Abstract
Background:
High body mass index (BMI) has been associated with worse clinical outcomes in patients with early-stage breast cancer (BC), and its negative effects could be mediated by hyperglycemia/diabetes. However, the prognostic impact of high BMI in early-stage HER2-positive (HER2+) BC patients remains controversial.
Methods:
We conducted a retrospective study to investigate the impact of baseline BMI or glycemia on relapse-free survival (RFS) and overall survival (OS) in patients with surgically resected, stage I–III HER2+ BC treated with standard-of-care, trastuzumab-containing adjuvant biochemotherapy. The optimal BMI and glycemia cut-off values for RFS were identified through maximally selected rank statistics. Cox regression models were used to assess the impact of BMI, glycemia and other relevant variables on clinical outcomes.
Results:
Among 505 patients included in the study, a BMI cut-off of 27.77 kg/m2 was identified as the best threshold to discriminate between patients with low BMI (n = 390; 77.2%) or high BMI (n = 115; 22.8%). At multivariable analysis, higher BMI was associated with significantly worse RFS [hazard ratio 2.26; 95% confidence interval (CI): 1.08–4.74, p = 0.031] and worse OS (hazard ratio 2.25, 95% CI 1.03–4.94, p = 0.043) in the whole patient population. The negative impact of high BMI was only observed in patients with hormone receptor (HR)-negative/HER2+ BC (hazard ratio 2.29; 95% CI: 1.01–5.20; p = 0.047), but not in patients with HR-positive (HR+)/HER2+ BC (hazard ratio 1.36; 95% CI: 0.61–3.07, p = 0.452). By contrast, hyperglycemia (⩾109 mg/dl) at baseline was associated with a trend toward significantly worse RFS at multivariable analysis only in patients with HR+/HER2+ BC (hazard ratio 2.52; 95% CI: 0.89–7.1; p = 0.080).
Conclusions:
High BMI is associated with worse clinical outcomes in early-stage HR−/HER2+ BC patients treated with trastuzumab-containing adjuvant biochemotherapy, while baseline hyperglycemia could be a predictor of worse RFS in HR+/HER2+ BC patients. Prospective studies are needed to investigate if modifying patient BMI/glycemia during treatment can improve clinical outcomes.
Keywords
Introduction
Human epidermal growth factor receptor 2-positive (HER2+) breast cancer (BC) accounts for approximately 20–25% of breast neoplasms and is characterized by an aggressive biologic and clinical behavior as a result of HER2 signaling-induced stimulation of tumor cell growth, proliferation and metastatization.1,2 However, the introduction of anti-HER2 targeted therapies, including trastuzumab, pertuzumab, lapatinib and the antibody–drug conjugate trastuzumab emtansine (T-DM1) in the last two decades has remarkably improved clinical outcomes in patients with both early-stage (i.e. increased tumor cure rates and overall survival) and advanced (i.e. survival prolongation) HER2+ BC.3–7 Trastuzumab, a humanized monoclonal antibody that binds HER2 extracellular domain, inhibits the oncogenic signaling downstream of HER2 by preventing HER2 dimerization and phosphorylation. 8 In early-stage HER2+ BC, combining trastuzumab with (neo)adjuvant anthracycline/taxane-based chemotherapy remarkably improved patient relapse-free survival (RFS) and overall survival (OS),4,9 and trastuzumab-based biochemotherapy combinations have become standard systemic treatments in this setting. 10
Overweight and obesity, as defined as a body mass index (BMI) ⩾25 kg/m2 and ⩾30 kg/m2, respectively, are established risk factors for BC development. 11 In addition, increasing evidence points to an association between high adiposity and worse clinical outcomes in BC patients, including an increased rate of tumor recurrences and lower patient survival.12–15 A large, prospective cohort study recently examined the impact of BMI on the outcome of patients with early-stage BC on the basis of the PAM50 gene expression subtype, showing that class II/III obesity at diagnosis (i.e. BMI ⩾35 kg/m2) is associated with significantly higher risk of disease recurrence and death in women with luminal A disease, but not in patients with luminal B, basal-like or HER2-enriched BC subtypes. 16 Consistent with these data, other studies showed an association between high BMI and worse prognosis in patients with hormone receptor positive (HR+) HER2-negative (HER2−) BC.14,17
By contrast, the impact of BMI on the prognosis of HER2+ BC patients remains controversial. A large retrospective analysis showed an increased risk of distant metastases and death in obese versus normal/underweight HER2+ BC patients, and in particular in the subgroup of HR−/HER2+ patients, but not in patients with HR+/HER2+ disease. 18 However, this study included patients diagnosed with HER2+ BC in the pre-trastuzumab era, who did not receive adjuvant trastuzumab therapy. Therefore, findings of this study more likely reflect the impact of overweight/obesity on the natural history of surgically resected HER2+ BC, rather than the effect of BMI on the efficacy of trastuzumab-based (neo)adjuvant biochemotherapy. Post-hoc analyses of prospective trials, which evaluated the prognostic impact of BMI in the trastuzumab era, showed conflicting results. On the one hand, the NSABP B-31 study 19 and, more recently, the HERA trial 20 failed to find a significant association between baseline BMI categories and patient RFS or OS. On the other hand, an analysis of the three-arms, randomized, phase III trial N9831 found that overweight/obese patients have significantly lower disease-free survival when compared with patients with normal weight, with a statistically significant difference only in patients who did not receive trastuzumab (control arm) and in patients treated with adjuvant trastuzumab after the completion of taxane-based chemotherapy, but not in patients receiving concomitant paclitaxel–trastuzumab, which represents the standard-of-care (with or without anthracyclines) adjuvant therapy for surgically resected HER2+ BC. 21
