenez-Fonseca, Alberto Carmona-Bayonas, Alba Martinez-Torron, Maria Alsina, Ana Custodio, Olbia Serra, Diego Cacho Lavin, María Luisa Limón, Tamara Sauri, Flora López, Laura Visa, Mónica Granja, Nieves Martínez Lago, Virginia Arrazubi, Rosario Vidal Tocino, Raquel Hernandez, Gema Aguado, Juana María Cano, Alfonso Martín Carnicero, Monserrat Mangas, Paola Pimentel, Ana Fernández Montes, Ismael Macias Declara, Federico Longo, Avinash Ramchandani, Marta Martín Richard, Alicia Hurtado, Aitor Azkarate, Carolina Hernández Pérez, Raquel Serrano and Javier Gallego in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-4-tam-10.1177_17588359211019672 – Supplemental material for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry
Supplemental material, sj-docx-4-tam-10.1177_17588359211019672 for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry by Paula Jimenez-Fonseca, Alberto Carmona-Bayonas, Alba Martinez-Torron, Maria Alsina, Ana Custodio, Olbia Serra, Diego Cacho Lavin, María Luisa Limón, Tamara Sauri, Flora López, Laura Visa, Mónica Granja, Nieves Martínez Lago, Virginia Arrazubi, Rosario Vidal Tocino, Raquel Hernandez, Gema Aguado, Juana María Cano, Alfonso Martín Carnicero, Monserrat Mangas, Paola Pimentel, Ana Fernández Montes, Ismael Macias Declara, Federico Longo, Avinash Ramchandani, Marta Martín Richard, Alicia Hurtado, Aitor Azkarate, Carolina Hernández Pérez, Raquel Serrano and Javier Gallego in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-10-tam-10.1177_17588359211019672 – Supplemental material for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry
Supplemental material, sj-jpg-10-tam-10.1177_17588359211019672 for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry by Paula Jimenez-Fonseca, Alberto Carmona-Bayonas, Alba Martinez-Torron, Maria Alsina, Ana Custodio, Olbia Serra, Diego Cacho Lavin, María Luisa Limón, Tamara Sauri, Flora López, Laura Visa, Mónica Granja, Nieves Martínez Lago, Virginia Arrazubi, Rosario Vidal Tocino, Raquel Hernandez, Gema Aguado, Juana María Cano, Alfonso Martín Carnicero, Monserrat Mangas, Paola Pimentel, Ana Fernández Montes, Ismael Macias Declara, Federico Longo, Avinash Ramchandani, Marta Martín Richard, Alicia Hurtado, Aitor Azkarate, Carolina Hernández Pérez, Raquel Serrano and Javier Gallego in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-5-tam-10.1177_17588359211019672 – Supplemental material for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry
Supplemental material, sj-jpg-5-tam-10.1177_17588359211019672 for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry by Paula Jimenez-Fonseca, Alberto Carmona-Bayonas, Alba Martinez-Torron, Maria Alsina, Ana Custodio, Olbia Serra, Diego Cacho Lavin, María Luisa Limón, Tamara Sauri, Flora López, Laura Visa, Mónica Granja, Nieves Martínez Lago, Virginia Arrazubi, Rosario Vidal Tocino, Raquel Hernandez, Gema Aguado, Juana María Cano, Alfonso Martín Carnicero, Monserrat Mangas, Paola Pimentel, Ana Fernández Montes, Ismael Macias Declara, Federico Longo, Avinash Ramchandani, Marta Martín Richard, Alicia Hurtado, Aitor Azkarate, Carolina Hernández Pérez, Raquel Serrano and Javier Gallego in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-6-tam-10.1177_17588359211019672 – Supplemental material for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry
Supplemental material, sj-jpg-6-tam-10.1177_17588359211019672 for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry by Paula Jimenez-Fonseca, Alberto Carmona-Bayonas, Alba Martinez-Torron, Maria Alsina, Ana Custodio, Olbia Serra, Diego Cacho Lavin, María Luisa Limón, Tamara Sauri, Flora López, Laura Visa, Mónica Granja, Nieves Martínez Lago, Virginia Arrazubi, Rosario Vidal Tocino, Raquel Hernandez, Gema Aguado, Juana María Cano, Alfonso Martín Carnicero, Monserrat Mangas, Paola Pimentel, Ana Fernández Montes, Ismael Macias Declara, Federico Longo, Avinash Ramchandani, Marta Martín Richard, Alicia Hurtado, Aitor Azkarate, Carolina Hernández Pérez, Raquel Serrano and Javier Gallego in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-7-tam-10.1177_17588359211019672 – Supplemental material for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry
