Abstract
Background:
The clinical validity of tumor-infiltrating lymphocytes (TILs) has been investigated extensively in triple-negative breast cancer (TNBC). However, inconsistent cutoff values across studies limit its clinical application.
Objective:
To determine the optimal TILs cutoff predicting pathological complete response (pCR) and event-free survival (EFS) in Vietnamese TNBC patients undergoing neoadjuvant chemotherapy (NACT).
Design:
A single-center, retrospective cohort study of 106 stage II–III TNBC patients treated with NACT at Ho Chi Minh City Oncology Hospital between January 2022 and May 2023.
Methods:
Stromal TILs (sTILs) were evaluated on pre-treatment biopsy specimens using the International TILs Working Group guideline. The optimal cutoff was determined using receiver operating characteristic curve analysis. Logistic regression and Cox proportional hazards models were used to analyze potentially predictive and prognostic factors.
Results:
An optimal 20% TILs cutoff stratified the cohort into high- and low-TIL groups, with significantly different pCR rates (50.9% vs 9.8%, p < 0.001). High TILs (odds ratio (OR) = 8.22, p < 0.001) and lower clinical tumor status (OR = 3.64, p = 0.033) independently predicted pCR. TILs (hazard ratio (HR) = 0.38, p = 0.017) and pCR status (HR = 0.08, p = 0.017) were independent prognostic factors for EFS.
Conclusion:
A sTIL cutoff of 20% effectively predicts chemotherapy response and survival outcome in Vietnamese TNBC patients, supporting its use in clinical decision-making.
Keywords
Introduction
Neoadjuvant therapy has become the standard of care for early-stage and locally advanced triple-negative breast cancer (TNBC), aiming to achieve pathological complete response (pCR)—the well-established surrogate for long-term survival outcomes. 1 In recent trials such as KEYNOTE-522, pCR rates have exceeded 50% with the combined chemo-immunotherapy. 2 However, in Vietnam, anthracycline-based chemotherapy remains the standard neoadjuvant regimen due to limited access to novel agents, which is attributed to immune-related adverse event concerns, financial constraints, and lack of insurance coverage. Therefore, the identification and standardization of robust predictive and prognostic biomarkers are essential to enable the stratification of patients into subgroups with a high likelihood of response to standard chemotherapy and those at risk of suboptimal response or early disease progression. This stratification may facilitate personalized treatment approaches, including therapy intensification, the consideration of alternative regimens, or closer monitoring during the neoadjuvant phase, with the ultimate goal of improving clinical outcomes.
Among the emerging biomarkers under investigation, tumor-infiltrating lymphocytes (TILs) reflect tumor immune microenvironment activity and are strongly correlated with response to neoadjuvant chemotherapy (NACT) and prognosis in TNBC. 3 TILs are mononuclear immune cells that extravasate from blood vessels and infiltrate the tumor microenvironment (TME). Various methods exist to assess TILs at different levels. Subsequently, the simplest and most widely used approach in clinical practice is the morphological assessment of TILs on hematoxylin and eosin (H&E)-stained slides. This method quantifies the percentage of area within the invasive tumor margin infiltrating mononuclear immune cells, and expresses it as a continuous variable with 10% increments. In 2014, the International TILs Working Group (ITWG) published standardized guidelines for H&E-based TIL assessment, which have exhibited a high analytic validity. 4 A meta-analysis conducted by the German Breast Group, which analyzed 1000 TNBC data from six randomized Gepar trials, demonstrated a significantly higher pCR rate in patients with TILs ⩾60%, which officially confirmed its clinical validity. 5
However, the optimal cutoff for defining high versus low-TILs varies significantly across studies, ranging from 10% to 60%, which limits its applicability in routine clinical decision-making.5 –8 In addition, population-specific variability potentially hinders the establishment of a universal threshold. 9 A locally validated TILs cutoff may enhance risk stratification and support personalized treatment strategies for Vietnamese TNBC patients.
We conducted this study to identify the optimal pre-treatment TILs cutoff to predict pCR and evaluate the prognostic value for event-free survival (EFS) in a Vietnamese TNBC cohort undergoing NACT.
Methods
Study population and data collection
A total of 106 consecutive core needle biopsy specimens from TNBC patients were retrospectively retrieved from the pathology database of Ho Chi Minh City Oncology Hospital between January 2022 and May 2023. Inclusion criteria were as follows: primary invasive TNBCs (defined as tumors with < 1% estrogen and progesterone receptor expression, and absence of human epidermal growth factor receptor 2 (HER2) protein overexpression or gene amplification); clinical stage II–III; and treated with NACT. Patient demographics, clinicopathological characteristics, and treatment outcomes were collected.
TILs evaluation
Pre-treatment stromal TILs (sTILs) were assessed on H&E-stained slides by a single experienced breast pathologist, blinded to clinical outcomes. Assessment followed the ITWG 2014 recommendations. 4 sTILs were defined as the proportion of stromal area occupied by mononuclear inflammatory cells, excluding areas of necrosis or artifacts. TILs were scored based on the full tumor section rather than hotspots. Scoring was performed in 10% increments for values ⩾20%, and as exact percentages (1%, 5%, or 10%) for lower values when morphologically countable (Figure 1).
