Abstract
Background:
Apatinib, a vascular endothelial growth factor receptor (VEGFR) blocker, has demonstrated encouraging antitumor activities and tolerable toxicities in various cancer types. Recurrent or metastatic adenoid cystic carcinoma of the head and neck (R/MACCHN) carries a poor prognosis, and treatment options are currently limited. This study was conducted to explore the antitumor activity and safety of apatinib in patients with R/MACCHN.
Methods:
In this phase II single-arm, prospective study, patients aged 15–75 years with incurable R/MACCHN received apatinib at a 500 mg dose once daily until intolerance or progression occurred. The primary endpoint was the 6-month progression-free survival (PFS) rate based on RECIST version 1.1. The secondary endpoints included response rate, overall survival (OS), and safety. Efficacy was assessed in all dosed patients with at least one post-baseline tumor assessment.
Results:
Among 68 patients treated with apatinib, 65 were evaluable for efficacy analysis, with a median follow-up time of 25.8 months. The 6-month, 12-month, and 24-month PFS rates were 92.3% [95% confidence interval (CI): 83–97.5%], 75.2% (95% CI: 61.5–84.0%) and 44.7% (95% CI: 32.3–57.5%), respectively. The objective response rate (ORR) and disease control rate (DCR), as assessed by investigators, were 46.2% (95% CI: 33.7–59.0%) and 98.5% (95% CI: 91.7–100.0%), respectively. The median duration of response was 17.7 months [interquartile range (IQR) 14.0–20.9]. The 12-month and 24-month OS rates were 92.3% (95% CI: 83.0–97.5%) and 82.3% (95% CI: 70–90.4%), respectively. The most common adverse events of grades 3–4 were hypertension (5.9%), proteinuria (9.2%), and hemorrhage (5.9%). One patient developed a fatal hemorrhage.
Conclusion:
An encouraging PFS, a high ORR, and a manageable safety profile were observed in this study. It seems that the administration of apatinib in R/MACCHN is likely to have a clinically meaningful therapeutic benefit and warrants further investigation.
This study was prospectively registered in ClinicalTrials.gov (NCT02775370; date of registration: 17 May 2016; date of first patient enrollment: 25 May 2016)
Introduction
Adenoid cystic carcinoma (ACC) most often develops in salivary glands, accounting for approximately 1% of head and neck malignancies. 1 It grows slowly but is prone to recurrence, and distant metastasis. 2 Recurrent or metastatic ACC of the head and neck (R/MACCHN) represents one of the most intractable tumors, for which the progression-free survival (PFS) rate at 6 months (PFS6mos) was less than 48%, 3 and the rate of response to systemic therapy was only 10–30%. 4 Currently, no specific treatment has consistently improved clinical outcomes in patients with R/MACCHN.
Apatinib is one of the latest orally active small-molecule tyrosine kinase inhibitors (TKIs) and highly selectively binds to and strongly inhibits vascular endothelial growth factor receptor (VEGFR)-2, resulting in a decrease in vascular endothelial growth factor (VEGF)-mediated endothelial cell migration and proliferation. It has shown encouraging antitumor activities and tolerable toxicities in preclinical settings and clinical trials.5–7 There is substantial evidence highlighting VEGF overexpression in ACC,8,9 representing a potential target for anti-angiogenic cancer therapy. 10 Hence, we conducted a single-arm, phase II trial to explore the feasibility, efficacy, and toxicity of this novel VEGFR-2 inhibitor in patients with R/MACCHN.
Methods
This investigator-initiated phase II prospective study was approved by the ethics committee of Shanghai Ninth People’s Hospital affiliated to Shanghai Jiao Tong University, School of Medicine (2016-74-T31), registered under http://www.clinicaltrials.gov [ClinicalTrials.gov identifier: NCT02775370], and performed according to good clinical practice guidelines. All patients provided written informed consent before enrollment.
