Abstract
Background:
Our goal was to organize the data from randomized controlled trials that evaluated first-line chemotherapy for chemo-naïve extensive disease small-cell lung cancer (ED-SCLC).
Methods:
The protocol following PRISMA methodology was submitted as PROSPERO 154049. We included individually randomized trials comparing two or more chemotherapy regimens as the first-line treatment for chemo-naïve ED-SCLC regardless of the age, sex, performance status, co-morbidities, and organ functions written in the English language since 2000. Molecular targeted agents and immune checkpoint inhibitors were considered chemotherapy along with cytotoxic medications. We pooled the logarithm of hazard ratio (HR) and its standard error using the frequentist weighted least squares approach random-model network meta-analysis.
Results:
A total of 46 eligible trials that involved 11,987 patients were included. The primary endpoint, HR of overall survival (OS, HRos) of the selected comparisons was as follows: carboplatin+amrubicin (HRos 0.56, 95% confidence interval (CI) 0.33–0.96), carboplatin+etoposide+atezolizumab (HRos 0.70, 95% CI 0.53–0.92), and carboplatin+irinotecan (HRos 0.73, 95% CI 0.58–0.91) were compared with carboplatin+etoposide. The carboplatin+etoposide+atezolizumab regimen was compared with carboplatin+irinotecan (HRos 0.97, 95% CI 0.68–1.37) and cisplatin+irinotecan regimen (HRos 0.87, 95% CI 0.58–1.31). “Selective carboplatin or cisplatin (CBDCA/CDDP)”+etoposide+durvalumab was compared with CBDCA/CDDP+etoposide (HRos 0.73, 95% CI 0.59–0.91). Platinum+etoposide+durvalumab was compared with platinum+irinotecan (HRos 0.88, 95% CI 0.67–1.15). Cumulative meta-analysis suggested that platinum+irinotecan was associated with better OS than platinum+etoposide as of 2010 through 40 out of 46 trials in our review that used platinum+etoposide as a reference regimen.
Conclusion:
Patients treated with carboplatin+amrubicin, carboplatin+etoposide+atezolizumab, CBDCA/CDDP+etoposide+durvalumab, and platinum+irinotecan showed better HRos than those treated with platinum+etoposide, one of the standard regimens.
Background
Small-cell lung cancer (SCLC) is a thoracic malignancy often caused by habitual smoking. 1 The majority of patients are diagnosed with SCLC after progression to extensive disease (ED) due to rapid growth and easy dissemination of the tumor cells. Chemotherapy is usually considered in ED-SCLC patients with a good performance status because SCLC is highly sensitive to the treatment. A Japanese study by Noda et al. reported that the cisplatin (CDDP)+irinotecan (CPT11) regimen resulted in better overall survival (OS) than the CDDP+etoposide (ETP) regimen. 2 Since then, many physicians have believed that the CDDP+ CPT11 regimen was the best to treat patients with ED-SCLC, especially the non-elderly Asian population. 3 However, the outcome was not confirmed as the primary endpoint in any of the subsequent phase III trials in the United States (US),4,5 Europe, 6 and Korea, 3 although CDDP+CPT11 showed a tendency of better OS in most of these trials. Therefore, others chose CDDP+ETP as the standard regimen. 7 Both CDDP+CPT11 and CDDP+ETP have been considered the standard regimen for more than a decade.
Recently, oncologists were excited to learn that addition of atezolizumab (ATZ) or durvalumab (DUR) to platinum+ETP improved both OS and progression-free survival (PFS) in patients with ED-SCLC.8,9 Programmed death-ligand 1 (PD-L1) inhibiting regimens are an excellent therapeutic option for lung cancer as well as several other malignancies. 10
Although the PD-L1 inhibiting agents including ATZ and DUR may be attractive options to treat patients with ED-SCLC, there could be other attractive regimens that are not currently featured in phase III trials.11–13 Therefore, the goal of this systematic review was to organize data from randomized clinical trials (RCT) evaluating first-line chemotherapy regimens for ED-SCLC that were published in the last 20 years and apply network meta-analysis to assess the obtained data.