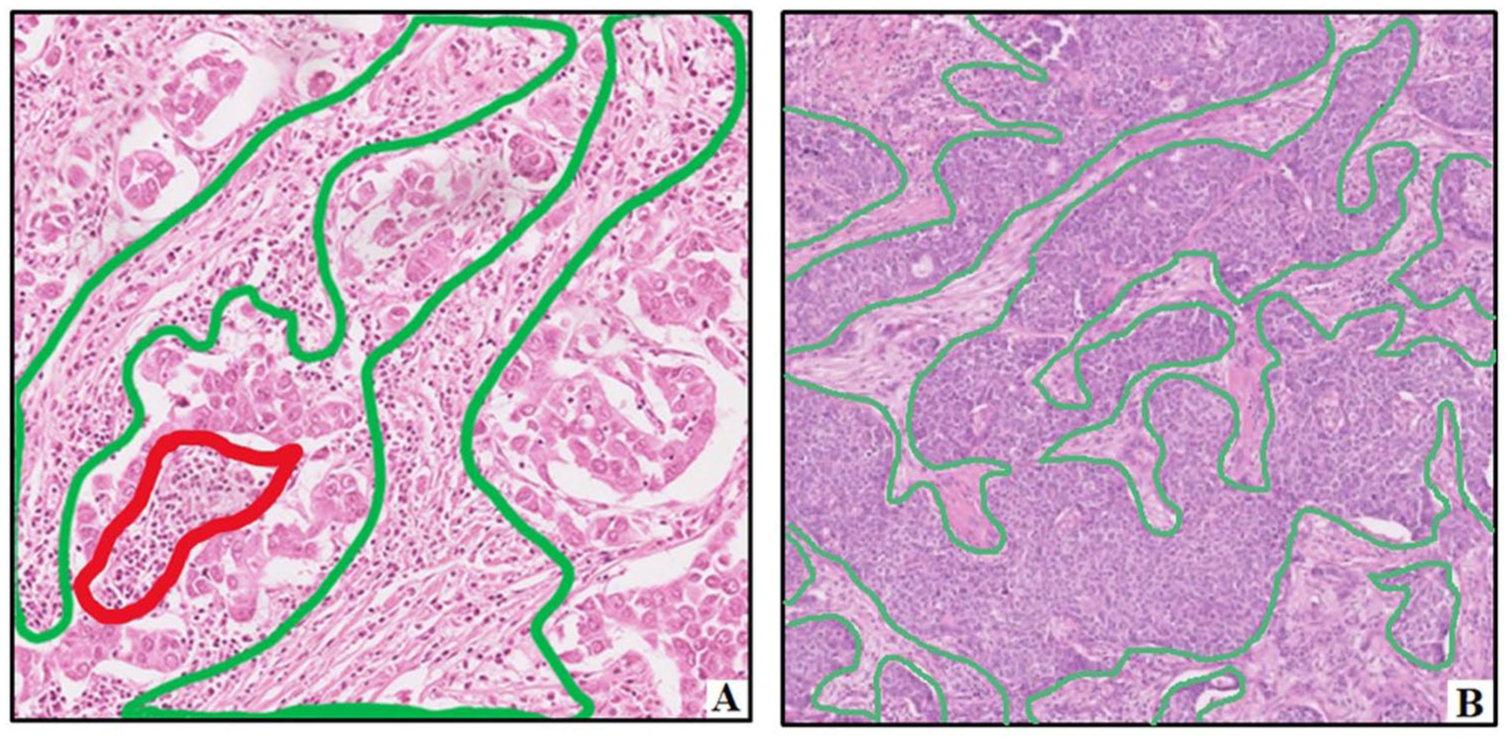
Illustration of TILs assessment on H&E-stained slides. (a) H&E-stained slide at 100× magnification shows ~40% stromal TILs (green outline), with intratumoral TILs in red. (b) At 40× magnification, a case with 1% stromal TILs displays a clean stroma with sparse cells. High-TIL cases (a) are typically estimated in 10% increments due to difficulty in precise quantification, while low-TIL cases (b) allow finer estimates (e.g., 1%, 5%, 10%). These practices align with 2014 ITWG guidelines, recommending “Pathologist should report their scores in as much detail as the pathologist feels comfortable with.”
Endpoints measurement
The primary endpoint was pCR, defined as the absence of residual invasive carcinoma in postoperative breast tissue and axillary lymph nodes specimens (ypT0/isN0). Patients who experienced progressive disease (PD) during NACT, making definitive surgery unfeasible, were also classified as non-pCR.
The secondary endpoint was EFS, defined as the time from the initiation of NACT to the first occurrence of any of the following events: PD, relapse, or death, whichever came first. Patients were followed up every 3–6 months through clinical visits and medical record review to document any of the above events. Patients without complete follow-up data were censored at the time of their last known contact. Based on recent guidelines for standardizing endpoint selection in neoadjuvant breast cancer studies, we selected EFS as an appropriate survival endpoint. 10 Disease-free survival (DFS) and recurrence-free survival were not chosen due to the risk of missing cases where the disease progresses during NACT, which could lead to an inaccurate assessment of the true efficacy of neoadjuvant treatment.