Patients
Patients aged 15–75 years with histologically confirmed ACC of the head and neck with metastatic and/or locally recurrent status, surgical incurability, and progression of a measurable lesion on previous palliative chemotherapy or documented radiological progression during or within 3 months of the most recent treatment were enrolled in the trial. Additional inclusion criteria were as follows: an Eastern Cooperative Oncology Group (ECOG) performance status ⩽2; adequate hematological and end-organ function, and adequate bone marrow function within 2 weeks before treatment.
The main exclusion criteria included: confirmed hypersensitivity to apatinib and/or its excipients; poorly controlled hypertension despite antihypertensive drug treatment; grade I or higher coronary heart disease, evidence of active bleeding or bleeding diathesis, suggesting a bleeding tendency; uncontrolled diabetes mellitus or an acute infection; a positive urine protein test; and the presence of multiple factors affecting oral treatment administration.
Study assessments
Demographic data and medical history were collected during screening. A physical examination, vital sign evaluations, and other safety assessments (ECOG performance status, registration of concomitant medication, hematology tests, biochemistry tests, thyroid function tests, and urine analysis) were performed at baseline. Hematology and biochemistry assessments were performed throughout the treatment. Measurable disease was assessed and documented before any treatment. Tumor response assessments according to RECIST version 1.1 11 were carried out by investigators using computed tomography (CT) or magnetic resonance imaging (MRI) at screening and then at weeks 4, 8, 16, and 24 and then every 12 weeks until disease progression. Additional scans were warranted if there was a need for confirmation of response (no sooner than 4 weeks and no later than 8 weeks after initial observation) or whenever disease progression was suspected.
For evaluation of safety, all adverse events (AEs) were monitored throughout the study and graded according to the National Cancer Institute common terminology criteria for adverse events (version 4.03). Patients were followed for an additional 30 days after discontinuing treatment.
Study design and treatment
In this open-label, non-randomized, phase II study, patients received apatinib 500 mg/day orally once a day in 28-day cycles continuously until documented progressive disease intolerance, a patient or physician decision to withdraw, or death. Treatment was interrupted if there was intolerable grade 2 toxicity, grade 3 toxicity of a significant clinical risk despite optimal management or any grade 4 toxicity. It was restarted if toxicity resolved to grade 1 or baseline levels for non-hematological toxicities and to grade 2 or below for hematological toxicities within 4 weeks. If the AE was unrelated to the study therapy, treatment was resumed with no dose change. If the AE was deemed related to the study treatment, treatment was resumed at a reduced dose of 250 mg/day or patients who needed to wait until their AEs were resolved; dose interruption for more than 4 weeks required discontinuation of the study treatment. Whenever possible, patients who discontinued treatment were encouraged to continue regular tumor assessment until documented progressive disease or death.
Outcomes
The primary endpoint was PFS6mos assessed by an investigator, according to RECIST version 1.1. PFS is an endpoint recommended for phase II studies of ACC and has been used in previous studies.12,13 The secondary endpoints included objective response rate (ORR), time to response, duration of response, disease control rate, the overall survival rate at 12 months, and toxicity, according to National Cancer Institute common toxicity criteria (version 4.0).
Statistical analysis
The safety data were analyzed based on the safety population, defined as all patients who received at least one study treatment dose. The efficacy data were analyzed based on an efficacy analysis population, defined as all patients who received at least one dose of the study treatment and had at least one post-baseline tumor assessment. The database lock-off date was 31 August 2019.
The target enrollment size was 65 patients. It was selected to provide 80% power when using the one-sample log-rank test to reject the null hypothesis that the PFS6mos was ⩽48% with a two-sided type I error of 0.05, assuming the true PFS6mos was 61%, the accrual period was 18 months, the follow-up period was 12 months, and the drop-out rate was 10%. The justification of the 48% cut-off was based on the result of a previously published phase II trial that assessed the activity of gemcitabine, in which the PFS6mos was 48%. 3 The study protocol’s writing committee regarded an increase from 48% to 61% as clinically relevant.
