Abstract
Background:
There is an unmet clinical need for the locoregional recurrent head and neck squamous cell carcinoma (HNSCC). Moreover, little data regarding the therapeutic survival outcomes are available for the unresectable recurrent setting that did not receive radiotherapy during the initial course of treatment.
Objectives:
To investigate the survival outcomes of radiation-naïve recurrent HNSCC who were treated with first-line immunotherapy-based systemic therapy in combination with radical locoregional radiotherapy.
Design:
This is a retrospective study.
Methods:
From January 2019 to December 2023, locoregional recurrent HNSCC patients receiving immune checkpoint inhibitor (ICI)-based systemic therapy plus locoregional radiotherapy as first-line treatment in our institution were selected. Median follow-up was 16.4 months.
Results:
A total of 23 patients with recurrent HNSCC met the inclusion criteria and were finally analyzed. The median time to progression from the beginning of initial course of treatment was 9.3 months. Nineteen patients (82.6%) harbored recurrent stage IV (rIV) disease according to AJCC eighth edition, and 17 (73.9%) patients were assessed unresectable. For overall cohort, the median progression-free survival (PFS) and locoregional progression-free survival (LRPFS) were 17.0 months and 27.2 months, while the median overall survival (OS) and distant metastasis-free survival (DMFS) were not reached. The 1-year OS, PFS, LRPFS, and DMFS were 100%, 79.5%, 79.5%, and 100%, respectively. Twenty patients obtained objective response during the treatment course, achieving the 1-year duration of response (DOR) of 75.3%, and the median of 16.7 months. Combined positive score (CPS) ⩾ 20 was unveiled to be correlated with significantly favorable PFS compared with CPS < 20 or unknown (1-year PFS: 100% vs 50.0%, p = 0.035).
Conclusion:
This study presented promising survival and tumor control with durable response in recurrent HNSCC, supporting the use of radical RT as the first-line treatment in addition to ICI-based systemic therapy, in particular for patients with CPS ⩾ 20.
Keywords
Introduction
Head and neck carcinoma (HNC) is the seventh most common malignancy globally. 1 According to GLOBALCAN report, an estimated 745, 000 new HNC cases and more than 364,000 HNC deaths occurred in 2020. 1 Accordingly, in China, there were an estimated 94,600 new cases and 52,100 deaths in 2022, 2 with squamous cell carcinoma accounting for over 95%. Approximately 60% of head and neck squamous cell carcinoma (HNSCC) patients harbored locally advanced disease (LA-HNSCC) at the initial diagnosis, with the reported 5-year overall survival (OS) being <50%. 3 There is significant locoregional failure in HNSCC. For resectable disease receiving radical surgery and postoperative radiotherapy, EORTC 22931 study demonstrated a 5-year locoregional recurrence of 31%. 4 For LA-HNSCC patients receiving curative chemoradiotherapy, locoregional recurrence remains the predominant pattern of failure, with over 50% of failure occurring within 2 years after treatment.3,5 –7
Locoregional recurrent HNSCC possesses detrimental survival outcomes. 8 Without distant progression of disease, local and regional management may play a more prominent role for the locoregional recurrent setting. So far, study reports investigating local therapy for this specific setting predominantly focused on salvage surgery or re-irradiation. Patients were primarily evaluated their eligibility for salvage surgery. For patients eligible for salvage surgery, the subsequent OS and disease-free survival (DFS) were much better than those without chance of surgery.9 –11 For recurrent patients who were not suitable for salvage surgery, the most discussed treatment modality was re-irradiation. In this circumstance, a series of retrospective studies reported the median OS of <1 year and approximate 5-year OS rate of only 10%.12 –15 However, scarce literature reported the therapeutic survival outcomes for the unresectable recurrent setting that did not receive radiotherapy (RT) during the initial course of treatment, with remarkably inferior OS and progression free survival (PFS) compared with resectable disease. 16
Systemic therapy is another important modality for the treatment of recurrent HNSCC. Before the emergence of immunotherapy, EXTREME regimen was the primary option for the metastatic HNSCC and was also selectively used in the recurrent setting. 17 In recent years, immune checkpoint inhibitor (ICI), represented by the PD1/PDL1 checkpoint inhibitors, has become the mainstay in recurrent/metastatic HNSCC (R/M HNSCC).18,19 By comparing with standard EXTREME regimen, KEYNOTE 048 study revealed that pembrolizumab plus platinum and 5-fluorouracil (PF) could significantly improve OS in R/M HNSCC, and even pembrolizumab monotherapy could markedly extend OS in R/M HNSCC with combined positive score (CPS) ⩾ 1.18,20 However, no PFS improvement was found in either pembrolizumab or pembrolizumab with chemotherapy groups. Additionally, it should also be noticed that the OS benefits from pembrolizumab were more pronounced in metastatic patients whereas no survival increase was obtained from immunotherapy over EXTREME regimen for the specific setting with locoregional failure only.
