Abstract
Background:
Pyrotinib, an irreversible pan-human epidermal growth (HER) inhibitor, has proven its antitumor efficacy as a second-line treatment for HER2-positive metastatic breast cancer (HER2+ MBC) when combined with capecitabine. However, real-world data concerning the pyrotinib, trastuzumab, and chemotherapy (PyroHC) combination remains scarce.
Objectives:
Our study is to report the treatment patterns, efficacy, and safety of the PyroHC combination in a real-world setting.
Design:
This study enrolled patients with HER2+ MBC from five institutions in China, treated with PyroHC between June 2017 and January 2023 (ClinicalTrials.gov, identifier: NCT05839288).
Methods:
We evaluated progression-free survival (PFS), objective response rate (ORR), toxicity profile, and utilized treatment regimens.
Results:
Of the 135 patients in our cohort, 91.9% had prior trastuzumab exposure and 52.2% underwent at least two systematic therapy lines before receiving PyroHC. The most prevalent chemotherapies paired with PyroH were capecitabine (36.3%). Patients receiving PyroHC achieved a median PFS of 8.67 months [95% confidence interval (CI): 6.84–10.51] and an ORR of 51.3% (95% CI: 42.1–61.5%). The first-line treatment with PyroHC led to a median PFS of 14.46 months (95% CI: 6.35–22.56). Patients with brain metastases showed a median PFS of 9.03 months (95% CI: 6.56–11.50), achieving an ORR of 52.17% (95% CI: 51.74–83.39). Longer previous trastuzumab (⩾6.37 months) or lapatinib (⩾10.05 months) therapies could indicate improved PFS, while prior pyrotinib exposure negatively influenced PFS. Notably, the most common grade 3/4 adverse events were diarrhea (37.8%), which were generally manageable.
Conclusion:
PyroHC shows promising efficacy and a satisfactory safety profile for treating HER2+ MBC, both as a first-line option and for heavily treated patients, including those with brain metastasis. Our findings suggest the duration and history of anti-HER2 therapy as potential predictors for PyroHC efficacy in advanced settings.
Keywords
Introduction
Breast cancer is the most prevalent malignancy and leading cause of cancer-related death among women worldwide. 1 Among the various subtypes of breast cancer, the human epidermal growth factor receptor-2-positive (HER2+) type accounts for 15–20% of all cases and is associated with poor prognosis due to its aggressive biological behavior. 2 The cornerstone of therapy for advanced HER2+ breast cancer has been established by the seminal M77001 and H0648g studies, affirming the pivotal role of targeted anti-HER2 treatment.3,4
Several therapeutic strategies have been developed to inhibit the HER2-mediated signal transduction pathway. Monoclonal antibodies, notably trastuzumab, have been widely approved for early and advanced HER2+ breast cancer treatment, by targeting the extracellular domain of HER2. 3 The combination of pertuzumab and trastuzumab with a taxane has been favored as the first-line treatment for HER2+ metastatic breast cancer (MBC), 5 while trastuzumab deruxtecan (T-Dxd) has become increasingly prevalent for those who experience disease progression following trastuzumab-based therapy. 6 However, due to economic constraints, the considerable financial burden of pertuzumab and T-Dxd has limited their use in China’s clinical practice. Alternatively, the utility of small molecule tyrosine kinase inhibitors (TKIs), such as lapatinib, neratinib, and pyrotinib, represent another route of HER2 inhibition. 7 The conjunction of TKIs with monoclonal antibodies, aimed at a dual blockade of HER2, presents an intriguing strategy. Preclinical investigations highlight the potential of lapatinib to augment antibody-dependent cellular cytotoxicity effects driven by trastuzumab. 8 Clinically, the EGF104900 study showcased that trastuzumab combined with lapatinib, in comparison to lapatinib monotherapy, could proffer superior overall survival (OS) benefit in patients with HER2+ MBC who had prior trastuzumab-based therapy. 9
Pyrotinib, an irreversible pan-HER2 TKI, has shown a significant prolongation of progression-free survival (PFS) by 5.7 months (median PFS: 12.5 versus 6.8 months, p < 0.0001) compared to lapatinib in combination with capecitabine in the PHOEBE study (a multicentre, open-label, randomised, controlled, phase 3 trial). 10 Pyrotinib’s role and status in the clinical management of this disease is of growing importance. In China, pyrotinib has become the commonly used anti-HER2 agents. Thus, pyrotinib in combination with trastuzumab remains to be explored for the treatment of HER2+ MBC. In the recent PHILA study, pyrotinib in combination with trastuzumab and docetaxel showed impressive results with the median PFS reaching 24.3 months in the first-line setting of HER2+ MBC. 11 On 21 April 2023, the National Medical Products Administration approved a new indication for pyrotinib in China, making it the third indication for pyrotinib, in combination with trastuzumab and docetaxel for first-line treatment of HER2+ MBC. 12
While clinical trials have evidenced the efficacy of the pyrotinib, trastuzumab, and chemotherapy (PyroHC) combination,13,14 the real-world data for this combination remains scant. Therefore, we conducted this multicenter, retrospective study to evaluate the real-world effectiveness, treatment pattern, and safety of PyroHC in patients with HER2+ MBC. To the best of our knowledge, this investigation represents the first multicenter, retrospective report of real-world data for the use of PyroHC.
Methods
Patients and treatment
This is a retrospective multicenter study, comprising all HER2+ MBC patients who received PyroHC treatment at five medical institutions, including the Fudan University Shanghai Cancer Center, San Huan Cancer Hospital, the Cancer Center of Sun Yat-sen University, West China Hospital of Sichuan University, and Tumor Hospital of the Chinese Academy of Medical Sciences, from June 2017 to January 2023. All investigations were conducted in accordance with the Declaration of Helsinki. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement 15 (Supplemental File 1). Our study has been registered on clinicaltrials.gov (NCT 05839288).