The Kaplan–Meier method was applied for estimates of PFS, time to response, duration of response, and OS. To investigate the relationship between PFS and some factors, Cox proportional hazards models were fitted, and the results are presented as hazard ratios and 95% confidence intervals (CIs) and p values. PFS was defined as the time from the start of treatment to disease progression or death from any cause (whichever occurred first) or last tumor assessment for patients alive without progression. The exact 95% two-sided CIs for ORR and DCR values were calculated using the Clopper–Pearson method.
All statistical analyses were performed using IBM SPSS 23 software (SPSS, Inc., Chicago, IL, USA) and R software (version 3.5.1).
Results
Patient characteristics and disposition
From May 2016 until December 2017, 79 patients with R/MACCHN were screened for enrollment. A total of 68 patients were enrolled in the trial and were eligible for inclusion in the safety population, and 65 (95.6%) patients were eligible for inclusion in the efficacy analysis population. Three of the 68 patients in the safety population were excluded from the efficacy analysis population because they withdrew from the study without any post-treatment tumor assessment (Figure 1).

Patient disposition.
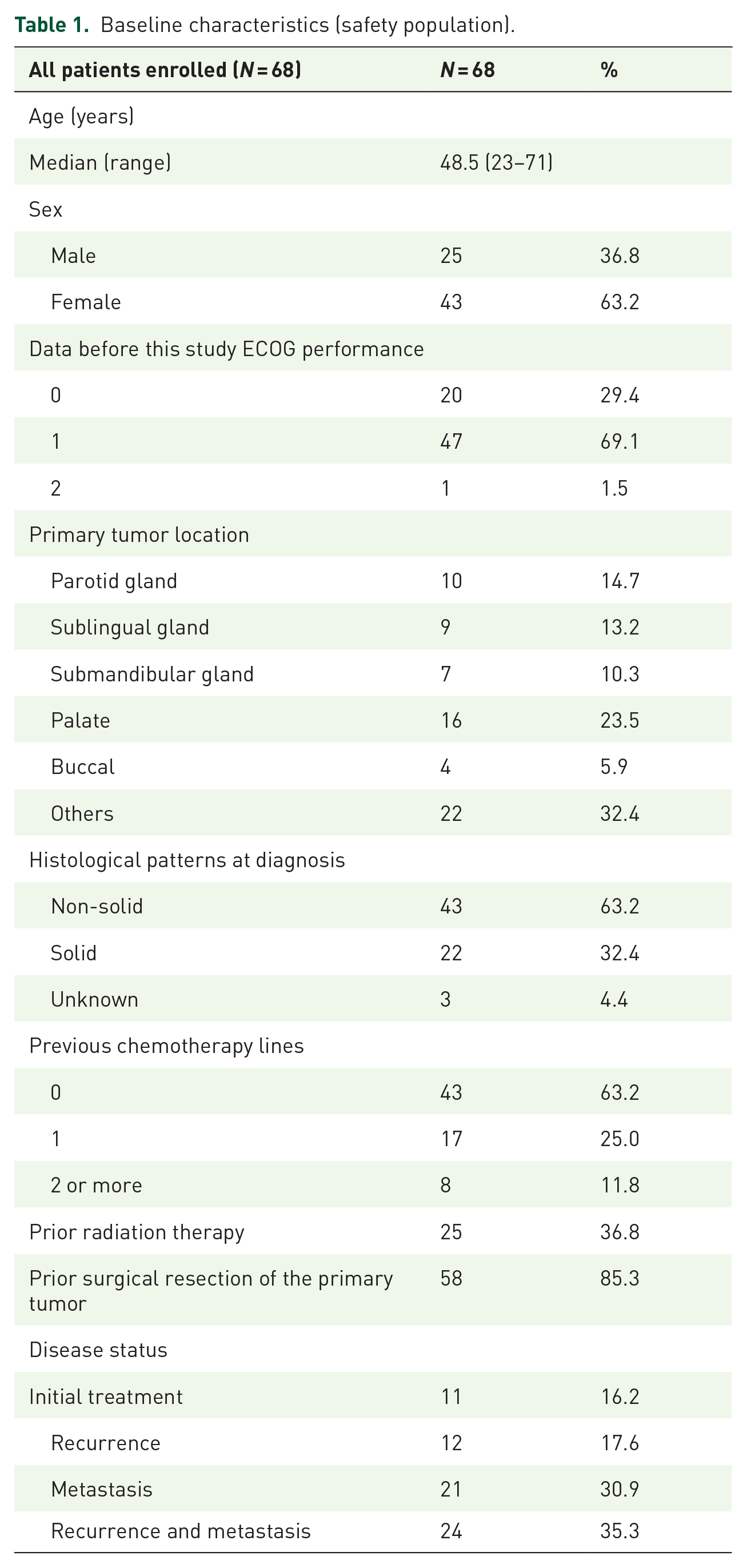
Baseline characteristics are shown in Table 1. The median age of the patients was 48.5 years (range 23–71); 63.2% of patients were women. The performance statuses at baseline were ECOG 0, 1, and 2 in 20 (29.4%), 47 (69.1%) and 1 (1.5%) patient, respectively. The primary tumor was localized to the major salivary glands, and minor salivary glands in 26 (38.2%), 42 (61.8%) patients, respectively. Forty-five (66.2%) patients had metastatic disease, of which the lung was the most frequent metastasis location (35 out of 45, 77.8%). Thirty-six (52.9%) patients showed local recurrence of the primary tumor. Fifty-eight (85.3%) patients had prior resection of the primary tumor, and 25 (36.8%) had previous treatment with radiotherapy. Twenty-five of 68 patients (36.8%) had one or more previous lines of treatment before enrollment in the study.
Baseline characteristics (safety population).
