Abstract
Background:
Lactate dehydrogenase (LDH) is a known prognostic biomarker for the endemic variant of nasopharyngeal carcinoma (NPC). Here, we investigate whether serial changes in LDH level between chemotherapy (CT) cycles are associated with tumour response to CT.
Methods:
Patients with biopsy-proven, recurrent or treatment-naïve metastatic NPC (mNPC) were recruited. All patients had received at least two cycles of platinum-based doublet or triplet CT, with serial assessment of LDH prior to every cycle of chemotherapy (CT1–6). Patients harbouring conditions that affect LDH levels (IU/L) were excluded. Tumour response was assessed after every two cycles of CT by RECIST v1.1.
Results:
A total of 158 patients were analysed, including 77 with recurrent and 81 with treatment-naïve mNPC. High pre-CT LDH was associated with an inferior overall survival [hazard ratio 1.93 for ⩾240 versus <240 (1.34–2.77), p < 0.001], which is consistent with published literature. We found that both absolute LDH levels and LDH ratios (LDHCTn: LDHCTn–1) were associated with tumour response [partial response versus progressive disease: median value across CT1–6 = 168–190 versus 222–398 (absolute); 0.738–0.988 versus 1.039–1.406 (ratio)], albeit LDH ratio had a tighter variance between patients. Finally, we showed that an LDH ratio cut-off of 1.0 at CT1, CT3 and CT5 was predictive of progressive disease at CT2, CT4, CT6 [area under the curve of 0.73 (0.65–0.80)].
Conclusion:
Herein, we characterised the longitudinal variation of LDH in response to CT in mNPC. Our findings suggest the potential utility of interval LDH ratio to predict subsequent tumour response to CT.
Introduction
Nasopharyngeal carcinoma (NPC) is a distinct entity of head and neck cancer that is invariably associated with Epstein–Barr virus (EBV) infection in the endemic parts of the world. While it is exquisitely sensitive to platinum-based chemotherapy (CT) and radiotherapy (RT), NPC still has a propensity for distant metastatic relapses, which occur in approximately 20% of patients treated with intensity-modulated RT (IMRT).1,2 For patients with treatment-naïve NPC, synchronous distant metastasis is also detected in 6–8% of patients.3,4 In these patients with systemic disease, platinum-based CT is the standard of care, with gemcitabine–cisplatin (GP) doublet regimen being the first-line treatment of choice, following the randomised controlled trial by Zhang and colleagues showing the superior efficacy over doublet cisplatin and 5-fluorouracil. 5 Despite the high initial response rates of GP, median progression-free survival (PFS) of recurrent metastatic NPC (rmNPC) patients following GP is only about 7.0 months, and more than half of these patients often do not complete a full course of six cycles of GP.
Conventionally, patients on palliative CT are monitored every two to three cycles with interval imaging. Response to treatment is evaluated using RECIST (Response Evaluation Criteria in Solid Tumours), 6 and depending on the radiological/clinical response, patients are either continued on the existing therapy or switched to second-line regimens. Nonetheless, gross responses on imaging and clinical examination are often not an accurate indication of the tumour clonal sensitivity to the CT agents. Moreover, mixed responses to CT between different lesions from the same patient are frequently elicited, which would support the concept of clonal heterogeneity of response to CT.6,7 Thus, there is a clinical need for more sensitive biomarkers to detect early responses to CT, which would in turn offer the opportunity for the treating physician to adapt treatment intensity and switch drugs, accordingly.