This study included eligible patients: (1) female patients aged more than or equal to 18 years with MBC confirmed by histology or cytology, and a record of HER2 overexpression (i.e. local assessment of immunohistochemistry 3+ and/or positive fluorescence in situ hybridization); (2) patients who received pyrotinib (320–400 mg, po, qd)plus trastuzumab at a dose of 6 mg/kg/week (after an initial loading dose of 8 mg/kg) and chemotherapy regimen (chosen by the physician), from June 2017 to January 2023 at the aforementioned five medical institutions; and (3) patients with complete medical records.
All data were collected in a retrospective manner from the medical records and laboratory findings of each participating institution. This task was coordinated and managed by the Fudan University Shanghai Cancer Center. MBC was defined as either de novo stage IV disease or recurrent breast cancer and confirmation was obtained through clinical assessments, imaging studies, or histological and cytological measures. The decision to initiate therapy was made by the treating physician, guided by the results of previous clinical trials, the general health status, and preferences of patients. Treatment with the designated regimen continued until disease progression occurred, toxicity became intolerable, or the patient voluntarily chose to withdraw from the treatment. Imaging examinations by enhanced magnetic resonance imaging (MRI) [or enhanced computed tomography (CT) for patients with MRI contraindication] for intracranial lesions and CT or enhanced MRI for extracranial lesions were done every 2–3 months. PET (Positron Emission Tomography)/CT and SPECT (Single Photon Emission Computed Tomography)/CT are also performed as needed. Tumor responses needed to be confirmed at next imaging examination and were assessed by the physician according to the Response Evaluation Criteria in Solid Tumors 1.1 (RECIST 1.1) during treatment until disease progression. Through a multicenter design, standardized outcome measures, comprehensive data collection, transparent reporting, and rigorous statistical approaches, our study made concerted efforts to minimize potential sources of bias in evaluating the PyroHC combination’s implications for HER2+ MBC patients.
Outcome measurements
PFS was the primary outcome measure of this study, defined as the time from initiation of PyroHC treatment to disease progression or death. The second outcome measure included OS, objective response rate (ORR), safety, and treatment pattern of PyroHC. OS was defined as the time between the initiation of treatment to death from any cause or censoring on January 2023. ORR was defined as the percentage of evaluable patients at baseline who had either complete response (CR) or partial response (PR) as the best objective tumor response. Tumor response evaluation was performed using the RECIST 1.1 criteria through CT, MRI, and physical examination. Adverse events (AEs) were graded using the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0. Disease-free interval (DFI) was defined as the time from surgery to the diagnosis of metastasis. Visceral metastasis was defined as visceral organ involvement, including lung, liver, peritoneal, or pleural and central nervous system (CNS) recurrence. Trastuzumab resistance was defined as new recurrences diagnosed during or within 12 months after adjuvant trastuzumab or progression at first radiological reassessment or within 3 months after first-line trastuzumab in the metastatic setting. 16 Trastuzumab refractoriness was defined as disease progression after two or more lines of trastuzumab-containing regimens that initially achieved disease response or stabilization at first radiological assessment. 16
Statistical analysis
Survival analyses, including median PFS and OS, were estimated using Kaplan–Meier curves, with corresponding 95% confidence intervals (CIs). Variables potentially predictive of treatment efficacy were evaluated using both univariate and multivariate Cox proportional hazard models. The variables included in the exploratory univariate analysis comprised age, hormone receptor (HR) status, DFI, number of metastatic sites, presence of visceral or brain metastases, advanced systematic therapy lines of PyroHC, trastuzumab resistance status, and prior exposure to treatments including trastuzumab, pyrotinib, lapatinib, TKIs, and pertuzumab. Missing data were addressed by treating them as null values in the analysis. All reported p values and CIs were two-tailed, with a p value <0.05 denoting statistical significance. All statistical analyses were performed using SPSS statistical software (version 24.0, IBM Corporation). The optimal cut-off values were determined using the ‘surv_cutpoint’ function in R (version 4.3.0, R Foundation for Statistical Computing).
Results
Baseline characteristics
Our study encompassed a cohort of 135 participants (Table 1). A total of 52.2% (70/135) of patients had undergone at least three lines of PyroHC advanced systematic therapy, while 24 (17.9%) patients received PyroHC as their first-line advanced therapy. In our study, 22.2% (30/135) of patients encountered a period of less than a year between completing adjuvant trastuzumab treatment and receiving a metastatic diagnosis, while 5.2% (7/135) experienced an interval extending beyond 2 years. A total of 84 (62.2%) patients did not undergo adjuvant trastuzumab treatment.
Patient characteristics at baseline.

Data are median (interquartile range) or n (%).
DFI, disease-free interval; ECOG, Eastern Cooperative Oncology Group; HR, hormone receptor; PyroHC, pyrotinib + trastuzumab + chemotherapy; T-DM1, Trastuzumab Emtansine.
Among 24 patients who underwent the PyroHC treatment as the first line of advanced therapy (Supplemental Table 1), 30.4% (7/24) had de novo metastasis. In terms of the interval between the completion of adjuvant trastuzumab treatment and the diagnosis of metastasis, 33.3% (8/24) of patients experienced an interval of less than 1 year, with 45.8% (11/24) of patients not undergoing adjuvant trastuzumab treatment.
In the analysis of metastatic sites, the liver (47.4%, 64/135) and lung (45.2%, 61/135) emerged as the most common, closely followed by the bone (40.0%, 54/135) and brain (34.8%, 47/135; Table 1). A total of 52.2% (70/135) patients reported metastases in three or more distinct locations, with 81.5% (110/135) revealing visceral metastases. Within 47 patients who exhibited brain metastases (with concurrent metastases in other locations or brain metastasis as their sole metastatic site; Supplemental Table 2), 40.7% (19/47) of patients showed symptoms of brain metastases before the initiation of PyroHC treatment. Furthermore, 85.2% (40/47) of these patients had previously received radiotherapy and none of them had surgery as CNS local therapy. The median duration from the completion of radiotherapy to enrollment was observed to be 6.24 months.