Obesity is often part of a more complex clinical scenario, known as metabolic syndrome, in which high BMI and dyslipidemia coexist with glucose intolerance/diabetes and systemic inflammation. Since diabetes, which is part of the metabolic syndrome spectrum, has been also associated with increased BC risk and worse BC patient prognosis, 22 clarifying the specific contribution of high BMI and hyperglycemia on patient outcomes in this clinical setting is a hard goal. Interestingly, a recent sub-analysis of the ALTTO study revealed a negative impact of diabetes on the clinical outcomes of early-stage HER2+ BC patients treated with single (trastuzumab, lapatinib) or dual (trastuzumab plus lapatinib) anti-HER2 therapies in combination with preoperative chemotherapy. 23
The aim of the present study was to investigate the prognostic role of high BMI and of hyperglycemia, in a consecutive series of early-stage HER2+ BC patients treated with trastuzumab-containing adjuvant biochemotherapy at our Institution. We also evaluated the impact of BMI and glycemia on the clinical outcomes of patients with HR−/HER2+ BC and HR+/HER2+ BC, as well as the potential impact of the interaction between BMI and glycemia on patient RFS.
Materials and methods
Study population
This was a monocentric, retrospective study that included consecutive patients diagnosed with stage I–III HER2+ BC and treated with surgery followed by adjuvant trastuzumab-containing biochemotherapy at Fondazione IRCCS Istituto Nazionale dei Tumori (Milan, Italy) between January 2008 and September 2018. Patients were considered eligible if they fulfilled all the following criteria: (1) women with pathologically/cytologically confirmed diagnosis of HER2+ BC, as defined by an immunohistochemistry (IHC) score for HER2 of 3+ or by in situ hybridization (ISH) indicative of HER2 gene amplification in patients with an equivocal (2+) HER2 score; 24 (2) age ⩾18 years; (3) treatment with adjuvant trastuzumab for at least three, consecutive, every-21-day cycles (9 weeks); (4) available information about baseline (i.e. within 30 days from the initiation of adjuvant treatment) weight and height; (5) available data regarding patient outcomes, including RFS, distant metastasis-free survival (DMFS) and OS.
Baseline glycemia was evaluated as part of routine blood chemistry analyses performed within 30 days before the initiation of adjuvant treatment, that is, between surgery and the initiation of adjuvant therapy. Since breast surgery is a low-morbidity surgery 25 and it is not followed by significant weight loss, 26 the time window in which we measured BMI and glycemia in patients included in this study can be reliably considered representative of these values as evaluated at surgery and/or at treatment initiation.
Patient blood samples were collected in the morning after at least 8 h of overnight fasting. All patients were followed-up until death, loss of contact, or time of data lock (1 March, 2020). HR status was assessed by IHC. HR+ BC was defined as equal to or higher than 1% of tumor cells staining positive for estrogen receptor (ER) and/or progesterone receptor (PgR). 27
The study protocol was approved by the Local Ethics Committee of Fondazione IRCCS Istituto Nazionale dei Tumori (INT 170/20). Patient data were collected according to the ethical principles for medical research involving human subjects adopted in the Declaration of Helsinki. Patients alive at the time of data collection and/or analysis signed an informed consent form for the use of their personal data for research purposes. The Ethics Committee also authorized the collection and analysis of data from patients who were not alive when the study was conducted; in the latter case, no additional consent forms were required for the analysis of these data, except for a general consent form that all patients sign when they start a treatment at our Institution, in which they authorize the use of data collected as per clinical practice for research purposes.
Statistical analysis
RFS was defined as the time between surgery and tumor recurrence or patient death from any cause, whichever occurred first. DMFS was defined as the time between surgery and the first occurrence of tumor relapse in one or more distant sites (i.e. outside the breast and draining lymph nodes). OS was defined as the time between surgery and patient death from any cause.
Descriptive statistics were used to analyze and report patients’ characteristics. The distribution of categorical or continuous variables (patient- or tumor-related) in the two BMI groups was evaluated through the Chi-squared or Wilcoxon test, respectively.
Patient BMI was calculated for all patients using the following formula: BMI = weight (kg)/height 2 (m 2 ). Maximally selected rank statistics were used to estimate the best BMI and glycemia cut-points to divide patient population in two subgroups according to the risk of recurrence. 28
Survival curves were represented through the Kaplan–Meier method, and survival distributions were compared with the log rank test. Median follow-up time was calculated using the reverse Kaplan–Meier method. Patients who had not undergone disease recurrence or death at the time of data cut-off and analysis were censored at the last date of follow-up.