Considering the limitations of the above-mentioned treatment strategies, there is an unmet clinical need for the locoregional recurrent HNSCC. Moreover, there are little data regarding the therapeutic survival outcomes in the unresectable recurrent setting that did not receive radiotherapy during the initial course of treatment. We hypothesize that the combination of upfront locoregional RT and ICI may have complementary effects and consequently augment the effectiveness of each therapy. Therefore, this study aimed to investigate whether de novo locoregional RT with curative intent in combination with ICI-based systemic therapy could delay progression and further prolong survival outcomes in locoregional recurrent HNSCC that did not receive RT previously.
Patients and methods
Study population
This is a single-center retrospective study. HNSCC patients who were treated with PD1/PDL1 ICI integrated systemic therapy plus locoregional radiotherapy in our institution between January 2019 and December 2023 were primarily selected to obtain a minimum follow-up duration of 6 months. Among these patients, those with merely locoregional recurrence and receiving radiotherapy and ICI as first-line treatment after the diagnosis of recurrence were included in the final analysis. Patients with distant metastases or secondary primary tumor were excluded. Patient, disease, treatment, and follow-up data were extracted from medical records and further analyzed. This study was approved by the local institutional review board (IRB Number 24/214-4494), and written consent was waived as a retrospective study. The reporting of this study conforms to the STROBE statement 21 (Supplemental Table 1).
Treatment regimen
All patients were discussed in multidisciplinary team (MDT). Basically, the “unresectable” patients included those with clinical rIVB disease, rIVA patients receiving surgical resection whilst with residual disease (namely R2 resection), and patients with recurrent lymph node encompassing vessels significantly. All enrolled patients received volumetric modulated arc therapy (VMAT)-based RT and PD1 inhibitor-based immunotherapy.
Target delineation shared the same principle with the locally-advanced setting. 22 Radiation volume included primary lesion, metastatic lymph nodes, and corresponding expansion to cover the potential subclinical tumor extension to the vicinity of the primary or lymph nodes as well as elective lymph node region. In patients with postoperative residual tumor or without surgery, the gross tumor volume (GTV) in primary lesion and lymph node was irradiated with 66 to 69.96 Gy in 30 to 33 fractions. In patients receiving R0 surgery, the tumor bed (GTVtb) was assigned 60 to 66 Gy in 30 to 33 fractions. All high-risk clinical tumor volumes (CTV1s), which included at least 1 cm expansion of GTV or GTVtb, as well as high-risk lymph node regions, received 60.06 Gy in 33 fractions. CTV2s, which consisted of low-risk lymph node levels, received 50.96 Gy in 28 fractions.
All ICI agents were administered with the dose and periodic interval as per respective instructions for use (IFU). The PDL1 expression examination was highly preferred but not mandatory for the inclusion of this study. There was no restriction for the sequence of PD1 inhibitors and RT. The use and selection of chemotherapy regimens depended on the treating physicians’ discretion.