Methods
Protocol registration
The protocol was submitted to the International Prospective Register of Systematic Reviews (submission ID, 154049). 14 The protocol was structured according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 15
Study search
Search strategies for PubMed, Web of Science Core Collection, Cochrane advanced search, and EMBASE are presented in Supplemental Text 1. The databases were searched on 10 October 2019. An additional manual search was also conducted independently by two investigators (NH and HC). Papers identified by hand search were carefully evaluated before being added to the list of candidate articles.
Candidate articles were first screened and subsequently scrutinized independently by two investigators. Any discrepancies were resolved through discussions between the two investigators.
Publication type and trial design
We included individually randomized trials that compared two or more chemotherapy regimens as the first-line treatment for chemo-naïve ED-SCLC patients that were published as full papers in English since 2000. Conference abstracts in English reported after 2015 were also included to review prevailing regimens. We contacted author groups of abstracts to obtain relevant data.
All superiority, non-inferiority, phase II, phase III, non-blinded, single-blinded, and double-blinded trials were included. However, patients included in these trials had to be randomized before the initiation of chemotherapy. Therefore, trials that randomized relapsed or responsive cases were excluded.
Trials evaluating a specific population defined by age, race, or performance status were permitted. Further, we excluded trials that did not report survival data. Trials that included both limited disease and ED were included as long as the data of ED cases could be extracted.
Treatments
All regimens that included cytotoxic, molecular targeted agents, or an immune checkpoint inhibitor were considered chemotherapy. However, any regimen that included so-called “first-generation” cytotoxic agents developed around 1950 namely methotrexate, mitomycin, vincristine, cyclophosphamide, doxorubicin, and ifosfamide was excluded from our analysis because such regimens are outdated. 7
Regimens that used the same medication were evaluated collectively regardless of the administration route, timing, frequency, dosage, and schedule. We equated placebo with “no treatment.” For example, CDDP + (placebo of ETP) arm was identical to the CDDP arm in our analysis. The best supportive care could not be a comparator of chemotherapy. Studies that reported multimodality treatments including chemoradiotherapy, radiotherapy, or surgery were not included. Similarly, studies that reported irregular regimens, such as alternative regimens, were excluded.
Study that permitted the physician to select either carboplatin (CBDCA) or CDDP were regarded as “selective CBDCA or CDDP (CBDCA/CDDP) regimens” in order to discriminate from regular CDDP and CBDCA.
We used two models to assess the primary endpoint, OS. In the first model, CBDCA and CDDP were distinct from each other. In the second model, CBDCA, CDDP, and CBDCA/CDDP were collectively considered platinum.
Patients
Chemo-naïve ED-SCLC patients who underwent first-line chemotherapy were included. The age, sex, performance status, co-morbidities, and organ functions of patients were not considered as differentiating parameters.
Quality assessment
We assessed the quality of the original studies using six domains of the Cochrane Risk of Bias tool: random sequence generation, allocation concealment, performance, detection, attrition, and reporting. 16
Outcomes
The primary outcome measure in our review was the hazard ratio (HR) for OS (HRos). The secondary outcomes were HR for PFS (HRpfs) and odds ratio (OR) of response rate (RR, ORrr). 17 Evaluation of disease progression to assess the PFS and the objective response to assess RR was done by ensuring minimal deviation from the Response Evaluation Criteria In Solid Tumors 2000 guidelines and the 2009 revised guidelines. 18 In studies where disease progression and objective response were evaluated both by physicians who treated patients and by the blinded Independent Central Review Board, we chose the data reviewed by the blinded evaluator. Time to progression was not regarded as PFS.
Additional cumulative meta-analysis comparing ETP and CPT11 regimens, head-to-head meta-analysis evaluating adding of PD-L1 inhibiting agent on platinum+ETP, and head-to-head meta-analysis comparing CBDCA and CDDP doublet with the same counterpart were conducted. Although these analyses were not part of the protocol, in our opinion, these analysis could provide useful data to the readers.