In this regard, plasma EBV DNA is an established biomarker for screening, risk stratification and tracking treatment response.8–11 Technically, this test is based on a polymerase chain reaction of the BAMH-I sequence of the EBV genome, and it has been shown that clinical implementation of the EBV DNA assay requires robust testing quality assurance to minimise inter-laboratory variation. 12 It is for this reason that EBV DNA testing remains localised to tertiary institutions, but it is not available to several other high-volume centres in the low- and middle-income countries (LMICs). On this note, lactate dehydrogenase (LDH), an enzyme in the glycolytic pathway that converts pyruvate to lactate during hypoxia, has previously been reported to be a biochemical parameter that is significantly associated with oncogenesis, proliferation and metastasis for several human cancers.13–15 In NPC, baseline LDH has been demonstrated to be prognostic for tumour recurrence and survival; Pan and colleagues have previously constructed a nomogram that incorporates American Joint Committee on Cancer/Union for International Cancer Control eighth edition TNM stage classification and pretreatment LDH to predict overall survival (OS) in locoregionally advanced NPC. 16 However, there is little information on the utility of LDH as a biochemical parameter for tracking on-treatment response in NPC and other tumour types. For instance, real-time monitoring of plasma EBV DNA had been shown to be correlated to induction chemotherapy and chemoradiotherapy responses. 7 More recently, Criscitiello et al. showed that pretreatment LDH levels were linked to immunotherapy response in a variety of tumour types, but still, they did not indicate if responses were correlated to serial changes of the LDH levels. 17
Here, we therefore investigate the utility of serum LDH as a biomarker to evaluate tumour response. In addition, we characterised the serial fluctuation of LDH with CT cycles, and determined whether this was correlated to tumour response. Finally, we probed whether early trends of this marker predict subsequent CT response.
Materials and methods
Study criteria and cohort
We enrolled patients who were diagnosed with biopsy-proven NPC, and have metastatic disease either at presentation or at the point of recurrence following definitive RT. All patients were treated at the Guangxi Medical University Cancer Hospital (GXMUCH). Inclusion criteria were: (1) presence of distant metastasis ascertained by either computed tomography of the thorax, abdomen, and pelvis, and skeletal scintigraphy, or 18F-fluorodeoxyglucose positron emission tomography–computed tomography scanning; (2) having undergone CT as first-line treatment with or without palliative RT; (3) having at least three serial serum LDH measurements (including pretreatment); and (4) undergoing imaging to assess tumour response following every two cycles of CT. Exclusion criteria were: (1) patients who did not receive CT as first-line treatment or received only one cycle of CT; (2) having an abnormal liver function test; (3) occurrence of any infective episodes during CT; and (4) no measurable disease for assessment of tumour response by RECIST, such as bone metastasis with no measurable soft tumour component or evaluation of metastatic lesions (e.g. liver or nonregional lymph node metastases) by ultrasound only. Figure 1 illustrates the patient selection process for this study.

Patient inclusion and exclusion criteria.
This retrospective research was approved by the Ethics Committee of the GXMUCH (AB18221007), and performed in accordance with the Declaration of Helsinki and Good Clinical Practice Guidelines. Informed consent was obtained from all living patients and next of kin of deceased patients. All patient records were anonymised before analysis.
CT regimens
CT regimens included doublet cisplatin [75 mg/m2 on day 1 (D1)] or nedaplatin (80–100 mg/m2 on D1) 18 with either docetaxel (75 mg/m2 on D1), paclitaxel (135 mg/m2 on D1), 5-fluorouracil (500 mg/m2/d as a continuous infusion from D1–5) or gemcitabine (1000 mg/m2 on D1 and D8).5,19,20 The triplet CT combination comprised of cisplatin (60 mg/m2 on D1), docetaxel (60 mg/m2 on D1) and 5-fluorouracil (600 mg/m2/d on D1–5). 19 These regimens were repeated every 3–4 weeks. Additional details on the institution dose adjustment protocol are elaborated in the Supplemental Methods.
RT treatment protocol
Patients with palliative RT were permitted on this study. For patients with rmNPC, RT was delivered to metastatic lesions for symptom control or when indicated during acute emergencies for example, bleeding or cord compression. Dose prescriptions were typically 30 Gy in 10 fractions (fr) or 20 Gy in 5 fr. For patients with treatment-naïve mNPC, high-dose RT to the primary nasopharynx and neck could be recommended in a subset of patients at the discretion of the treating physician. The prescribed doses were 66–72 Gy/30–32 fr to the nasopharynx gross tumour volume, 66–70 Gy/30–32 fr to the lymph node gross tumour volume, 60–64 Gy/30–32 fr to the high-risk clinical target volume (CTV1), 54–58 Gy/30–32 fr to the low-risk CTV (CTV2). A detailed RT-planning protocol is summarised in the Supplemental Methods.
Tumour response assessment
Appropriate radiological imaging, including computed tomography or magnetic resonance imaging, were performed every two cycles of CT to monitor objective tumour response. Complete response (CR), partial response (PR), stable disease (SD) or progressive disease (PD) were assessed based on RECIST version 1.1. 6 The baseline for each evaluation of tumour response was based on the most recent preceding radiological scan, except for PD, which was evaluated with respect to the minimum tumour size achieved during treatment.