Regarding trastuzumab resistance status, 65.9% (89/135) of patients were characterized as refractory, 23.7% (32/135) were resistant, and 10.4% (14/135) were sensitive (Table 1). Lastly, in terms of prior HER2-targeted therapy, the majority of patients (91.9%, 124/135) had been treated with trastuzumab, followed by lapatinib (34.1%, 46/135) and pertuzumab (32.5%, 44/135).
Treatment patterns
The most common chemotherapy combination was pyrotinib, trastuzumab, and capecitabine (PyroHX), given to 49 patients, among which 14.2% (7/49) received it as the first-line treatment, 39.6% (19/49) as the second, and 46.9% (23/49) during the third or later treatment lines (Table 2).
Treatment administration.
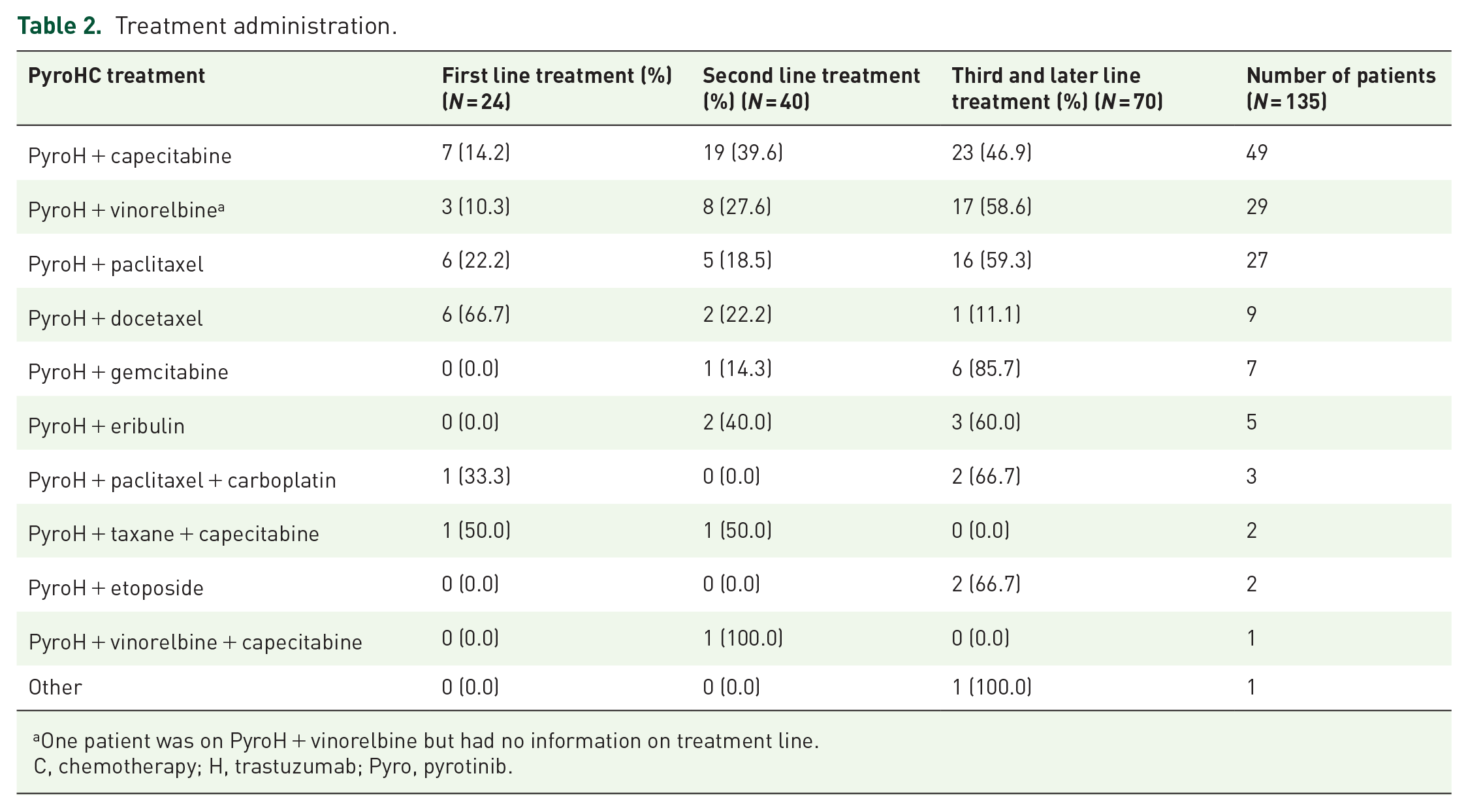
One patient was on PyroH + vinorelbine but had no information on treatment line.
C, chemotherapy; H, trastuzumab; Pyro, pyrotinib.
Following PyroHX, the PyroH + vinorelbine combination was used by 29 patients, distributed as 10.3% (3/29) in the first line, 27.6% (8/29) in the second, and 58.6% (17/29) in subsequent treatments.
PyroH along with taxane, encompassing paclitaxel and docetaxel, was applied to treat 36 patients, with 33.3% (12/36) of them as the first line, 19.4% (7/36) as the second, and 47.2% (17/36) as the third or later treatment lines.
Efficacy
After a median follow-up of 27 months, 95 patients experienced progressive disease, resulting in a median PFS of 8.67 months [95% CI: 6.84–10.51, Figure 1(a)]. The median OS was not reached at the time of analysis. A total of 117 patients were included in the analysis of ORR, with 18 patients excluded due to lack of measurable lesions or missed visits (Table 3). One patient (0.8%) achieved CR, while 59 patients (50.4%) had PR, resulting in an ORR of 51.3% (95% CI: 42.1–61.5%).

Kaplan–Meier curves representing PFS: (a) for the entire patient cohort, (b) for patients grouped according to treatment lines (first line versus second line versus third line or beyond), and (c) for patients categorized into two groups based on treatment lines (first or second line versus third line or beyond).