Supplemental material, sj-jpg-7-tam-10.1177_17588359211019672 for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry by Paula Jimenez-Fonseca, Alberto Carmona-Bayonas, Alba Martinez-Torron, Maria Alsina, Ana Custodio, Olbia Serra, Diego Cacho Lavin, María Luisa Limón, Tamara Sauri, Flora López, Laura Visa, Mónica Granja, Nieves Martínez Lago, Virginia Arrazubi, Rosario Vidal Tocino, Raquel Hernandez, Gema Aguado, Juana María Cano, Alfonso Martín Carnicero, Monserrat Mangas, Paola Pimentel, Ana Fernández Montes, Ismael Macias Declara, Federico Longo, Avinash Ramchandani, Marta Martín Richard, Alicia Hurtado, Aitor Azkarate, Carolina Hernández Pérez, Raquel Serrano and Javier Gallego in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-8-tam-10.1177_17588359211019672 – Supplemental material for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry
Supplemental material, sj-jpg-8-tam-10.1177_17588359211019672 for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry by Paula Jimenez-Fonseca, Alberto Carmona-Bayonas, Alba Martinez-Torron, Maria Alsina, Ana Custodio, Olbia Serra, Diego Cacho Lavin, María Luisa Limón, Tamara Sauri, Flora López, Laura Visa, Mónica Granja, Nieves Martínez Lago, Virginia Arrazubi, Rosario Vidal Tocino, Raquel Hernandez, Gema Aguado, Juana María Cano, Alfonso Martín Carnicero, Monserrat Mangas, Paola Pimentel, Ana Fernández Montes, Ismael Macias Declara, Federico Longo, Avinash Ramchandani, Marta Martín Richard, Alicia Hurtado, Aitor Azkarate, Carolina Hernández Pérez, Raquel Serrano and Javier Gallego in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-9-tam-10.1177_17588359211019672 – Supplemental material for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry
Supplemental material, sj-jpg-9-tam-10.1177_17588359211019672 for External validity of clinical trials with diverse trastuzumab-based chemotherapy regimens in advanced gastroesophageal adenocarcinoma: data from the AGAMENON-SEOM registry by Paula Jimenez-Fonseca, Alberto Carmona-Bayonas, Alba Martinez-Torron, Maria Alsina, Ana Custodio, Olbia Serra, Diego Cacho Lavin, María Luisa Limón, Tamara Sauri, Flora López, Laura Visa, Mónica Granja, Nieves Martínez Lago, Virginia Arrazubi, Rosario Vidal Tocino, Raquel Hernandez, Gema Aguado, Juana María Cano, Alfonso Martín Carnicero, Monserrat Mangas, Paola Pimentel, Ana Fernández Montes, Ismael Macias Declara, Federico Longo, Avinash Ramchandani, Marta Martín Richard, Alicia Hurtado, Aitor Azkarate, Carolina Hernández Pérez, Raquel Serrano and Javier Gallego in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors would like to thank the investigators of the AGAMENON study, the Spanish Society of Medical Oncology (SEOM), Priscilla Chase Duran for editing the manuscript and Natalia Cateriano, Gustavo Reporte, Miguel Vaquero, IRICOM S.A. for supporting the registry website.
Authors’ contributions
A.C.B., P.J.F., J.G. and A.M.T. developed the project, analyzed the data and drafted the manuscript. The other authors recruited patients and provided clinical information, comments, and improvements to the manuscript. All authors participated in the interpretation and discussion of data, and the critical review of the manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethic approval and consent to participate
All procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions. Informed consent was obtained from all patients before being included in the study. This work is original and has not been previously presented elsewhere.
Ethical statement
This study was performed in accordance with the ethical standards of the Declaration of Helsinki and its subsequent amendments. This observational, non-interventional trial was approved by the Research Ethics Committee of all centers.
Informed consent statement
Signed informed consent was obtained from all alive patients.
Consent for publication
Informed consent and approval by the competent national authorities includes permission for publication and dissemination of the data.
Data availability
All the data generated or analyzed in this study are included in the manuscript or the supplementary information.
Code availability
The R Code is available upon request to the authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