Statistical analysis
Receiver operating characteristic (ROC) analysis was used to evaluate the predictive performance of the model for pCR. The optimal TILs cutoff was defined as the value with the highest Youden index (J = sensitivity + specificity – 1). Univariate and multivariate logistic regression analyses were performed to identify independent predictors of pCR. Variables with p value <0.2 in the univariate analysis were included in the multivariate model.
The Kaplan–Meier method was used to estimate the EFS. The log-rank test was applied to compare survival outcomes between subgroups. Multivariate Cox proportional hazards models were performed to identify independent prognostic factors for EFS.
Chi-square or Fisher’s exact test was used for categorical variables, and t-test or Mann–Whitney U test for continuous variables. All statistical analyses were performed using the SPSS software version 20.0 (IBM, Armonk, New York, USA) and GraphPad Prism version 10.0 (GraphPad Software, La Jolla, California, USA). Statistical significance was set at p < 0.05.
This study was completely checked according to the STROBE checklist, 11 which was included in Table S1.
Results
Patient characteristics and treatment outcomes
Baseline characteristics of the 106 TNBC patients are summarized in Table 1. The median age at diagnosis was 45.4 years (range: 29–78), with one-third diagnosed before age 40. Clinical stage was distributed relatively balanced from stage IIA to IIIC, with stage IIA (cT2N0M0) accounting for 21.7% of cases. Invasive ductal carcinoma of no special type (NST) comprised 96.2% of tumors. Histological grade was predominantly grade 2 (51.9%) and grade 3 (46.2%). The mean Ki-67 index was 62.8% (95% CI: 58.7–66.8), most commonly in the 60%–80% range. HER2 status was HER2-zero in 86.8% and HER2-low in 13.2% of patients.
Patient demographics, clinicopathologic, and treatment characteristics.

AC-T, doxorubicin plus cyclophosphamide followed by taxane; ALND, axillary lymph node dissection; BCS, breast-conserving surgery; cN, clinical nodal stage; cT, clinical tumor stage; ET, epirubicin plus taxane; HER2, human epidermal growth factor receptor 2; IDC of NST, invasive ductal carcinoma of no special type; SLNB, sentinel lymph node biopsy; TC, docetaxel plus cyclophosphamide; TILs, tumor-infiltrating lymphocytes.
Statistically significant (p < 0.05).
The majority of patients received anthracycline-based NACT, including AC-T (doxorubicin–cyclophosphamide followed by taxane) and ET (epirubicin–taxane) regimens. Two elderly patients with cardiovascular comorbidities were treated with a non-anthracycline regimen consisting of docetaxel and cyclophosphamide.
Following NACT, 91.5% (97/106) patients underwent surgical intervention, with 94 received curative-intent surgery and 3 patients undergoing palliative mastectomy due to metastatic progression. The pCR rate was 31.1% (33/106), including three cases with residual in situ carcinoma in the breast. Among the 68.9% (73/106) of non-pCR patients, 12 experienced PD during NACT, and 61 had residual invasive disease in the surgical specimen following curative surgery.
Predictive value of TILs for pCR
Optimal TILs cutoff for predicting pCR
The median pre-treatment sTILs level was 20% ± 4.58%, predominantly ranging from 1% to 30%. Only 15.1% of patients exhibited TIL levels ⩾50% (Figure 2). Higher baseline TILs were significantly associated with increased pCR probability. For each 10% increase in TILs, the odds of achieving pCR increased by 62% (odds ratio (OR) = 1.62; 95% CI: 1.26–2.09; p < 0.001). The area under the ROC curve (AUC) was 0.74 (95% CI: 0.64–0.84; p = 0.001), demonstrating good discriminatory performance (Figure 3). The 20% of TILs was identified as the optimal cutoff for predicting pCR based on the maximum Youden index (Table 2). It stratified the cohort into high-TIL (⩾20%) and low-TIL (<20%) groups. The two groups exhibited broadly comparable clinicopathologic and treatment characteristics, except the high-TILs group showed a significantly lower clinical T stage (cT) (Table 1). Patients in the high-TILs group achieved significantly higher pCR rates compared to those in the low-TILs group (50.9% vs 9.8%; p < 0.001; Figure 4). This cutoff demonstrated high sensitivity (84.85%) and negative predictive value (90.2%) in predicting pCR following NACT (Table 3).

Distribution of TIL levels in the study population (n = 106).