ORR in all patients.
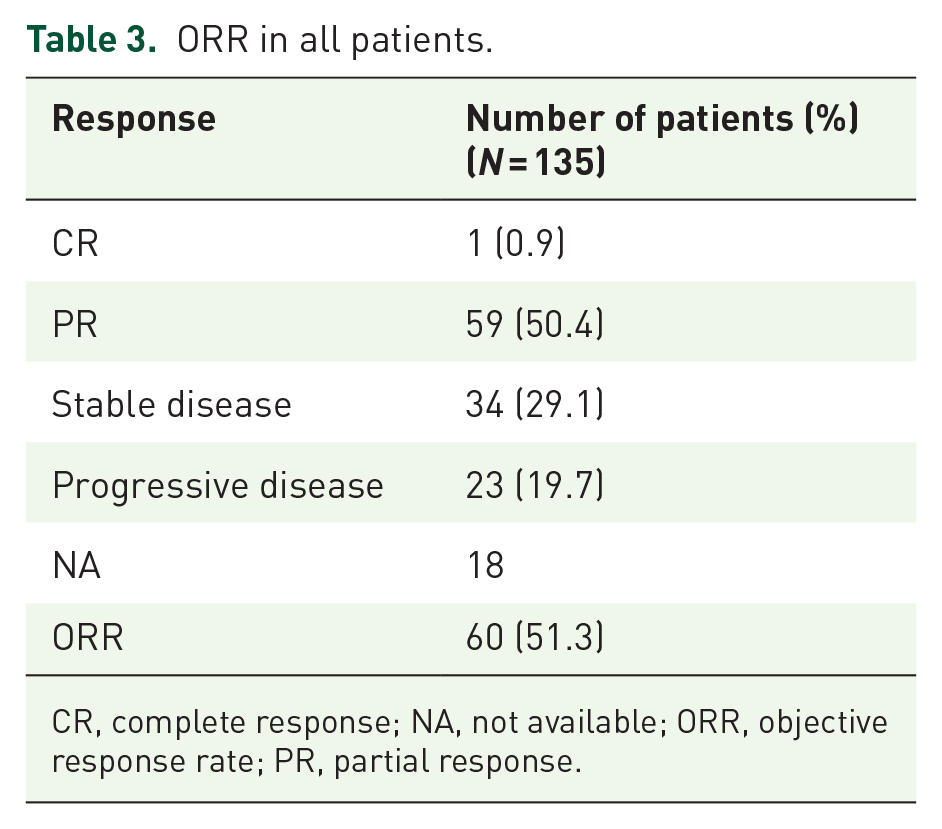
CR, complete response; NA, not available; ORR, objective response rate; PR, partial response.
Median PFS among patients who underwent the PyroHX regimen (a combination of pyrotinib, trastuzumab, and capecitabine), following progression on trastuzumab therapy was 11.17 months (Figure 2). With the similar distribution of treatment lines for PyroH + capecitabine in comparison with both PyroH + vinorelbine and PyroH + taxane (Supplemental Table 3), the efficacy of PyroHX showed similar outcomes to both PyroH with vinorelbine [median PFS: 11.17 versus 7.75 months, p = 0.303, Figure 3(a)] and PyroH with taxane [median PFS: 11.17 versus 10.02 months, p = 0.319, Figure 3(b)].
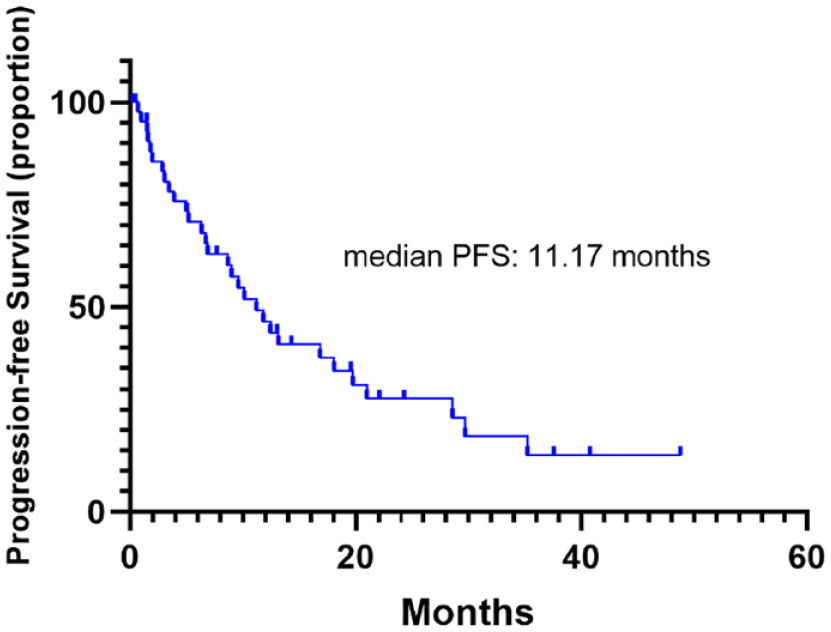
Kaplan–Meier curve illustrating PFS in patients who received PyroHX, a regimen of pyrotinib, trastuzumab, and capecitabine, following progression on trastuzumab therapy.