Data extraction
Data, such as author name, publication year, country of origin, number of patients randomized, chemotherapy regimen, and outcomes were extracted from the included studies independently by two investigators (NH and HC). The data were cross-checked, and any discrepancies were discussed and resolved by the two investigators. Data from non-inferiority trials were extracted using the same method as applied for the superiority trials. For three-arm studies, data of each pair-wise comparison were extracted. We adopted Parmar’s method to extract data from Kaplan–Meier curves, where required. 19 Intention-to-treat analysis was preferred over full analysis set analysis and per-protocol analysis when two or more of them were available.
Statistical analyses
We pooled the logarithm of OR, HR, and their standard errors using the frequentist weighted least squares approach random-model network meta-analysis.20,21 A league table of the HRos, HRpfs, ORrr, and their 95% confidence intervals (CI) was presented. For the network meta-analysis, the “netmeta” command in the “netmeta” package of R Project was used. The head-to-head meta-analysis and generation of a funnel plot were performed using Review Manager ver5.3 (Cochrane, London, UK). GraphPad Prism ver. 7.02 was used to draw figures as necessary (GraphPad Software, San Diego, CA, USA).
Results
Study selection and characteristics
We found 1431 and 5 articles through search engine and hand searching, respectively. Of the 1436 articles that met the preliminary criteria, 569, 669, and 152 were excluded due to duplication, title/abstract screening, and full-article scrutiny, respectively (Figure 1). We identified 46 eligible articles that provided data of HRos (45 studies), HRpfs (33 studies), and ORrr (43 studies), respectively (Supplemental Text 2, Table 1). Furthermore, we obtained data for the CASPIAN trial from the author group. 9
Characteristics of included studies.

n, number of patients randomized to concerned arms.
Median age: *indicates mean age instead of median age.
WHO PS 0–2 is roughly equivalent to ECOG PS 0–2; KPS, Karnofsky’s index of PS.
ROB H/U/L (High/Unclear/Low): Six domains of Cochrane risk of bias were assessed. 6/0/0, meaning 6 domains with high risk of bias, is the poorest score. 0/0/6, meaning 6 domains with low risk of bias, is the best score.
Outcome missing: an indicated outcome was not reported in the article. Blank means all of OS, PFS, and RR were available.
/m2, per body surface area square meter; AUC, area under curve by Calvart formula; CBDCA, carboplatin; CDDP, cisplatin; d, day; ECOG, Eastern Cooperative Oncology Group; ETP, toposide; CPT11, irinotecan; GEM, gemcitabine; OS, overall survival; PEM, pemetrexed; PFS, progression-free survival; PS, performance status; PTX, paclitaxel; q3w, every 3 weeks; ROB, risk of bias; RR, response rate; TOP, topotecan; WHO, World Health Organization. In a two-arm trial, arm 1 and arm 2 may be switched from the original publication to place the reference arm to arm2.

PRISMA flow diagram for the study search.
Of the included studies, 18 were from the US, 15 were from the European Union (EU), 6 were from Japan, 4 were from China, 2 were from Korea, and 1 was from India; 24 studies were phase III and 20 were phase II trials. Two studies did not declare the study phase. Number of patients randomized in each trial ranged from 56 to 1132 with a median of 155, and the total number of cases was 11,987. A total of 26 trials included the ECOG performance status 0–2. Of the 46 trials, 3 were three-arm studies, and the remaining 43 adopted a two-arm study design. The most common regimen was CDDP+ETP, which was used in 20 RCTs, followed by CBDCA+ETP, which was used in 13 trials; 7 trials considered ETP+selective CDDP/CBDCA as the reference arm. Therefore, 40 out of the 46 trials (87%) compared platinum+ETP with other regimens. The median years of publication of these three regimens were as follows: 2012 (IQR 2007.5-2016) for CDDP+ETP, 2014 (IQR 2008-2017) for CBDCA+ETP, and 2017 (IQR 2013.5-2018) for CBDCA/CDDP+ETP (Table 1). Therefore, we concluded that preferred reference regimen shifted from CDDP+ETP to CBDCA+ETP, and subsequently to CBDCA/CDDP+ETP.