The impact of different prognostic factors on RFS and OS was first assessed at univariate analysis. Factors showing a statistically significant association with the risk of disease recurrence or patient death (p < 0.1) were included in a multivariable Cox proportional hazard model to assess their independent association with survival.
A p-value of 0.05 was set as the significance threshold for all statistical analyses, with the exception of univariate survival analyses, for which we used a threshold of 0.1 with the aim of reducing the chances to miss potentially relevant variables to be subsequently evaluated at multivariable analysis. R software (Version 3.5.3) and RStudio software (Version 1.2.5033) were used for statistical analyses.
Results
Patient characteristics
Patient and tumor characteristics are summarized in Table 1. Of 505 patients included in this study, 312 (61.8%) were underweight or normal weight, 122 (24.2%) were overweight and 71 (14%) were obese. Median baseline glycemia was 93 mg/dl (range: 68–176 mg/dl). Regarding tumor characteristics, the majority of patients had HR+/HER2+ BC (n = 330; 65.3%), absence of lymph node involvement (n = 304; 60.2%), tumors of ductal histotype (n = 441; 87.7%), histological grade 3 (n = 341; 68.9%) and high (⩾20%) Ki-67 expression (n = 400; 90.7%).
Patient characteristics.

Data are presented as n (%) except where otherwise noted.
Defined positive if >1% of tumor cells express hormone receptors.
Missing data for 64 patients.
Missing data for 10 patients.
Missing data for 73 patients.
Missing data for 87 patients.
Missing data for 263 patients.
A, anthracycline-based CT; A+T, anthracycline–taxane combination CI, confidence interval; CT; BMI, body mass index; CT, chemotherapy; ER, estrogen receptors; HR, hormone receptors; IHC, immunohistochemistry; PgR, progesterone receptors; PVI, peritumoral vascular invasion; T, taxane-based CT.
All patients received adjuvant trastuzumab in combination with standard cytotoxic chemotherapy, with a median number of 18 triweekly trastuzumab cycles administered (range: 4–19). Adjuvant chemotherapy consisted of concomitant or sequential anthracycline-taxane (71.7%), taxane-alone (16.4%) and anthracycline-alone (10.3%) chemotherapy (Supplemental Table 1).
Impact of baseline BMI and glycemia on clinical outcomes
With a median follow-up of 76.7 (72.4–81.8) months, 55 tumor relapse events (10.8%) occurred, including 48 distant recurrences and seven local recurrences. Overall, 35 death events occurred. Median RFS and OS were not reached at the time of data lock and analysis. Positive lymph node status was associated with significantly worse RFS and OS, while there was trend toward an association between HR-negative biology, or high tumor grade (G3), and worse patient RFS, but not with worse OS (Supplemental Figure 1). Finally, clinical tumor stage was not associated with RFS or OS (Supplemental Figure 1).
The maximally selected rank statistics method identified a value of 27.77 kg/m2 as the cut-off level of BMI associated with the most significant differences between RFS Kaplan–Meier curves. Based on this cut-off, 390 (77.2%) patients were classified as low-BMI patients (<27.77 kg/m2), while 115 (22.8%) patients were classified as high-BMI (⩾27.77 kg/m2) patients. We found no significant differences, in terms of HR status, size of primary tumor, lymph node involvement, Ki-67 score, tumor grade or perivascular invasion, between patients with high versus low BMI (Supplemental Table 2). Conversely, high-BMI patients were significantly older than low-BMI patients, and were more likely to have higher baseline blood glucose and triglyceride concentration (Supplemental Table 2).
Patients with high BMI had significantly worse RFS (p = 0.024, log rank test) and a trend toward worse OS (p = 0.081, log rank test) when compared with low-BMI patients (Figure 1a, b). In high-BMI versus low-BMI patients, 5-year RFS rate was 84.6% [95% confidence interval (CI): 78.0–91.9%] versus 90.1% (95% CI: 86.9–93.3%) (Figure 1a), while 5-year OS rate was 90.4% (95% CI: 84.6–96.6%) versus 96.0% (95% CI: 93.9–98.2%), respectively (Figure 1b). As for distant tumor recurrences, 5-year DMFS trended to be worse in patients with high BMI when compared with patients with low BMI (85.7%, 95% CI: 79.2–92.7% versus 90.9%, 95% CI: 87.8–94.1%, respectively; p = 0.056) (Supplemental Figure 2).

Relapse-free survival and overall survival according to BMI and glycemia categories. Kaplan–Meier curves of (a) RFS and (b) OS in the whole population by BMI category. Kaplan–Meier curves of (c) RFS and (d) OS in the whole population by glycemia category.
Then, we investigated the potential impact of baseline glycemia, as evaluated as a dichotomous variable, on patient outcomes. The maximally selected rank statistics method identified 109 mg/dl as the blood glucose threshold associated with the most significant separation of RFS Kaplan–Meier curves. With this threshold, patients with higher baseline glycemia (⩾109 mg/dl) had significantly worse RFS when compared with patients with low (<109 mg/dl) baseline glycemia (5-year RFS rate: 80.3% and 93.6%, respectively; 95% CI: 68.9–93.6% and 91.0–96.3%, respectively; p = 0.015) (Figure 1c). On the other hand, we found no statistically significant association between patient glycemia and OS (p = 0.2) (Figure 1d).