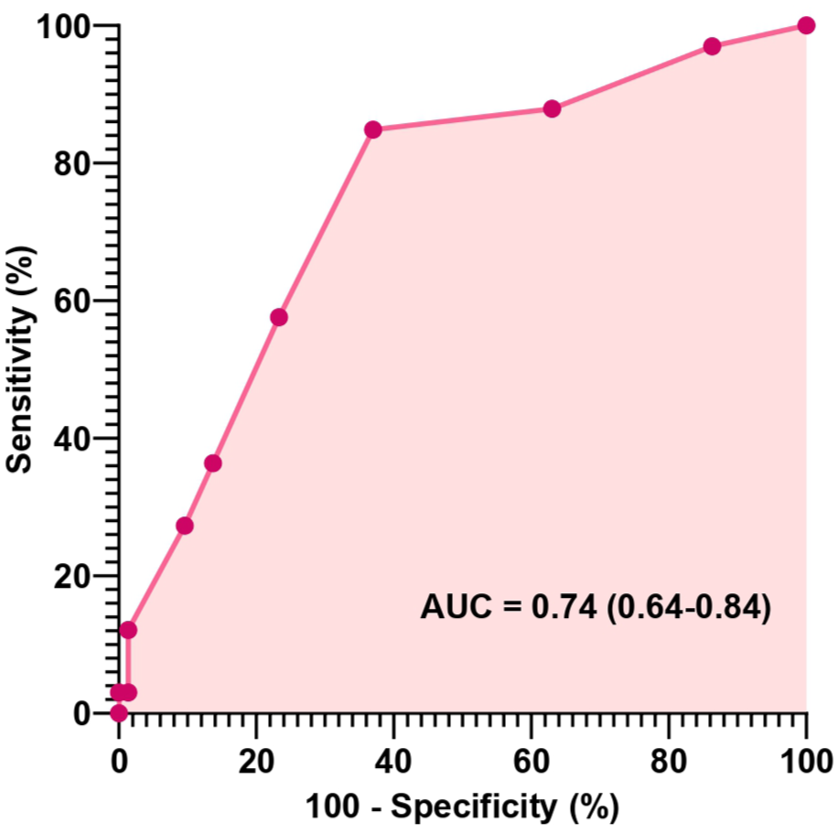
ROC curve for predicting pCR based on the TILs cutoff value. The ROC curve demonstrated the diagnostic performance of TILs in predicting pCR following neoadjuvant chemotherapy in triple-negative breast cancer patients. The area under the curve (AUC) is 0.74, indicating acceptable discriminative ability. Sensitivity and 1 − specificity are plotted for various TIL thresholds.
Diagnostic performance of TILs at different cutoff values on ROC curve analysis.

Sensitivity, 1 – specificity, and Youden’s index are reported for selected TILs cutoff values. The optimal threshold was identified at 20%, yielding the highest Youden’s index (0.479).
pCR, pathological complete response; ROC, receiver operating characteristic; TILs, tumor-infiltrating lymphocytes.
The values shown in bold indicate the optimal TILs cutoff value, as determined by the highest Youden’s index on ROC curve analysis. At a cutoff of 20%, TILs achieved the maximum Youden’s index (0.479), representing the best balance between sensitivity (0.848) and specificity (0.63) for predicting pathological complete response (pCR).

pCR rate according to TIL groups. A significantly higher pCR rate was observed in the high-TIL groups (50.9%) compared to the low-TIL groups (9.8%) (p < 0.001), highlighting the predictive value of TILs for treatment response in TNBC.
Diagnostic performance of TILs ⩾20% in predicting pCR.
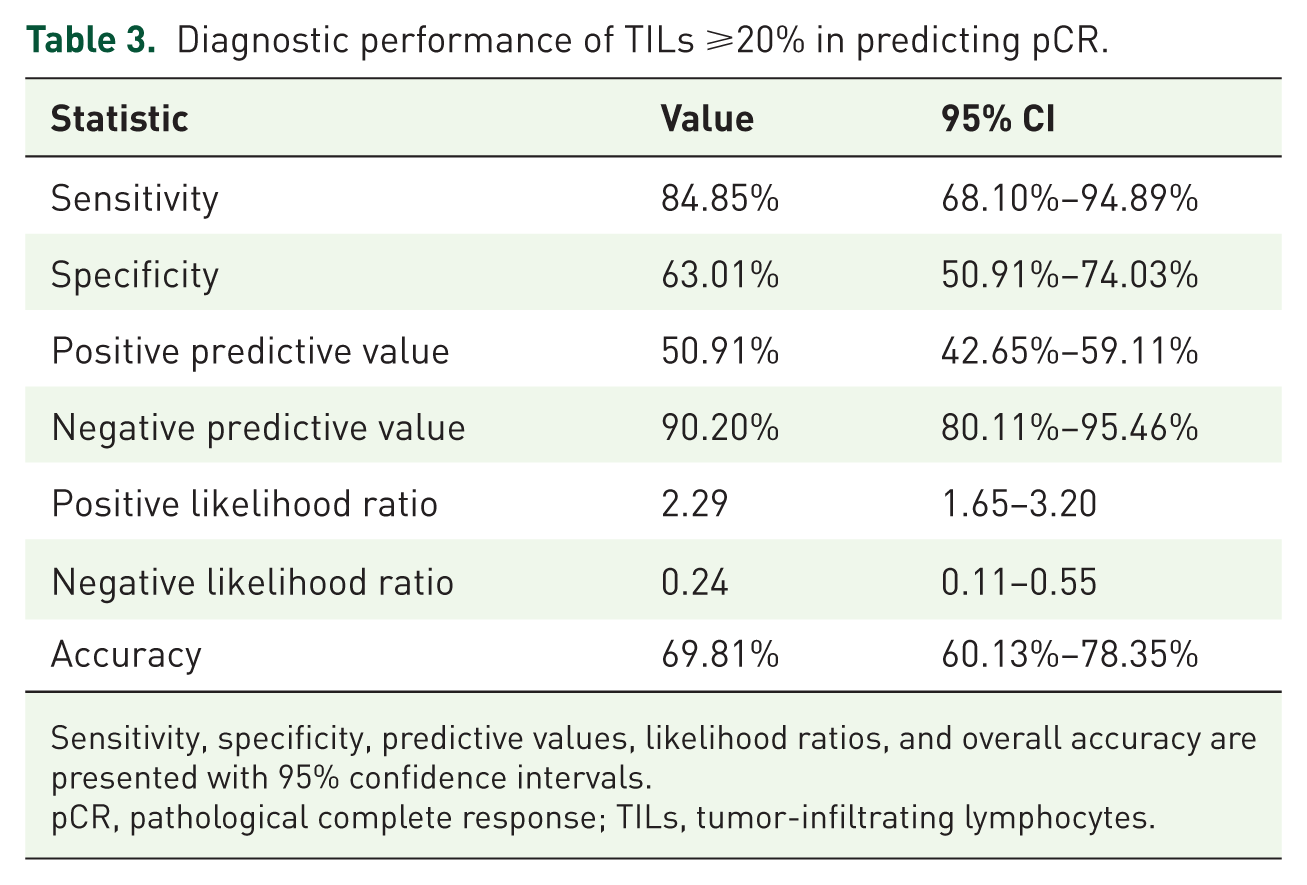
Sensitivity, specificity, predictive values, likelihood ratios, and overall accuracy are presented with 95% confidence intervals.