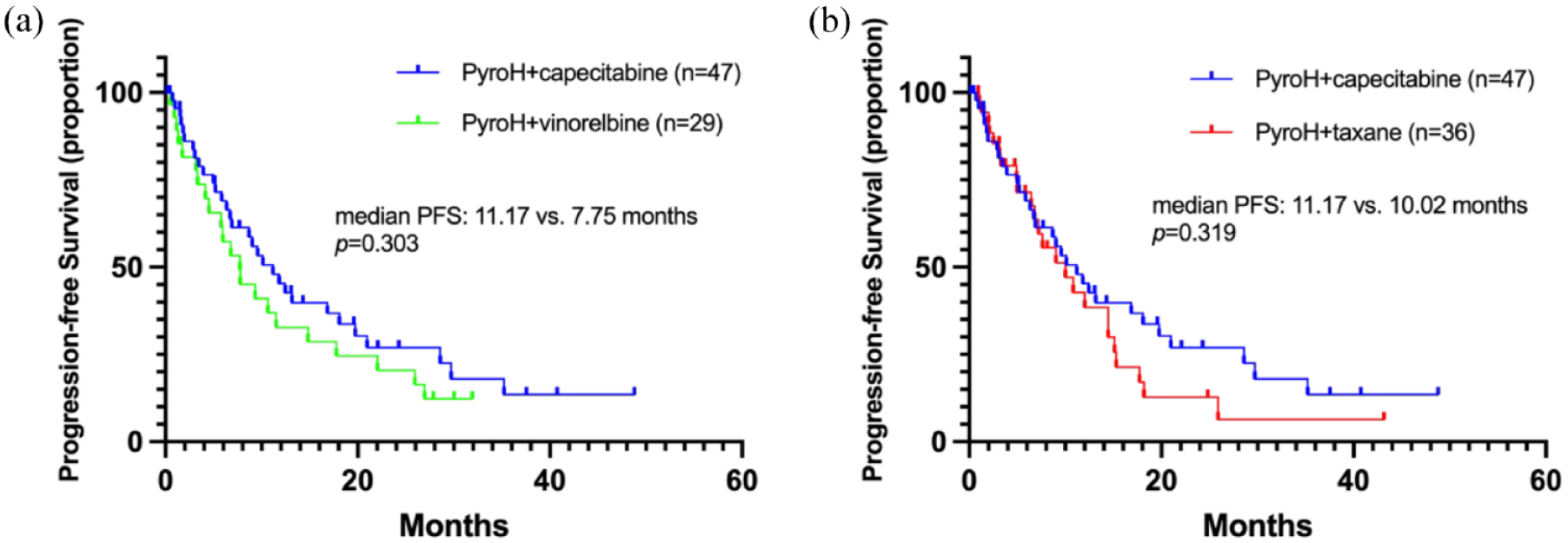
Kaplan–Meier curves depicting PFS for (a) patients treated with PyroH in combination with capecitabine as compared to those treated with vinorelbine; (b) patients treated with PyroH and capecitabine as compared to those treated with taxane.
Among patients with brain metastases, the median PFS was observed to be 9.03 months (95% CI: 6.56–11.50), similar with the PFS of 8.21 months (95% CI: 6.29–10.13) in patients who did not have brain metastases (p = 0.881, Figure 4). Patients with brain metastases had a systemic ORR of 52.17% (95% CI: 51.74–83.39; Supplemental Table 5).

Kaplan–Meier curve illustrating PFS for patients, comparing those with and without brain metastases.
When considering the treatment lines, the median PFS was 14.46 months (95% CI: 6.35–22.56) for the first line, 9.59 months (95% CI: 7.43–11.76) for the second line, and 6.80 months (95% CI: 4.390–9.212) for the third line or later [log-rank Mantel–Cox test: p = 0.171, Gehan–Breslow–Wilcoxon test: p = 0.042, Figure 1(b)]. The median PFS for patients receiving first or second line treatment was 11.17 months (95% CI: 8.33–14.01), showing a difference when compared to those receiving third line or later treatments [log-rank Mantel–Cox test: p = 0.051, Gehan–Breslow–Wilcoxon test: p = 0.008, Figure 1(c)]. The ORR for the first, second, and third (or subsequent) treatment lines were 75.00%, 52.63%, and 43.10%, respectively (Supplemental Table 4). A negative correlation was identified between the ORR and the number of PyroHC treatment lines, as suggested by a Spearman’s correlation coefficient of −0.211 (p = 0.023).
We identified optimal cut-off values of 6.37 months for last trastuzumab-based therapy, 10.05 months for prior lapatinib-based therapy, and 7.56 months for previous TKI-based therapy. Interestingly, prior trastuzumab exposure did not significantly affect median PFS [12.57 versus 8.05 months, p = 0.541, Figure 5(a)], yet a longer duration (⩾6.37 months) showed a trend toward improved PFS [12.02 versus 5.32 months, p = 0.023, Figure 5(b)]. In contrast, prior exposure to pyrotinib led to a lower PFS [11.66 versus 5.32 months, p < 0.001, Figure 5(c)], although the duration of previous pyrotinib-based therapy did not significantly impact PFS [7.90 versus 3.94 months, p = 0.394, Figure 5(d)]. As for lapatinib, prior exposure did not affect PFS [8.67 versus 7.2 months, p = 0.813, Figure 5(e)], but a longer exposure (⩾10.05 months) did improve PFS significantly [17.71 versus 6.8 months, p = 0.023, Figure 5(f)]. Finally, absence of previous TKIs exposure trended toward a higher PFS [11.17 versus 6.7 months, p = 0.063, Figure 5(g)], and a longer duration of previous TKI-based therapy (⩾7.56 months) significantly increased PFS of PyroHC [10.48 versus 4.90 months, p = 0.029, Figure 5(h)].