Factors independently associated with patient prognosis
Factors associated with an increased risk of BC recurrence at univariate analysis were: high BMI (hazard ratio 1.88; 95% CI: 1.08–3.27, p = 0.027), high baseline glycemia (hazard ratio 2.61; 95% CI 1.17–5.81, p = 0.019), lymph node involvement (hazard ratio 3.85; 95% CI: 2.15–6.89, p < 0.001) and high tumor grade (hazard ratio 1.79; 95% CI: 0.92–3.46, p = 0.086), while positive HR status (i.e. ER/PgR expression) correlated with better RFS (hazard ratio 0.62; 95% CI: 0.37–1.06, p = 0.078) (Table 2, upper part). Multivariable analysis confirmed an independent association between high BMI and an increased risk of tumor recurrence (hazard ratio 2.26; 95% CI: 1.08–4.74, p = 0.031); positive lymph node status was also independently associated with worse RFS (hazard ratio 2.29; 95% CI: 1.12–4.72, p = 0.024). On the other hand, baseline glycemia did not show an independent association with the risk of disease relapse (hazard ratio 1.58; 95% CI: 0.66–3.81, p = 0.308) (Table 2, lower part).
Univariate and multivariable Cox proportional hazards model for RFS.

The p-value is indicated in bold numbers when statistically significant.
Defined positive if >1% of tumor cells express hormone receptors.
Low BMI: <27.77 kg/m2; High BMI ⩾27.77 kg/m2. Low glycemia: <109 mg/dl; High glycemia: ⩾109 mg/dl.
A+T, anthracycline–taxane combination CT; BMI, body mass index; CI, confidence interval; CT, chemotherapy; HR hormone receptors; IHC, immunohistochemistry; PVI, peritumoral vascular invasion.
Since baseline BMI and glycemia are not independently distributed (Supplemental Table 2), and since we cannot exclude that the impact of each of these variables on RFS is at least in part mediated by the prognostic effect of the other variable, we investigated if the interaction between BMI and glycemia could at least account for the observed association of each of these variables with the risk of relapse. To this aim, we fitted another multivariable Cox model, in which we also included a covariate accounting for the interaction between baseline BMI and glycemia. This model confirmed an independent association between high BMI and worse RFS, while glycemia, or the interaction between BMI and glycemia, were not associated with the risk of disease relapse (Supplemental Table 3). Together, these data confirm results of our previous analyses, while at the same time indicating that the impact of BMI on RFS is not affected by baseline glycemia.