The median follow-up duration at the time of data analysis (data cut-off was on 31 August 2019) was 25.8 months [interquartile range (IQR) 21–30.2]. At the cut-off data point, a total of 54 patients discontinued the study. Eleven of 68 treated patients remained on study. The median duration of treatment exposure was 11.5 months (IQR 4.1–19.3). The primary reason for treatment discontinuation was disease progression [15 (23.1%) of 65 patients]. Nine (13.8%) patients dropped out of the trial at the physician’s discretion because these patients responded poorly to drug therapy due to lack of efficacy and were assessed to be at risk of more serious toxicities. Treatment was also discontinued in 12 (18.5%) patients due to treatment-related AEs, including hemorrhage (4), pulmonary infection (3), proteinuria (2), fatigue (2), and hand–foot syndrome (1). Other reasons for trial discontinuation are summarized in Figure 1.
Efficacy
Survival analysis was performed in the efficacy analysis population. Figure 2 shows the Kaplan–Meier curves for PFS and OS. Forty-three [66.2% (95% CI 53.4–77.4%)] PFS events and 14 deaths were documented. The median PFS was 19.7 months (IQR 11.8–26.0). The 6-month, 12-month, and 24-month PFS rates were 92.3% (95% CI: 83–97.5%), 75.2% (95% CI: 61.5–84.0%), and 44.7% (95% CI: 32.3–57.5%), respectively. The median survival was not reached. The 6-month, 12-month, and 24-month OS rates were 93.8% (95% CI: 85.0–98.3%), 92.3% (95% CI: 83.0–97.5%), and 82.3% (95% CI: 70–90.4%), respectively. Thirteen patients died before the last follow-up due to disease progression, and one patient developed treatment-related fatal bleeding. In multivariable analyses with the Cox regression model, patients who received a higher intensity of apatinib treatment over 6 months (reached or exceeded 66.7% of the planned dosage) had significantly longer PFS than those who received a lower intensity of apatinib (p = 0.024, hazard ratio 0.033, 95% CI 0.002–0.639). Other possible prognostic factors included in the multivariable model, including sex, age, the status of the disease (recurrent or distant metastasis), and first-line or multiple-line previous treatments, did not reveal a statistically significant influence on PFS.