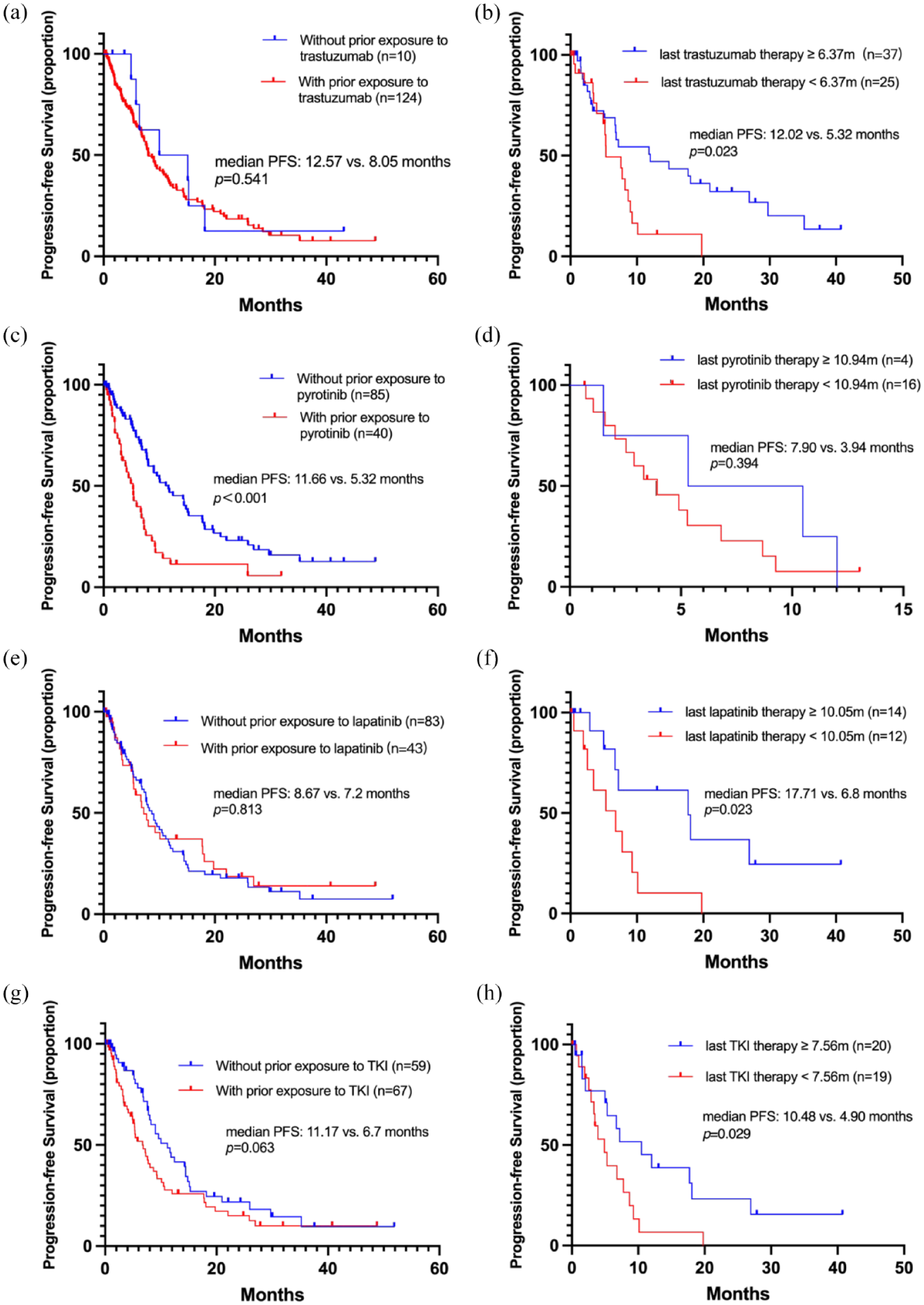
Kaplan–Meier curves depicting PFS as follows: (a) for patients with and without prior exposure to trastuzumab; (b) for patients stratified by the duration of last trastuzumab therapy (⩾6.37 versus <6.37 months)*; (c) for patients with and without prior exposure to pyrotinib; (d) for patients stratified by the duration of their last pyrotinib therapy (⩾10.94 versus <10.94 months)*; (e) for patients with and without prior exposure to lapatinib; (f) for patients stratified by the duration of their last lapatinib therapy (⩾10.05 versus <10.05 months)*; (g) for patients with and without prior exposure to TKI; and (h) for patients stratified by the duration of their last trastuzumab therapy (⩾7.56 versus <7.56 months)*.
In the univariate analysis, the number of metastatic sites (⩽2 versus >2) showed a significant association with PFS with an HR of 0.659 (95% CI: 0.438–0.991, p = 0.045) (Table 4). The lines of advanced systematic therapy of PyroHC (1–2 versus ⩾3) approached significance with an HR of 0.673 (95% CI: 0.447–1.015, p = 0.059). Strong associations with PFS were demonstrated by prior exposure to pyrotinib (no versus yes) and pertuzumab (no versus yes), with HRs of 0.447 (95% CI: 0.285–0.700, p < 0.001) and 0.398 (95% CI: 0.252–0.630, p < 0.001), respectively. In the multivariate analysis, the lines of advanced systematic therapy of PyroHC (1–2 versus ⩾3) was identified as significantly associated with PFS with an HR of 0.359 (95% CI: 0.139–0.923, p = 0.033; Table 4).
Univariate and multivariate Cox regression analysis of factors associated with PFS.