Statistical analysis
Tumor response was evaluated using Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST 1.1) 23 . Duration of response (DOR) was defined as the time from first obtained CR or PR to first documented disease progression or death from disease. Overall survival (OS), locoregional progression-free survival (LRPFS), and distant metastasis-free survival (DMFS) were defined as the time from the diagnosis of recurrence until the first occurrence of specific event: death, locoregional recurrence, or distant metastasis, respectively. Progression-free survival (PFS) was defined as the duration between the onset of recurrence and the date of any progression or cancer-related death. Kaplan–Meier method was performed to estimate survival and the log-rank test was used to examine the significance of difference. Cox hazard regression model was employed for univariate analyses to identify factors associated with survival variables and to calculate hazard ratios (HR). Patients without specific progression were censored at the date of last follow-up or non-cancer-related death. Immunotherapy-related adverse event (irAE) was extracted from the medical record and graded according to the ASCO practice guideline. 24 p < 0.05 was considered statistically significant. Statistical analyses were conducted with SPSS (Version 27.0, IBM Inc.) and R (Version 4.3.3, R Foundation for Statistical Computing).
Results
General characteristics
From January 2019 to December 2023, a total of 23 patients with locoregional recurrent HNSCC met the inclusion criteria and were finally analyzed. The consort diagram of patient identification is shown in Figure 1. The median recurrent age was 55 years, and the majority were male (82.6%). The median time to progression from the beginning of initial course of treatment was 9.3 months. The predominant site of primary lesion was oral cavity (69.6%). Nineteen patients (82.6%) harbored stage rIV disease according to AJCC eighth edition. The 17 unresectable patients included those with clinical rIVB disease (n = 10), rIVA patients receiving surgical resection whilst with residual disease (n = 2), and patients with recurrent lymph node encompassing vessels significantly (n = 5). Among 17 patients with available CPS, 11 with CPS ⩾ 20, 5 with CPS 1–19, and only 1 patient with CPS < 1. Detailed characteristics of the study cohort are presented in Table 1.

The consort diagram of patient identification.
General characteristics.

CPS, combined positive score; ICI, immune checkpoint inhibitors; RT, radiotherapy.
Post-progression treatment
A total of 12 patients received salvage surgery, including 4 out of 6 resectable patients receiving surgical resection as the primary salvage management and achieving R0 resection, 4 unresectable patients suffering from R1 or R2 resection, and 4 patients obtaining excellent pathological response (3 with pathological complete response, PCR) after induction immunotherapy and chemotherapy. Median RT dose was 66.0 Gy with conventional dose fractionation. Detailed locoregional management is summarized in Supplemental Table 2.
With regard to the PD1 inhibitors, pembrolizumab was the most commonly administered agent (43.5%). The combination pattern of immunotherapy and RT consisted of induction immunotherapy in 16 (69.5%), concurrent immunotherapy with RT in 9 patients (39.1%), and consolidation immunotherapy in 11 patients (47.8%). The median cycle of administered PD1 inhibitors was 4. Detailed treatment courses of study patients are demonstrated in Figure 2.

Clinical courses for 23 patients who received radiation plus immunotherapy for recurrent head and neck cancer. The time duration was calculated from the diagnosis of recurrence.
Survival outcomes
By May 2024, the median follow-up time was 16.4 months. Three patients died, and nine patients suffered from tumor progression, including one with distant metastasis and eight with locoregional failure. For overall cohort, the median PFS and LRPFS were 17.0 and 27.2 months, while the median OS and DMFS were not reached. The 12-month OS, PFS, LRPFS, and DMFS were 100%, 79.5%, 79.5%, and 100%, respectively. The 18-month OS, PFS, LRPFS, and DMFS were 85.6%, 47.2%, 55.5%, and 87.5%, respectively, Figure 3(a)–(d). Twenty patients obtained the best response of CR or PR during the treatment course. The 12-month and 18-month DOR were 75.3% and 43.0%, respectively, with the median DOR of 16.7 months (Figure 3(e)).