Overall survival: CDDP/CBDCA distinguishing model
The network graph of the main loop had two dominant hubs, CBDCA+ETP and CDDP+ETP (Figure 2A). The league table is presented in Supplemental Table S1. Publication bias was not suspected based on the funnel plot (Supplemental Figure S1. In this analysis, the CBDCA+ETP regimen was used as the common comparator to present a forest plot because this regimen has been recently preferred over CDDP+ETP (Table 1, Figure 2A). CBDCA+amurubicin (AMR) (HR 0.56, 95% CI 0.33–0.96), CBDCA+ETP+ATZ (HR 0.70, 95% CI 0.53–0.92), and CBDCA+CPT11 (HR 0.73, 95% CI 0.58–0.91) showed better OS than CBDCA+ETP. Two PEM regimens, CBDCA+PEM (HR 1.56, 95% CI 1.25–1.95) and CDDP+PEM (HR 2.23, 95% CI 1.26–3.90) clearly demonstrated deterioration in OS.

Network graph for HR of OS. (A) In the CDDP/CBDCA distinguished model, CDDP and CBDCA are recognized as different agents. (B) In the CDDP/CBDCA distinguished model, some trials that allowed selective administration of CBDCA or CDDP (CBDCA/CDDP) made an independent loop. (C) CBDCA = CDDP model. CDDP and CBDCA are recognized switchable Pt reagent.
The CBDCA+ETP+ATZ regimen demonstrated improved OS compared with CBDCA+ETP (HR 0.70, 95% CI 0.53–0.92) and a similar OS outcome compared with CBDCA+CPT11 (HR 0.97, 95% CI 0.68–1.37) (Supplemental Table S1).
Seven trials permitted selective administration of CDDP and CBDCA in all arms. Therefore, these trials composed an independent small loop (Figure 2B). Platinum+ETP+DUR regimen resulted in improved OS than common comparator CBDCA/CDDP+ETP (HR 0.73, 95% CI 0.59–0.91) (Figure 3B, Supplemental Table S2).
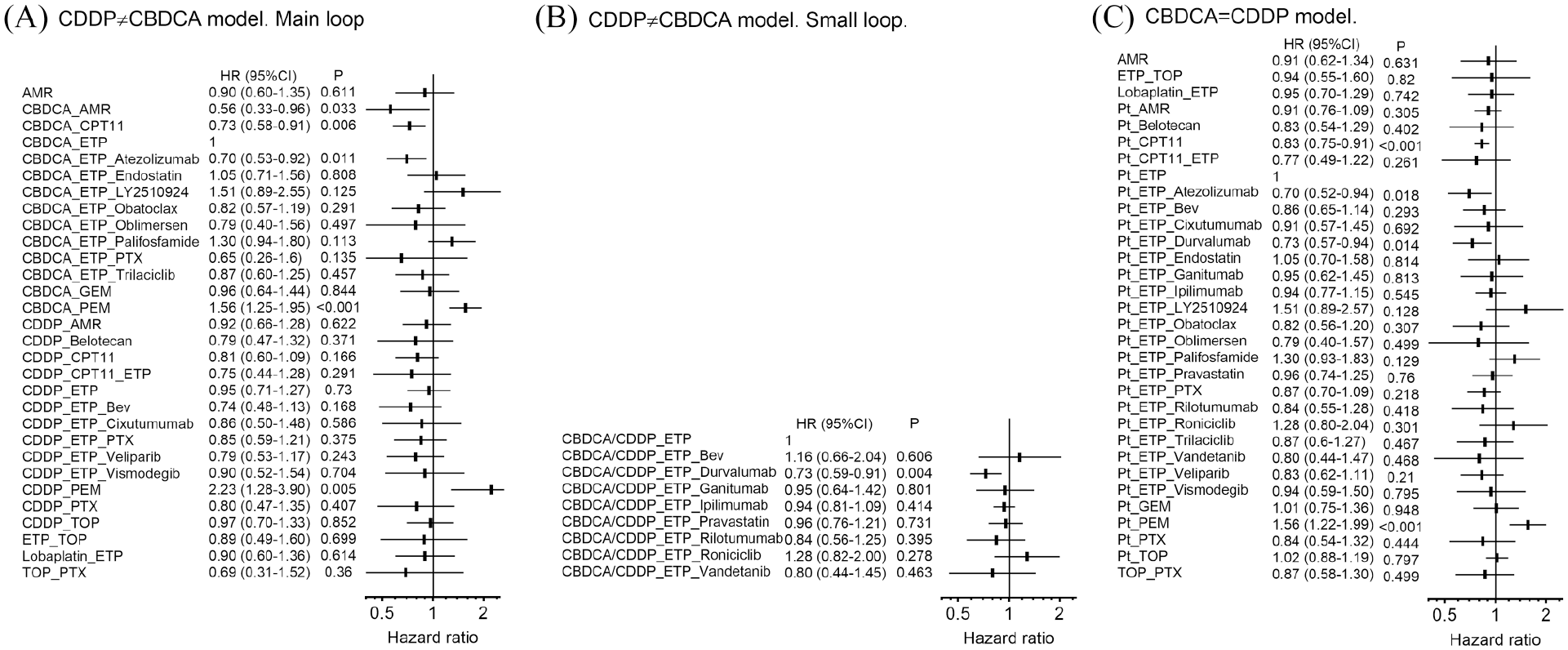
Network meta-analysis for HR of OS. (A) In the CDDP/CBDCA distinguished model, CDDP and CBDCA are recognized as different reagents. (I2 = 9.5%, P for heterogeneity 0.353). CBDCA+ETP is the common comparator. (B) In the CDDP/CBDCA distinguished model, some trials that allowed selective administration of CBDCA or CDDP (CBDCA/CDDP) made an independent loop. (I2 = 0%, P for heterogeneity not available). CBDCA/CDDP+ETP is the common comparator. (C) CBDCA = CDDP model. CDDP and CBDCA are recognized switchable Pt reagent (I2 = 22.3%, P for heterogeneity 0.200). Pt+ETP was the common comparator. A horizontal error bar indicates 95% CI.
