Abstract
Epithelial ovarian cancer (EOC) is the most important cause of gynecological cancer-related mortality. Despite improvements in medical therapies, particularly with the incorporation of drugs targeting homologous recombination deficiency, EOC survival rates remain low. Adoptive cell therapy (ACT) is a personalized form of immunotherapy in which autologous lymphocytes are expanded, manipulated
Keywords
Introduction
Epithelial ovarian cancer (EOC) is the most lethal gynecologic malignancy and the fifth deadliest cancer in women. 1 In recent years, high-grade serous ovarian carcinoma (HGSOC), the most common histological subtype, has experienced many practice-changing treatment advances. These advances were mainly related to the incorporation of poly-[adenosine diphosphate (ADP)-ribose] polymerase (PARP) inhibitors into the treatment paradigm for both BRCA-mutant and for BRCA wild-type EOC tumors. 2 Despite significant improvements in surgical and medical treatment, most EOC patients experience disease recurrence within 2–3 years, and successive progressions with shorter treatment-free periods, and eventually, death. 3 Hence, there is an unmet need for novel and effective therapeutic options for patients with EOC.
Drug-based immunotherapy, such as immune-checkpoint blockade (ICB), has brought practice-changing advances in many hematological and solid tumors. Although EOC is considered a potentially immunoreactive tumor type, with the presence of intratumoral tumor-infiltrating lymphocytes (TILs) correlating with good clinical outcomes,4,5 the use of immunotherapy for EOC patients has failed to deliver clinically meaningful results so far. 6 Indeed, despite durable responses in a subset of patients, many patients do not respond to the immunotherapies in current use.7,8
Advanced therapeutic medicinal products (ATMPs), such as adoptive cell therapy (ACT), represent an emerging potential treatment option for solid tumors, which could be of interest for EOC patients. This review aims to give a comprehensive overview of the previous developments and the current status of ACT in EOC.
Adoptive cell therapies: an overview
ACT treatments are a type of personalized ATMP, manufactured specifically for each patient using their own cellular material (e.g. immune effector cells). This process utilizes autologous intra-tumoral or peripheral blood immune effector cells, which are extracted, expanded
ACT can broadly be classified into three different types: (a) expanded natural TILs; (b) T-cell receptor (TCR)-engineered T cells; and (c) chimeric antigen receptor (CAR)-modified T cells. 10 All three of these cellular therapy types are currently being explored for EOC treatment, along with other strategies such as natural killer (NK) cells.
In one approach to ACT, which has been pioneered in patients with metastatic melanoma, TILs are cultured from resected cancer deposits, then expanded to large numbers
High-dose chemotherapy and autologous stem-cell transplantation (HDCT-ASCT)
The preconditioning LTD, apheresis, and re-infusion scheme used in ACT protocols are partially inspired by treatment protocols developed in 1980 in hematological stem-cell transplantation for the treatment of solid tumors. In this period, many solid tumors, including breast and EOC, were treated by high-dose chemotherapy (HDCT).15,16 This strategy was based on the observation that the cytotoxic effect of chemotherapy would increase proportionally with increased doses and eventually overcome tumor chemoresistance. 17 The dose intensity of these protocols was five to ten times the usual standard and was leading to protracted hematological toxicity and bone marrow aplasia. Thus, HDCT required autologous stem-cell transplantation (ASCT) as a hematologic rescue.
Several phase I and II trials of HDCT using bone-marrow ASCT
18
and one phase III trial have been carried out in EOC in the past.
19
A survey of 421 patients who had undergone HDCT with ASCT in the US, identified 20 different chemotherapy regimens, usually a combination of agents such as melphalan, carboplatin, cyclophosphamide, and thiotepa. The 2-year progression-free survival (PFS) for this treatment was 12%, and the 2-year overall survival (OS) was 35%.
20
The only randomized trial comparing sequential high
Simultaneously, a less toxic regimen of ‘mini ablation’ developed for allogeneic bone-marrow transplantation showed immune-mediated graft-
Adoptive cell therapy with peripheral blood lymphocytes
The PBLs were amongst the first forms of ACT used for EOC treatment. In this technique, PBLs were isolated and expanded from peripheral blood mononuclear cell (PBMC) aphaeresis. The precursor CD8+ lymphocytes were then isolated and stimulated with specific tumor antigens to obtain a tumor-reactive ACT product. The ACT re-infusion was administered intravenously (i.v.) or intraperitoneally (i.p.).