K-M survival curve. (a) overall survival, (b) progression-free survival, (c) locoregional progression-free survival, (d) distant metastasis-free survival, (e) duration of response, and (f) progression-free survival for patients with CPS ⩾ 20 or CPS < 20 or unknown.
Univariate analysis
Cox hazard regression model was employed for univariate analysis in light of PFS (Table 2). Given the limited number of events, univariate analysis was not conducted with regard to OS, LRPFS, or DMFS. CPS ⩾ 20 was unveiled to be correlated with favorable PFS compared with CPS < 20 or unknown (HR = 0.176, 1-year PFS: 100% vs 50.0%, p = 0.035) (Figure 3(f)). Other factors, such as age, gender, primary site, resectability of recurrent disease, PD1 inhibitors, interval between two courses of treatment, and the administration of salvage surgery, were not found to be associated with PFS.
Univariate analysis for progression-free survival.
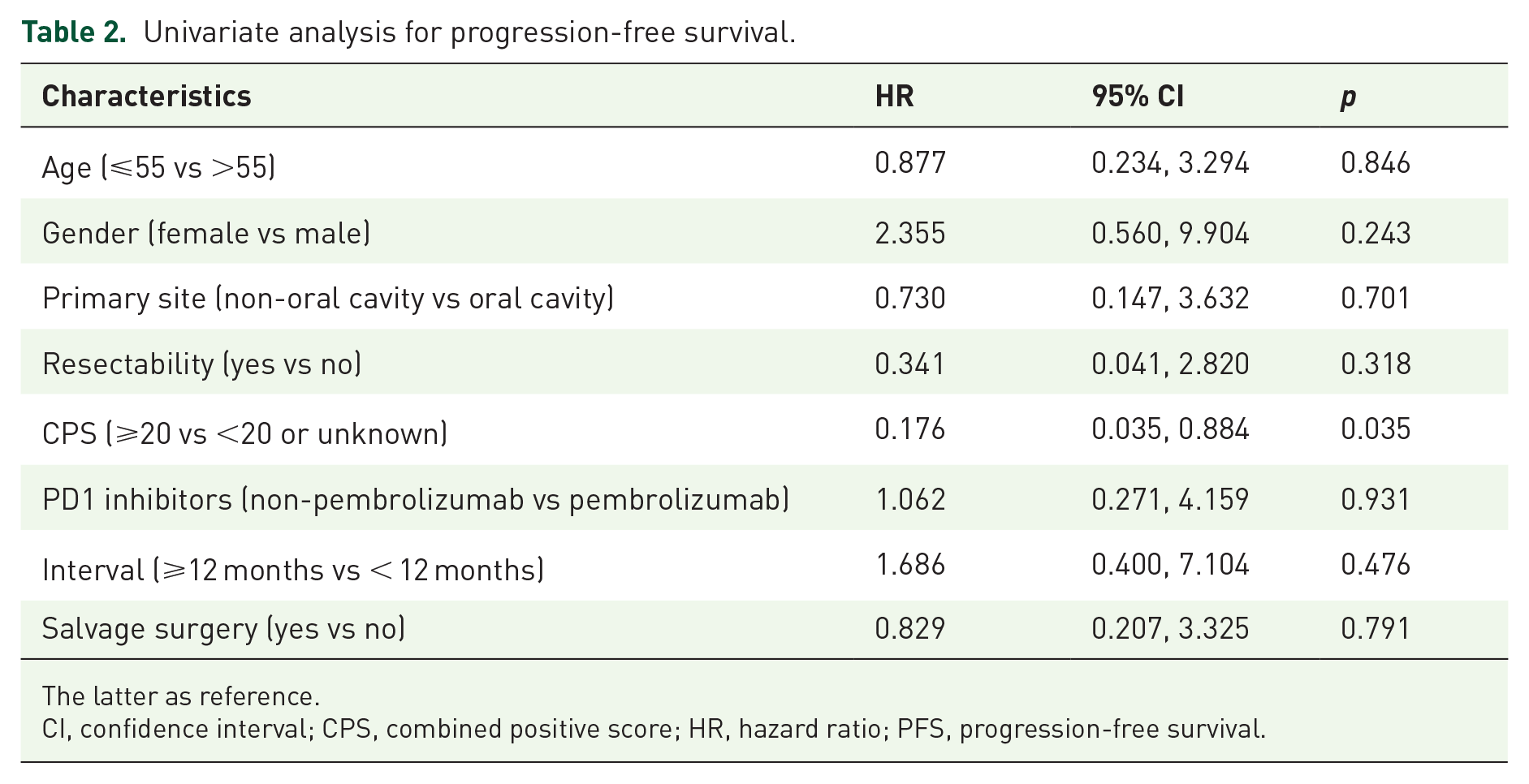
The latter as reference.