Sequential measurement of serum LDH
Serum concentration of LDH was measured using enzymatic spectrophotometric method with an automatic continuous monitoring platform (Kwork Inc, Zhejiang, China). The upper limit normal value of our LDH assay was 240 IU/L. LDH was measured prior to CT and before each cycle of CT.
Statistical considerations
Primary analysis of this study was to test the association between serial LDH levels at the different CT cycles and tumour response (CR, PR, SD and PD) as assessed at the time points of CT2, CT4 and CT6. Due to the small number of CR cases, we aggregated CR with PR cases, which are collectively labelled as PR. Secondary analyses include longitudinal change in LDH with CT cycles, and association of the clinical covariates (age, disease status and burden, CT regimen and cycle) and LDH levels with OS and PFS. Age was considered as both a continuous and categorical variable (<45 y versus ⩾45 y), while disease status (recurrent versus de novo) and burden (⩽3 versus >3 lesions; 1, 2 versus ⩾3 sites), CT regimens and cycles (2, 4 versus 6) and LDH [<240 IU/L (low) versus ⩾240 IU/L (high)] were considered as categorical variables. OS was defined by the date of diagnosis to death from any cause; PFS was defined by the date of diagnosis to PD or death from any cause. Kaplan–Meier survival curves were used to estimate OS and PFS for the LDH low and high groups; they were generated with Python 3.7.4 and the lifelines (version 0.24) library. Hazard ratio (HR) for the respective groups was estimated using the Cox proportional hazards modelling, with multivariable adjustment against the clinical covariates. Cox modelling on survival curves was performed in R 3.6.1 with the survival library (version 3.1–8). 21
For the predictive model, sensitivity and specificity measurements were evaluated by systematically varying the LDH ratio cut-off using an in-house Python script with the scikit-learn 0.22 library. 22 Confidence intervals (CIs) for the area under the receiver operating characteristic curve (AUC) were derived using the DeLong method, as evaluated in R by the pROC R library version 1.16. 23
Results
Patient cohort characteristics
We recruited 158 patients to this study; of which, 77 were patients with de novo mNPC and 81 were patients with rmNPC. Baseline demographics and clinical characteristics of our study cohort are summarised in Table 1; TNM staging information for the de novo patients are in Supplemental Table 1. Median age was 46.5 [interquartile range (IQR) 39–53] years. Of the 158 patients, 51.9% (82 of 158) had disease involving a single metastatic site; bone involvement was the most common site of involvement (93 of 158, 58.9%), followed by lung metastasis (69 of 158, 43.7%). 46.8% (74 of 158) of the patients had oligometastatic disease (defined as ⩽3 lesions), although this was more common in treatment-naïve patients than those with rmNPC (53.2% versus 40.7%). Median pretreatment LDH level was 228 (IQR 182–305) IU/L; 44.9% (71 of 158) had LDH levels of ⩾240 IU/L. Tumour burden was also associated with pretreatment LDH levels, which was independent of anatomic sites of involvement (see Supplemental Table 2).
Clinical and treatment characteristics of the study cohort.