The following factors were associated with higher risk of death at univariate analysis: high BMI (hazard ratio 1.85; 95% CI: 0.92–3.71, p = 0.085), lymph node involvement (hazard ratio 5.67; 95% CI: 2.47–12.98, p < 0.001) and the presence of peritumoral vascular invasion (hazard ratio 2.60; 95% CI: 1.20–5.65, p = 0.016) (Table 3, upper part). Multivariable analysis adjusting for these covariates confirmed an independent association between high BMI and worse OS (hazard ratio 2.25, 95% CI 1.03–4.94, p = 0.043) (Table 3, lower part).
Univariate and multivariable Cox proportional hazards model for OS.
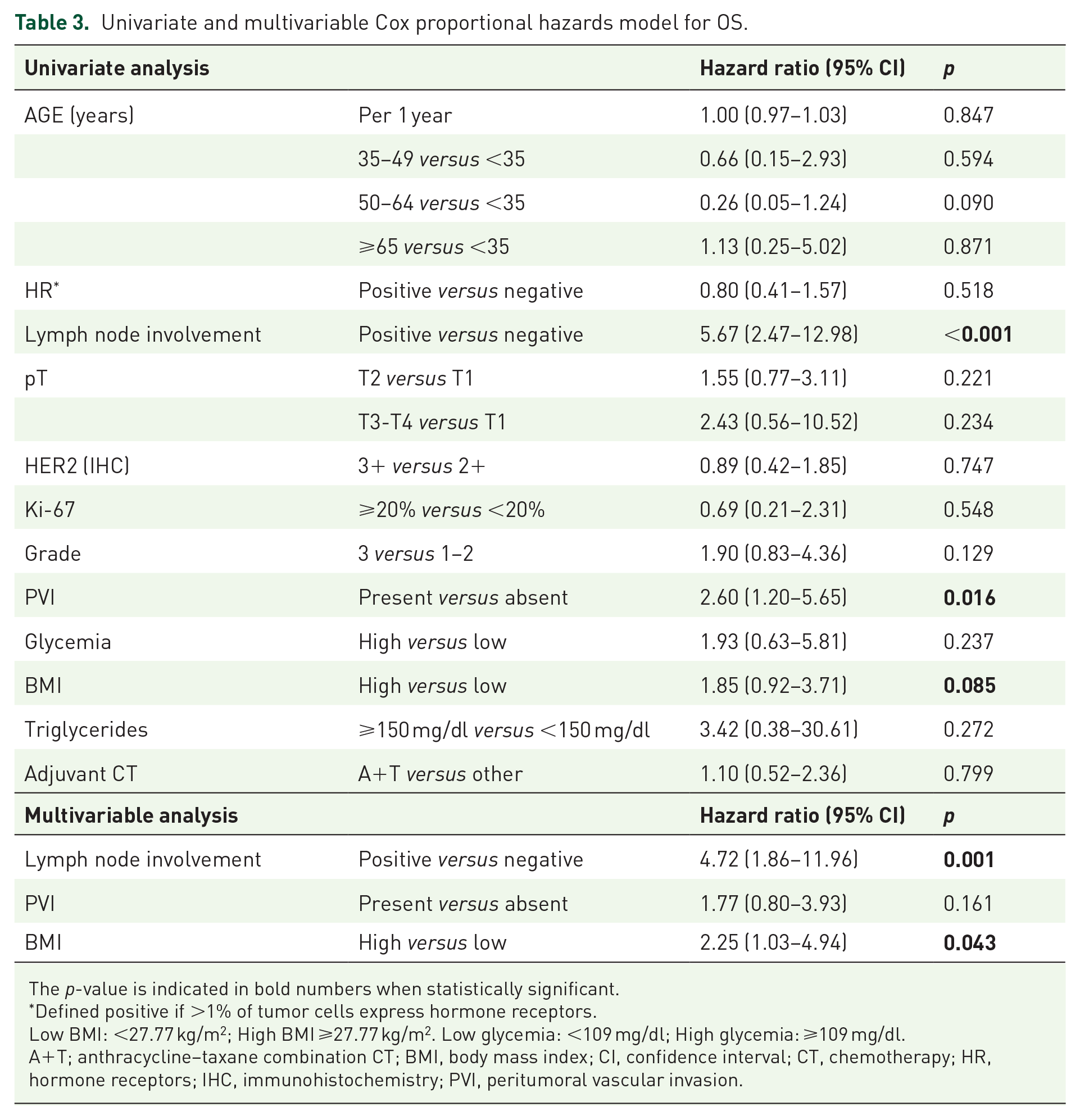
The p-value is indicated in bold numbers when statistically significant.
Defined positive if >1% of tumor cells express hormone receptors.
Low BMI: <27.77 kg/m2; High BMI ⩾27.77 kg/m2. Low glycemia: <109 mg/dl; High glycemia: ⩾109 mg/dl.
A+T; anthracycline–taxane combination CT; BMI, body mass index; CI, confidence interval; CT, chemotherapy; HR, hormone receptors; IHC, immunohistochemistry; PVI, peritumoral vascular invasion.
Impact of BMI and glycemia on patient RFS according to hormone receptor status
To investigate the prognostic impact of BMI and glycemia according to HR status, we separately evaluated the association between BMI and the risk of tumor relapse among patients with HR-/HER2+ or HR+/HER2+ BC. During the study follow-up, 25 recurrence events occurred among 175 patients with HR−/HER2+ BC, while 30 recurrence events were observed among 330 patients with HR+/HER2+ BC.
In the HR−/HER2+ BC cohort, high BMI (⩾27.77 kg/m2) and positive lymph node status were associated with significantly higher risk of disease recurrence (hazard ratio 2.52; 95% CI: 1.14–5.55, p = 0.022 and hazard ratio 3.45; 95% CI: 1.49–8.00, p = 0.004, respectively), and these data were confirmed by multivariable analysis (hazard ratio 2.29; 95% CI: 1.01–5.20, p = 0.047 and hazard ratio 3.32; 95% CI: 1.42–7.78, p = 0.006, respectively) (Figure 2; Table 4). In these patients, hyperglycemia (⩾109 mg/dl) was not independently associated with patient RFS (Figure 2; Table 4). By contrast, in the HR+/HER2+ BC cohort we found no statistically significant association between BMI and RFS (hazard ratio 1.36; 95% CI: 0.61–3.07, p = 0.452), while hyperglycemia was associated with worse RFS at univariate analysis (hazard ratio 2.85; 95% CI: 1.02–8.00, p = 0.047), with a clear trend toward worse RFS also at multivariable analysis (hazard ratio 2.52; 95% CI: 0.89–7.1; p = 0.080) (Figure 2; Table 4).