pCR, pathological complete response; TILs, tumor-infiltrating lymphocytes.
Associations between TILs, clinicopathologic parameters, and pCR
Univariate logistic regression analysis identified several variables significantly associated with pCR, including TILs, cT, cN, clinical stage, histological grade, and chemotherapy regimen. Variables with p values >0.2 were excluded from further analysis. Due to strong collinearity with cT and cN, clinical stage was omitted from the multivariate model to avoid multicollinearity. Multivariate analysis of the remaining potential predictors identified high-TILs (OR = 8.22; 95% CI: 2.58–26.25; p < 0.001) and lower cT (OR = 3.64; 95% CI: 1.11–11.96; p = 0.033) as independent predictive factors for pCR (Table 4). The model correctly classified 79.8% of cases as pCR or non-pCR.
Correlations between TILs and pCR in TNBCs treated with NACT.

AC-T, doxorubicin/cyclophosphamide followed by taxane; CI, confidence interval; cN, clinical nodal stage; cT, clinical tumor stage; ET, epirubicin/taxane; NACT, neoadjuvant chemotherapy; NE, not estimated; OR, odds ratio; pCR, pathological complete response; TC, docetaxel/cyclophosphamide; TIL, tumor-infiltrating lymphocyte; TNBC, triple-negative breast cancer.
Statistically significant (p < 0.2).
Statistically significant (p < 0.05).
Prognostic value of TILs for EFS
After a median follow-up time of 11.3 months, 38 events were recorded, including 23 PD events and 15 cases of relapse. The estimated 2-year EFS rate in the overall TNBC cohort was 52.6%, with 65.5% in the high-TILs group compared to 38.6% in the low-TILs group. The median EFS was 14.5 ± 2.7 months in the low-TILs group, but did not reach the high-TILs group in the final analysis. High-TIL levels were associated with a 75% reduction in the risk of disease progression, relapse, or death (HR = 0.25; 95% CI: 0.13–0.47; p < 0.0001; Figure 5).

Kaplan–Meier curves for event-free survival by TIL groups. Kaplan–Meier estimates of EFS stratified by TILs group. The 2-year EFS rates were 65.5% in the high-TILs group and 38.6% in the low-TILs group. Median follow-up was 11.3 months. The hazard ratio (HR) for PD, recurrence, or death in the high-TILs group compared to the low-TILs group was 0.25 (95% CI: 0.13–0.47; p < 0.0001).
In univariate analysis, pCR, high-TILs, cT1–2, cN0–1, clinical stage II, and grade 3 tumors were associated with improved EFS. In multivariate analysis, only high-TILs (HR = 0.38; 95% CI: 0.17–0.84; p = 0.017) and pCR (HR = 0.08; 95% CI: 0.01–0.64; p = 0.017) remained independent prognostic factors (Table 5). Stratifying patients by TILs level and pCR status yielded four subgroups. Among residual disease groups, patients with high-TILs had significantly better EFS than those with low-TILs, with a median EFS of only 11 months in the low-TIL group (p = 0.01) (Figure 6).
Prognostic value of clinicopathologic factors for EFS.

CI, confidence interval; cN, clinical nodal stage; cT, clinical tumor stage; EFS, event-free survival; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; NR, not reached; pCR, pathological complete response; TILs, tumor-infiltrating lymphocytes.
Statistically significant (p < 0.2).
Statistically significant (p < 0.05).