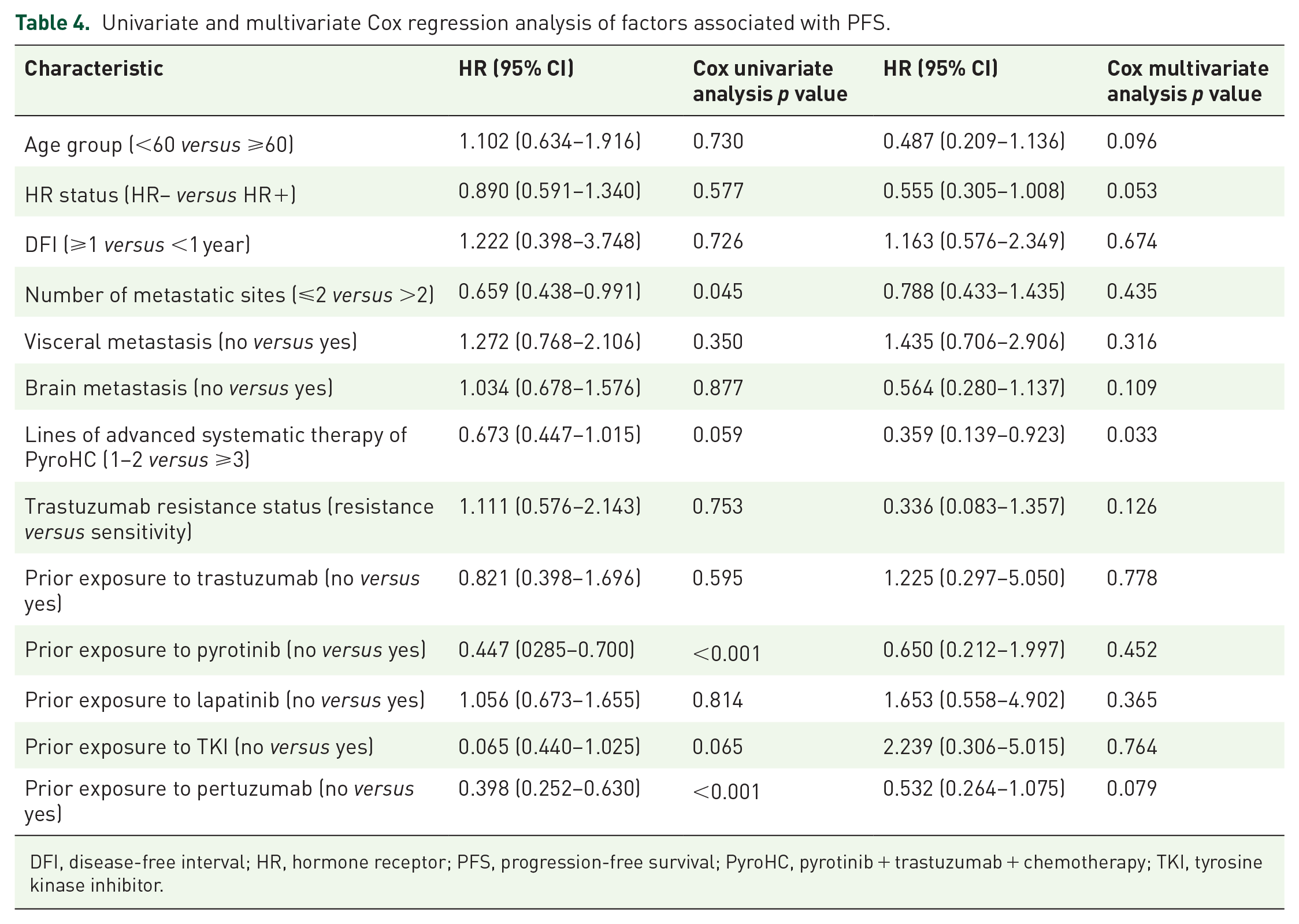
DFI, disease-free interval; HR, hormone receptor; PFS, progression-free survival; PyroHC, pyrotinib + trastuzumab + chemotherapy; TKI, tyrosine kinase inhibitor.
Safety
Among the 137 patients, diarrhea was the most prevalent grade 3/4 AEs, affecting 37.8% of patients (Table 5). Palmar–plantar erythrodysesthesia syndrome and vomiting were the next most common grade 3/4 AEs, both occurring in 6.7% of patients. The AEs reported in this study were manageable, and no treatment-related fatalities were observed during PyroHC.
AEs (grade 3/4).

AE, adverse event; ALT, alanine transaminase; AST, aspartate transaminase.
Discussion
Our research offers a comprehensive real-world assessment of the PyroHC regimen’s efficacy and safety in treating HER2+ MBC. Our findings of a median PFS of 8.67 months and an ORR of 51.3% align with those of a previous phase II trial 13 but provide a more nuanced understanding given our larger patient population and the broader range of chemotherapy combinations. The phase II trial reported a median PFS of 7.5 months and an ORR of 50.0% but was limited by its small sample size and the limited prior exposure to pertuzumab among its participants. 13 In contrast, our study’s multicenter design and larger patient cohort, many of whom had received prior pertuzumab treatment, better reflect real-world clinical scenarios. The variety of chemotherapeutic agents used in combination with PyroHC in our study further enhances the comprehensiveness and applicability of our findings. Additionally, our diverse patient characteristics and treatment histories offer a rich dataset for exploring various factors’ impact on treatment outcomes.
Our study revealed the application of the PyroHC regimen as first-line advanced therapy for HER2+ MBC in 24 patients, resulting in favorable outcomes, evidenced by a median PFS of 14.46 months. Importantly, a significant proportion of these patients (66.7%, 16/24) had undergone prior trastuzumab therapy, with the majority (50.0%, 12/24) having an interval between adjuvant trastuzumab and diagnosis of metastasis less than 2 years. Contrarily, the PHILA study presented at the 2022 ESMO (European Society for Medical Oncology) Congress, while exhibiting a higher median PFS of 24.3 months with first-line PyroH and docetaxel, incorporated a smaller proportion of patients with prior trastuzumab (15.49%) and a majority with a treatment-free interval exceeding 2 years following prior therapy. 14 Furthermore, the M77001 study, assessing trastuzumab plus docetaxel, indicated a slightly shorter median PFS (11.7 months) compared to our study. 4 Given that 66.7% of our first-line patients had prior trastuzumab exposure, the inclusion of pyrotinib in our regimen may confer enhanced therapeutic benefits. The CLEOPATRA trial (a double-blind, randomised, placebo-controlled, phase 3 study), however, reported a median PFS of 18.7 months, slightly longer than in our study, but included a lower proportion (12%) of patients with prior trastuzumab adjuvant treatment. 5 Considering that treatment guidelines prior to September 2022 did not recommend PyroHC as a first-line treatment option for advanced stages both internationally or locally in China, the number of patients receiving PyroHC as a first-line treatment in our study remains limited. Although pyrotinib has become a standard second-line treatment option for HER2+ MBC in China, exploration of its application in the first-line treatment is still ongoing. This necessitates further research to validate these findings and optimize the treatment strategy for this patient population.