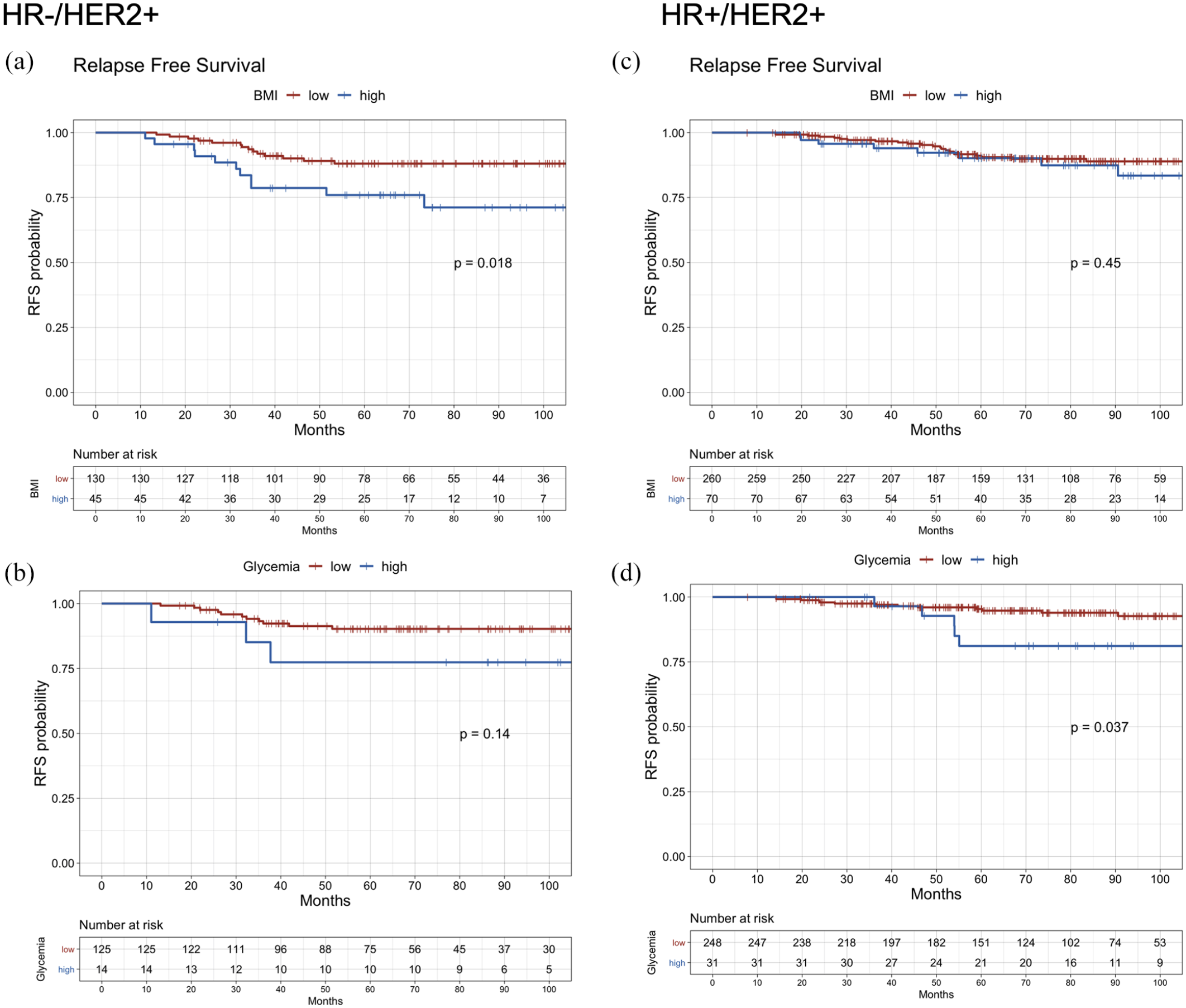
RFS in different hormone receptor subgroups according to BMI and glycemia categories. Kaplan–Meier curves of RFS in HR− patients by (a) BMI and (b) glycemia categories. Kaplan–Meier curves of RFS in HR+ patients by (c) BMI and (d) glycemia categories.
Univariate and multivariable Cox proportional hazards model for RFS on the basis of HR status (negative, positive).

The p-value is indicated in bold numbers when statistically significant.
Defined positive if >1% of tumor cells express hormone receptors.
Low BMI: <27.77 kg/m2; High BMI: ⩾27.77 kg/m2. Low glycemia: <109 mg/dl; High glycemia: ⩾109 mg/dl.
A+T, anthracycline–taxane combination CT; BMI, body mass index; CI, confidence interval; CT, chemotherapy; HR, hormone receptors; IHC, immunohistochemistry; PVI, peritumoral vascular invasion.
Discussion
Overweight and obesity are established risk factors for BC development, and have been associated with worse prognosis in patients with surgically resected BC.11,15 However, there is conflicting evidence regarding the prognostic impact of BMI in patients with limited-stage HER2+ BC.
Here we showed that high BMI is an independent predictor of worse RFS and OS in the whole population of patients with surgically resected, stage I–III HER2+ BC receiving adjuvant trastuzumab-based biochemotherapy, as well as in the subgroup of HR−/HER2+ BC patients, but not in patients with HR+/HER2+ BC. We also found that the impact of high BMI on patient outcome is not affected by baseline glycemia, and that hyperglycemia could be a predictor of worse RFS specifically in the subgroup of HR+/HER2+ BC patients.