Overall survival: CDDP/CBDCA identifying model
In this model, we collectively regarded CDDP and CBDCA as platinum. The majority of the trials used platinum+ETP as the reference arm (Figure 2C, Supplemental Table S3). This analysis revealed that platinum+ETP+ATZ (HR 0.70, 95% CI 0.52–0.94), platinum+ETP+DUR (HR 0.73, 95% CI 0.57–0.94), and platinum+CPT11 (HR 0.83, 95% CI 0.75–0.91) improved OS and that platinum+PEM (HR 1.56, 95% CI 1.22–1.99) led to the deterioration in the OS (Figure 3C, Supplemental Table S3). The platinum+ETP+DUR regimen showed a tendency toward improved HRos compared with platinum+CPT11 (HR 0.88, 95% CI 0.67–1.15) (Supplemental Table S3).
Cumulative meta-analysis of HRos comparing CPT11 and ETP regimens
Seven trials compared the CDDP+CPT11 versus CDDP+ETP regimens and two compared CBDCA+CPT11 and CBDCA+ETP regimens (Table 1, Figure 4). We constructed four models of cumulative forest plots, which suggested that, since 2010, the CPT11 regimen demonstrated better HRos, and the trend persists to date. The current cumulative HRos were 0.88 (95% CI 0.80–0.96), 0.72 (95% CI 0.59–0.90), 0.85 (95% CI 0.78–0.93), and 0.87 (95% CI 0.78–0.96), in the CDDP, CBDCA, platinum, and non-Asian platinum models, respectively.

Cumulative meta-analysis for HR of OS comparing CPT11 regimen and ETP regimen. In a cumulative meta-analysis, each trial is merged into previous data. For example, in CDDP model, “Lara_2009” in the figure suggests pooled HR from Noda_2002, Hana_2006, and Lara_2009 is 0.85. (A) Seven trials compared cisplatin (CDDP)+CPT11 and CDDP+ETP were analyzed. (B) Two trials compared carboplatin (CBDCA)+CPT11 and CBDCA+ETP were analyzed. (C) Both CDDP and CBDCA were regarded Pt and all nine trials were analyzed. (D) Noda 2002 and Kim 2019, which were reports from Asia, were excluded. A horizontal error bar indicates 95% CI.