Wright
The difficulties in obtaining high-affinity CTL in sufficient numbers prompted the investigation of
Adoptive cell therapy using tumor-infiltrating lymphocytes (TILs)
ACT using TILs is based on the infusion of autologous CD4+ and CD8+ T lymphocytes expanded from tumors in the presence of high-dose interleukin-2 (IL-2) alone or in combination with other interleukins (Figure 1). TIL-ACT is based on the concept that the cellular immune response to cancer largely depends on conventional T cells, CD8+ and CD4+, that specifically target tumor-specific antigens such as cancer/testis antigens or cancer neoantigens (NeoAgs) that are derived from somatic genomic alterations that lead to the expression of immunogenic neo-epitopes.28–31
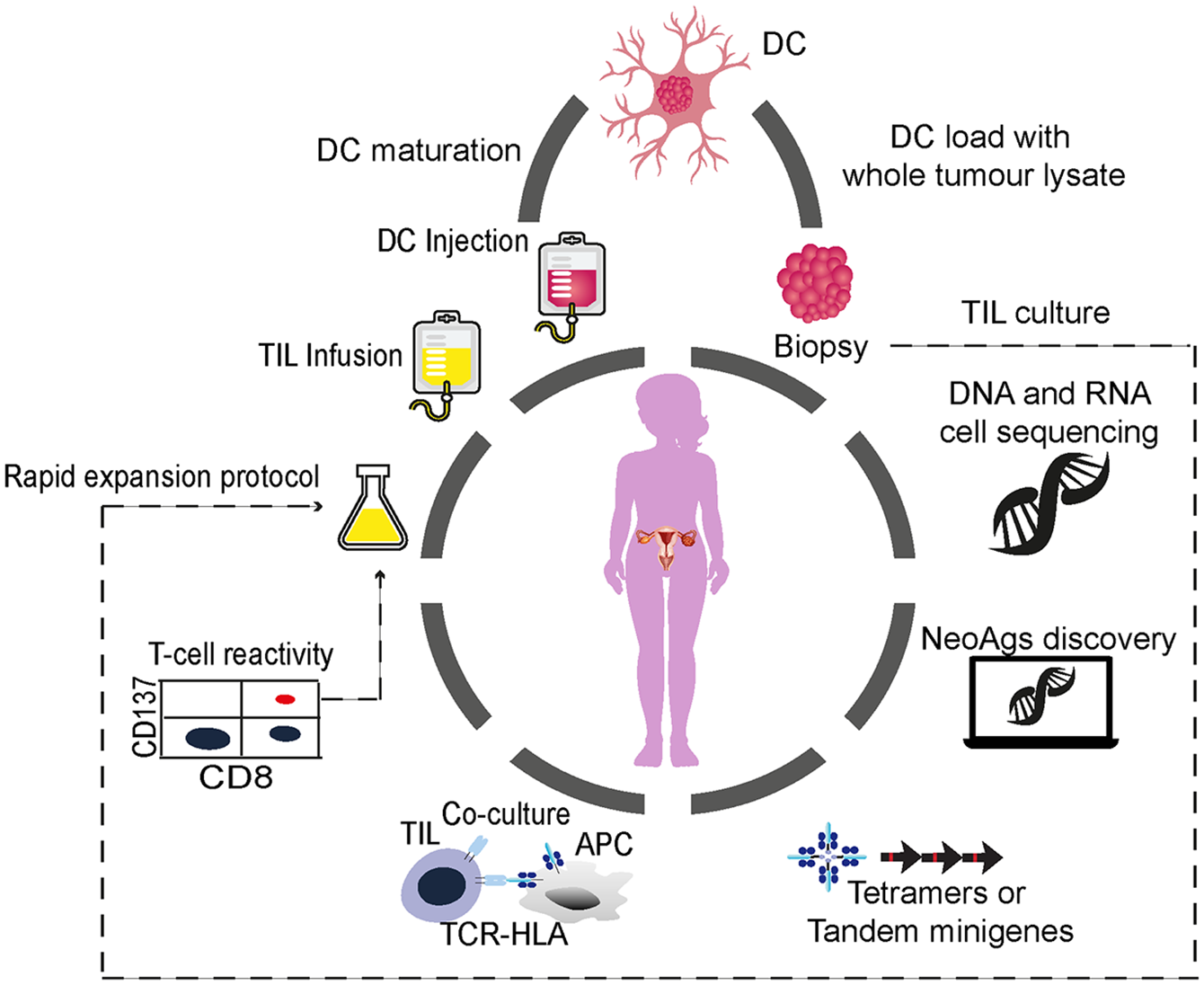
General schematic for using DC vaccines and TIL-ACT for patients with OC.