Previous studies showed a statistically significant and consistent association between high BMI and worse clinical outcomes in patients with early-stage luminal-type (HR+/HER2−) BC.14,16,17,29 These findings are coherent with the fact that overweight/obese patients typically have higher average blood insulin and IGF-1 levels, which can result in an enhanced activation of the IR-IGFR1/PI3K/AKT/mTORC1 axis in cancer cells, thus potentially bypassing ER inhibition and causing primary/acquired HR+ BC resistance to endocrine therapies.30,31 Although the PI3K/AKT/mTORC1 pathway also contributes to the growth, proliferation and survival of HER2+ BC cells, thus establishing a potential connection between body weight, systemic metabolism and patient outcomes, 32 the association between BMI and clinical outcomes in HER2+ BC patients remains controversial. For instance, a post-hoc analysis of the HERA and NSABP B-31 trials failed to reveal a significant association between overweight/obesity and the risk of tumor relapse in surgically resected HER2+ BC patients treated with adjuvant trastuzumab-containing biochemotherapy. On the other hand, some retrospective analyses suggested that overweight and obesity could be associated with worse clinical outcomes in HER2+ BC patients.18,21 However, these studies were limited by several factors, including the fact that patients did not receive adjuvant trastuzumab, 18 or that trastuzumab was not administered concomitantly with taxane chemotherapy, 21 which actually represents the standard-of-care treatment in these patients. When compared with results of previous studies, the findings of our analysis, which included quite a homogeneous cohort of early-stage HER2+ BC patients treated with standard-of-care, adjuvant trastuzumab-containing biochemotherapy, are more representative of the current clinical practice, and indicate that baseline BMI can affect the rate of HER2+ BC relapses and the prognosis of HER2+ BC patients also when HER2—the oncogenic driver of most of these neoplasms—is pharmacologically inhibited.
Excess weight is often associated with insulin resistance, which results from systemic metabolic adaptation to an increased blood concentration of free fatty acids (FFAs) and other endocrine factors released by the adipose tissue. 33 In turn, insulin resistance causes hyperglycemia/diabetes and compensatory hyperinsulinemia. 33 An increased extracellular concentration of glucose can directly fuel tumor bioenergetics and anabolism by increasing intracellular levels of ATP and amino acid/FA precursors; in parallel, hyperglycemia can cause compensatory hyperinsulinemia, which, in turn, can result in aberrant activation of the insulin receptor (IR)-PI3K-AKT-mTORC1 pathway, which activates anabolic processes, such as protein synthesis and lipogenesis, and stimulates HER2+ BC cell growth and proliferation. 34 In addition, insulin stimulates IGF-1 biosynthesis in the liver, thus contributing to activate the PI3K-AKT-mTORC1 pathway in cancer cells. 35 By activating the PI3K-AKT-mTORC1 axis, an increased concentration of blood insulin and IGF-1 can finally cause HER2+ BC cell resistance to anti-HER2 therapies, as convincingly indicated by preclinical studies. 36
To test whether the impact of high BMI on clinical outcomes is at least in part mediated by overweight-associated hyperglycemia (and, potentially, of hyperinsulinemia), we investigated the association between hyperglycemia and RFS. Although high baseline blood glucose levels were associated with higher risk of disease recurrence, multivariable analysis did not confirm this association. We also failed to find a significant interaction between BMI and hyperglycemia in affecting patient RFS, thus excluding that the impact of BMI on clinical outcomes is influenced by baseline glycemic status. Together, these data indicate that the impact of BMI on the risk of tumor relapse in early-stage HER2+ BC patients is independent of baseline blood glucose concentration. In addition, the observation that high BMI is associated with significantly worse RFS and OS only in the subgroup of HR−/HER2+ BC patients, together with the observed trend toward an association between hyperglycemia and worse RFS among HR+/HER2+ BC patients, makes us hypothesize that the impact of BMI and glycemia on clinical outcomes in HER2+ BC patients could be affected by HR status (i.e. HR+ versus HR−).
In HR+/HER2+ BC, higher extracellular glucose and insulin concentration can activate the PI3K-AKT-mTORC1 pathway regardless of pharmacological HER2 inhibition, thus increasing the risk of disease relapse after curative surgery. This hypothesis is in line with results of a post-hoc analysis of the adjuvant ALTTO trial, in which diabetes was associated with significantly higher risk of disease relapse only in patients with surgically resected HR+/HER2+ BC, but not in patients with HR−/HER2+ BC; 23 in the same study, the use of metformin, which lowers blood glucose, insulin and IGF-1 concentration, was associated with better clinical outcomes only in diabetic patients belonging to the HR+/HER2+ BC subgroup. On the other hand, in HR−/HER2+ BC, in which HER2 signaling in not affected by ER signaling, 37 the IGFR1/PI3K/AKT/mTORC1 pathway could be intrinsically more active (as a result of HER2-mediated oncogenic signaling), and consequently less susceptible of being affected by environmental factors, such as glucose- and insulin-induced activation of growth factor-induced proliferation pathways. In this BC subtype other metabolic factors, such as fatty acids (FAs), could play a major role in stimulating tumor cell growth and proliferation, and in inducing resistance to anti-HER2 therapies.