Efficacy of apatinib based on RECIST assessed by investigator review: (a) Best overall response: waterfall plot for the maximum percentage change in target lesion size in each patient. The waterfall plot for the maximum percentage change in target lesion size in 65 patients had at least one post-baseline efficacy assessment. Six patients had a 0% change from baseline. The color indicates the type of response. Red represents progressive disease, yellow represents stable disease, and blue represents a partial response. (b) Percentage change from baseline in target lesion size over time. The Y-axis values at 20% represent the boundary for determination of progressive disease, and the values on the Y-axis at −30% represent the boundary for determination of partial response.
The ORR analysis included 65 patients who were eligible for the efficacy analysis population. We did not observe complete response (CR), but 30 [46.2% (95% CI 33.7–59.0%)] patients achieved the best overall response of partial response (PR) with apatinib. Disease control was achieved in 64 [98.5% (95% CI: 91.7–100.0%)] patients (Table 2). After 6 months of treatment and follow-up, the ORR and disease control rate (DCR) decreased to 33.8% (95% CI: 22.6–46.6%) and 70.8% (95% CI: 59.7–81.8%), respectively. Among patients who achieved a response, the median response duration with apatinib was 17.7 months (IQR 14.0–20.9). Overall, 55 patients with at least one post-baseline tumor assessment had a decrease from baseline in the size of their target lesions. The median change from baseline was –32.5% (95% CI 26.6–38.4%) (Figure 3).
Response and survival data (efficacy analysis population).

BOR, best of response; CI, confidence interval; CR, complete response; DCR, disease control rate; IQR, interquartile range; NE, not evaluated; ORR, objective response rate; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease.

Kaplan–Meier graph for progression-free survival and overall survival.
Safety
In 66 of 68 (98.3%) patients treated with at least one cycle of apatinib (the safety population), at least one AE was reported for each patient. Serious adverse events (SAEs) were reported in 10 (14.7%) patients. A total of 194 AEs were recorded, including 23 AEs of grade 3 in 18 (26.5%) patients and four AEs of grade 4 in four (5.9%) patients. One case of treatment-related fatal hemorrhage of grade 5, resulting in death, was recorded. The patient showed recurrent maxillary sinus ACC and underwent surgery and 66 Gy external beam radiation therapy. After relapse, radioactive particles were implanted; however, the disease could not be controlled. During apatinib treatment, necrosis and infection developed, which resulted in fatal bleeding. Five patients also experienced hemorrhagic complications, among whom four had episodes of hemorrhage occurring at the recurrent lesion site, and one had ulcerative bleeding at the previous radiotherapy site. A list of the AEs is shown in Table 3. The most frequently occurring AEs were hypertension (64.7%), hand–foot syndrome (39.7%), proteinuria (29.2%), transferase increase (25.3%), fatigue (23.5%), and thyroid-stimulating hormone (TSH) increase (19.1%), while neutropenia (8.8%), thrombocytopenia (7.4%), anemia (7.4%), pulmonary infection (4.4%) and diarrhea (1.5%) were observed occasionally.
Treatment-related adverse events (safety population, N = 68).

AE, adverse event; TSH, thyroid-stimulating hormone.
Treatment-related AEs
Treatment- related AE refers to AEs that started or worsened in severity on or after the first dose of apatinib and no later than 30 days from the date of last study treatment administration.
The percentages were calculated based on the total number of subjects in the safety population (N = 68).