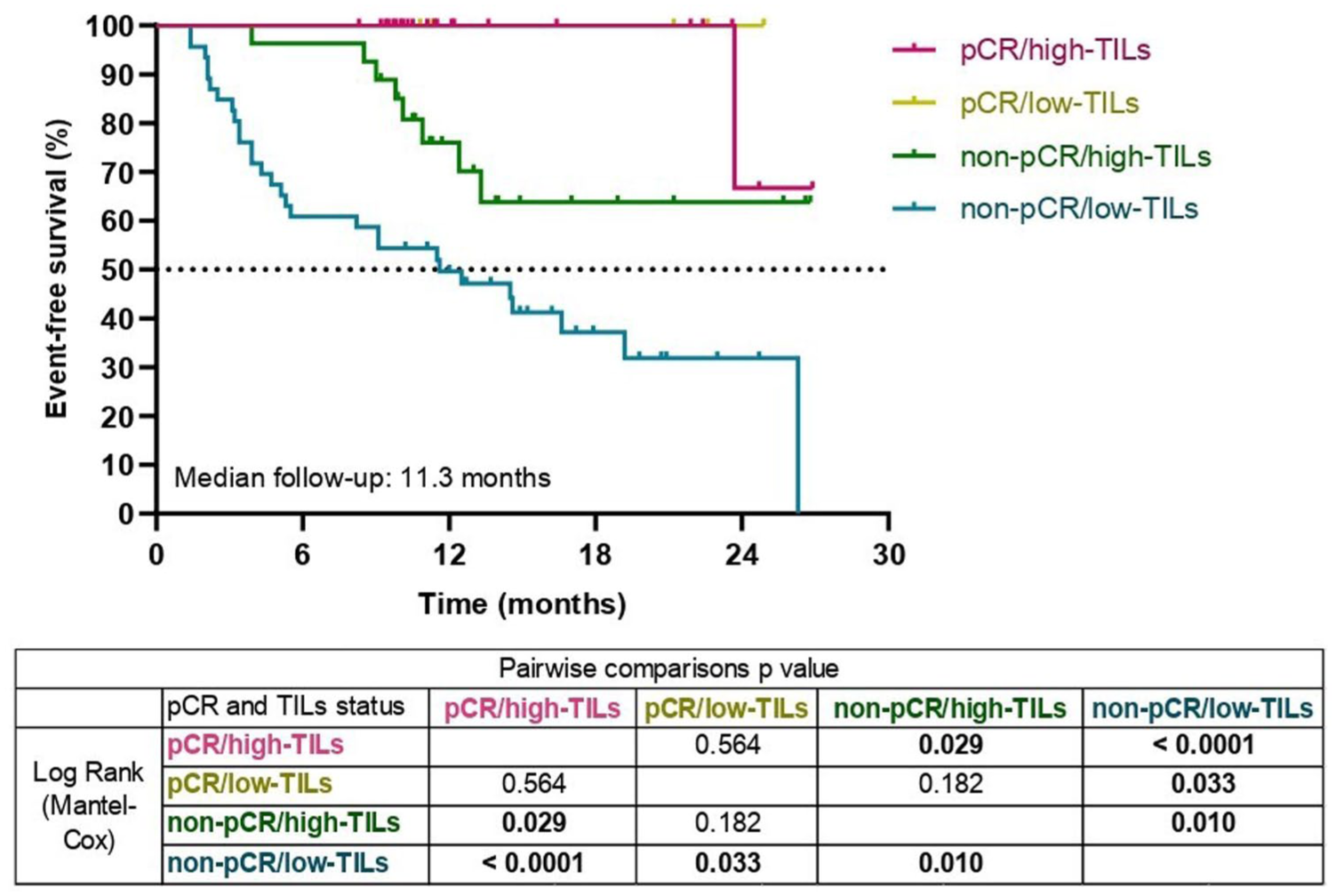
Kaplan–Meier curves for EFS stratified by TILs and pCR status. Kaplan–Meier curves depicting EFS for patients stratified into four subgroups based on pCR status and pre-treatment TILs levels: (1) pCR with high-TILs, (2) pCR with low-TILs, (3) non-pCR with high-TILs, and (4) non-pCR with low-TILs. Patients achieving pCR with high-TILs showed the most favorable survival outcomes. Median follow-up was 11.3 months. Pairwise comparisons using the Mantel–Cox log-rank test are summarized in the accompanying table, with significant differences observed between key subgroups (p < 0.05).
Discussion
Predictive value of TILs for pCR
This study confirms the predictive value of TILs in Vietnamese TNBC patients treated with NACT. Baseline TIL levels were increased linearly with the rate of pCR, consistent with prior evidence from the first landmark Gepar Duo trial in 2010 and subsequent studies.5,7,12,13 Two hypothetical biological mechanisms, extrapolated from prior studies, may underlie this association. First, a high density of TILs, particularly CD8+ cytotoxic T cells, within the TME indicates a robust cell-mediated antitumor immune response. The abundant presence of these cytotoxic T lymphocytes enables direct tumor cell lysis, thereby enhancing the effectiveness of chemotherapy. Moreover, studies at the protein level have demonstrated that a higher CD8+/FOXP3+ ratio or a lower proportion of FOXP3+ regulatory T cells (Tregs)—a immunosuppressive mediators—indicates a more permissive immune environment for tumor eradication. 14 Second, a recent study reported that higher TIL levels were positively correlated with systemic pro-inflammatory mediators such as interleukins, TNF-α, and IFN-γ, which may augment chemotherapy sensitivity through immune activation pathways. 15
Although evaluating TILs as a continuous variable preserves biological information, 4 it presents practical challenges in clinical settings, including interpretation variability and lack of a standardized threshold for decision-making. Therefore, defining a cutoff value is crucial for stratifying TNBC patients into clinically meaningful subgroups.