TIL-ACT has shown impressive results in metastatic melanoma and has reproducibly demonstrated having a curative potential with overall response rates of approximately 50%, including durable CR in up to 25% of treated patients. 32
TIL-ACT has also been tested in EOC. However, the trials reporting data so far have shown a limited success of this strategy (Table 1).
Clinical studies with TIL-ACT in EOC.
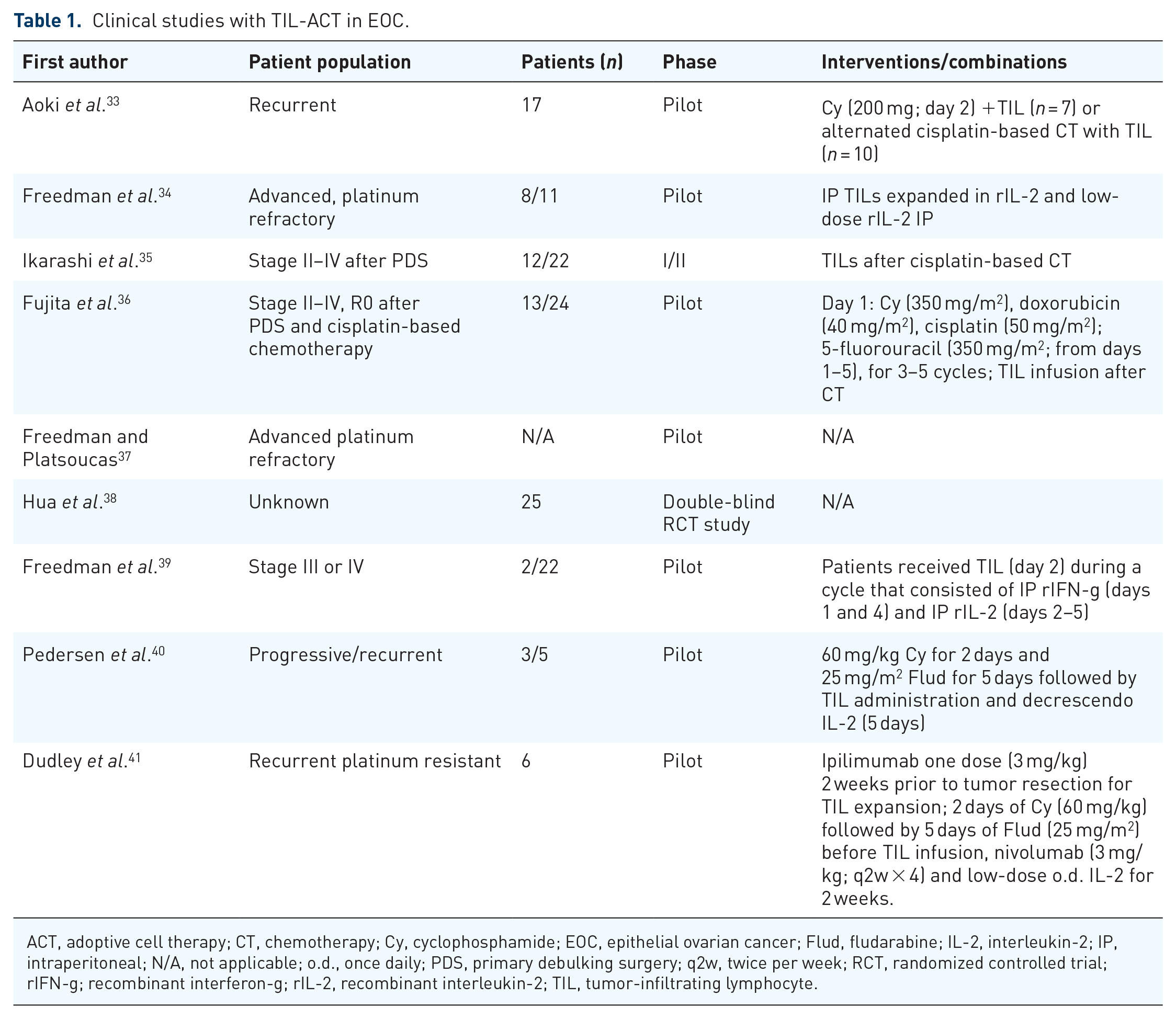
ACT, adoptive cell therapy; CT, chemotherapy; Cy, cyclophosphamide; EOC, epithelial ovarian cancer; Flud, fludarabine; IL-2, interleukin-2; IP, intraperitoneal; N/A, not applicable; o.d., once daily; PDS, primary debulking surgery; q2w, twice per week; RCT, randomized controlled trial; rIFN-g; recombinant interferon-g; rIL-2, recombinant interleukin-2; TIL, tumor-infiltrating lymphocyte.