Our study not only showcases the efficacy of the PyroHC approach but also highlights its versatility in accommodating a variety of chemotherapy regimens. The most frequently administered regimen was PyroHX, which consists of pyrotinib, trastuzumab, and capecitabine, utilized in 36.3% of the patients. This regimen, with a median PFS of 11.17 months, remained effective even in patients showing progression on trastuzumab therapy, aligning with findings from previous studies.10,17 In parallel, other combinations, specifically PyroH with vinorelbine and PyroH with taxane, were administered to 21.5% and 26.7% of our cohort, respectively. These combinations demonstrated a median PFS almost parallel to the PyroHX regimen. Patients treated with PyroH and vinorelbine in our study exhibited a median PFS of 7.75 months, a finding that closely mirrors a median PFS of 7.8 months reported in a multicenter retrospective study. 18 This demonstrates the comparable efficacy of various PyroHC regimens, underscoring the importance of personalizing therapeutic strategies based on factors like patient health status and treatment response.
Brain metastases were identified as a common complication in HER2+ MBC, with an incidence of one-third of all cases. 19 This rate is consistent with our study, where 34.8% (47/135) of patients were diagnosed with brain metastases. This is a significant clinical challenge given that effective drug delivery to brain metastases is often impeded by the blood–brain barrier. Despite this hurdle, we found that patients with brain metastases under the PyroHC regimen exhibited a median PFS of 9.03 months similar with their counterparts without brain metastases. This trend mirrors the beneficial outcomes reported in previous studies, where patients with brain metastases undergoing pyrotinib-based therapy showed a median PFS ranging from 8.00 to 9.4 months.13,20,21 This brings to light the documented efficacy of TKIs and various chemotherapeutic drugs that have the ability to penetrate the blood–brain barrier in managing HER2+ MBC patients with brain metastases. 22 The context of prior radiotherapy was considered in our study. A substantial majority (85.2%) of our patients with brain metastases had undergone previous radiotherapy and yet still achieved an ORR of 52.63%. It’s noteworthy to mention the PERMEATE trial, 23 which documented an intracranial ORR of 42.1% in patients with disease progression post-radiotherapy. Taken together, our findings underscore the promising potential of the PyroHC regimen in managing HER2+ MBC patients with brain metastases.
Furthermore, our study delved deeply into the implications of previous therapy exposure and its duration on subsequent treatment outcomes. Interestingly, we found that patients receiving PyroHC exhibited comparable PFS irrespective of whether they had prior exposure to trastuzumab, thereby highlighting the potential benefits of cross-line trastuzumab use in PyroHC regimens. This observation aligns with the findings of prior study, 24 specifically exploring the implications of continuous trastuzumab treatment for HER2+ MBC patients who showed disease progression during the previous trastuzumab regimen. In addition to this, our findings indicate that an extended PFS resulting from previous trastuzumab-based therapy (⩾6.37 months), lapatinib-based therapy (⩾10.05 months), or TKI-based therapy (⩾7.56 months) could significantly enhance PFS upon subsequent PyroHC regimen administration. Such insights might inform the development of therapeutic strategies, suggesting that better sensitivity to previous lapatinib or TKI therapies could lead to a better response to subsequent PyroHC therapy. This result resonates with the findings of a previous real-world study in lapatinib-resistant HER2+ MBC patients, which showed an extended PFS for patients who had a prior benefit from lapatinib for 6 months or longer. 25 Another retrospective analysis of post-lapatinib treatment demonstrated that continuing lapatinib yielded more favorable median PFS results compared to non-anti-HER2 treatments, providing evidence for the cross-line use of lapatinib. 26 Contrarily, our study identified that prior exposure to pyrotinib significantly reduced PFS, and the duration of previous pyrotinib-based therapy did not significantly influence subsequent PFS. These insights indicate that any prior use of pyrotinib could potentially hinder the effectiveness of subsequent PyroHC therapy, regardless of the patient’s initial sensitivity to pyrotinib, thus hinting at the potential limitations of PyroHC as an effective therapeutic strategy for patients who have previously used pyrotinib. Collectively, our study emphasizes the role of a patient’s history of previous anti-HER2 therapy in influencing the efficacy of PyroHC regimens, thus underlining the importance of considering the history of prior therapy and its duration when planning treatment strategies for HER2+ MBC.
Our study’s safety analysis of the PyroHC regimen revealed diarrhea as the most prevalent grade 3/4 AE, affecting 37.8% of patients. This observation aligns with the known adverse effect profile of pyrotinib. 10 Crucially, despite the occurrence of such events, all were manageable and our study observed no treatment-related fatalities, suggesting an acceptable tolerance to the PyroHC regimen among patients.
Notwithstanding these findings, our study does bear several limitations that must be acknowledged. Its retrospective design inherently presents potential biases, including selection and information biases. Additionally, our study lacks a comparison group, which limits our ability to definitively assert the efficacy and safety of the PyroHC regimen. Further confirmation of our findings would thus require prospective randomized trials. Moreover, patient heterogeneity and variations in treatment regimens might have also influenced the study outcomes.
Conclusions
In conclusion, our study provides real-world evidence for the promising efficacy, treatment pattern, and appreciable safety of PyroHC in treating HER2+ MBC, applicable to both first-line treatment, heavily treated patients, and those with brain metastasis. We also suggest that the duration and prior exposure to anti-HER2 therapy could serve as valuable predictors for the efficacy of PyroHC in an advanced treatment setting.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231217972 – Supplemental material for Real-world data of triplet combination of pyrotinib, trastuzumab, and chemotherapy in HER2-positive metastatic breast cancer: a multicenter, retrospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359231217972 for Real-world data of triplet combination of pyrotinib, trastuzumab, and chemotherapy in HER2-positive metastatic breast cancer: a multicenter, retrospective study by Shuhui You, Die Sang, Fei Xu, Ting Luo, Peng Yuan, Yizhao Xie and Biyun Wang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors express their gratitude toward all participating patients, nurses, clinicians, and CSCO YOUNG BC for their invaluable support and contribution to this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