CI, confidence interval; CPS, combined positive score; HR, hazard ratio; PFS, progression-free survival.
Treatment-related toxicities
Dermitis (17.4%), mucositis (17.4%), thyroid disfunction (13%), and hepatitis (8.7%) were the most frequent irAE, with ⩾G3 toxicities occurring in four patients (17.4%).
Discussion
In locoregional recurrent HNSCC treated with PD1 inhibitors and locoregional radiotherapy, the present study demonstrated promising overall survival and tumor control with durable response. Moreover, patients with CPS ⩾ 20 may achieve even more favorable survival outcome from this combined modality of treatment, in particular for the prolongation of PFS.
Locoregional recurrent HNSCC is a unique entity, which is biologically different from metastatic disease. Without distant spreading, the locoregional recurrent setting may gain more pronounced benefit from local and regional therapy. So far, the therapeutic decision-making for the recurrent setting is primarily according to the resectability of disease. For patients eligible for salvage surgery, the subsequent OS was much better than those without the chance of surgery.9,25 An early meta-analysis reported the efficacy of salvage surgery in recurrent HNSCC (2-year DFS and median DFS: 44% and 17.9 months) and further revealed that DFS were significantly associated with the recurrent stage (2-year DFS and median DFS: 73% and 22.1 months for stage I vs 22% and 5.5 months for stage IV) and recurrence site (2-year DFS and median DFS: 76% and 24.5 months for larynx; 47% and 9.2 months for oral cavity; 27% and 7.8 months for pharynx; 25% and 7.2 months for the neck). 9 A systematic review summarized the 2-year OS ranged from 43% to 75% for the salvage surgery-treated recurrent HNSCC, compared to 0%–40% for the non-surgical management group. 11 Based on 1088 patients with recurrent HNSCC, Ganan et al. reported the 5-year DFS were 58.3% and 26.4% for patients treated with salvage surgery and radiotherapy. The median DFS for supported care and palliative chemotherapy were only 3.1 and 6.0 months, respectively. According to the survival curves, the 18-month DFS for surgery, radiotherapy, chemotherapy, and supportive care were 74%, 41%, 14%, and 3%, respectively. 16
However, not all recurrent HNSCC are amenable to salvage surgery. Multiple retrospective studies explored the potential candidates for salvage resection. A relatively recent study from Pittsburgh University reported an acceptable 1-year survival rate of 72%. However, a subset of patients died within 12 months or underwent a substantially long duration of hospital stay, indicating that these patients actually did not benefit from salvage surgery. The following factors, such as primary T3 or T4 stage, short disease failure interval, comorbidity, and age should be taken into consideration when selecting patients for salvage surgery treatment. 10 As per these selection factors, more than 75% patients in our cohort were considered unresectable but exhibited the 1-year OS of 100% and 1-year PFS of 79.5%, supporting the therapeutic efficacy of this combined modality of immunotherapy and radiotherapy. To the best of our knowledge, a median of 17.0 months of PFS is the longest in the unresectable recurrent setting of HNSCC.