Dose reductions were performed in 50 (76.9%) of the 65 patients, of whom 25 (50%) patients took medication intermittently. Seven (14%) patients required only one dose reduction, 18 (36%) patients stopped taking drugs for more than 2 weeks, and only one of them recovered after 3 weeks. The average intensity of apatinib in the first 6 months was 70% of the planned dosage. After the first six cycles, 35 patients (53.8%) had an apatinib intensity higher than the average intensity level, and 30 patients (46.2%) did not reach the average intensity level.
Discussion
Treatment of R/MACCHN remains a challenge. Given the rarity and indolent nature of ACC, few clinical trials define the optimal approach in patients with R/MACCHN. There is no clear evidence that survival is prolonged by systemic therapy. It is also unclear whether prolonged stable disease (SD) represents a real drug effect or a manifestation of the natural history of an indolent malignancy, which is difficult to discern, particularly as these trials did not require clear evidence of disease progression before enrollment. 4
In our study, we required radiological evidence of disease progression before enrollment as part of the eligibility criteria to ensure the contribution of the study therapy to disease stabilization. The primary hypothesis was proved: apatinib had clinical efficacy in patients with R/MACCHN who showed disease progression before treatment. The 92.3% PFS6mos reported here compares favorably with the rates of 39.5–48% observed in several phase II trials of other chemotherapeutic drugs and multitargeted TKIs tested in ACC, including axitinib, 14 sorafenib, 15 and gemcitabine. 3 The median PFS duration was 19.8 months, which compares favorably to the median PFS values associated with sunitinib 16 and sorafenib, 15 which were 7.2 and 11.3 months, respectively.
PFS may be a more meaningful endpoint than the ORR for the evaluation of non-cytotoxic agents in ACC. In our study, 98.5% of patients achieved disease control, and tumor shrinkage within the PR criteria was observed in 46.2% of patients. The response rate was much higher than that reported in several phase II trials (0–11%) of other multitargeted TKIs tested for ACC, including sunitinib, 16 sorafenib, 15 dovitinib, 17 and lenvatinib. 18 There have been few objective responses in previous trials, but several studies have demonstrated high rates and prolonged disease stabilization. Considering the excellent efficacy, it maybe benefits from the strict screening of patients to exclude ones with stable disease or slow progression. On the other hand, the inhibition rate of apatinib on VEGFR-2 is much higher than those of sorafinib, sunitinib, or pazopanib.19–22
Limited data suggest that cytotoxic chemotherapy agents appear to have sufficient activity to treat metastatic or locally recurrent ACC.23,24 This study included 25 patients (36.8%) who had received chemotherapy before. The main benefit of apatinib lies in its theoretically expected efficacy in chemoresistant ACC. The efficacy of apatinib was promising compared with that in other studies that tested similar anti-angiogenic drugs. Another strength is that apatinib was orally administered without the need for hospitalization or an infusion pump, which might have improved patient adherence and economic effectiveness. We remain hopeful that the novel anti-angiogenic treatment option will play a role in R/MACCHN.
Hypertension, proteinuria, and hand–foot syndrome are considered the most common AEs related to anti-angiogenic agents. 25 In our trial, most of these adverse reactions were of grades 1 to 2, which is generally consistent with those reported in previous studies. 26 In this study, one fatal hemorrhage was observed and was considered to be associated with the treatment. The occurrence of a grade 5 bleeding event requires further study to examine any association between prior radiation and subsequent bleeding risk. A careful selection of patients is mandatory to minimize severe bleeding risk, especially in pretreated areas. Toxicities attributable to VEGFR TKIs have been reported as predictive biomarkers for treatment efficacy. 26 We found no toxicities correlated with improved PFS in a post hoc exploratory analysis of toxicity and efficacy.
Cox regression analysis did not show that the duration of medication changed the survival prognosis of patients. Conversely, the intensity of apatinib administered within 6 months strongly suggested that patients taking apatinib at the full dose had a better PFS6mos. Nevertheless, most patients underwent dose modifications because of AEs in our study. The AEs were usually manageable with appropriate interventions, and the treatment was continued in nearly all patients after dose modifications. Generally, with medication prolongation, the side effects observed and recorded are more serious and frequent. Taking these findings into account, we suggest that careful selection of patients for therapy with angiogenesis inhibitors (i.e. those patients with good performance status and controlled blood pressure) and close monitoring and prompt intervention are necessary to alleviate the risks posed by these toxicities.