The first results of TIL-ACT in EOC were reported back in 1991 by Aoki
Freedman
In 1994, Ikarashi
Fujita
More recently, Pedersen
Interestingly, the TIL-ACT trials in EOC dating from the 1990s have reported better outcomes than more recent trials (Table 1). Despite the difficulty in reproducing older datasets due to different LTD preparatory regimens,
Neoantigen-based TIL-ACT in EOC
As highlighted in Table 1, none of the TIL-ACT trials in EOC to date have tested the TIL product for reactivity against tumor NeoAgs prior to infusion. NeoAg-specific T cells have been identified in several solid cancers, including EOC,47,48 and they have been identified as a major factor in the activity of T-cell responses both in patients treated with ICB49,50 and those treated with TIL-ACT.30,51
The NeoAgs discovery pipeline is an extensive process and is briefly summarized in Figure 1.52,53
In the context of EOC, the relatively low somatic point mutation load, high aneuploidy levels, and high levels of copy number alterations (CNAs) have been associated with low immunogenicity.54,55 However, as previously mentioned, a high density of intraepithelial CD8+ TILs is associated with an increased overall survival, as well as the presence of an immunoreactive gene expression signature.4,55 Moreover, these epithelial CD8+ TILs are negatively associated with tumor clonal diversity, reflecting immunological pruning of tumor clones by T cells with associated NeoAg depletion and loss of human leukocyte antigen (HLA)-I heterozygosity. 56 This implies the existence of a close relationship between genotype, TME, and T-cell immune response in EOC, 57 suggesting an important role of cytolytic T cells directed against cancer EOC NeoAgs during tumor evolution.
Intratumoral neoantigenome-directed T-cell responses have not been comprehensively evaluated in EOC until recently. In a pilot study on three patients with EOC, only one mutation (1/79, 0.01%) was detected to elicit a T-cell response.
47
More recently, three other studies assessed the NeoAg landscape of EOC tumors. Deniger
The same group subsequently reported the presence of intratumoral and PBL T-cell responses to defined TP53 hotspot mutations in a large cohort of patients with multiple tumor types.59,60 The idea of having TILs recognizing a key driver event in EOC, as well as in other tumors, is indeed fascinating. However, it is evident through the lack of cancer-cell elimination and tumor immune escape, that mechanisms capable of preventing T-cell-mediated death against driver mutations might be an extremely early event in cancer evolution. In fact, the presence of widespread driver mutations in healthy tissue, such as pathogenic TP53 mutations in normal endometrium epithelium, 61 may promote immune tolerance early in life against these potential neo-epitopes and thus hampering the possibility as immunogenic targets.