Immunotherapy-based systemic therapy is an encouraging modality for the treatment of R/M HNSCC. Before the emergence of immunotherapy, EXTREME regimen was the primary option for the R/M HNSCC not amenable to surgery.3,17,26 In recent years, ICI represented by the PD1/PDL1 checkpoint inhibitors has become the mainstay in R/M HNSCC, which is characterized by high PDL1 expression, with nearly 50% of cases showing a CPS of ⩾20 and around 80% harboring a CPS of ⩾1.18,19 By comparing with standard EXTREME regimen, KEYNOTE 048 study revealed that pembrolizumab plus platinum and 5-fluorouracil (PF) could significantly improve OS in R/M HNSCC (13.0 vs 10.7 months).18,20 Furthermore, pembrolizumab monotherapy could markedly extend OS in R/M HNSCC in patients with CPS ⩾ 1 (12.3 vs 10.4 months). Therefore, multiple guidelines consistently recommend pembrolizumab combined with PF chemotherapy as the preferred first-line systemic treatment option.17,27,28 However, no PFS improvement was found in either pembrolizumab or pembrolizumab with chemotherapy groups. Additionally, subgroup analysis showed that the OS benefits from pembrolizumab were more pronounced in metastatic patients whilst no survival improvement was obtained from immunotherapy over EXTREME regimen for the specific setting with locoregional failure. These disconcerting results challenged the role of solitary systemic therapy in recurrent HNSCC and indicated more attempt should be made to improve the outcome for this setting.
There are few studies addressing the survival assessment and treatment optimization in unresectable and radiotherapy-naïve recurrent HNSCC. These patients share certain characteristics in common with the locally advanced setting whilst harboring more aggressive and destructive behavior, justifying more intensive modality of treatment. Theoretically, the combination of local management and advanced systemic therapy is warranted for these patients, such as the concurrent chemoradiotherapy and subsequent consolidation systemic therapy. Nevertheless, there is a lack of high-level evidence-based recommendation in terms of this challenging population. The UK national guideline recommended that patients with non-resectable recurrent HNSCC should be offered the opportunity to participate in phase I–III clinical trials. 29
Recurrent HNSCC without previous irradiation may be considered an intermediate entity between the newly diagnosed locally advanced phase and the distant metastasis. Notwithstanding no evidenced survival improvement from the simultaneous combination of PD1/PDL1 inhibitors in addition to concurrent chemoradiotherapy in the locally advanced HNSCC,30 –32 this therapeutic modality may be effective in the recurrent setting due to more advanced nature of the recurrent disease. In addition, there are emerging data from multiple prospective studies showing proof of concept that neoadjuvant immunotherapy and chemotherapy could elicit promising and unprecedent pathological response in locally-advanced HNSCC, with the reported PCR rate between 17% and 42%.33 –38 Therefore, the modality of neoadjuvant immunotherapy-based systemic therapy followed by radical local therapy may produce elevation of survival for unresectable disease and is justified to be extrapolated to the recurrent setting. In total, it is plausible to expect an improvement of survival from the combination of local therapy and immunotherapy integrated first-line systemic therapy.
Treated by the combination of local radiotherapy and immunotherapy integrated first-line systemic therapy, current study demonstrated promising OS and tumor control with durable response. First, with 73.9% harboring unresectable disease, our cohort achieved a median PFS of 17.0 months and the unreached median OS. These survival data were apparently superior to the previous studies reporting median OS of around 1 year and even shorter PFS. For instance, the PFS of all arms in KEYNOTE 048 was around only 5.0 months.12–15,18,20 Next, it should be noticed that four patients including three with initially non-resectable disease received induction immunochemotherapy and obtained subsequent R0 surgical resection, attaining three PCR and one MPR. Notwithstanding the limited number of patients, this result still indicated the excellent response to immunochemotherapy and shed light on the combination pattern of the multiple treatment modalities for recurrent HNSCC. In addition, 87% (20/23) patients in our cohort achieved objective response and exhibited a 12-month DOR of 75.3%, with the median of 16.7 months. This persistent response duration was also much longer than KEYNOTE 048 study, which reported the respective DOR of 7.0 and 4.3 months in the pembrolizumab plus chemotherapy group and EXTREME group. 20 These results consistently endorsed the addition of RT to the first-line immunotherapy-based systemic therapy for recurrent HNSCC.