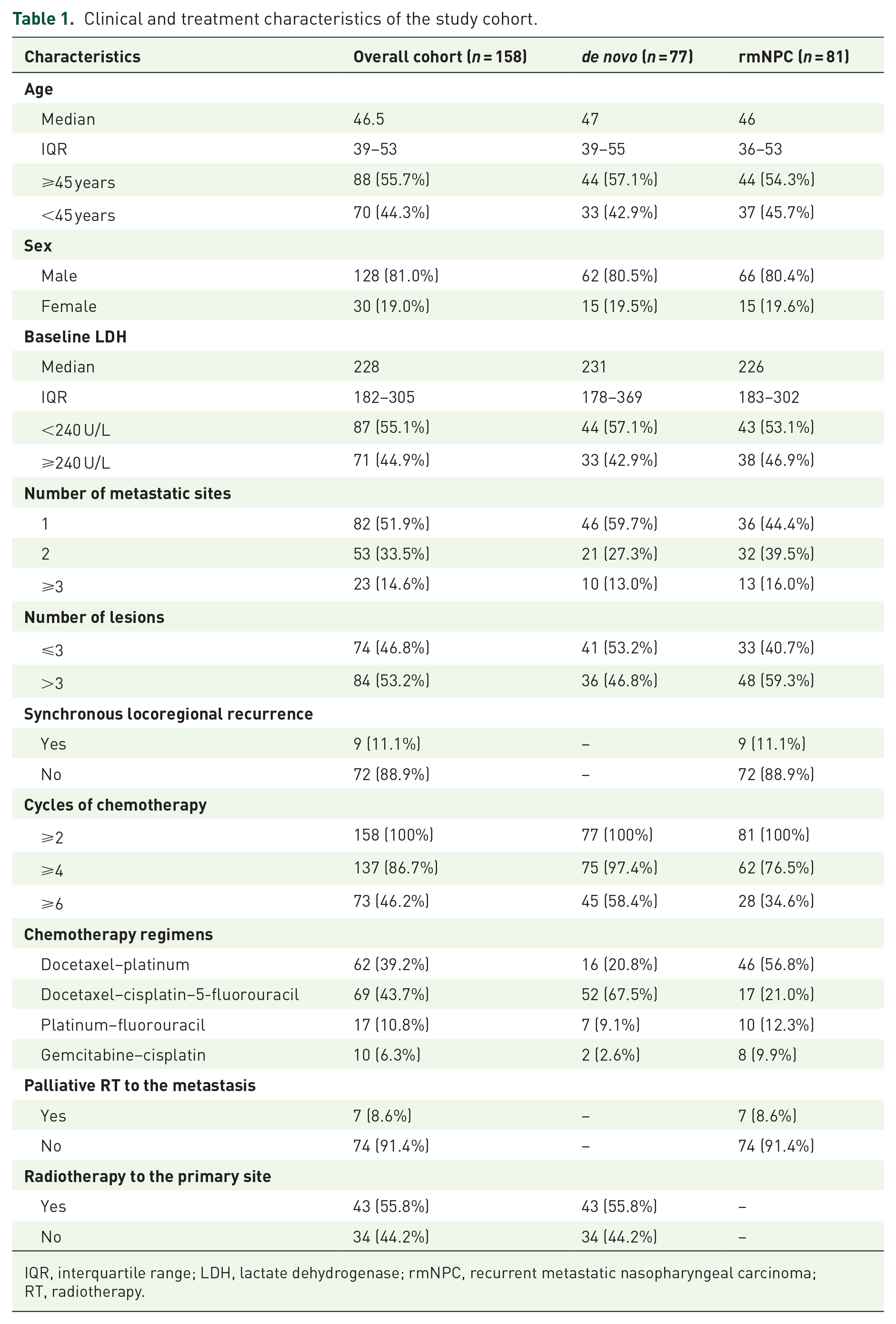
IQR, interquartile range; LDH, lactate dehydrogenase; rmNPC, recurrent metastatic nasopharyngeal carcinoma; RT, radiotherapy.
CT regimens differed between the de novo and rmNPC subgroups; triplet docetaxel–cisplatin–5-fluorouracil was the first-line CT of choice for the former (52 of 77, 67.5%), while patients with rmNPC were mostly treated with doublet regimens (64 of 81, 79.0%): docetaxel–platinum (n = 46), platinum–fluorouracil (n = 10) and GP (n = 8). Of the 77 de novo patients, 43 (55.8%) received high-dose RT to the primary site. Separately, seven (8.6%) patients with rmNPC received palliative RT during their CT. Overall, 86.7% (137 of 158) of all patients received at least four cycles of CT, and 46.2% (73 of 158) of all patients completed six cycles of CT (58.4% in the de novo mNPC subgroup versus 34.6% in the rmNPC subgroup).
Pretreatment LDH level is prognostic in metastatic NPC patients
Consistent with published literature,24–26 we observed that patients with an elevated LDH level (⩾240 IU/L) prior to treatment had an inferior OS (HR 1.93, 95% CI: 1.34–2.77, p < 0.001; Figure 2) and PFS [HR 2.07 (1.47–2.92), p < 0.001; Supplemental Figure 1] compared with individuals with normal LDH levels, albeit this did not achieve statistical significance for OS in the de novo subgroup of patients. Separately, number of metastatic sites and lesions and number of CT cycles were associated with OS (see Supplemental Table 3).

Kaplan–Meier curves of overall survival between high (⩾240 IU/L) and low (<240 IU/L) pre-CT LDH subgroups for our cohort.