Bobisse
All three studies reached important conclusions. First, TILs recognizing tumor NeoAgs can be specifically identified in a large proportion of EOC patients with different techniques, even if such tumor type harbors a relatively low mutational load. Conversely, the rate of NeoAg immunogenicity filtered by the current bioinformatics pipeline is very low (<0.1%) and similar/lower to that seen in other studies,64,65 despite cross-validation of NeoAg prediction being very low at the moment. 66
In conclusion, TIL-ACT has been shown as feasible, but with modest results, in treating patients with EOC. The lack of TIL-ACT efficacy in this setting may result from several factors such as an inefficient expansion of TILs
CAR-T and TCR-based therapy in EOC
TCR and CAR-T therapy are two types of ACT that genetically modify T cells to treat cancers. In both these ACT procedures, naïve T cells are isolated by aphaeresis of peripheral blood, but they differ in their antigen recognition machinery. 68
TCRs employ a glycoprotein alpha–beta chain heterodimer that recognizes peptides presented on the cell surface by HLA molecules. CARs, on the other hand, are engineered to display antigen-binding fragments of a specific antibody fused to intracellular T-cell signaling domains. Thus, the bioengineered TCR can elicit an HLA-restricted immune response, while the CAR-T cells can recognize tumor-cell-surface antigens and elicit an immune response independently of HLA-antigen recognition.69,70 Moreover, the TCRs confer recognition to epitopes derived from proteins residing within any subcellular compartment while CAR-T cells can only recognize cell-surface antigens.
Much of the work on CAR-T and TCR therapy in EOC has been directed at tumor-associated antigens (TAAs), which are self-proteins abnormally expressed by cancer cells. These shared antigens can be separated into three main groups: the overexpressed antigens, which are normal surface proteins expressed in elevated levels on cancer cells, but in lower levels in normal cells (e.g. hTERT, HER2, mesothelin, and MUC-1); tissue-specific TAAs, which are shared between tumors and the normal tissue of their origin [e.g. tyrosinase, gp100, MART-1, prostate-specific antigen (PSA)]; and TAAs whose expression is normally restricted to male germline cells [i.e. cancer testis (CT) antigens, such as MAGE-A1, MAGE-A3, and NY-ESO-1]. 71
Some of the EOC antigens investigated
CAR-T against mesothelin (CART-meso), a consistently overexpressed protein carrying a negative prognostic role in EOC, was tested in a phase I/II trial to treat metastatic cancer patients, including EOC. Unfortunately, the trial was terminated due to slow/insufficient accrual [ClinicalTrials.gov identifier: NCT01583686]. Preliminary results of second-generation CART-meso are encouraging, 74 although final data are not available yet.
More recently, Haas
Other targets, such as MUC16 (CA125), were tested in a phase I trial using an i.v. and i.p. CAR-T modified to secrete IL-12. Although no significant toxicity was seen, the best observed clinical response was SD in a cohort of 18 heavily pretreated EOC. 76 A vast range of clinical trials using third-generation CAR-T against known EOC TAAs, such as the one cited earlier, are ongoing and have been recently reviewed. 77
Similarly, HLA-A2-restricted TCRs specific for epitopes from known EOC antigens such as NY-ESO-1, WT1, p53, MAGE-A4 are available for clinical application.44,78 In particular, NY-ESO-1, a CT antigen, over-expressed in more than 40% of EOC patients,79,80 has been tested in several phase I/II clinical trials, with TCR-ACT showing clinical benefits. Several other trials against NY-ESO-1 are currently ongoing in EOC patients [ClinicalTrials.gov identifiers: NCT01567891, NCT03691376, NCT02457650, NCT02869217, NCT03159585]
In contrast to CAR-T cells’ striking successes in treating patients with hematological malignancies, no equivalent achievements have been demonstrated to date in patients with solid tumors, including EOC. The limited number of targetable membrane antigens and their heterogeneous expression, difficulties in T-cell fitness and survival before reaching the tumor sites, and impaired trafficking to tumor sites have proven key obstacles for the success of CAR-T and TCR-based therapies in solid tumors and EOC. Technological advances in immune-engineering with viral vectors or for gene-editing with CRISPR–Cas9 technology 81 might open important avenues to tackle some of these key barriers to ACT, such as overcoming T-cell inhibitory signal or T-cell immunosuppressive metabolic insults82,83 from the TME. An ongoing clinical trial, which includes patients with EOC, is testing the effect of CRISPR-Cas9mediated PD-1 knocked-out CAR-T cells against mesothelin [ClinicalTrials.gov identifier: NCT03747965], and results are awaited.
The problem of antigen heterogeneity in solid tumors has led to the attempt of strategies that target multiple TAAs simultaneously. The use of aFRα bispecific T-cell engager has been shown to increase CAR-T cells’ efficacy in preclinical models of ovarian, colon, or pancreatic cancer. 84 Other examples include avidin-linked CARs combined with biotinylated antibodies 85 or fluorescein isothiocyanate fluorophore conjugated to TAA-binding molecules, 86 in an effort to successfully target heterogeneous solid tumors such as EOC.