Progression-free survival
Network graphs for HRpfs are similar to those of OS (Supplemental Figure S2).
The CBDCA+AMR (HR 0.43, 95% CI 0.24–0.78), CBDCA+ETP+ATZ (HR 0.77, 95% CI 0.60–0.99), and CBDCA+CPT11 (HR 0.73, 95% CI 0.56–0.96) regimens showed better PFS than the common comparator CBDCA+ETP (Supplemental Figure S3A, Supplemental Table S4).
In a small independent loop, addition of Bevacizumab (HR 0.53, 95% CI 0.32–0.87), DUR (HR 0.78, 95% CI 0.65–0.94), and ipilimumab (HR 0.85, 95% CI 0.75–0.97) to CBDCA/CDDP+ETP led to improved PFS (Supplemental Figure S3B, Supplemental Table S5).
Response rate
Network meta-analysis was performed for ORrr (Supplemental Figures S4 and S5, Supplemental Tables S6 and S7). In the main loop, the forest plot suggested that patients treated with the CDDP+ETP+PTX (OR 6.57, 95% CI 1.17–37.1) regimen demonstrated a more objective response compared with those treated with CBDCA+ETP.
In the independent small loop, addition of DUR (HR = 1.56, 95% CI 1.09–2.21) to CBDCA/CDDP resulted in a higher ORrr.
Efficacy outcome from PD-L1 inhibiting regimen
Two trials evaluated the effects of addition of PD-L1 inhibitor to platinum+ETP regimens. Adding PD-L1 inhibitor improved HRos (0.72, 95% CI 0.61–0.85, I2 = 0, P for heterogeneity 0.81) and HRpfs (0.78, 95% CI 0.67–0.89, I2 = 0, P for heterogeneity 0.93), while no increase was observed in the ORrr (1.15, 95% CI 0.63–2.11, I2 = 81%, P for heterogeneity 0.020) (Figure 5).

Key efficacy outcomes in trials adding PD-L1 inhibitor on Pt doublet. A horizontal error bar indicates 95% CI.
OS comparison between CBDCA and CDDP doublet using the same counterpart
Only two trials directly compared CBDCA and CDDP with the same counterpart. Pooled HRos was 1.14 (95% CI 0.86–1.51, I2 = 11%, P for heterogeneity 0.29) using CBDCA as reference (Supplemental Figure S6).
Discussion
In this systematic review, we examined the efficacy data from 46 RCTs that were published since 2000 and included a total of 11,987 patients with ED-SCLC. The robustness of the data was supported by the use of the standard PRISMA methodology, sufficiently large number of trials and patients, low heterogeneity in the employed models, and the multifaceted meta-analysis approach. This review covered chemotherapy trials conducted in the last two decades in chemo-naïve ED-SCLC patients and raised several issues.
Based on results of the studies conducted in the last decade (Figure 4), it was evident that CPT11 regimen led to better OS than the ETP regimens (Figure 2). However, as many as 40 out of 46 trials in our review used the ETP doublet regimen as the reference. We had a long-lasting argument comparing platinum+ETP and platinum+CPT11. This discussion was in part “dogmatic.” Some insisted that CDDP+ETP was the standard regimens because CDDP+CPT11 did not show statistically significant superiority compared with CDDP+ETP in any study, with the exception of a Japanese trial.2–6 Other groups argued that relying on significance based on p value alone was not scientific,22,23 and preferred to accept “the trend” and the result from meta-analyses.11–13 Schmittel et al. claimed that the CBDCA+ETP regimen remained the standard treatment because the CBDCA+CPT11 regimen could not demonstrate statistically significant superiority in OS of patients with ED-SCLC (HR 1.29, 95% CI 0.96–1.73, p = 0.095; median OS 10.0 month versus 9.0 months, p = 0.06). 24 Although the pooled HRos calculated using data from Hermes and Schmittel was 0.72 (95% CI 0.59–0.90, p = 0.002) without heterogeneity (P for heterogeneity = 0.2, I2 = 0%) at the time of the publication of the study Schmittel (Figure 4B), 11–13,24,25 several groups agreed with the results of the Schmittel and continued to use CBDCA+ETP as the standard reference treatment (Table 1, Figure 2). Two recent phase III trials reported addition of ATZ or DUR to the ETP regimen, the results of which persisted to date.8,9 However, survival outcome of patients treated with the ETP regimen was clearly inferior to the CPT11 regimen (Figure 2, Supplemental Figure S3, Figure 4), as long as we can accept the data from meta-analysis. Consequently, although addition of ATZ or DUR to platinum+ETP provided better OS than platinum+ETP alone, the HRos of these PD-L1 inhibiting regimens was not greatly better compared with those of the platinum+CPT11 regimen (Figure 3).