However, applying a universal cutoff across all TNBC populations may be impractical. Despite adherence to the same 2014 ITWG guidelines across studies, comparative data reveal substantial inter-population variability in TIL distributions. Studies conducted in various Western countries (including Italy, Spain, and Germany, etc.) reported relatively high rate of lymphocyte-predominant breast cancer (LPBC—first defined as TILs ⩾60% by Denkert et al. in Gepar Duo study), ranging from 21.9% to as high as 88%.5,8,16 By contrast, data from Asian populations—particularly from China—showed substantially lower LPBC prevalence as 4%.6,7 These findings prompted the question of whether differences in the TME, particularly TIL levels, exist across populations and ethnic groups. Moreover, a recently published multiethnic cohort study in Hawaii found a significant difference in TIL levels among Asians, Native Hawaiians, and White patients. 9 This further reinforced the hypothesis that immune profiles and TIL distributions may be population-specific. Our study, alongside emerging evidence from other Vietnamese cohorts, highlights a relatively low median TILs level in Vietnamese TNBC patients—typically between 20% and 25%.17,18 These results were comparable to European populations,18,19 slightly higher than Chinese cohorts (15%–17%),6,20 but significantly lower than Latin American populations, where median TILs can reach 40%. 21 These disparities likely reflect ethnic, genetic, and environmental variations in the immune landscape, reinforcing the need for population-specific TILs cutoffs to enhance the predictive and prognostic utility of this biomarker in TNBC. Therefore, the 20% cutoff identified in our study may serve as a reference for validation in larger cohorts and has the potential to become a population-specific threshold for predicting pCR following NACT in Vietnamese TNBC patients.
The clinical relevance of TILs varies depending on the treatment context and healthcare resources of each setting. Globally, TILs are increasingly explored as biomarkers to identify TNBC patients who may benefit from neoadjuvant chemo-immunotherapy. Patients with high-TILs often exhibit stronger immune activation and better response to immunotherapy, supporting their inclusion in intensified treatment strategies. However, in resource-constrained settings such as Vietnam, where immune checkpoint inhibitors are often inaccessible due to financial limitations, the clinical utility of TILs may be best oriented toward identifying patients at high risk of chemotherapy resistance. In this context, our proposed 20% TILs cutoff demonstrated high sensitivity (84.85%) and negative predictive value (90.2%) for predicting pCR following NACT. This threshold is particularly useful for identifying patients with poor response to standard chemotherapy, who may require closer monitoring, earlier reassessment, or consideration for treatment escalation (e.g., carboplatin addition, dose-dense regimens, or chemo-immunotherapy) if clinically feasible. Patients with low-TILs should be counseled about their low probability of achieving pCR (9.8% in our study) and informed of potential benefits from treatment intensification strategies based on cost–benefit considerations. Although current international guidelines do not yet recommend TILs as a biomarker for tailoring NACT intensity due to insufficient clinical utility evidence, our findings support its practical relevance in low- and middle-income settings. A TIL-based stratification model offers a simple, low-cost, and biologically grounded tool to aid treatment decisions. Ultimately, our study provides a population-specific perspective that complements global research while addressing local treatment limitations in real-world oncology practice.
The need for a combination model in pCR prediction
Despite its strong negative predictive value, the 20% TILs cutoff has only moderate positive predictive value (50.91%), indicating that a substantial proportion of patients with high-TILs do not ultimately achieve pCR. Increasing the TILs threshold improved specificity—reaching 90.4% and 98.6% at cutoffs of 50% and 60%, respectively—but substantially reduced sensitivity, potentially excluding patients who might still benefit from standard NACT. These findings suggest that TILs alone may not sufficiently capture the biological complexity underlying treatment response in TNBC, integrating the need for combination of several clinicopathologic or molecular markers and optimization of prediction models. Several studies have proposed multifactorial models incorporating TILs alongside biomarkers such as programmed cell death ligand 1 (PD-L1), CD73, Ki-67, androgen receptor expression, and multi-gene immune signatures.22 –24 In this study, TILs and cT were demonstrated as independent predictors of pCR, supporting the feasibility of developing a combined predictive nomogram to improve individualized treatment planning in future studies.
Prognostic value of TILs for EFS
Multiple studies have highlighted the prognostic significance of TILs in TNBC patients treated with NACT, particularly in relation to DFS and overall survival (OS). Hong et al. reported superior 5-year DFS and OS in high-TIL patients compared to low-TIL counterparts (DFS: 79.7% vs 47.8%, p = 0.002; OS: 83.1% vs 57.5%, p = 0.009). 6 Castaneda found that high-TIL levels were associated with improved 32-month DFS (40% vs 20%; p = 0.0076). 20 Similarly, in our study, Kaplan–Meier curves demonstrated an obvious EFS advantage in patients with high-TIL levels, supporting the long-term survival prognostic value of TILs. Notably, the early and sustained separation between the curves reflects the disproportionately high rate of PD during NACT in the low-TILs group, accounting for 72.4% of EFS events. These findings underscore the value of EFS as a clinically meaningful endpoint, as used in our study, by capturing early treatment failures that may not be reflected in traditional DFS outcomes, thereby avoiding overestimation of therapeutic efficacy.