Natural killer cells (NK)-based immunotherapy
Natural killer (NK) cells are another type of immune cell that can kill target cells through similar cytotoxic mechanisms as CD8+ cells. There has been a renewed interest in the use of NK cells as a source for genetically modified immune cell-based immunotherapy (CAR-NK) due to the reduced risk of off-target toxicity compared with CAR-T cells, the possibility of both CAR-dependent and NK cell receptor-dependent mechanisms of tumor killing, and their reduced risk of alloreactivity and thus graft
NK-based immunotherapy strategies have been studied for a long time in treating EOC,
89
also based on the
Geller
NK-ACT is also under evaluation for i.p. administration in the APOLLO phase I study [ClinicalTrials.gov identifier: NCT03213964]. Preliminary results presented, but not yet published, with the i.p. delivery of FATE-NK100 cells, show safety, and a clinical benefit in 3/9 patients treated. A summary of all clinical trials employing NK and NK-related cells for the treatment of EOC is presented in Table 2.
Clinical studies with NK-cell based immunotherapies in OC.

AE, adverse event; CAR, chimeric antigen receptor; CMV, cytomegalovirus; HGSOC, high-grade serous ovarian carcinoma; IL, interleukin; N/A, not available; NK, natural killer; OC, ovarian cancer; PD, progressive disease; PFS, progression-free survival; PR, partial response; RFS, recurrence-free survival; SD, stable disease.
As of January 2021, 17 ongoing CAR-NK cells studies were registered in the ClinicalTrials.gov, of which seven were targeting metastatic solid tumors expressing TAAs. In particular, a phase I trial is evaluating the role of anti-mesothelin NK-CAR cells in patients with EOC expressing >50% of mesothelin after standard chemotherapy [ClinicalTrials.gov identifier: NCT03692637]. An NK-CAR targeting NKG2DL (a molecule commonly upregulated in solid tumors) is being evaluated in solid tumors, including EOC [ClinicalTrials.gov identifier: NCT03415000].
Dendritic-cell vaccination
The application of vaccination for cancer treatment relies on the capability of vaccines to train the immune system. Through vaccine-training the host immune system can accurately detect and kill a variety of offending noxae, including cancer. This strategy was broadly tested in the field of infectious diseases. Cancer vaccines can be classified into different categories: (a) cell-based vaccines; (b) peptide/protein; (c) epigenetic; and (d) genetic vaccines, according to the method of choice to deliver the selected antigens. 98
So far, around 50 trials with vaccination approaches have been performed in patients with EOC with no statistically clinical differences detected by vaccine type or treatment schema.99,100 Some of these vaccines have targeted a single antigen such as CA125, MUC1, Her2, p53, and NY-ESO-1. In contrast, others used multiple peptides from various antigens or a whole-tumor antigen approach created with tumor cells, autologous tumor lysate, or tumor-derived ribonucleic acid.101–103 Overall, most trials demonstrated low therapeutic effects with a median PFS of 13.0 months, mainly due to the low expression of the target antigen, tumor heterogeneity, and the patient population.100,104
A prolongation of PFS and OS in recurrent EOC was demonstrated in several clinical trials investigating the efficacy of NY-ESO-1-based vaccines,105,106 dendritic cell (DC) vaccines based on a pool of peptides (Her2/neu, hTERT, and PADRE peptides), 107 or whole-tumor vaccines of autologous tumor cells infected with Newcastle disease virus, 108 or viral oncolysate vaccine generated from EOC cell lines infected with influenza-A virus. 109
DC vaccination with whole-tumor lysate might be a relevant strategy for EOC treatment, as surgery is a crucial component of EOC treatment, and therefore sufficient tumoral material should be available (Figure 1). Advantages of DC vaccination with autologous tumors and lysates include the agnostic targeting of the full repertoire of patient-specific TAAs and NeoAgs. This can stimulate a broad polyclonal T-cell response with priming of CD4+ and CD8+ T cells independent from the patient’s HLA haplotype.110,111 The pretreatment of EOC cells with hypochlorous acid (HOCl) oxidation (OCDC vaccine) 112 has been shown to increase the overall vaccine immunogenicity.113,114 The intranodal injection of autologous DCs pulsed with the OCDC vaccine was evaluated in a pilot study of five subjects with recurrent EOC. 114 Subsequently, other groups confirmed the safety of DC vaccination in EOC.115,116
We previously reported a two-step pilot clinical study testing the feasibility, safety, and clinical outcomes of DC vaccination using autologous tumor-cell-lysate supernatants in combination with bevacizumab and oral metronomic cyclophosphamide in recurrent OC. 27 Subjects who achieved at least SD after the vaccine but failed to exhibit a complete remission were enrolled in a second trial, involving LTD and ACT of autologous vaccine-primed CD3/CD28-costimulated T cells. Despite the relative low frequency of tumor-specific T cells elicited by this vaccine (<1 tumor-reactive T cell per 500 PBLs), an overall clinical benefit of 50% was seen in this heavily pretreated population. This strategy demonstrated T-cell responses against known EOC antigens and, more importantly, a close correlation between clinical benefit and immune response.