Longitudinal LDH trends over the course of CT
Next, we investigated whether serum LDH levels change over the course of CT cycles. A total of 158 patients had sequential LDH records before CT (pre-CT), after cycle 1 (CT1) and after cycle 2 (CT2). Of these, 123 and 60 had complete records up to CT4 and CT6, respectively. To characterise the trend of how LDH fluctuates with CT cycles, we utilised the subset of patients with complete records from pre-CT to CT6 (n = 60; one patient had missing data at CT3, and hence was excluded from this time-series analysis). First, we did not observe a difference in LDH levels between de novo patients (n = 32) and those with rmNPC (n = 28) across all the time points [Figure 3(a)], suggesting that LDH fluctuation is independent of prior treatment. Next, we observed that LDH fluctuation was most pronounced between pre-CT and CT1, which was primarily contributed by patients with an elevated LDH level pre-CT [Figure 3(b)]. However, LDH levels demonstrated less pronounced shifts beyond post-CT1.

Longitudinal fluctuation of LDH levels over the course of CT in a subset of 60 patients with complete data-points from CT1–6.
Regarding the longitudinal tumour response, we observed that initial tumour response at CT2 was not correlated with subsequent tumour responses at CT4 and CT6. The majority of patients had SD between CT2 and CT4, which was independent of initial tumour response at CT2. Some patients with initial SD eventually were able to elicit a PR at CT4, while a small proportion of patients (11 of 123; 8.9%) developed PD in the same time period [Figure 3(c)]. Interestingly, 28.3% (17 of 60) patients continued to manifest PR between CT4 to CT6, although a higher number of patients developed PD in the same time period [predominantly in patients who had SD at CT4; Figure 3(d)]. Given the dynamic changes in disease states over time, we hypothesise that changes in LDH levels between preceding CT cycles are predictive of subsequent tumour responses, as opposed to a single LDH measurement at a specified time point.
Temporal LDH measurements were associated with tumour response to CT
Figure 4(a) illustrates the absolute LDH levels at CT1–6 for the different tumour response subgroups. Here, patients were assessed after every two cycles of CT for PR, SD and PD. We observed that absolute LDH levels at all time points are associated with tumour response; patients with PR had the lowest median LDH as opposed to individuals with PD, who had the highest median LDH. Consistent with existing literature in NPC and other cancer types, absolute LDH was associated with tumour response [Figure 4(a)]. However, utilising raw LDH values to predict chemotherapy response is limited by the fact that LDH values can vary substantially between patients of different ages and medical comorbidities, which would render identifying an optimal cut-off for clinical prediction challenging. To address this, we explored whether a relative assessment of LDH using the ratio defined by LDHCTn: LDHCTn–1, where n ranges from 1 to 6 [Figure 3; CT0 corresponds to pretreatment (pre-CT)], was associated with CT response.

Longitudinal LDH measurements at different CT time points for the different response groups.
As illustrated in Figure 4(b), LDH ratios at CT1–6 were significantly associated with tumour response to CT; this was independent of CT regimen (see Supplemental Figure 2). Notably, the inter-patient variance of LDH ratios was reduced compared to absolute LDH values, thus addressing the conundrum of large LDH variation between patients. Median LDH ratio was <1.0 for patients with PR across all time points (range 0.738–0.988). Conversely, patients with PD had a median LDH ratio of >1.0 from CT1–6 (range 1.039–1.406).
Temporal LDH ratio for early prediction of PD
Given the association between serial LDH trends and CT response, we next ask the question whether LDH ratio of the preceding CT cycles predicts for subsequent responses (i.e. using LDHCT1: LDHpre–CT and LDHCT3: LDHCT2 to predict for tumour response at CT2 and CT4, respectively). In practice, unless the patient develops PD, it is not routine to alter the CT regimen, even in the presence of SD. We therefore dichotomised our patient cohort by their PD status (PD versus non-PD), and interrogated if the preceding LDH ratio predicts for likelihood of PD.