Preclinical studies conducted in recent years have revealed that HER2+ BC is characterized by the activation of an epigenetic program leading to the upregulation of the de novo FA biosynthesis, the so-called “lipogenic phenotype.”38,39 Fatty acid synthase (FASN) is a key lipogenic enzyme that catalyzes the final step of FA biosynthesis from acetyl-CoA units. Of note, FASN is overexpressed in more than 85% of HER2+ BCs, and its expression and activity are positively regulated at both transcriptional and post-translational levels by HER2 and its downstream signaling. 39 Therefore, trastuzumab-induced inhibition of HER2 also results in reduced FASN activity and in the downmodulation of FA biosynthesis, thus making trastuzumab-treated HER2+ BC cells potentially dependent on FA uptake from the extracellular environment. 40 Most blood lipids are transported in the form of triglycerides or phospholipids within plasma lipoproteins. Therefore, FA uptake by cancer cells requires the sequential activity of lipoprotein lipase (LPL), which hydrolyzes extracellular triglycerides into individual FAs, and the subsequent uptake of FAs through CD36, a FA transporter that is located in cells’ plasma membrane. Previous reports showed a negative prognostic impact of LPL or CD36 overexpression in BC patients, thus suggesting a more aggressive behavior of tumors with enhanced activation of FA uptake from the extracellular environment.41,42 More recently, CD36 overexpression was implicated in acquired resistance to lapatinib in HER2+ BC cell lines, and high baseline LPL and CD36 intratumor expression was associated with statistically significantly worse prognosis in HER2+ BC patients treated with anti-HER2 neoadjuvant therapies. 43 Mechanistically, an increased concentration of plasma lipids (including triglycerides and FFAs), which is frequently observed in overweight/obese patients, could fuel HER2+ BC metabolism, thus contributing to HER2+ BC resistance to anti-HER2 therapies. In our study, patients with high BMI had significantly higher blood triglycerides at baseline, as expected. However, due to the low number of patients for whom baseline triglyceride measurement was available, we were unable to reliably evaluate the association between baseline triglycerides and patient RFS or OS in the whole patient population and, in particular, in the subset of HR−/HER2+ BC patients. We are now initiating a prospective study to specifically investigate the impact of baseline and on-treatment peripheral blood FA and lipid profiles on the efficacy of preoperative chemotherapy-trastuzumab therapy in HER2+ BC patients.
If validated by future studies, our results could have clinically relevant implications, as BMI and glycemia can be potentially modified through the use of specific dietary or pharmacological interventions. In particular, low-calorie or low-lipid diets, or pharmacological treatments capable of reducing blood triglyceride levels, such as statins or metformin,44,45 could improve the efficacy of anti-HER2 therapies in patients with HR−/HER2+ BC, while interventions aimed at reducing blood glucose levels, such as fasting-mimicking diets or metformin,46,47 could synergize with anti-HER2 therapies in HR+/HER2+ BC patients.
Strengths of this study consist in: (1) its monocentric design, which guarantees homogeneous patient treatment and follow-up; (2) the high number of patients included; (3) the long follow-up; (4) a well-balanced distribution of clinically relevant patient- and tumor-related variables in high-BMI and low-BMI patient subgroups (Supplemental Table 2). Finally, we analyzed a population of cancer patients homogeneously treated with adjuvant trastuzumab-based biochemotherapy, thus reflecting the clinical practice of adjuvant treatment of HER2+ BC patients. The following are limitations of this study: (1) the retrospective nature; (2) the absence of a pre-specified statistical power calculation; (3) the lack of available data on lipid profiles from many patients, which did not allow us to test the hypothesis that the levels of blood lipids can at least in part explain the association between high patient BMI and worse clinical outcomes.
Conclusions
In conclusion, this is the first study to show that high BMI is associated with worse RFS and OS in HER2+ BC patients treated with trastuzumab-containing adjuvant biochemotherapy according to clinical practice. This is also the first study to show a potentially different impact of high BMI and hyperglycemia in patients with HR−/HER2+ BC and HR+/HER2+ BC, respectively. Further studies are needed to confirm our findings, as well as to investigate potential molecular mechanisms at the basis of these results.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359211006960 – Supplemental material for Hormone receptor status influences the impact of body mass index and hyperglycemia on the risk of tumor relapse in early-stage HER2-positive breast cancer patients
Supplemental material, sj-pdf-1-tam-10.1177_17588359211006960 for Hormone receptor status influences the impact of body mass index and hyperglycemia on the risk of tumor relapse in early-stage HER2-positive breast cancer patients by Francesca Ligorio, Luca Zambelli, Achille Bottiglieri, Lorenzo Castagnoli, Emma Zattarin, Riccardo Lobefaro, Arianna Ottini, Andrea Vingiani, Serenella M. Pupa, Giulia V. Bianchi, Giuseppe Capri, Giancarlo Pruneri, Filippo de Braud and Claudio Vernieri in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We would like to thank the “Associazione Italiana per la Ricerca sul Cancro” (AIRC) (MFAG 2019. Id. 22977. PI: Claudio Vernieri) and the Scientific Directorate of Fondazione IRCCS Istituto Nazionale dei Tumori for supporting our research.
Author contributions
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