More recently, we reported further results of a pilot clinical trial testing a personalized vaccine generated by autologous DCs pulsed with OCDCs.
26
The vaccine was injected intranodally in platinum-treated, immunotherapy-naïve recurrent EOC patients. OCDC was administered alone (cohort 1,
This is particularly important, as the OCDC vaccine could be integrated as a priming modality before other immunotherapeutic strategies. The consequent anti-tumoral post-vaccination T-cell responses could be interrogated for the subsequent design of synthetic vaccines or T-cell therapies against newly recognized TAAs and/or tumor neo-epitopes. This combination of DC vaccines and TIL therapy has shown encouraging results in patients with melanoma. 117 Moreover, OCDC may prime patients, especially those lacking TILs, by inducing antitumor immunity, which could benefit from ICB treatment.
Similar to what is seen for T-cell therapy, the ability to identify NeoAgs through tumor deoxyribonucleic acid (DNA)-genomic sequencing has shifted the focus to investigate the clinical feasibility of personalized recombinant NeoAgs vaccines.118,119 For this reason, a phase I/II, randomized two-cohort, single-center study was developed to compare the immunogenicity and assess the safety of a personalized peptide (with a pool of 10 prioritized NeoAg-based peptides), pulsed DC vaccine (PEP-DC) alone, or OCDC vaccine followed by PEP-DC (PEP-DC2), in combination with low-dose cyclophosphamide. 120
Despite the reported limited clinical efficacy of DC-based therapeutic vaccination, this treatment modality has been associated with clinical activity in a subset of EOC patients representing a promising therapeutic option, alone or in combination, with other immunotherapies. Further studies are needed to define the best patient-candidate characteristics for therapeutic vaccine treatment, along with the optimal selection of the route of administration and neo-antigen selection.
Challenges of cell therapy in ovarian cancer
Despite targeting various antigens (Ags) and combining ACT with chemotherapy and anti-angiogenic agents, the exciting results of cell therapies in hematological malignancies and melanoma have not yet been reproduced in EOC, showing so far limited anti-tumor activity in early-phase clinical trials. 121
An ‘ideal’ target antigen should be highly and homogeneously expressed throughout the tumor, across multiple patients, and have minimal to no expression in normal tissues. Unfortunately, these characteristics do not apply to most currently explored targets for either CAR-T or TCR-T therapies in EOC. Current targets are abhorrently expressed antigens such as TAAs, which may be present at low levels in normal tissues; they might have heterogeneous Ag expression and have undergone thymic tolerance. 122 Therefore, research has focused on targeting cancer stem cells or tumor-initiating cells. A peculiarity of EOC, however, compared with other types of tumors, is its unknown putative site and cell of origin. 123 This might still preclude the identification of lineage markers as seen in hematological malignancies.
NeoAgs derived from non-synonymous mutations are instead tumor specific; they have not undergone central tolerance, and thus, are ideal candidates to exploit for cell therapy. However, a genomic peculiarity of HGSOC is its relatively low somatic point mutation load, high aneuploidy levels, and high levels of CNAs. 55 These features have been associated with low immunogenicity due to the low presence of NeoAgs. Moreover, the majority of EOCs are diagnosed at an advanced stage, FIGO III–IV with multiple metastatic sites that are heterogeneous in terms of genomics and T-cell infiltration patterns. 56 Thus, identifying clonal NeoAgs, potentially capable of simultaneously targeting multiple lesions, is a challenging task that might require access to various biopsies, which might not be clinically manageable in late-setting diseases.