First, we quantified the accuracy of using the preceding LDH ratio for predicting PD with the AUC obtained by varying the LDH ratio cut-off. We achieved an AUC of 0.74 (CT1: Pre-CT), 0.69 [CT3: CT2] and 0.70 [CT5: CT4] for predicting PD at CT2, CT4 and CT6, respectively [Figure 5(a)]. It is particularly impressive that the prediction power of this index is largely constant across the early and late phases of treatment. To derive an optimal cut-off of LDH ratio for predicting the likelihood of PD, we probed the trade-off in sensitivity and specificity over a range of LDH ratios [Figure 5(b)]. We ascertained that an LDH ratio cut-off of 1.0 yielded a sensitivity of 0.79 and specificity of 0.62 in our cohort. Choosing a cut-off of 1.0 is therefore practical and intuitive, as a ratio of >1.0 equates to any increase in LDH as being predictive of PD.
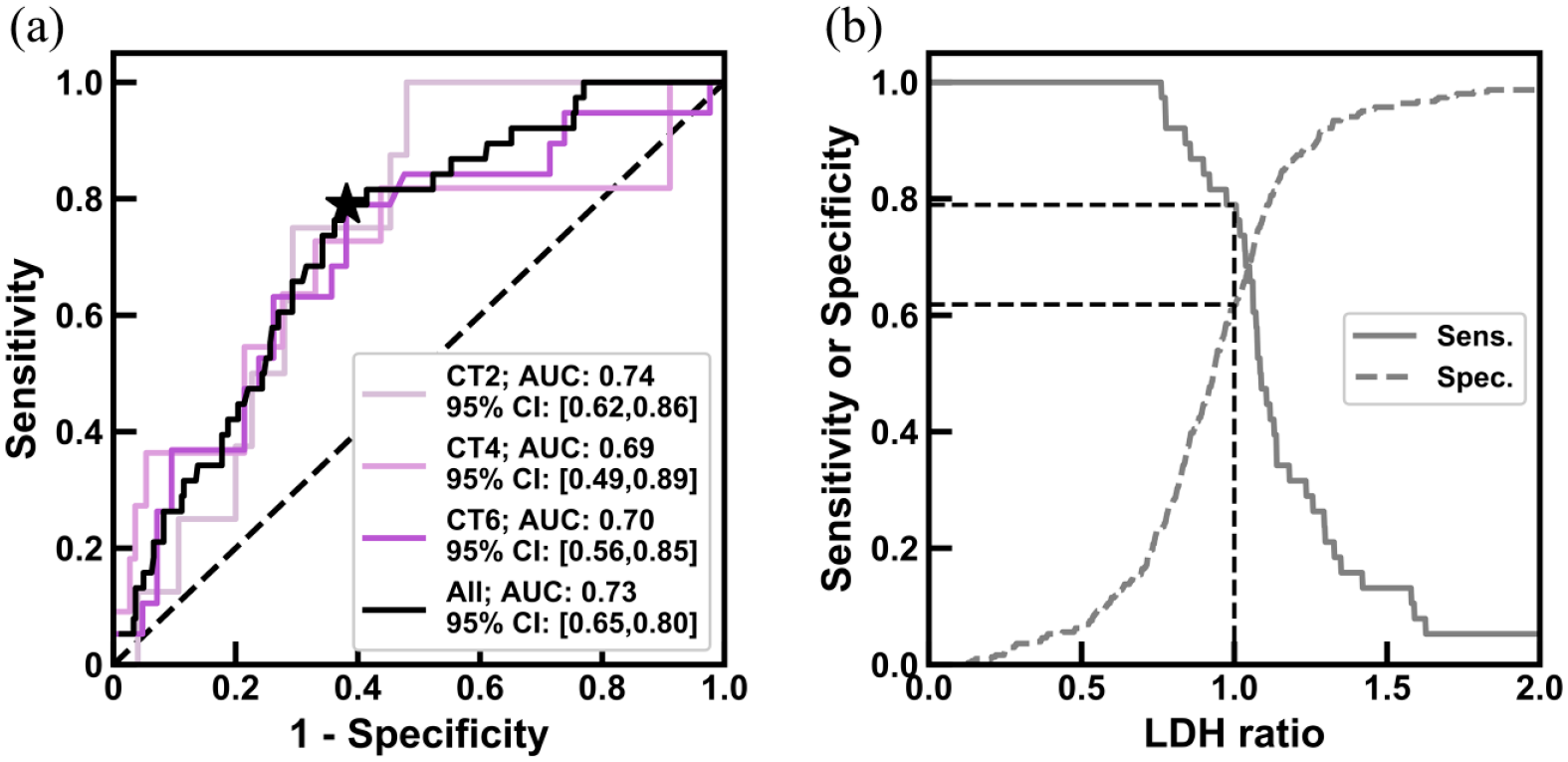
LDH ratio predicts progressive disease during CT.