To further investigate the beneficial factors related with this combined modality of therapy in recurrent HNSCC, we did univariate analysis, whilst multivariate analysis was not applicable due to the limited patient number. After evaluating multiple potential factors, CPS ⩾ 20 was found to be related with significantly superior PFS over those with CPS < 20 or unknown, with the 1-year PFS of 100% versus 50.0%. Age, gender, primary site, resectability of recurrent disease, PD1 inhibitors, interval between two courses of treatment and the administration of salvage surgery were not found to be associated with PFS. So far, there is no biomarker that could perfectly predict the therapeutic response to PD1 inhibitors in HNSCC and CPS is still the suboptimal consideration for the candidate selection of PD1 inhibitors. Similar trends were also reported in KEYNOTE 048 and KEYNOTE 412 studies in both R/M and locally advanced settings.18,30
There are several limitations in this study. First, as a retrospective study, it is inevitably affected by selection biases, multiple ICI agents as well as the various combination sequence of ICI with RT. To minimize the affect of this heterogeneity, we only selected patients who were treated with anti-PD1 agents while excluded those treated with other ICIs. Second, the patient number is limited and therefore we could not draw solid conclusion for the recurrent HNSCC. Moreover, due to the patient number, it is not feasible to make multivariate analysis to identify the beneficial factors related with the patient selection for the combination of immunotherapy-based systemic therapy and radical local therapy in recurrent HNSCC. The univariate analysis-based results should be interpreted with caution due to the underpowered statistical comparisons. Larger studies, especially prospective cohort-based studies focusing on recurrent HNSCC other than mixed with metastatic diseases, are warranted to guide decision-making, such as sequence of multiple disciplinary approaches, duration of systemic therapy, dose-fractionation of radiotherapy, and so on.
Conclusion
For locoregional recurrent HNSCC, the combined modality of first-line immunotherapy-based systemic therapy and locoregional radiotherapy obtained promising overall survival and tumor control with durable response. These results supported the integration of radical RT as the first-line treatment in addition to immunotherapy-based systemic therapy, in particular for patients with CPS ⩾ 20. Prospective clinical trials are justified to further assess the efficacy of this combined modality of therapy in recurrent HNSCC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251367358 – Supplemental material for Combination of immunotherapy-based systemic therapy and radiotherapy achieves durable response and favorable survival in recurrent head and neck cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359251367358 for Combination of immunotherapy-based systemic therapy and radiotherapy achieves durable response and favorable survival in recurrent head and neck cancer by Jingbo Wang, Gulidanna Shayan, Xin Guo, Xiaodong Huang, Ye Zhang, Runye Wu, Lin Gui, Sheng Yang, Xuesong Chen, Yuan Qu, Kai Wang, Jianghu Zhang, Yuchao Ma, Jingwei Luo, Shengyu Zhou, Xiaohui He and Junlin Yi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251367358 – Supplemental material for Combination of immunotherapy-based systemic therapy and radiotherapy achieves durable response and favorable survival in recurrent head and neck cancer
Supplemental material, sj-docx-2-tam-10.1177_17588359251367358 for Combination of immunotherapy-based systemic therapy and radiotherapy achieves durable response and favorable survival in recurrent head and neck cancer by Jingbo Wang, Gulidanna Shayan, Xin Guo, Xiaodong Huang, Ye Zhang, Runye Wu, Lin Gui, Sheng Yang, Xuesong Chen, Yuan Qu, Kai Wang, Jianghu Zhang, Yuchao Ma, Jingwei Luo, Shengyu Zhou, Xiaohui He and Junlin Yi in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