Although the identification of neo-epitope-specific CD8+ T cells has been reported in ~90% of patients evaluated, 62 these are patient specific, limiting this approach’s broad application. Moreover, most epitopes showing significant immune response in solid cancer are derived from intracellular proteins that are difficult to target with standard CAR-T cells. 124
Another important barrier for the success of cell therapies in solid tumors is the capacity of antitumor effector immune cells to infiltrate and persist at the tumor site.
The EOC TME is intrinsically heterogeneous among both patients and tumors. Furthermore, beyond tumor heterogeneity, it has been reported that both immune-cell-excluded and inflammatory phenotype microenvironments may exist within the same tumor.57,125 This is an important challenge for the successful application of therapies that target the TME, such as ICB, and therapies requiring a permissive TME to act, such as cell-therapies. Moreover, a huge plethora of biochemical pathways and molecules present in the TME, and ascitic fluid in patients with EOC has been linked to immunosuppressive phenotypes which can lead to T-cell anergy and desertification.54,126 It appears evident that a better understanding of the key mechanisms leading to inflammation/desertification, as well as leading to T-cell exhaustion/anergy in the EOC TME, will be important to rationally design genome-engineered strategies and combination therapies to overcome this obstacle.
In particular, the choice of the right Ags and the improvement of T-cell characteristics for enhanced anti-tumor activity despite immunosuppressive TME are key factors for the further success of EOC cell therapies.
Conclusions
Despite high rates of response to initial treatment, EOC has a high recurrence rate and has yet to show a significant response to available immunotherapeutic agents. Cell therapies have transformed the treatment paradigm for patients with hematologic malignancies; however, the translation of this success to the unmet need of EOC patients is ongoing. Cell-based therapies in EOC have been explored in early-phase studies from a few highly specialized centers, with modest clinical results, raising concerns for the future development of these potentially curative therapeutic approaches.
Previous experience with ACT-TIL has shown the feasibility of this complex approach in the setting with EOC. The incorporation of new technologies such as DNA sequencing, DNA synthesis, and genetic screening tools to identify individual patient-specific NeoAgs with high sensitivity, specificity, and at scale, might broaden the application of this type of treatment to a broader group of EOC patients in coming years. Vaccination strategies have, to date, mainly encompassed shared TAAs and have been met with limited success.
A better understanding of the T-cell biology (T-cell exhaustion) driven by the peculiar EOC TME will be a crucial step in developing ACT for these patients. It will drive advances in T-cell engineering and clinical trial design with combination therapies. The immunosuppressive TME needs to be tackled to improve CAR-T and TIL-ACT products’ activity and persistence, and immune-engineering innovative solutions are currently being developed. Similarly, as T-cell engineering technology is streamlined, it might gain a prominent role in combination with other strategies. We believe that a better understanding of these key biological questions, along with technological developments will be key to broadening the use of cell therapeutics and ultimately improve EOC patients’ clinical outcomes.
Footnotes
Conflict of interest statement
Dr. Coukos has received grants from Celgene, Boehringer-Ingelheim, Roche, BMS, Iovance Therapeutics, Kite Pharma. The institution Dr. Coukos is affiliated with has received fees for Dr Coukos’ participation in advisory board or for presentation at a company sponsored symposium from Genentech, Roche, BMS, AstraZeneca, NextCure, Geneos Tx and Sanofi/Avensis. Dr. Coukos has patents in the domain of antibodies and vaccines targeting the tumor vasculature as well as technologies related to T-cell expansion and engineering for T-cell therapy. Dr Coukos holds patents around antibodies and receives royalties from the University of Pennsylvania regarding technology licensed to Novartis.
Dr Sarivalasis has received research grants from Roche. The institution Dr Sarivalasis is affiliated with has received consultancy and advisory fees from Roche, Novartis, GSK, Tesaro, BMS, Celgene, BMS, AstraZeneca. The institution Dr Sarivalasis is affiliated with has received travel fees from Roche, GSK, Tesaro, Clovis, AstraZeneca, MSD, Pfizer, Amgen, Celgene, Novartis.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