Discussion
In the current era of personalised medicine, much of the research efforts have been focused on addressing the possibility of treatment adaptation based on baseline patient phenotyping and longitudinal response to treatment. For real-time on-treatment surveillance, molecular profiling of serial tumour samples and liquid biopsies have been explored. In the case of NPC, circulating plasma EBV DNA is a frontrunner given its utility as a sensitive biomarker in assessing early responses to CT and RT.9,27,28 However, molecular testing remains under-utilised in several parts of the world, especially in LMICs, where robust testing quality assurance is difficult to implement. Here, we demonstrate that serum LDH may be a low-cost alternative to EBV DNA for early prediction of PD during chemotherapy. Using a cohort of 158 patients with mNPC with serial LDH measurements, we foremost characterised the temporal changes of LDH in response to CT and observed that the most pronounced changes in LDH occurred during the early phase of treatment. Next, we compared both the absolute and relative LDH values for their association with tumour response; the rationale of using the latter was because of wide inter-patient variability of LDH levels in our cohort. We probed the concept of LDH ratio (LDHCTn: LDHCTn–1), and showed that this parameter was significantly associated with tumour response; median LDH ratio <1.0 in partial responders, implying that LDH generally drops in these patients, while in patients with PD, due to the rising LDH titres, median LDH ratio was >1.0. Based on these observations, we queried whether interval LDH ratio at the midpoint of two CT cycles could predict the likelihood of PD in our cohort. We showed that our LDH ratio model performed well over the full course of CT (AUC of 0.69–0.74), and proposed an LDH ratio cut-off of 1.0, which is practical and intuitive, with a reasonable sensitivity of 79% and a specificity of 62%. Collectively, our findings showcased the potential of serial LDH tracking as an inexpensive and efficient method to monitor CT response.
The utility of LDH as a biomarker for tracking tumour response is attributed to the significantly different cellular metabolism between tumour and normal cells.13,29,30 In particular, the majority of tumour cells would manifest a decrease in mitochondrial oxidative phosphorylation, resulting in an increase of both glucose consumption and lactate production regardless of oxygenation.14,31 Among the enzymes involved in glycolysis, LDH (which converts pyruvate to lactate) is regarded as one of the main metabolic enzymes. Many studies in different types of cancers, such as NPC, liver, lung, breast and prostate cancers have demonstrated the importance of LDH for tumourigenesis, proliferation, and progression in vitro and in vivo, whereby inhibition of LDH could reverse such tumour phenotypes.13,30,32 These experimental findings corroborate the clinical correlations of an elevated serum LDH with a poor prognosis and suboptimal tumour response.13,15,33,34 However, it is unknown if this observed correlation between LDH activity and cancer outcomes is isoenzyme-specific (LDH 1–5). Given that LDH isoenzymes are highly tissue-specific, it is plausible to consider that different isoenzymes could be predictive of tumour response to CT at specific anatomical organs, which could be investigated going forward.
In metastatic patients, conventional radiological examination with CT remains the gold standard for treatment response evaluation for most cancer types. Currently, we lack the ability to determine early PD, which is in part due to the lenient criterion of at least 20% increase in size, as set in the RECIST criteria. This void is where liquid biopsy has demonstrated to be useful in several cancers.7,35,36 The scientific rationale being that liquid biopsy, which purports to measure the amount of tumour biomaterial in the circulatory system, is correlated to tumour burden, and the fluctuation in titres is therefore representative of the tumour clonal sensitivity to treatment. Plasma EBV DNA represents one of such tests that is available for clinical use in patients with NPC. As aforementioned, implementation of the EBV DNA assay in LMICs is challenging, and hence a simpler alternative is needed. Here, we show the potential utility of tracking the temporal change of LDH in a patient, which could inform us of the disease trajectory of the individual in real time. In doing so, this simple test, if validated, can help patients avoid unnecessary ineffective treatments and their associated toxicities, and perhaps prompt an early switch of treatment. That said, it would be interesting to investigate if our results could be improved with the combination of both LDH and EBV DNA, given that these biochemical and molecular indices are mutually exclusive.