Many trials permitted physicians to choose between CBDCA and CDDP (Table 1), which could mean that CBDCA and CDDP are interchangeable, especially when using ETP as the doublet counterpart. However, only two trials directly compared the CBDCA and CDDP regimens using the same doublet counterpart.26,27 In 2006, Socinski et al. randomized patients into CDDP+PEM and CBDCA+PEM arms and reported a median OS of 7.6 and 10.4 months, respectively. 27 However, the results of this study do not suggest that CDDP may be used as an alternative to CBDCA. Another trial (JCOG9702) that compared CDDP+ETP and CBDCA+ETP did not indicate a large difference in the efficacy endpoint between the two arms (HRos 1.04, HRpfs 0.92, and ORrr 1.0), using CDDP+ETP as reference. 26 However, this trial recruited only elderly Japanese patients; therefore, it remains unclear whether the results can be generalized to other patients with ED-SCLC (Supplemental Figure S6). Individual patient data meta-analysis of four trials by Rossi et al. in 2012 compared the CBDCA and CDDP regimens and revealed that HRos of 1.10 (95% CI 0.92–1.27) in the CBDCA arm compared with the reference CDDP arm. 28 However, this analysis included RCTs with different counterparts, for example CDDP+ETP versus CBDCA+GEM, and RCTs using chemoradiotherapy. Therefore, the results should be interpreted with caution. Although we should admit that CBDCA regimen that does not require prolonged hydration is a useful choice to treat outpatients in daily practice, available data do not support unthoughtful use of CBDCA as a substitute of CDDP.
The most desirable outcome for a trial evaluating chemotherapy for advanced lung cancer is OS, 17 because it is easy to assess without bias and directly reflects the benefit to the patient. However, PFS and RR are often considered substitutes because assessment of OS requires a longer follow up. When PFS and RR are considered surrogate outcomes, the surrogacy should be confirmed beforehand. Nonetheless, there is lack of clarity regarding the accuracy of PFS and RR as surrogates of OS at a trial level, especially in those evaluating molecular-targeted therapies. Compared with platinum+ETP alone, addition of bevacizumab (HRpfs 0.53, HRos 1.16), trilaciclib (HRpfs 0.70, HRos 0.87), and ipilimumab (HRpfs 0.85, HRos 0.94) improved PFS (Supplemental Figure S3); however, the OS outcomes were not satisfactory (Figure 3). We should not be excited about the positive PFS in cases of molecular targeted therapies. On the contrary, excellent PFS reported by trials on PD-L1 inhibitor were endorsed by promising OS (Figures 3 and 5; Supplemental Figures S3 and S5). As PD-L1 inhibiting regimens improve OS of patients with ED-SCLC, it is becoming difficult to obtain data on OS. According to our recent analysis of 29 phase III first-line trials that included 8573 patients with SCLC, HR of PFS excellently correlated with HRos with rank correlation coefficient of 0.87. 29 PFS might be a good surrogate endpoint for trials in cases where OS is prolonged.