Furthermore, we acknowledge that this study has some limitations. First, it lacked validation with randomization and included a single arm with no control group for comparison. All the external comparators were derived from the published literature. Second, most previous studies used ORR as the primary endpoint because objective responses and stabilization of progressive disease infrequently suggest a real therapeutic effect. We selected the probability of PFS at 6 months, considering that the disease control time was more important than the short period of tumor stabilization or retraction. However, for PFS, the data were not mature enough. The calculated median PFS and median OS values might not be stable. Nevertheless, the study results demonstrated that the calculated sample size based on the PFS6mos was sufficient for ORR evaluation, a key secondary endpoint. The exact 95% two-sided CI for ORR excluded an ORR of 33% as a lower bound, deemed clinically meaningful. Further follow-up for PFS and OS is ongoing. Third, the biomarker analysis (e.g. analyses of VEGF, c-kit, and histological patterns) was not comprehensive. The association between the activities of apatinib and tumor antigenicity and aggressiveness remains inconclusive.
Conclusion
The study shows the promising efficacy of apatinib in R/MACCHN patients compared to the disappointing results of previously studied cytotoxic and targeted agents. Further investigation to support the administration of apatinib in R/MACCHN patients is needed.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359211013626 – Supplemental material for Apatinib in patients with recurrent or metastatic adenoid cystic carcinoma of the head and neck: a single-arm, phase II prospective study
Supplemental material, sj-pdf-1-tam-10.1177_17588359211013626 for Apatinib in patients with recurrent or metastatic adenoid cystic carcinoma of the head and neck: a single-arm, phase II prospective study by Guopei Zhu, Lin Zhang, Shengjin Dou, Rongrong Li, Jiang Li, Lulu Ye, Wen Jiang, Minjun Dong, Min Ruan, Wenjun Yang and Chenping Zhang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359211013626 – Supplemental material for Apatinib in patients with recurrent or metastatic adenoid cystic carcinoma of the head and neck: a single-arm, phase II prospective study
Supplemental material, sj-pdf-2-tam-10.1177_17588359211013626 for Apatinib in patients with recurrent or metastatic adenoid cystic carcinoma of the head and neck: a single-arm, phase II prospective study by Guopei Zhu, Lin Zhang, Shengjin Dou, Rongrong Li, Jiang Li, Lulu Ye, Wen Jiang, Minjun Dong, Min Ruan, Wenjun Yang and Chenping Zhang in Therapeutic Advances in Medical Oncology
Footnotes
Author contributions
Chenping Zhang, Guopei Zhu, Lin Zhang, and Shengjin Dou designed the study and wrote the manuscript. Jiang Li conducted pathology consultations and reviews. Minjun Dong carried out independent imaging assessments. Shengjin Dou and Lulu Ye performed the bioinformatics analysis. Guopei Zhu and Shengjin Dou prepared the figures. Guopei Zhu, Rongrong Li, Lin Zhang, Min Ruan, Wenjun Yang, Jian Sun and Chenping Zhang gathered patient data and provided clinical interpretations. All authors contributed to and approved the final manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shanghai Municipal Commission of Health and Family Planning (grant number 201640158), the Clinical Research Program of 9th People’s Hospital, Shanghai Jiao Tong University School of Medicine and the Shanghai Shenkang Hospital Development Center Refractory Diseases Project (project number 16CR2004A); Clinical Research Program of 9th People’s Hospital, Shanghai Jiao Tong University School of Medicine (program number JYLJ201825).
Prior presentation
This study was presented in part as a poster at the 2018 ASCO Annual Meeting (abstract no. 6026).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