A few limitations of our work ought to be highlighted. First, our study lacked a validation cohort. It is extremely difficult to source for another cohort that had serial LDH measurements for every CT cycle, as well as fulfilled the strict screening criteria of this study (absence of infection and other confounders; Figure 1). This is also partly because LDH is not a standard pre-CT assessment investigation. It is worth noting that the clinical characteristics and outcomes of our present cohort are comparable to other rmNPC cohorts,3,5,37 and LDH ratio demonstrated consistency in PD prediction across CT cycles, which would arguably serve as an internal validation. Next, CT regimens varied between patients in our cohort, and the utilisation of the triplet docetaxel–cisplatin–5-fluorouracil regimen, while effective in NPC, is not the standard of care as first-line treatment in rmNPC. Nonetheless, there was no significant difference in treatment efficacies for the different regimens in our cohort (with the exception of GP for PFS; Supplemental Tables 3 and 4), and thus it is reasonable to suggest that our findings are unlikely to be biased by treatment. Finally, LDH is highly concentrated in the liver, kidney, heart and muscle, which are vulnerable to insult by CT. However, patients with functional impairment of these organs were excluded, and interval between LDH measurement and CT was at least 3 weeks apart.
Conclusion
To conclude, we confirmed the findings of others that serum LDH is a prognostic biomarker for OS in endemic NPC. In addition, we demonstrated significant inter-patient heterogeneity in temporal fluctuation of LDH that is matched to tumour response to CT. The relative difference in LDH with CT cycles may have clinical utility in predicting resistance to CT, and warrants validation in other prospective or real-world datasets.
Supplemental Material
20200903_Supplementary_TAMO_FINAL – Supplemental material for Lactate dehydrogenase kinetics predict chemotherapy response in recurrent metastatic nasopharyngeal carcinoma
Supplemental material, 20200903_Supplementary_TAMO_FINAL for Lactate dehydrogenase kinetics predict chemotherapy response in recurrent metastatic nasopharyngeal carcinoma by Luo Huang, Adelene Y. L. Sim, Yongzhong Wu, Zhongguo Liang, Kaiguo Li, Youqin Du, Enya H. W. Ong, Hong Qi Tan, Joseph T. S. Wee, Yue Xie, Xiaolei Shu, Ying Wang, Melvin L. K. Chua and Xiaodong Zhu in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors thank the patients and their families for their participation in this study.
Author contributions
Conception and design: Melvin L.K. Chua, Xiaodong Zhu, Yongzhong Wu.
Financial support: Ying Wang, Yongzhong Wu, Melvin L.K. Chua.
Administrative support: Xiaodong Zhu, Melvin L.K. Chua, Yongzhong Wu.
Provision of study materials or patients: Xiaodong Zhu, Luo Huang, Zhongguo Liang, Kaiguo Li, Youqin Du.
Collection and assembly of data: Adelene Y.L. Sim, Luo Huang, Zhongguo Liang, Hong Qi Tan, Kaiguo Li, Yue Xie, Enya H.W. Ong, Joseph T.S. Wee, Xiaolei Shu.
Data analysis and interpretation: Adelene Y.L. Sim, Luo Huang, Hong Qi Tan.
Manuscript writing: All authors.
Final approval of manuscript: All authors.
Conflict of interest statement
MC reports personal fees from Astellas, Janssen, Bayer, Pfizer, MSD, personal fees and non-financial support from AstraZeneca, personal fees and grants from Ferring, personal fees and non-financial support from Varian, non-financial support from Decipher Biosciences, non-financial support from Medlever, and consults for immunoSCAPE Inc., outside the submitted work.
Data availability
All data included in this study are available upon request.
Ethics approval and consent to participate
This retrospective research was approved by the Ethics Committee of the GXMUCH (AB18221007), and performed in accordance with the Declaration of Helsinki and Good Clinical Practice Guidelines. Informed consent was obtained from all living patients. All patient records were anonymised before analysis.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Luo Huang, Yongzhong Wu and Ying Wang are funded by the Chongqing Science and Technology Innovation Guidance Project Led by Academician (cstc2017jcyj-yszx0001), Chongqing Science and Technology Innovation Guidance Project Led by Academician (2018), National Natural Science Foundation of China (81972857). Melvin L.K. Chua is supported by the National Medical Research Council Clinician-Scientist Award (CSA/0027/2018).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