pCR is a well-established prognostic factor in TNBC. Patients with residual disease generally face higher recurrence rates and poorer long-term outcomes, often necessitating extended systemic adjuvant therapy. However, our findings reveal substantial prognostic heterogeneity within this non-pCR population, with the high-TILs subgroup demonstrating significantly longer EFS. This suggests that TILs may enhance risk stratification beyond pCR status alone and help identify subsets with more favorable prognoses. By contrast, Abuhadra et al., using data from the ARTemis trial, found no EFS difference between high- and low-TIL patients with residual disease following NACT (p = 0.7). 24 This discrepancy may be explained by the difference in treatment protocols. In their study, patients exhibiting suboptimal response—defined as less than 70% reduction in tumor volume after four cycles of Anthracycline and Cyclophosphamide—were enrolled in phase II trials investigating targeted therapies or immunotherapy. The potential efficacy of these escalated treatment regimens may have mitigated the survival differences typically observed between TIL subgroups. Collectively, these findings underscore the need for novel post-NACT therapeutic strategies and support the incorporation of immune biomarkers such as TILs into risk-adapted treatment algorithms. Given their poor prognosis, patients in the low-TILs/non-pCR subgroup may represent the ideal candidates for biomarker-guided treatment escalation.
Study strengths, limitations, and future perspectives
To the best of our knowledge, this is the first comprehensive study in a Vietnamese TNBC population to evaluate the predictive and prognostic value of TILs using a population-specific cutoff. Previous studies in Vietnam primarily explored the analytical validity of TILs and their correlation with clinicopathologic variables, without integrating standardized outcome measures. 25 By contrast, our research employed clinically meaningful endpoints recommended by the NeoSTEEP framework, 10 thereby improving the interpretability of NACT outcomes and enhancing comparability with future international studies. To minimize interobserver variability and sampling bias, all H&E-stained sections were analyzed within 1.5 years of fixation by a single experienced breast pathologist using standardized TILs assessment criteria. However, the median follow-up duration for survival outcomes is relatively short, which may underestimate late recurrences and limit the assessment of long-term prognostic value. Extended follow-up is warranted to validate the durability of TILs as an independent prognostic biomarker for EFS and OS.
Despite advances in systemic therapy, TNBC remains a highly aggressive subtype with early relapse and poor survival outcomes. Future efforts should focus on standardizing the well-known and identifying novel biomarkers for patient stratification, developing integrative predictive models, and exploring individualized treatment strategies beyond conventional cytotoxic chemotherapy.
Standardization of TILs evaluation
While TILs are established predictive and prognostic markers in TNBC, their routine clinical use is hindered by variability in quantification. A recent Artificial Intelligent (AI)-based model presented at the 2024 ASCO annual meeting, using H&E and immunohistochemistry images, demonstrated strong correlation with EFS (AUC = 0.7, p < 0.05) and offers a reproducible and efficient approach to TILs assessment. 26 Integrating AI into pathology workflows may enhance standardization and scalability in TILs evaluation.
Integrating predictive and prognostic factors
TILs alone may not fully reflect the complex tumor–host interactions in TNBC. Several multivariable models—such as Cerbelli’s Tissue Immune Profile (TILs, PD-L1, CD73) or the MD Anderson model (TILs, TNM, Ki-67, AR)—have shown superior predictive performance.21,22 In our study, both TILs and cT stage independently predicted pCR, supporting the feasibility of building locally adapted predictive tools. While advanced genomic assays like DNA Damage Immune Response (DDIR) and DetermaIO show promise in guiding immunotherapy, their current application in Vietnam remains limited by cost and availability. 23
Optimization of neoadjuvant systemic therapy
The integration of pembrolizumab (Keytruda®) into NACT, as validated in the KEYNOTE-522 trial, has improved outcomes in early-stage TNBC. 2 However, key questions remain regarding post-pCR duration and escalation strategies for poor responders. Ongoing trials such as OptiMICE-pCR, OptiMICE-RD, and TROPION-Breast-03 are exploring novel agents in these contexts.27,28 With pembrolizumab now available at our institution, real-world studies are essential to evaluate its effectiveness, toxicity, and the utility of biomarkers for guiding personalized therapy and optimizing resource use in low- and middle-income settings.
Conclusion
This study confirms the predictive and prognostic value of TILs in Vietnamese patients with TNBC receiving NACT. A cutoff of 20% effectively stratified patients by response and survival outcomes, supporting its potential clinical utility. TILs and clinical tumor stage were identified as independent predictors of pCR, while both TILs and pCR independently predicted EFS. These findings underscore the importance of integrating immune biomarkers into treatment planning, particularly in resource-limited settings. Long-term follow-up and validation in larger, multicenter cohorts are warranted to confirm the durability and generalizability of these results.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251403410 – Supplemental material for Predictive and prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancer: a real-world study from Vietnam
Supplemental material, sj-docx-1-tam-10.1177_17588359251403410 for Predictive and prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancer: a real-world study from Vietnam by Linh Dang Ngoc Tran, Kha Kinh Nguyen, Tam Minh Pham, Quy Hoang Nguyen, Tu Anh Thai, Duc Thi Hong Phan and Anh Thi Tuyet Cung in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