In our network meta-analysis, the CBDCA+AMR regimen demonstrated the best HRos of 0.55 (95% CI 0.33–0.96) compared with the CBDCA+ETP regimen. This result was obtained from a single phase II trial by Morikawa et al. 30 Although this finding was significant, the trial did not validate CBDCA+AMR as the standard regimen because this phase II trial adopted RR as the primary endpoint. Nonetheless, this regimen appeared promising in some Japanese trials.30–33 However, efficacy of AMR is dependent on the race. Our previous analysis revealed that second-line AMR therapy was highly effective in the Japanese population compared with Euro-American populations. 34 In fact, the median OS of 10 months estimated in another single-arm phase II trial from the US was not promising. 35
Addition of PD-L1 inhibiting agents to the platinum doublet seems a reasonable strategy (Figure 5). Given the similar efficacy (Figure 3C) and the high drug price of DUR, some may suppose ATZ is a better option. Addition of ATZ to other platinum doublets such as CDDP+CPT11 may be an attractive option in the future.
There are some limitations of our study. First, the network between CBDCA and CDDP regimens is scarce. Therefore, comparison between these regimens was not solid. Second, most of RCTs included only PS 0-1 or 0-2 cases though patients with higher PS should also be treated in the real world. Notably, ICI trials lack data of patients with PS score of two or higher.8,9 Third, we could not perform country-, PS-, age-based subgroup network meta-analyses due to the insufficient sample size and data format.
In conclusion, we organized the data of 46 RCTs evaluating the first-line chemotherapies for patients with ED-SCLC that were published in the last 20 years using network meta-analysis. Although platinum+ETP regimen appeared inferior to platinum+CPT11 regimen in terms of OS, a large majority of the published trials considered the former regimen as the standard. Patients treated with CBDCA+AMR, CBDCA+ETP+ATZ, platinum+ETP+DUR, and platinum+CPT11 showed better HRos than those treated with platinum+ETP doublet.
Supplemental Material
NMA_SCLC_suppl_v9 – Supplemental material for Systematic review of first-line chemotherapy for chemo-naïve extensive-stage small-cell lung cancer: network meta-analysis
Supplemental material, NMA_SCLC_suppl_v9 for Systematic review of first-line chemotherapy for chemo-naïve extensive-stage small-cell lung cancer: network meta-analysis by Hao Chen, Nobuyuki Horita, Kentaro Ito, Hideyuki Nagakura, Yu Hara, Nobuaki Kobayash, Masaki Yamamoto, Makoto Kudo and Takeshi Kaneko in Therapeutic Advances in Medical Oncology
Supplemental Material
PRISMA_2009_checklist_1 – Supplemental material for Systematic review of first-line chemotherapy for chemo-naïve extensive-stage small-cell lung cancer: network meta-analysis
Supplemental material, PRISMA_2009_checklist_1 for Systematic review of first-line chemotherapy for chemo-naïve extensive-stage small-cell lung cancer: network meta-analysis by Hao Chen, Nobuyuki Horita, Kentaro Ito, Hideyuki Nagakura, Yu Hara, Nobuaki Kobayash, Masaki Yamamoto, Makoto Kudo and Takeshi Kaneko in Therapeutic Advances in Medical Oncology
Supplemental Material
Supplementary_Table_1-7._League_tables – Supplemental material for Systematic review of first-line chemotherapy for chemo-naïve extensive-stage small-cell lung cancer: network meta-analysis
Supplemental material, Supplementary_Table_1-7._League_tables for Systematic review of first-line chemotherapy for chemo-naïve extensive-stage small-cell lung cancer: network meta-analysis by Hao Chen, Nobuyuki Horita, Kentaro Ito, Hideyuki Nagakura, Yu Hara, Nobuaki Kobayash, Masaki Yamamoto, Makoto Kudo and Takeshi Kaneko in Therapeutic Advances in Medical Oncology
Footnotes
Author contributions
HC supported data acquisition and drafting. NH led conception, designing, acquisition, analysis, interpretation, and drafting. KI, HN, YH, NK, MY, MK, and TK supported interpretation and revision. All the authors provided final approval and agreement for accountability for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Conflict of interest statement
The authors have the following conflicts of interest to disclose: KI: lecture fee from AstraZeneca and Chugai Pharmaceutical co. ltd. YH: lecture fee from AstraZeneca. NK: lecture fee from AstraZeneca and Chugai Pharmaceutical co. ltd. MK: lecture fee from AstraZeneca. KT: lecture fee from AstraZeneca and Chugai Pharmaceutical co. ltd.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
